Anthelmintic Activities of Aporphine from Nelumbo nucifera Gaertn. cv. Rosa-plena against Hymenolepis nana
Abstract
:1. Introduction
2. Results and Discussion
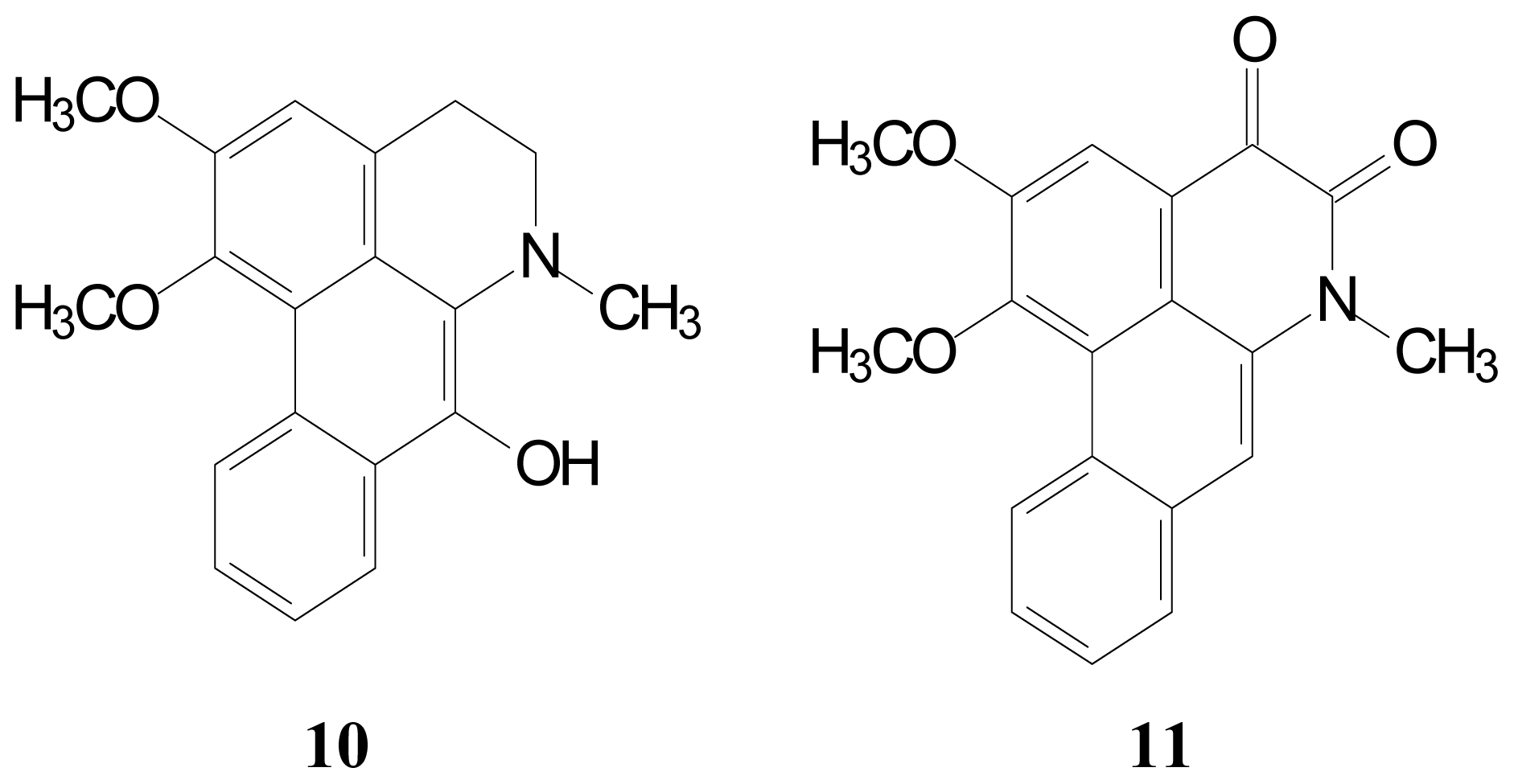
2.1. Isolation and Characterization of Aporphine Derivatives
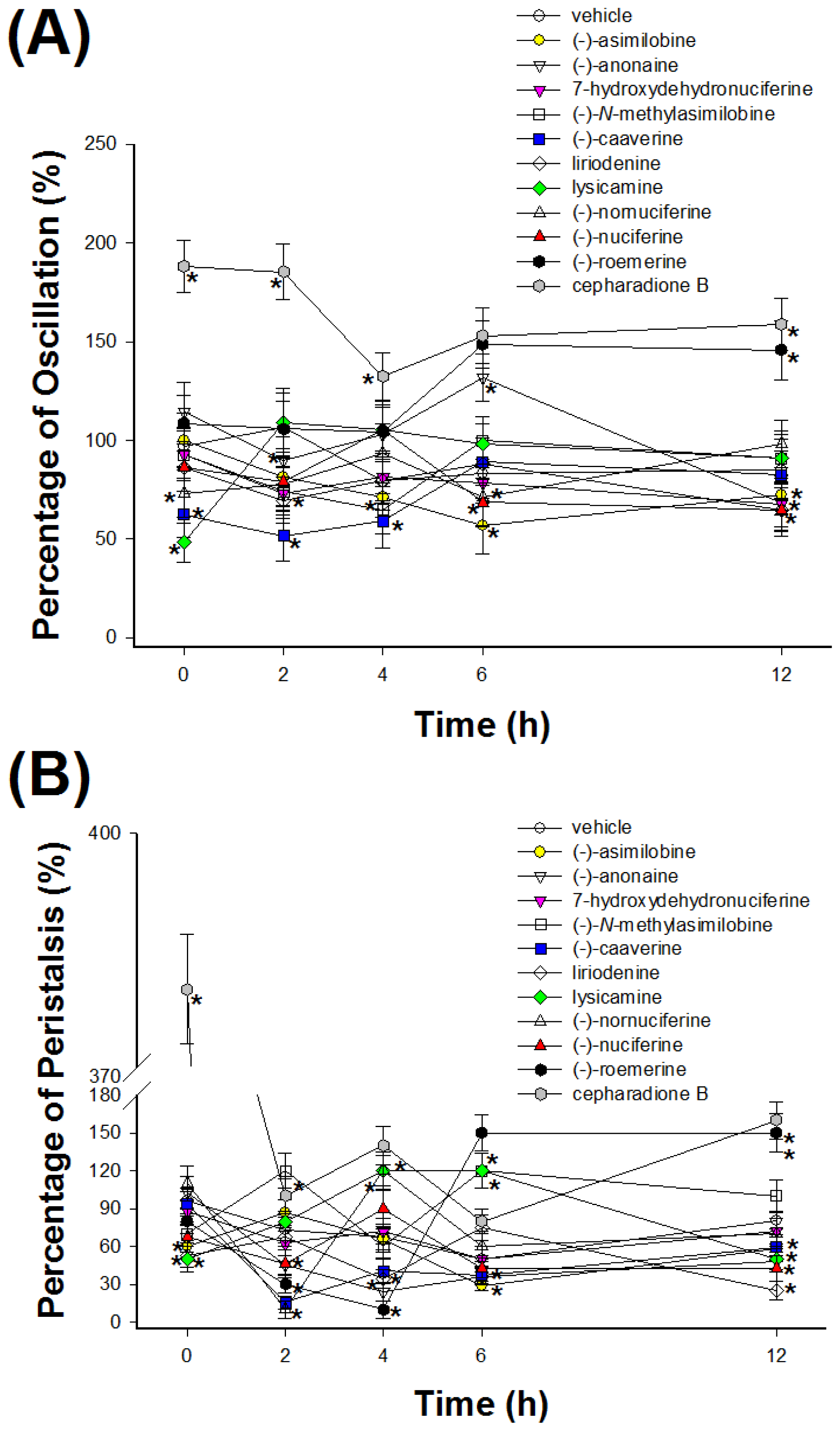
2.2. Cestocidal Activity against H. nana
2.3. Nematocidal Activity against A. simplex
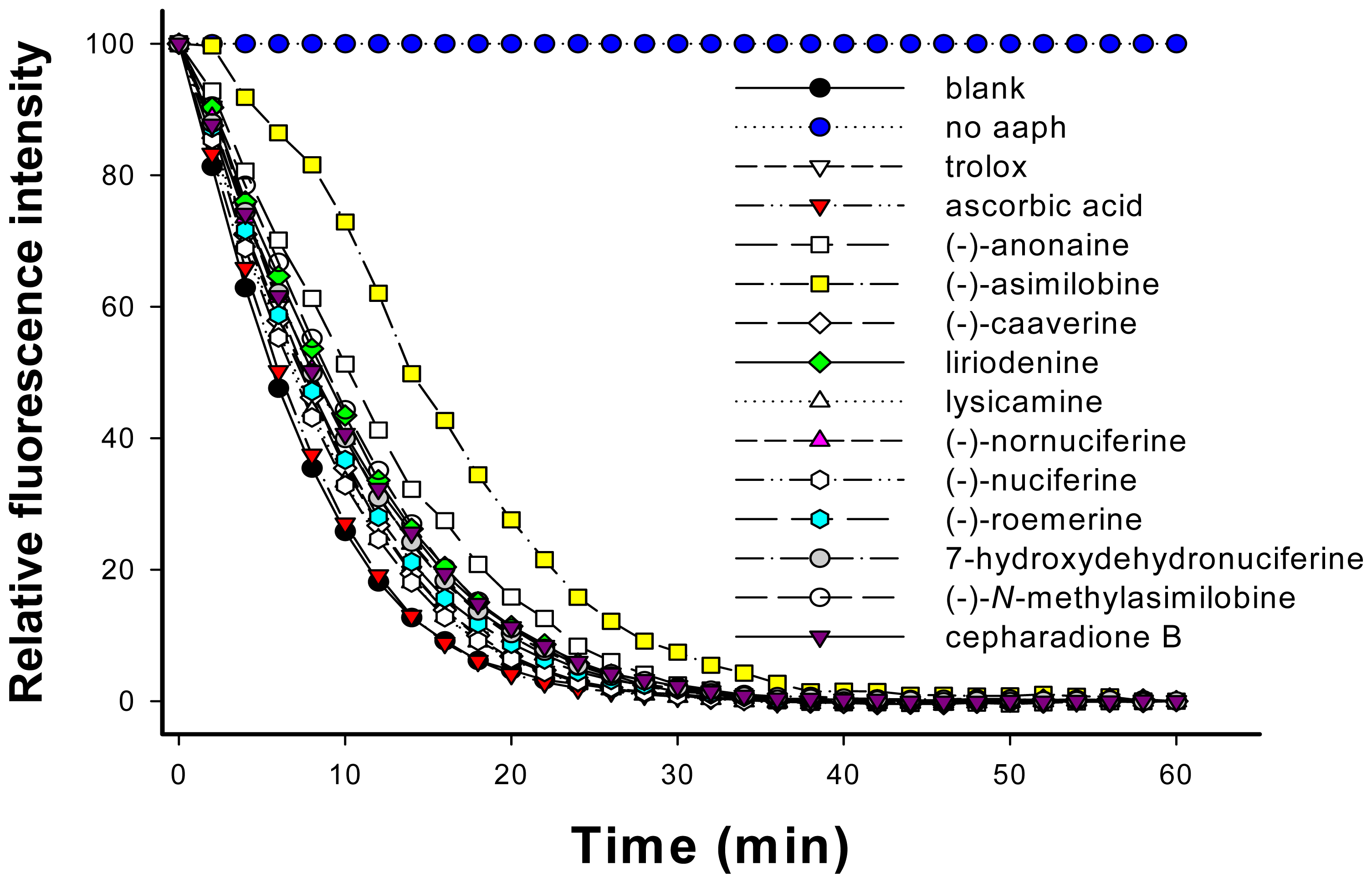
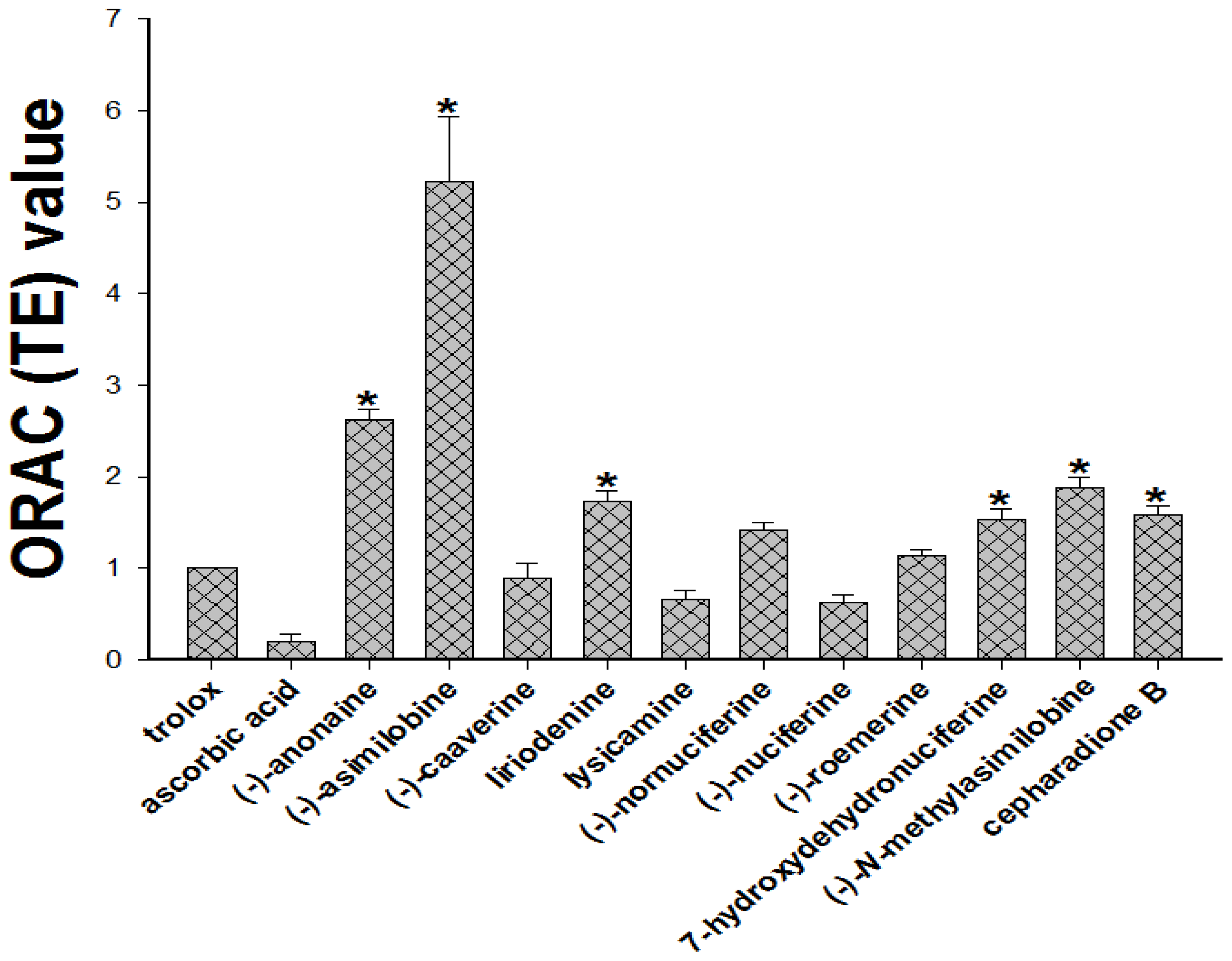
2.4. Determination of Oxygen Radical Absorbing Capacity (ORAC)
3. Materials and Methods
3.1. Drugs and Chemicals
3.2. Extraction and Isolation
3.3. Preparation of H. nana Adult Worms
3.4. Assay of Cestocidal Activity of Oscillation and Peristalsis Test on H. nana
3.5. A. simplex Larvae Preparation
3.6. Assay of Nematocidal Activity on A. simplex
3.7. Evaluation of Oxygen Radical Absorbing Capacity (ORAC)
3.8. Statistical Analysis
4. Conclusions
Acknowledgments
- Author ContributionsR.-J.L., M.-H.W., and Y.-H.M.: acquisition of data; analysis and interpretation of data; statistical analysis; drafting of the manuscript; funding obtained; and study supervision. All authors read and approved the final manuscript. R.-J.L. and C.-M.Y.: administrative support; and study supervision. L.-Y.C. and C.-Y.C.: review of the manuscript.
Conflicts of Interest
References
- Kashiwada, Y.; Aoshima, A.; Ikeshiro, Y.; Chen, Y.P.; Furukawa, H.; Itoigawa, M.; Fujioka, T.; Mihashi, K.; Cosentino, L.M.; Morris-Natschke, S.L.; et al. Anti-HIV benzylisoquinoline alkaloids and flavonoids from the leaves of Nelumbo nucifera and structure-activity correlations with related alkaloids. Bioorgan. Med. Chem. 2005, 13, 443–448. [Google Scholar]
- Do, T.C.; Nguyen, T.D.; Tran, H.; Stuppner, H.; Ganzera, M. Analysis of alkaloids in Lotus (Nelumbo nucifera Gaertn) leaves by non-aqueous capillary electrophoresis using ultraviolet and mass spectrometric detection. J. Chromatogr. 2013, 1302, 174–180. [Google Scholar]
- Yang, T.H.; Chen, C.M.; Lu, C.S.; Liao, C.L. Studies on the alkaloids of lotus receptacle. J. Chin. Chem. Soc. 1972, 19, 143–147. [Google Scholar]
- Chitchang, S.; Piamjinda, T.; Yodmani, B.; Radomyos, P. Relationship between severity of the symptom and the number of Hymenolepis nana after treatment. J. Med. Assoc. Thail. 1985, 68, 423–426. [Google Scholar]
- Ito, A. Basic and applied immunology in cestode infections: From Hymenolepis to Taenia and Echinococcus. Int. J. Parasitol. 1997, 27, 1203–1211. [Google Scholar]
- Audicana, M.T.; Kennedy, M.W. Anisakis simplex: From obscure infectious worm to inducer of immune hypersensitivity. Clin. Microbiol. Rev. 2008, 21, 360–379. [Google Scholar]
- Martin-Blazquez, A.; Chalumeau, C.; Ortega-Deballon, P. Anisakiasis and anisakidosis. Gastroenterol. Clin. Biol. 2009, 33, 185–186. [Google Scholar]
- Setyobudi, E.; Jeon, C.H.; Lee, C.H.; Seong, K.B.; Kim, J.H. Occurrence and identification of Anisakis spp (Nematoda:Anisakidae) isolated from chum salmon (Oncorhynchus keta) in Korea. Parasitol. Res. 2011, 108, 585–592. [Google Scholar]
- Kim, J.; Jo, J.O.; Choi, S.H.; Cho, M.K.; Yu, H.S.; Cha, H.J.; Ock, M. Seroprevalence of antibodies against Anisakis simplex larvae among health-examined residents in three hospitals of southern parts of Korea. Korean J. Parasitol. 2011, 49, 139–144. [Google Scholar]
- Anadon, A.M.; Romaris, F.; Escalante, M.; Rodriguez, E.; Garate, T.; Cuellar, C.; Ubeira, F.M. The Anisakis simplex Ani s 7 major allergen as an indicator of true Anisakis infections. Clin. Exp. Immunol. 2009, 156, 471–478. [Google Scholar]
- Lopes, N.P.; Chicaro, P.; Kato, M.J.; Albuquerque, S.; Yoshida, M. Flavonoids and lignans from Virola surinamensis twigs and their in vitro activity against Trypanosoma cruzi. Planta Med. 1998, 64, 667–668. [Google Scholar]
- Diallo, D.; Marston, A.; Terreaux, C.; Toure, Y.; Paulsen, B.S.; Hostettmann, K. Screening of Malian medicinal plants for antifungal larvicidal molluscicidal antioxidant and radical scavenging activities. Phytother. Res. 2001, 15, 401–406. [Google Scholar]
- Chang, F.R.; Chen, C.Y.; Hsieh, T.J.; Cho, C.P.; Wu, Y.C. Chemical constituents from Annona glabra III. J. Chin. Chem. Soc. 2000, 47, 913–920. [Google Scholar]
- Chen, C.Y.; Chang, F.R.; Wu, Y.C. The chemical constituents from the stems of Annona cherimola. J. Chin. Chem. Soc. 1997, 44, 313–319. [Google Scholar]
- Han, B.H.; Park, M.H.; Park, J.H. Chemical and pharmacological studies on sedative cyclopeptide alkaloids in some Rhamnaceae plants. Pure Appl. Chem. 1989, 61, 443–448. [Google Scholar]
- Wafo, P.; Nyasse, B.; Fontaine, C.; Sondengam, B.L. Aporphine alkaloids from Enantia chlorantha. Fitoterapia 1999, 70, 157–160. [Google Scholar]
- Kim, S.K.; Ryu, S.Y.; No, J.; Choi, S.U.; Kim, Y.S. Cytotoxic alkaloids from Houttuynia cordata. Arch. Pharm. Res. 2001, 24, 518–521. [Google Scholar]
- Lin, R.J.; Yen, C.M.; Chou, T.H.; Chiang, F.Y.; Wang, G.H.; Tseng, Y.P.; Wang, L.; Huang, T.W.; Wang, H.C.; Chan, L.P.; et al. Antioxidant anti-adipocyte differentiation antitumor activity and anthelmintic activities against Anisakis simplex and Hymenolepis nana of yakuchinone A from Alpinia oxyphylla. BMC Complement. Altern. Med. 2013, 13, 237. [Google Scholar]
- Kundu, S.; Roy, S.; Lyndem, L.M. Cassia alata L: Potential role as anthelmintic agent against Hymenolepis diminuta. Parasitol. Res. 2012, 111, 1187–1192. [Google Scholar]
- Lin, R.J.; Chen, C.Y.; Lee, J.D.; Lu, C.M.; Chung, L.Y.; Yen, C.M. Larvicidal constituents of Zingiber officinale (ginger) against Anisakis simplex. Planta Med. 2010, 76, 1852–1858. [Google Scholar]
- Iglesias, L.; Valero, A.; Benitez, R.; Adroher, F.J. In vitro cultivation of Anisakis simplex: Pepsin increases survival and moulting from fourth larval to adult stage. Parasitology 2001, 123, 285–291. [Google Scholar]
- Gillespie, K.M.; Chae, J.M.; Ainsworth, E.A. Rapid measurement of total antioxidant capacity in plants. Nat. Protoc. 2007, 2, 867–870. [Google Scholar]



© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lin, R.-J.; Wu, M.-H.; Ma, Y.-H.; Chung, L.-Y.; Chen, C.-Y.; Yen, C.-M. Anthelmintic Activities of Aporphine from Nelumbo nucifera Gaertn. cv. Rosa-plena against Hymenolepis nana. Int. J. Mol. Sci. 2014, 15, 3624-3639. https://doi.org/10.3390/ijms15033624
Lin R-J, Wu M-H, Ma Y-H, Chung L-Y, Chen C-Y, Yen C-M. Anthelmintic Activities of Aporphine from Nelumbo nucifera Gaertn. cv. Rosa-plena against Hymenolepis nana. International Journal of Molecular Sciences. 2014; 15(3):3624-3639. https://doi.org/10.3390/ijms15033624
Chicago/Turabian StyleLin, Rong-Jyh, Mei-Hsuan Wu, Yi-Hsuan Ma, Li-Yu Chung, Chung-Yi Chen, and Chuan-Min Yen. 2014. "Anthelmintic Activities of Aporphine from Nelumbo nucifera Gaertn. cv. Rosa-plena against Hymenolepis nana" International Journal of Molecular Sciences 15, no. 3: 3624-3639. https://doi.org/10.3390/ijms15033624





