8-Alkylcoumarins from the Fruits of Cnidium monnieri Protect against Hydrogen Peroxide Induced Oxidative Stress Damage
Abstract
:1. Introduction
2. Results and Discussion
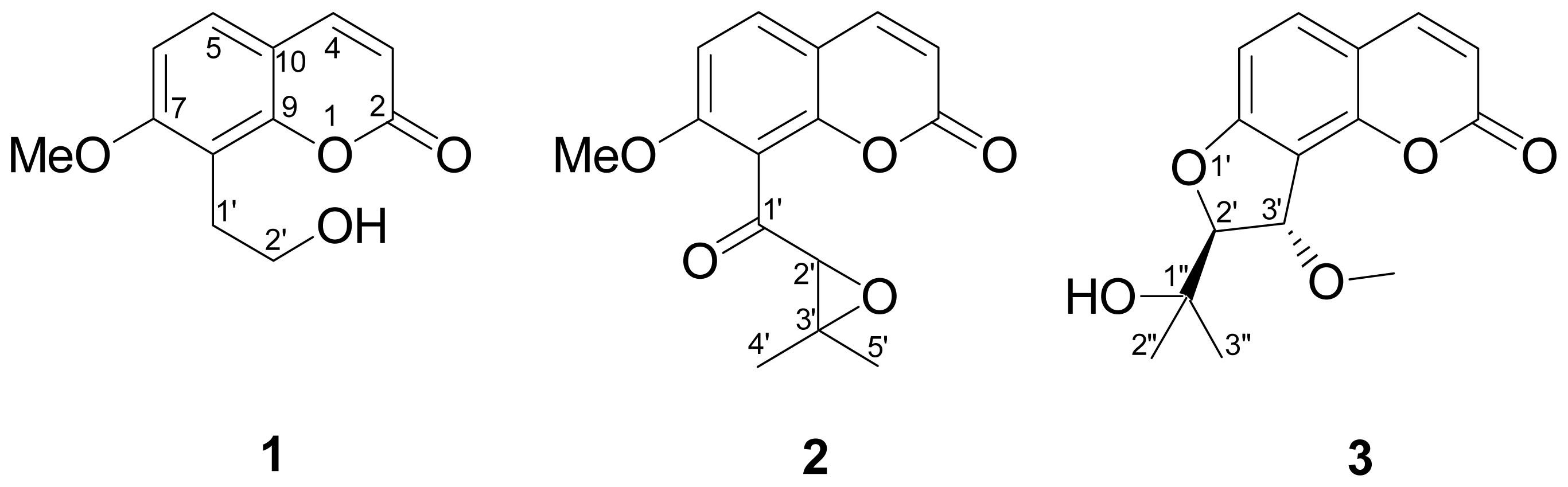
2.1. Isolation and Structural Elucidation
2.2. Cytoprotective Activity against Oxidative Stress
3. Experimental Section
3.1. Chemicals
3.2. General
3.3. Plant Material
3.4. Extraction and Isolation
3.5. Cell Culture
3.6. Cell Viability Assay
3.7. Statistical Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
- Author ContributionsYueh-Hsiung Kuo and Wei-Yong Lin designed research; Chi-I Chang, Wan-Chiao Hu, and Wei-Yong Lin performed research; Che-Piao Shen, Ban-Dar Hsu, Ping-Jyun Sung, Wei-Hsien Wang, and Jin-Bin Wu contributed new analytical tools and reagents; Yueh-Hsiung Kuo, Wei-Yong Lin, and Chi-I Chang wrote the paper.
References
- Ou, M. Chinese-English Manual of Commonly Used Traditional Chinese Medicine; Joint Publishing: Hong Kong, China, 1989; p. 522. [Google Scholar]
- Honda, G.; Tabata, M.; Baba, K.; Kozawa, M. On the antidermatophytic constituents and the original plants of the traditional Chinese drug “Shechuangzi”. Shoyakugaku Zasshi 1984, 38, 221–226. [Google Scholar]
- Basnet, P.; Yasuda, I.; Kumagai, N.; Tohda, C.; Nojima, H.; Kuraishi, Y.; Komatsu, K. Inhibition of itch-scratch response by fruits of Cnidium monnieri in mice. Biol. Pharm. Bull. 2001, 24, 1012–1015. [Google Scholar]
- Matsuda, H.; Tomohiro, N.; Ido, Y.; Kubo, M. Anti-allergic effects of Cnidium monnieri Fructus (dried fruits of Cnidium monnieri) and its major component osthol. Biol. Pharm. Bull. 2002, 25, 809–812. [Google Scholar]
- Zhang, Q.Y.; Qin, L.P.; He, W.D.; Puyvelde, L.V.; Adams, D.M.; Zheng, H.C.; Kimpe, N.D. Coumarins from Cnidium monnieri and their antiosteoporotic activity. Planta Med 2007, 73, 13–19. [Google Scholar]
- Juh, G.H.; Yu, S.M.; Ko, F.N.; Wu, T.S.; Teng, C.M. Antiproliferative effect in rat vascular smooth muscle cells by osthole isolated from Angelica pubescens. Eur. J. Pharmacol. 1996, 298, 191–197. [Google Scholar]
- Chiou, W.F.; Huang, Y.L.; Chen, C.F.; Chen, C.C. Vasorelaxing effect of coumarins from Cnidium monnieri on rabbit corpus cavernosum. Planta Med. 2001, 67, 282–284. [Google Scholar]
- Shin, E.; Lee, E.; Sung, S.H.; Kim, Y.C.; Hwang, B.Y.; Lee, M.K. Antifibrotic activity of coumarins from Cnidium monnieri fruits in HSC-T6 hepatic stellate cells. J. Nat. Med. 2011, 65, 370–374. [Google Scholar]
- Kim, S.B.; Ahn, J.H.; Han, S.B.; Hwang, B.Y.; Kim, S.Y.; Lee, M.K. Anti-adipogenic chromone glycosides from Cnidium monnieri fruits in 3T3-L1 cells. Bioorg. Med. Chem. Lett. 2012, 22, 6267–6271. [Google Scholar]
- Cai, J.N.; Basnet, P.; Wang, Z.T.; Komatsu, K.; Xu, L.S.; Tani, T. Coumarins from the fruits of Cnidium monnieri. J. Nat. Prod. 2000, 63, 485–488. [Google Scholar]
- Zhao, J.; Zhou, M.; Liu, Y.; Zhang, G.; Luo, Y. Chromones and coumarins from the dried fructus of Cnidium monnieri. Fitoterapia 2011, 82, 767–771. [Google Scholar]
- Oh, H.; Kim, J.S.; Song, E.K.; Cho, H.; Kim, D.H.; Park, S.E.; Lee, H.S.; Kim, Y.C. Sesquiterpenes with hepatoprotective activity from Cnidium monnieri on tacrine-induced cytotoxicity in Hep G2 cells. Planta Med. 2002, 68, 748–749. [Google Scholar]
- Zhou, F.; Zhong, W.; Xue, J.; Gu, Z.L.; Xie, M.L. Reduction of rat cardiac hypertrophy by osthol is related to regulation of cardiac oxidative stress and lipid metabolism. Lipids 2012, 47, 987–994. [Google Scholar]
- Zhang, J.; Xue, J.; Wang, H.; Zhang, Y.; Xie, M. Osthole improves alcohol-induced fatty liver in mice by reduction of hepatic oxidative stress. Phytother. Res. 2011, 25, 638–643. [Google Scholar]
- Zhao, Y.; Zhao, B. Oxidative stress and the pathogenesis of Alzheimer’s disease. Oxid. Med. Cell. Longev. 2013. [Google Scholar] [CrossRef]
- Gillmore, A.; Lauret, C.; Roberts, S.M. A route to the structure proposed for puetuberosanol and approaches to the natural products marshrin and phebalosin. Tetrahedron 2003, 59, 4363–4375. [Google Scholar]
- Ito, C.; Matsuoka, M.; Mizuno, T.; Sato, K.; Kimura, Y.; Ju-ichi, M.; Inoue, M.; Kajiura, I.; Omura, M.; Furukawa, H. New coumarins from some citrus plants. Chem. Pharm. Bull. 1988, 36, 3805–3810. [Google Scholar]
- Ito, C.; Furukawa, H. Two new coumarins from murraya plants. Chem. Pharm. Bull. 1989, 37, 819–820. [Google Scholar]
- Gray, A.I.; Meegan, C.J.; O’Callaghan, N.B. Coumarins and their taxonomic value in the genus. Phebalium. Phytochem. 1992, 31, 3083–3090. [Google Scholar]
- Ito, C.; Otsuka, T.; Ruangungsi, N.; Furukawa, H. Chemical constituents of Micromelum minutum Isolation and structural elucidation of new coumarins. Chem. Pharm. Bull. 2000, 48, 334–338. [Google Scholar]
- Imai, F.; Kinoshita, T.; Sankawa, U. Constituents of the leaves of Murraya paniculata collected in Taiwan. Chem. Pharm. Bull. 1989, 37, 358–362. [Google Scholar]
- Joshi, P.C.; Mandal, S.; Das, P.C.; Chatterjee, A. Two minor coumarins of Boenninghausenia albiflora. Phytochemistry 1993, 32, 481–483. [Google Scholar]
- Garg, T.K.; Chang, J.Y. Oxidative stress causes ERK phosphorylation and cell death in cultured retinal pigment epithelium: Prevention of cell death by AG126 and 15-deoxy-Δ12 14-PGJ2. BMC Ophthalmol. 2003, 3, 5:1–5:15. [Google Scholar]
- Kostoryz, E.L.; Zhu, Q.; Zhao, H.; Glaros, A.G.; Eick, J.D. Assessment of cytotoxicity and DNA damage exhibited by siloranes and oxiranes in cultured mammalian cells. Mutat. Res. 2007, 634, 156–162. [Google Scholar]
- Melnick, R.L. Carcinogenicity and mechanistic insights on the behavior of epoxides and epoxide-forming chemicals. Ann. N. Y. Acad. Sci. 2002, 982, 177–189. [Google Scholar]


| Position | Compound 1 | Compound 2 | Compound 3 | |||
|---|---|---|---|---|---|---|
| δH a | δC b | δH c | δC d | δH c | δC d | |
| 1 | ||||||
| 2 | 161.3 | 159.8 | 159.9 | |||
| 3 | 6.23 (d, J = 9.5) | 113.1 | 6.27 (d, J = 9.6) | 113.4 | 6.23 (d, J = 9.4) | 112.4 |
| 4 | 7.61 (d, J = 9.5) | 143.8 | 7.62 (d, J = 9.6) | 142.8 | 7.62 (d, J = 9.4) | 143.4 |
| 5 | 7.32 (d, J = 8.6) | 127.0 | 7.51 (d, J = 8.8) | 130.9 | 7.37 (d, J = 8.4) | 131.8 |
| 6 | 6.84 (d, J = 8.6) | 107.3 | 6.89 (d, J = 8.8) | 104.3 | 6.80 (d, J = 8.4) | 107.3 |
| 7 | 160.7 | 159.2 | 163.9 | |||
| 8 | 114.9 | 116.2 | 112.9 | |||
| 9 | 153.4 | 152.4 | 151.7 | |||
| 10 | 113.0 | 112.7 | 113.9 | |||
| 1′ | 3.15 (t, J = 6.7) | 26.3 | 195.2 | |||
| 2′ | 3.84 (t, J = 6.7) | 62.0 | 3.83 (s) | 66.8 | 4.51 (d, J = 2.8) | 96.1 |
| 3′ | 63.7 | 5.21 (d, J = 2.8) | 79.4 | |||
| 4′ | 1.45 (s) | 18.5 | ||||
| 5′ | 1.52 (s) | 24.8 | ||||
| 1″ | 71.4 | |||||
| 2″ | 1.25 (s) | 25.2 | ||||
| 3″ | 1.31 (s) | 25.7 | ||||
| OCH3 | 3.91 (s) | 56.1 | 3.92 (s) | 56.6 | 3.63 (s) | 57.9 |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chang, C.-I.; Hu, W.-C.; Shen, C.-P.; Hsu, B.-D.; Lin, W.-Y.; Sung, P.-J.; Wang, W.-H.; Wu, J.-B.; Kuo, Y.-H. 8-Alkylcoumarins from the Fruits of Cnidium monnieri Protect against Hydrogen Peroxide Induced Oxidative Stress Damage. Int. J. Mol. Sci. 2014, 15, 4608-4618. https://doi.org/10.3390/ijms15034608
Chang C-I, Hu W-C, Shen C-P, Hsu B-D, Lin W-Y, Sung P-J, Wang W-H, Wu J-B, Kuo Y-H. 8-Alkylcoumarins from the Fruits of Cnidium monnieri Protect against Hydrogen Peroxide Induced Oxidative Stress Damage. International Journal of Molecular Sciences. 2014; 15(3):4608-4618. https://doi.org/10.3390/ijms15034608
Chicago/Turabian StyleChang, Chi-I, Wan-Chiao Hu, Che-Piao Shen, Ban-Dar Hsu, Wei-Yong Lin, Ping-Jyun Sung, Wei-Hsien Wang, Jin-Bin Wu, and Yueh-Hsiung Kuo. 2014. "8-Alkylcoumarins from the Fruits of Cnidium monnieri Protect against Hydrogen Peroxide Induced Oxidative Stress Damage" International Journal of Molecular Sciences 15, no. 3: 4608-4618. https://doi.org/10.3390/ijms15034608





