Comparative Proteomic Analysis of Differential Responses of Pinus massoniana and Taxus wallichiana var. mairei to Simulated Acid Rain
Abstract
:1. Introduction
2. Results and Discussion
2.1. Results
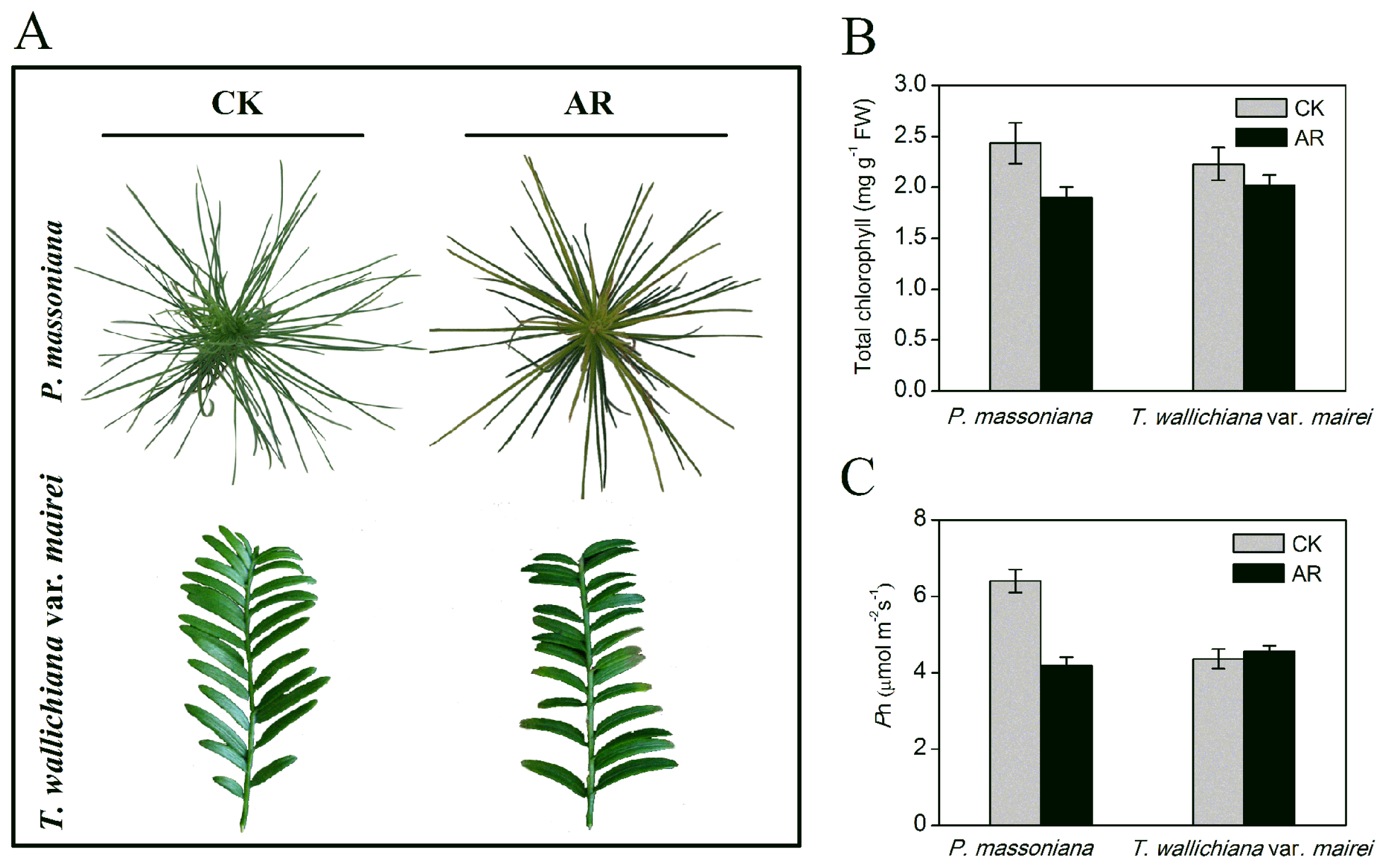
2.1.1. Phenotype and Physiological Changes in P. massoniana and T. wallichiana var. mairei under AR
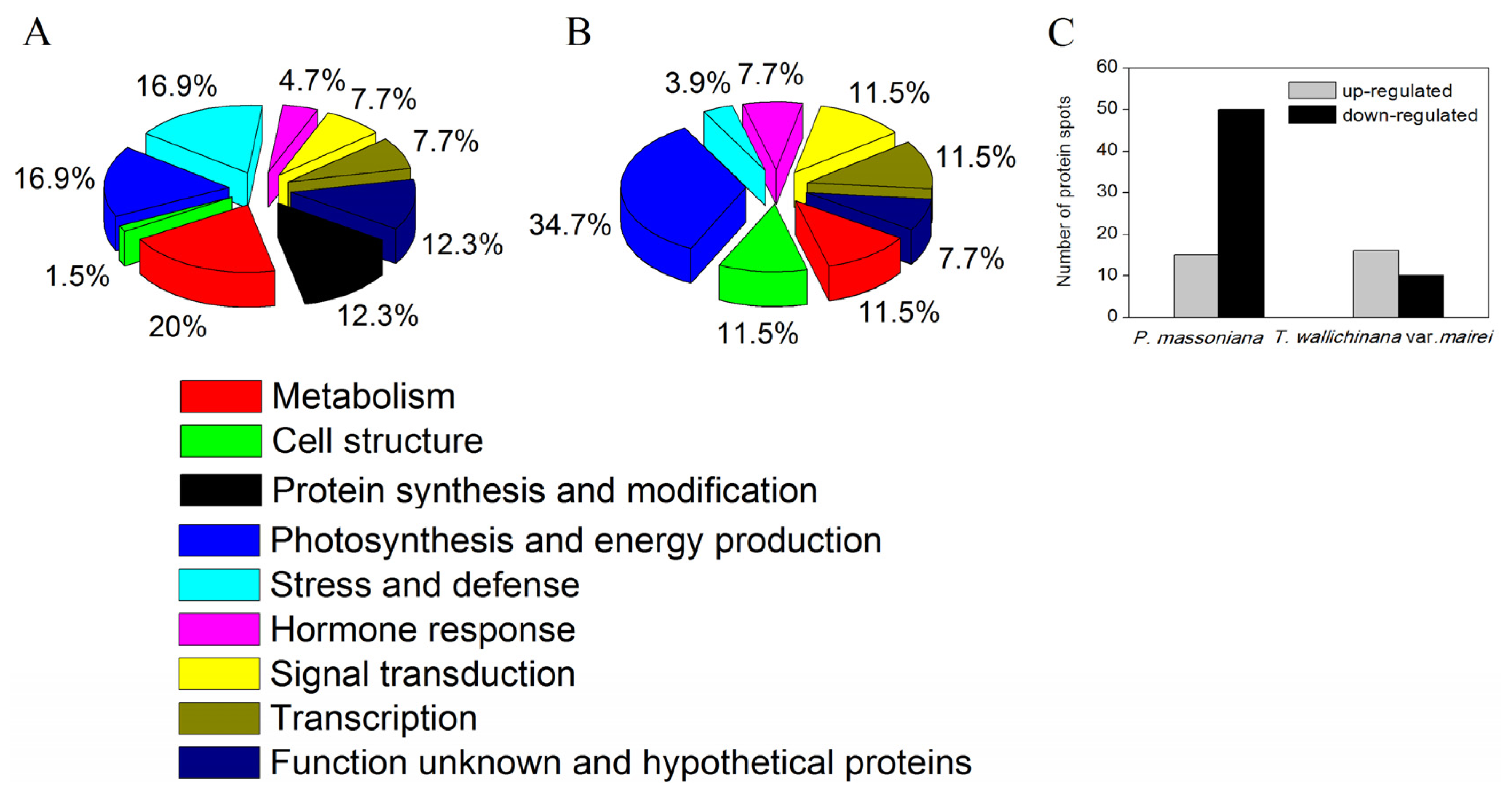
2.1.2. Identification and Functional Classification of AR-Responsive Proteins in P. massoniana and T. wallichiana var. mairei
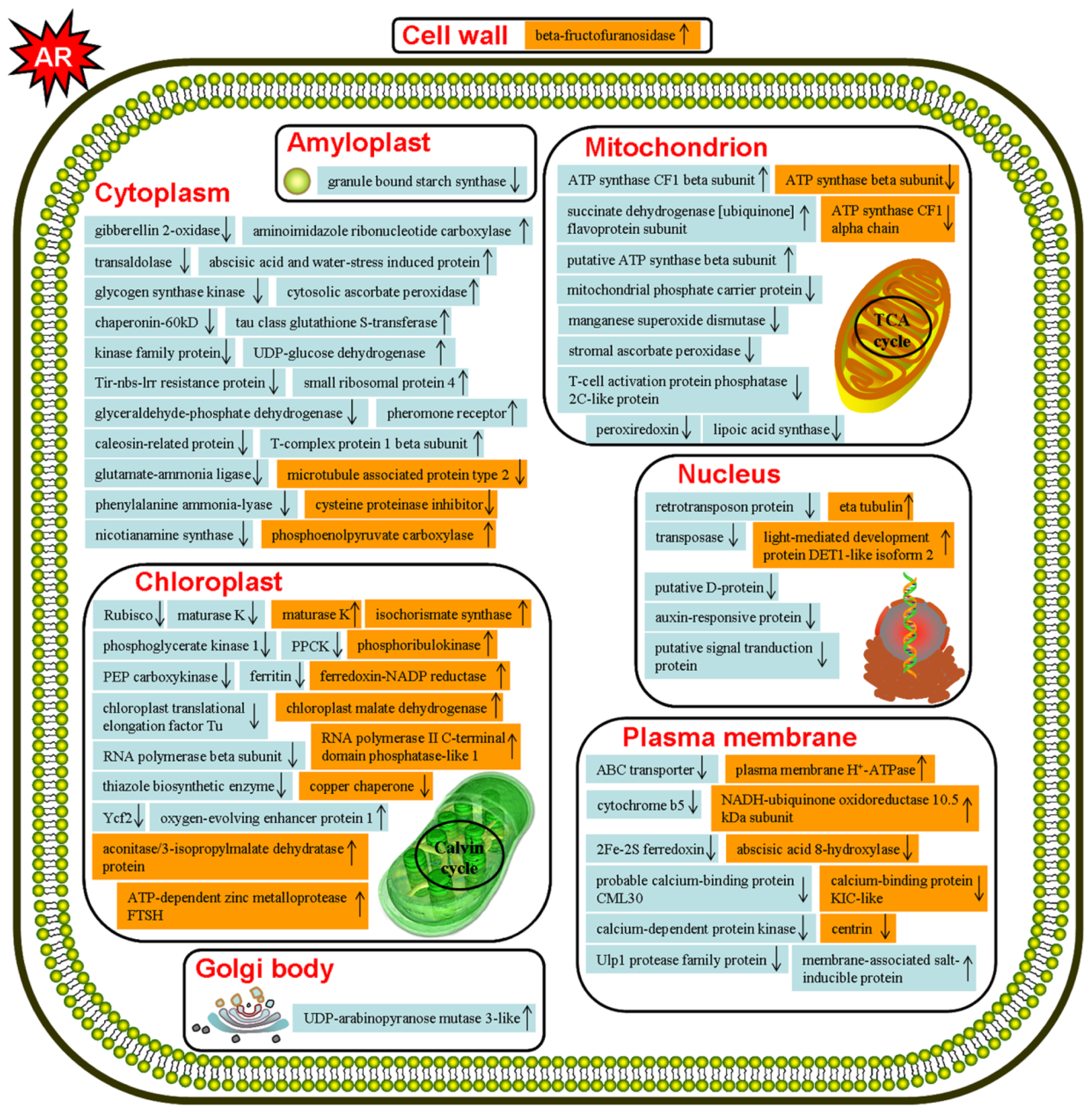
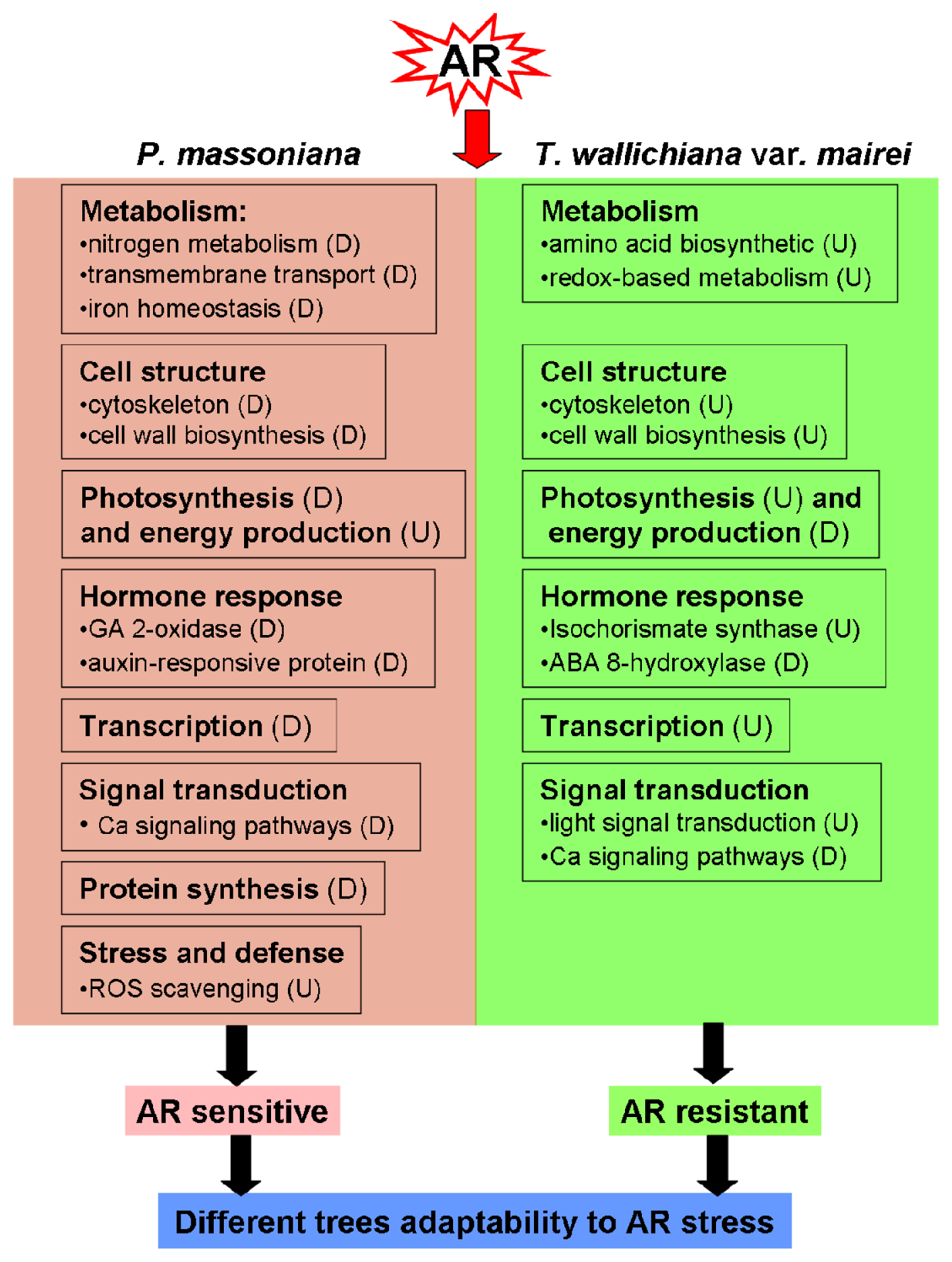
2.2. Discussion
2.2.1. Metabolism Related Proteins
2.2.2. Cell Structure Related Proteins
2.2.3. Protein Synthesis and Modification Related Proteins
2.2.4. Photosynthesis and Energy Production Related Proteins
2.2.5. Stress and Hormone Response Related Proteins
2.2.6. Signal Transduction Related Proteins
2.2.7. Transcription Related Proteins
3. Experimental Section
3.1. Plant Materials and Experimental Procedure
3.2. Chlorophyll Content and Net Photosynthetic Rate Measurements
3.3. Protein Extraction, 2-DE and 2-DE Gel Data Analysis
3.4. Protein Digestion and Identification
3.5. Statistical Analysis
4. Conclusions
Supplementary Information
ijms-15-04333-s001.pdfAcknowledgments
Conflicts of Interest
References
- Liu, J.; Diamond, J. China’s environment in a globalizing world. Nature 2005, 435, 1179–1186. [Google Scholar]
- Larssen, T.; Lydersen, E.; Tang, D.; He, Y.; Gao, J.; Liu, H.; Duan, L.; Seip, H.M.; Vogt, R.D.; Mulder, J. Acid rain in China. Environ. Sci. Technol. 2006, 40, 418–425. [Google Scholar]
- Likens, G.E.; Driscoll, C.T.; Buso, D.C. Long-term effects of acid rain: Response and recovery of a forest ecosystem. Science 1996, 272, 244–245. [Google Scholar]
- Malakoff, D. Taking the sting out of acid rain. Science 2010, 330, 910–911. [Google Scholar]
- Fan, H.B.; Wang, Y.H. Effects of simulated acid rain on germination foliar damage chlorophyll contents and seedling growth of five hardwood species growing in China. For. Ecol. Manag. 2000, 126, 321–329. [Google Scholar]
- Liu, J.; Zhou, G.; Yang, C.; Ou, Z.; Peng, C. Responses of chlorophyll fluorescence and xanthophyll cycle in leaves of Schima superba Gardn & Champ and Pinus massoniana Lamb to simulated acid rain at Dinghushan Biosphere Reserve China. Acta Physiol. Plant. 2007, 29, 33–38. [Google Scholar]
- Lee, Y.; Park, J.; Im, K.; Kim, K.; Lee, J.; Lee, K.; Park, J.A.; Lee, T.K.; Park, D.S.; Yang, J.S.; et al. Arabidopsis leaf necrosis caused by simulated acid rain is related to the salicylic acid signaling pathway. Plant Physiol. Biochem. 2006, 44, 38–42. [Google Scholar]
- Kong, F.; Liu, Y.; Hu, W.; Shen, P.; Zhou, C.; Wang, L. Biochemical responses of the mycorrhizae in Pinus massoniana to combined effects of Al Ca and low pH. Chemosphere 2000, 40, 311–318. [Google Scholar]
- Larssen, T.; Carmichael, G. Acid rain and acidification in China: The importance of base cation deposition. Environ. Pollut. 2000, 110, 89–102. [Google Scholar]
- Liu, T.W.; Wu, F.H.; Wang, W.H.; Chen, J.; Li, Z.J.; Dong, X.J.; Patton, J.; Pei, Z.M.; Zheng, H.L. Effects of calcium on seed germination seedling growth and photosynthesis of six forest tree species under simulated acid rain. Tree Physiol. 2011, 31, 402–413. [Google Scholar]
- Gao, L.; Möller, M.; Zhang, X.M.; Hollingsworth, M.; Liu, J.; Mill, R.; Gibby, M.; Li, D.Z. High variation and strong phylogeographic pattern among cpDNA haplotypes in Taxus wallichiana (Taxaceae) in China and North Vietnam. Mol. Ecol. 2007, 16, 4684–4698. [Google Scholar]
- Liu, K.; Yu, F.; Peng, S.; Fang, Y.; Li, F. Effects of simulated acid on saplings of Taxus species. Ecol. Environ. 2007, 16, 309–312. [Google Scholar]
- Liu, J.; Yang, C.; Wang, M. Effect of simulated acid rain on physiological characteristics in Taxus chinensis var. Mairei. Chin. J. Trop. Crop. 2012, 33, 1046–1050. [Google Scholar]
- Liu, T.; Jiang, X.; Shi, W.; Chen, J.; Pei, Z.; Zheng, H. Comparative proteomic analysis of differentially expressed proteins in β-aminobutyric acid enhanced Arabidopsis thaliana tolerance to simulated acid rain. Proteomics 2011, 11, 2079–2094. [Google Scholar]
- Liu, T.W.; Fu, B.; Niu, L.; Chen, J.; Wang, W.H.; He, J.X.; Pei, Z.M.; Zheng, H.L. Comparative proteomic analysis of proteins in response to simulated acid rain in Arabidopsis. J. Proteome Res. 2011, 10, 2579–2589. [Google Scholar]
- Wang, X.Q.; Liu, Z.; Niu, L.; Fu, B. Long-term effects of simulated acid rain stress on a staple forest plant Pinus massoniana Lamb: A proteomic analysis. Trees-Struct. Funct. 2013, 27, 297–309. [Google Scholar]
- Chen, J.; Wang, W.H.; Liu, T.W.; Wu, F.H.; Zheng, H.L. Photosynthetic and antioxidant responses of Liquidambar formosana and Schima superba seedlings to sulfuric-rich and nitric-rich simulated acid rain. Plant Physiol. Biochem. 2013, 64, 41–51. [Google Scholar]
- Bernard, S.M.; Habash, D.Z. The importance of cytosolic glutamine synthetase in nitrogen assimilation and recycling. New Phytol. 2009, 182, 608–620. [Google Scholar]
- Wang, H.; Ahan, J.; Wu, Z.; Shi, D.; Liu, B.; Yang, C. Alteration of nitrogen metabolism in rice variety ‘Nipponbare’ induced by alkali stress. Plant Soil 2012, 355, 131–147. [Google Scholar]
- Yousfi, S.; Serret, M.D.; Márquez, A.J.; Voltas, J.; Araus, J.L. Combined use of δ13C δ18O and δ15N tracks nitrogen metabolism and genotypic adaptation of durum wheat to salinity and water deficit. New Phytol. 2012, 194, 230–244. [Google Scholar]
- Hanke, G.T.; Kimata-Ariga, Y.; Taniguchi, I.; Hase, T. A post genomic characterization of Arabidopsis ferredoxins. Plant Physiol. 2004, 134, 255–264. [Google Scholar]
- Neuhaus, H.; Emes, M. Nonphotosynthetic metabolism in plastids. Annu. Rev. Plant Biol. 2000, 51, 111–140. [Google Scholar]
- Ceccarelli, E.A.; Arakaki, A.K.; Cortez, N.; Carrillo, N. Functional plasticity and catalytic efficiency in plant and bacterial ferredoxin-NADP (H) reductases. BBA-Proteins Proteom. 2004, 1698, 155–165. [Google Scholar]
- Rodriguez, R.E.; Lodeyro, A.; Poli, H.O.; Zurbriggen, M.; Peisker, M.; Palatnik, J.F.; Tognetti, V.B.; Tschiersch, H.; Hajirezaei, M.R.; Valle, E.M. Transgenic tobacco plants overexpressing chloroplastic ferredoxin-NADP (H) reductase display normal rates of photosynthesis and increased tolerance to oxidative stress. Plant Physiol. 2007, 143, 639–649. [Google Scholar]
- Kushnir, S.; Babiychuk, E.; Storozhenko, S.; Davey, M.W.; Papenbrock, J.; de Rycke, R.; Engler, G.; Stephan, U.W.; Lange, H.; Kispal, G. A mutation of the mitochondrial ABC transporter Sta1 leads to dwarfism and chlorosis in the Arabidopsis mutant starik. Plant Cell 2001, 13, 89–100. [Google Scholar]
- Bona, E.; Marsano, F.; Cavaletto, M.; Berta, G. Proteomic characterization of copper stress response in Cannabis sativa roots. Proteomics 2007, 7, 1121–1130. [Google Scholar]
- Ravet, K.; Touraine, B.; Boucherez, J.; Briat, J.F.; Gaymard, F.; Cellier, F. Ferritins control interaction between iron homeostasis and oxidative stress in Arabidopsis. Plant J. 2008, 57, 400–412. [Google Scholar]
- Jiang, Y.; Yang, B.; Harris, N.S.; Deyholos, M.K. Comparative proteomic analysis of NaCl stress-responsive proteins in Arabidopsis roots. J. Exp. Bot. 2007, 58, 3591–3607. [Google Scholar]
- Singh, B.; Mishra, R.; Agarwal, P.K.; Goswami, M.; Nair, S.; Sopory, S.; Reddy, M. A pea chloroplast translation elongation factor that is regulated by abiotic factors. Biochem. Biophys. Res. Commun. 2004, 320, 523–530. [Google Scholar]
- Jonak, C.; Hirt, H. Glycogen synthase kinase 3/SHAGGY-like kinases in plants: An emerging family with novel functions. Trends Plant Sci. 2002, 7, 457–461. [Google Scholar]
- Wan, X.Y.; Liu, J.Y. Comparative proteomics analysis reveals an intimate protein network provoked by hydrogen peroxide stress in rice seedling leaves. Mol. Cell. Proteomics 2008, 7, 1469–1488. [Google Scholar]
- Ohta, H.; Shibata, Y.; Haseyama, Y.; Yoshino, Y.; Suzuki, T.; Kagasawa, T.; Kamei, A.; Ikeuchi, M.; Enami, I. Identification of genes expressed in response to acid stress in Synechocystis sp PCC 6803 using DNA microarrays. Photosynth. Res. 2005, 84, 225–230. [Google Scholar]
- Ozturk, Z.N.; Talamé, V.; Deyholos, M.; Michalowski, C.B.; Galbraith, D.W.; Gozukirmizi, N.; Tuberosa, R.; Bohnert, H.J. Monitoring large-scale changes in transcript abundance in drought-and salt-stressed barley. Plant Mol. Biol. 2002, 48, 551–573. [Google Scholar]
- Gao, L.; Yan, X.; Li, X.; Guo, G.; Hu, Y.; Ma, W.; Yan, Y. Proteome analysis of wheat leaf under salt stress by two-dimensional difference gel electrophoresis (2D-DIGE). Phytochemistry 2011, 72, 1180–1191. [Google Scholar]
- Apel, K.; Hirt, H. Reactive oxygen species: Metabolism oxidative stress and signal transduction. Annu. Rev. Plant Biol. 2004, 55, 373–399. [Google Scholar]
- Magome, H.; Yamaguchi, S.; Hanada, A.; Kamiya, Y.; Oda, K. The DDF1 transcriptional activator upregulates expression of a gibberellin-deactivating gene GA2ox7 under high-salinity stress in Arabidopsis. Plant J. 2008, 56, 613–626. [Google Scholar]
- Wildermuth, M.C.; Dewdney, J.; Wu, G.; Ausubel, F.M. Isochorismate synthase is required to synthesize salicylic acid for plant defence. Nature 2001, 414, 562–565. [Google Scholar]
- Hepler, P.K. Calcium: A central regulator of plant growth and development. Plant Cell 2005, 17, 2142–2155. [Google Scholar]
- Kim, Y.Y.; Jung, K.W.; Yoo, K.S.; Jeung, J.U.; Shin, J.S. A stress-responsive caleosin-like protein AtCLO4 acts as a negative regulator of ABA responses in Arabidopsis. Plant Cell Physiol. 2011, 52, 874–884. [Google Scholar]
- DeHayes, D.H.; Schaberg, P.G.; Hawley, G.J.; Strimbeck, G.R. Acid rain impacts on calcium nutrition and forest health. BioScience 1999, 49, 789–800. [Google Scholar]
- Wellburn, R. The spectral determination of chlorophylls a and b as well as total carotenoids using various solvents with spectrophotometers of different resolution. J. Plant Physiol. 1994, 144, 307–313. [Google Scholar]
- Carpentier, S.C.; Witters, E.; Laukens, K.; Deckers, P.; Swennen, R.; Panis, B. Preparation of protein extracts from recalcitrant plant tissues: An evaluation of different methods for two-dimensional gel electrophoresis analysis. Proteomics 2005, 5, 2497–2507. [Google Scholar]
- Hu, W.J.; Chen, J.; Liu, T.W.; Liu, X.; Wu, F.H.; Wang, W.H.; He, J.X.; Xiao, Q.; Zheng, H.L. Comparative proteomic analysis on wild type and nitric oxide-overproducing mutant (nox1) of Arabidopsis thaliana. Nitric Oxide 2014, 36, 19–30. [Google Scholar]

| Spot a | NCBI accession b | Protein identity c | Thero.kDa/pI d | Exper.kDa/pI e | SC f | MP/TP g | Score h | C i | Species |
|---|---|---|---|---|---|---|---|---|---|
| Metabolism | |||||||||
| 4 | gi|308807529 | aminoimidazole ribonucleotide carboxylase | 62.31/6.2 | 27.37/5.18 | 17% | 9/14 | 97 | U | Ostreococcus tauri |
| 12 | gi|90718161 | granule bound starch synthase | 37.38/7.11 | 27.42/5.44 | 31% | 7/13 | 91 | D | Ficus hispidioides |
| 16 | gi|357122042 | UDP-arabinopyranose mutase 3-like | 41.34/6.02 | 48.06/5.46 | 25% | 9/12 | 95 | U | Brachypodium distachyon |
| 24 | gi|224122152 | mitochondrial phosphate carrier protein | 38.64/9.18 | 44.04/5.56 | 21% | 8/13 | 103 | D | Populus trichocarpa |
| 27 | gi|435103 | glyceraldehyde-phosphate dehydrogenase | 44.66/7.55 | 51.45/6.65 | 32% | 10/12 | 145 | D | Pinus sylvestris |
| 28 | gi|396547 | glutamate-ammonia ligase | 39.79/6.42 | 52.00/6.55 | 20% | 18/23 | 86 | D | Pinus sylvestris |
| 29 | gi|308806413 | putative D-protein | 22.80/6.74 | 31.71/6.42 | 32% | 7/9 | 98 | D | Ostreococcus tauri |
| 34 | gi|357448955 | nicotianamine synthase | 32.03/8.11 | 35.56/5.88 | 25% | 6/6 | 105 | D | Medicago truncatula |
| 39 | gi|303281782 | ABC transporter | 130.84/6.55 | 85.07/5.89 | 10% | 9/11 | 92 | D | Micromonas pusilla |
| 41 | gi|13873338 | lipoic acid synthase | 41.75/8.61 | 84.86/5.95 | 23% | 6/7 | 91 | D | Bruguiera gymnorhiza |
| 48 | gi|190899164 | 2Fe-2S ferredoxin | 18.33/8.52 | 20.94/6.21 | 38% | 4/4 | 81 | D | Populus tremula |
| 54 | gi|15240625 | transaldolase | 47.96/6.08 | 49.44/6.61 | 13% | 6/6 | 92 | D | Arabidopsis thaliana |
| 57 | gi|126583387 | ferritin | 28.26/5.66 | 49.03/5.29 | 24% | 4/4 | 75 | D | Triticum urartu |
| Cell structure | |||||||||
| 62 | gi|29028306 | UDP-glucose dehydrogenase | 53.52/6.06 | 87.31/4.70 | 21% | 7/8 | 101 | D | Colocasia esculenta |
| Protein synthesis and modification | |||||||||
| 6 | gi|357111489 | oxygen-evolving enhancer protein 1 | 24.81/4.97 | 37.64/4.72 | 26% | 6/10 | 91 | U | Brachypodium distachyon |
| 11 | gi|46811008 | small ribosomal protein 4 | 22.28/10.24 | 33.33/5.43 | 38% | 6/9 | 87 | U | Haplomitrium blumei |
| 20 | gi|308801835 | Ulp1 protease family protein | 68.17/9.21 | 22.11/6.90 | 22% | 12/15 | 117 | D | Ostreococcus tauri |
| 21 | gi|297830742 | kinase family protein | 43.77/5.79 | 33.72/5.55 | 31% | 7/10 | 97 | D | Arabidopsis lyrata subsp |
| 31 | gi|6525065 | chloroplast translational elongation factor Tu | 50.55/6.05 | 139.96/5.73 | 27% | 9/11 | 109 | D | Oryza sativa Japonica Group |
| 37 | gi|159477317 | T-complex protein 1 beta subunit | 57.40/5.37 | 58.03/5.88 | 30% | 14/18 | 152 | U | Chlamydomonas reinhardtii |
| 45 | gi|233142272 | glycogen synthase kinase | 47.06/8.64 | 31.97/6.00 | 25% | 8/12 | 100 | D | Glycine max |
| 49 | gi|255560267 | chaperonin-60kD | 61.48/6.20 | 24.54/6.25 | 14% | 6/7 | 76 | D | Ricinus communis |
| Photosynthesis and energy production | |||||||||
| 3 | gi|228016009 | ATP synthase CF1 beta subunit | 52.92/5.19 | 60.32/4.79 | 43% | 18/30 | 161 | U | Pinus resinosa |
| 9 | gi|357481701 | Ycf2 | 108.70/8.98 | 32.16/6.10 | 13% | 8/10 | 88 | D | Medicago truncatula |
| 17 | gi|56784992 | putative ATP synthase beta subunit | 45.27/5.26 | 64.05/5.08 | 47% | 13/19 | 167 | U | Oryza sativa Japonica Group |
| 18 | gi|220938463 | phosphoenolpyruvate carboxykinase (PEP carboxykinase) | 55.85/6.63 | 89.72/5.19 | 19% | 7/9 | 95 | D | Hyparrhenia hirta |
| 35 | gi|18073888 | phosphoenolpyruvate carboxylase(PPCK) | 41.34/7.74 | 52.73/5.88 | 24% | 6/7 | 94 | D | Leptotes bicolor |
| 44 | gi|31281466 | ribulose-1,5-bisphosphate carboxylase/oxygenase large subunit(Rubisco) | 52.11/6.09 | 21.97/6.05 | 16% | 8/14 | 88 | D | Pinus monophylla |
| 50 | gi|166714465 | ribulose-1,5-bisphosphate carboxylase/oxygenase large subunit(Rubisco) | 51.60/6.00 | 59.41/6.11 | 36% | 21/27 | 223 | D | Pinus parviflora |
| 53 | gi|332591479 | phosphoglycerate kinase 1 | 52.94/8.84 | 50.50/6.73 | 36% | 15/22 | 153 | D | Pinus pinaster |
| 55 | gi|34733684 | ribulose-1,5-bisphosphate carboxylase/oxygenase large subunit(Rubisco) | 48.97/6.34 | 21.49/4.63 | 14% | 6/6 | 90 | D | Gnetum hainanense |
| 60 | gi|264160443 | ribulose 1,5-bisphosphate carboxylase | 46.11/6.08 | 82.57/4.66 | 14% | 8/11 | 87 | D | Callistephus chinensis |
| 65 | gi|357111628 | succinate dehydrogenase [ubiquinone] flavoprotein subunit | 68.79/6.18 | 84.59/5.93 | 14% | 7/8 | 90 | U | Brachypodium distachyon |
| Stress and defense | |||||||||
| 5 | gi|255575353 | peroxiredoxin | 23.94/7.63 | 19.53/5.23 | 22% | 5/6 | 83 | D | Ricinus communis |
| 10 | gi|192912966 | cytosolic ascorbate peroxidase | 27.550/5.42 | 33.28/5.22 | 38% | 7/9 | 98 | U | Elaeis guineensis |
| 13 | gi|289187423 | tau class glutathione S-transferase | 26.47/5.56 | 29.44/5.44 | 36% | 11/22 | 95 | U | Pinus brutia |
| 14 | gi|289187423 | tau class glutathione S-transferase | 26.47/5.56 | 29.21/5.25 | 35% | 10/14 | 103 | U | Pinus brutia |
| 30 | gi|66841104 | manganese superoxide dismutase | 13.31/5.85 | 29.40/6.52 | 46% | 5/6 | 97 | D | Larix gmelinii |
| 33 | gi|154101561 | phenylalanine ammonia-lyase | 24.86/6.54 | 32.30/5.86 | 27% | 5/7 | 84 | D | Scutellaria baicalensis |
| 36 | gi|380863088 | stromal ascorbate peroxidase | 80.54/9.10 | 63.15/5.81 | 57% | 4/4 | 92 | D | Dimocarpus longan |
| 43 | gi|357513733 | Tir-nbs-lrr resistance protein | 140.72/5.31 | 140.40/5.95 | 12% | 12/15 | 110 | D | Medicago truncatula |
| 51 | gi|195620494 | membrane-associated salt-inducible protein | 43.66/9.02 | 114.61/6.19 | 20% | 7/9 | 102 | U | Zea mays |
| 59 | gi|308804281 | cytochrome b5 | 16.34/7.90 | 55.92/5.86 | 45% | 5/6 | 92 | D | Ostreococcus tauri |
| 64 | gi|56481813 | thiazole biosynthetic enzyme | 36.63/6.01 | 35.00/5.51 | 29% | 8/11 | 85 | D | Pseudotsuga menziesii |
| Hormone response | |||||||||
| 7 | gi|212725010 | abscisic acid and water-stress induced protein | 16.30/9.55 | 40.39/4.95 | 28% | 4/5 | 81 | U | Pinus sylvestris |
| 40 | gi|224108798 | gibberellin 2-oxidase | 34.47/5.44 | 84.30/5.98 | 29% | 8/10 | 106 | D | Populus trichocarpa |
| 47 | gi|357485291 | auxin-responsive protein | 36.69/8.52 | 21.95/6.15 | 18% | 6/6 | 80 | D | Medicago truncatula |
| Signal transduction | |||||||||
| 8 | gi|145336050 | caleosin-related protein | 23.89/9.62 | 116.00/4.47 | 40% | 6/7 | 97 | D | Arabidopsis thaliana |
| 25 | gi|22128710 | putative signal tranduction protein | 97.72/6.16 | 54.98/5.62 | 13% | 9/11 | 101 | D | Oryza sativa Japonica Group |
| 42 | gi|226494574 | T-cell activation protein phosphatase 2C-like protein | 34.59/7.70 | 140.00/5.88 | 22% | 5/5 | 84 | D | Zea mays |
| 52 | gi|225425656 | probable calcium-binding protein CML30 | 20.64/4.66 | 32.35/6.55 | 30% | 5/5 | 97 | D | Vitis vinifera |
| 63 | gi|357440111 | calcium-dependent protein kinase | 54.85/5.85 | 21.74/5.42 | 17% | 7/8 | 84 | D | Medicago truncatula |
| Transcription | |||||||||
| 15 | gi|384584973 | maturase K | 31.08/10.17 | 38.94/5.31 | 30% | 6/8 | 100 | D | Cynodon nlemfuensis |
| 22 | gi|313199657 | RNA polymerase beta subunit | 120.90/6.41 | 32.90/5.60 | 9% | 8/9 | 78 | D | Isoetes flaccida |
| 23 | gi|372482380 | RNA polymerase beta subunit | 158.24/9.38 | 38.65/5.46 | 11% | 14/20 | 109 | D | Sansevieria trifasciata |
| 26 | gi|18419497 | transposase | 15.77/8.93 | 69.37/5.69 | 44% | 5/6 | 89 | D | Oryza nivara |
| 56 | gi|108862655 | retrotransposon protein | 21.22/8.62 | 56.71/6.93 | 32% | 6/8 | 93 | D | Oryza sativa Japonica Group |
| Function unknown and hypothetical proteins | |||||||||
| 1 | gi|326501884 | predicted protein | 53.01/5.32 | 18.56/4.20 | 18% | 7/11 | 84 | D | Hordeum vulgare subsp |
| 2 | gi|21593511 | pheromone receptor | 39.28/4.44 | 33.13/4.56 | 24% | 5/6 | 83 | D | Arabidopsis thaliana |
| 19 | gi|118486611 | unknown | 43.94/6.43 | 35.36/6.07 | 19% | 6/9 | 78 | D | Populus trichocarpa |
| 32 | gi|116780007 | unknown | 25.59/5.82 | 30.20/5.92 | 45% | 13/29 | 120 | D | Picea sitchensis |
| 38 | gi|148906365 | unknown | 48.34/5.78 | 63.66/5.90 | 29% | 11/16 | 109 | U | Picea sitchensis |
| 46 | gi|116789937 | unknown | 54.01/6.00 | 57.72/6.04 | 23% | 12/17 | 116 | D | Picea sitchensis |
| 58 | gi|357488033 | hypothetical protein MTR_5g047930 | 54.35/4.92 | 47.79/5.26 | 19% | 6/9 | 78 | D | Medicago truncatula |
| 61 | gi|242033729 | hypothetical protein SORBIDRAFT_01g015060 | 18.87/4.75 | 36.24/4.67 | 32% | 5/6 | 85 | U | Sorghum bicolor |
| Spot a | NCBI accession b | Protein identity c | Thero.kDa/pI d | Exper.kDa/pI e | SC f | MP/TP g | Score h | C i | Species |
|---|---|---|---|---|---|---|---|---|---|
| Metabolism | |||||||||
| 9 | gi|3913651 | ferredoxin-NADP reductase | 40.71/8.37 | 11.75/5.40 | 35% | 6/9 | 88 | U | Nicotiana tabacum |
| 13 | gi|15228869 | copper chaperone | 13.08/4.91 | 39.62/5.55 | 33% | 4/4 | 87 | D | Arabidopsis thaliana |
| 21 | gi|334184891 | aconitase/3-isopropylmalate dehydratase protein | 23.88/6.16 | 28.02/4.47 | 36% | 6/8 | 96 | U | Arabidopsis thaliana |
| Cell structure | |||||||||
| 14 | gi|2500930 | beta-fructofuranosidase | 62.90/7.07 | 46.39/5.49 | 13% | 6/6 | 94 | U | Pisum sativum |
| 23 | gi|357521323 | microtubule associated protein type 2 | 70.55/9.05 | 39.64/5.49 | 20% | 9/12 | 102 | D | Medicago truncatula |
| 25 | gi|159490038 | eta tubulin | 35.80/7.75 | 23.31/5.80 | 29% | 6/8 | 94 | U | Chlamydomonas reinhardtii |
| Photosynthesis and energy production | |||||||||
| 1 | gi|226498532 | NADH-ubiquinone oxidoreductase 10.5 kDa subunit | 11.28/9.15 | 15.08/4.64 | 47% | 4/4 | 85 | U | Zea mays |
| 3 | gi|138277483 | ATP synthase beta subunit | 51.67/5.11 | 63.61/4.90 | 42% | 18/30 | 170 | D | Taxus brevifolia |
| 5 | gi|138277483 | ATP synthase beta subunit | 51.67/5.11 | 62.00/5.08 | 45% | 20/37 | 167 | D | Taxus brevifolia |
| 10 | gi|357137138 | phosphoribulokinase | 45.31/5.97 | 40.14/5.21 | 27% | 7/11 | 95 | U | Brachypodium distachyon |
| 11 | gi|225459844 | ATP-dependent zinc metalloprotease FTSH | 75.76/6.36 | 84.83/5.33 | 27% | 15/28 | 124 | U | Vitis vinifera |
| 12 | gi|7592732 | plasma membrane H+-ATPase | 22.04/8.92 | 31.30/5.47 | 38% | 6/6 | 102 | U | Nepenthes alata |
| 15 | gi|150251443 | ATP synthase CF1 alpha chain | 55.36/5.38 | 57.82/5.53 | 15% | 9/12 | 99 | D | Cycas taitungensis |
| 16 | gi|154146830 | phosphoenolpyruvate carboxylase | 49.31/6.21 | 29.94/5.82 | 29% | 8/10 | 118 | U | Cymbopogon citratus |
| 20 | gi|350536787 | chloroplast malate dehydrogenase | 47.79/5.92 | 28.80/6.19 | 23% | 8/8 | 110 | U | Solanum lycopersicum |
| Stress and defense | |||||||||
| 6 | gi|357520455 | cysteine proteinase inhibitor | 25.78/7.78 | 28.19/5.04 | 21% | 6/6 | 84 | D | Medicago truncatula |
| Hormone response | |||||||||
| 4 | gi|76009223 | isochorismate synthase | 13.34/8.47 | 11.83/5.11 | 32% | 4/4 | 82 | U | Solanum lycopersicum |
| 19 | gi|335346406 | abscisic acid 8-hydroxylase | 53.18/8.77 | 30.53/6.17 | 17% | 7/9 | 88 | D | Ipomoea nil |
| Signal transduction | |||||||||
| 2 | gi|357132195 | light-mediated development protein DET1-like isoform 2 | 48.61/8.53 | 49.88/4.76 | 17% | 7/9 | 95 | U | Brachypodium distachyon |
| 8 | gi|384245525 | calcium-binding protein CML19 (centrin) | 19.59/4.75 | 19.99/5.44 | 31% | 4/4 | 78 | D | Coccomyxa subellipsoidea |
| 17 | gi|356573251 | calcium-binding protein KIC-like | 14.00/4.18 | 28.30/5.78 | 47% | 5/5 | 85 | D | Glycine max |
| Transcription | |||||||||
| 7 | gi|154082680 | maturase K | 57.14/9.40 | 10.70/5.33 | 12% | 6/6 | 85 | U | Haplophyllum buxbaumii |
| 24 | gi|79481163 | RNA polymerase II C-terminal domain phosphatase-like 1 | 109.15/5.81 | 30.99/5.62 | 15% | 10/13 | 103 | U | Arabidopsis thaliana |
| 26 | gi|15341050 | maturase K | 33.04/9.65 | 25.36/5.83 | 31% | 7/11 | 99 | U | Persicaria runcinata |
| Function unknown and hypothetical proteins | |||||||||
| 18 | gi|125547814 | hypothetical protein OsI_15422 | 45.62/6.20 | 38.91/6.03 | 22% | 7/10 | 94 | U | Oryza sativa Indica Group |
| 22 | gi|296087931 | unnamed protein product | 79.30/7.32 | 15.63/5.96 | 20% | 11/16 | 106 | D | Vitis vinifera |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hu, W.-J.; Chen, J.; Liu, T.-W.; Simon, M.; Wang, W.-H.; Chen, J.; Wu, F.-H.; Liu, X.; Shen, Z.-J.; Zheng, H.-L. Comparative Proteomic Analysis of Differential Responses of Pinus massoniana and Taxus wallichiana var. mairei to Simulated Acid Rain. Int. J. Mol. Sci. 2014, 15, 4333-4355. https://doi.org/10.3390/ijms15034333
Hu W-J, Chen J, Liu T-W, Simon M, Wang W-H, Chen J, Wu F-H, Liu X, Shen Z-J, Zheng H-L. Comparative Proteomic Analysis of Differential Responses of Pinus massoniana and Taxus wallichiana var. mairei to Simulated Acid Rain. International Journal of Molecular Sciences. 2014; 15(3):4333-4355. https://doi.org/10.3390/ijms15034333
Chicago/Turabian StyleHu, Wen-Jun, Juan Chen, Ting-Wu Liu, Martin Simon, Wen-Hua Wang, Juan Chen, Fei-Hua Wu, Xiang Liu, Zhi-Jun Shen, and Hai-Lei Zheng. 2014. "Comparative Proteomic Analysis of Differential Responses of Pinus massoniana and Taxus wallichiana var. mairei to Simulated Acid Rain" International Journal of Molecular Sciences 15, no. 3: 4333-4355. https://doi.org/10.3390/ijms15034333




