Docetaxel-Loaded Chitosan Microspheres as a Lung Targeted Drug Delivery System: In Vitro and in Vivo Evaluation
Abstract
:1. Introduction
2. Results and Discussion
2.1. Preparation and Microsphere Characterization
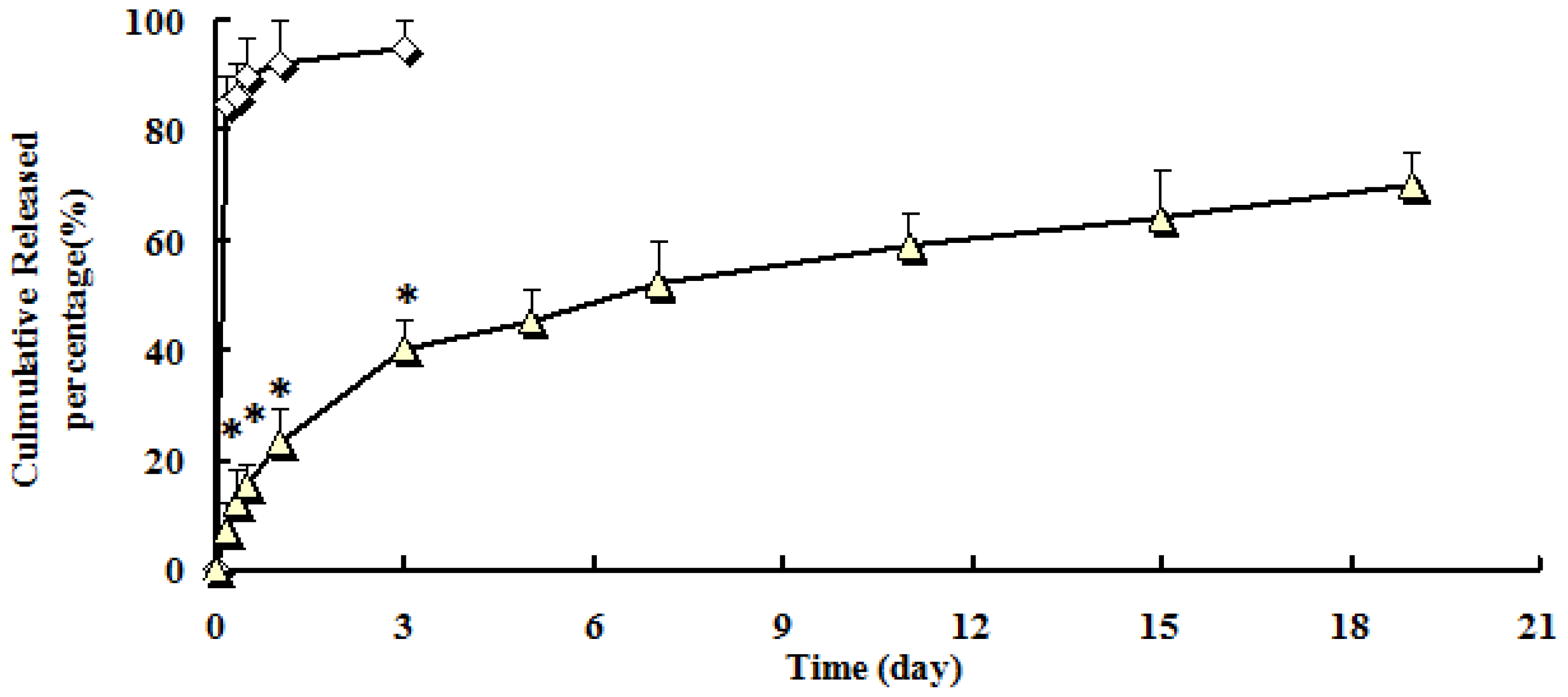
2.2. In Vitro Drug Release Behavior
2.3. Pharmacokinetic Studies
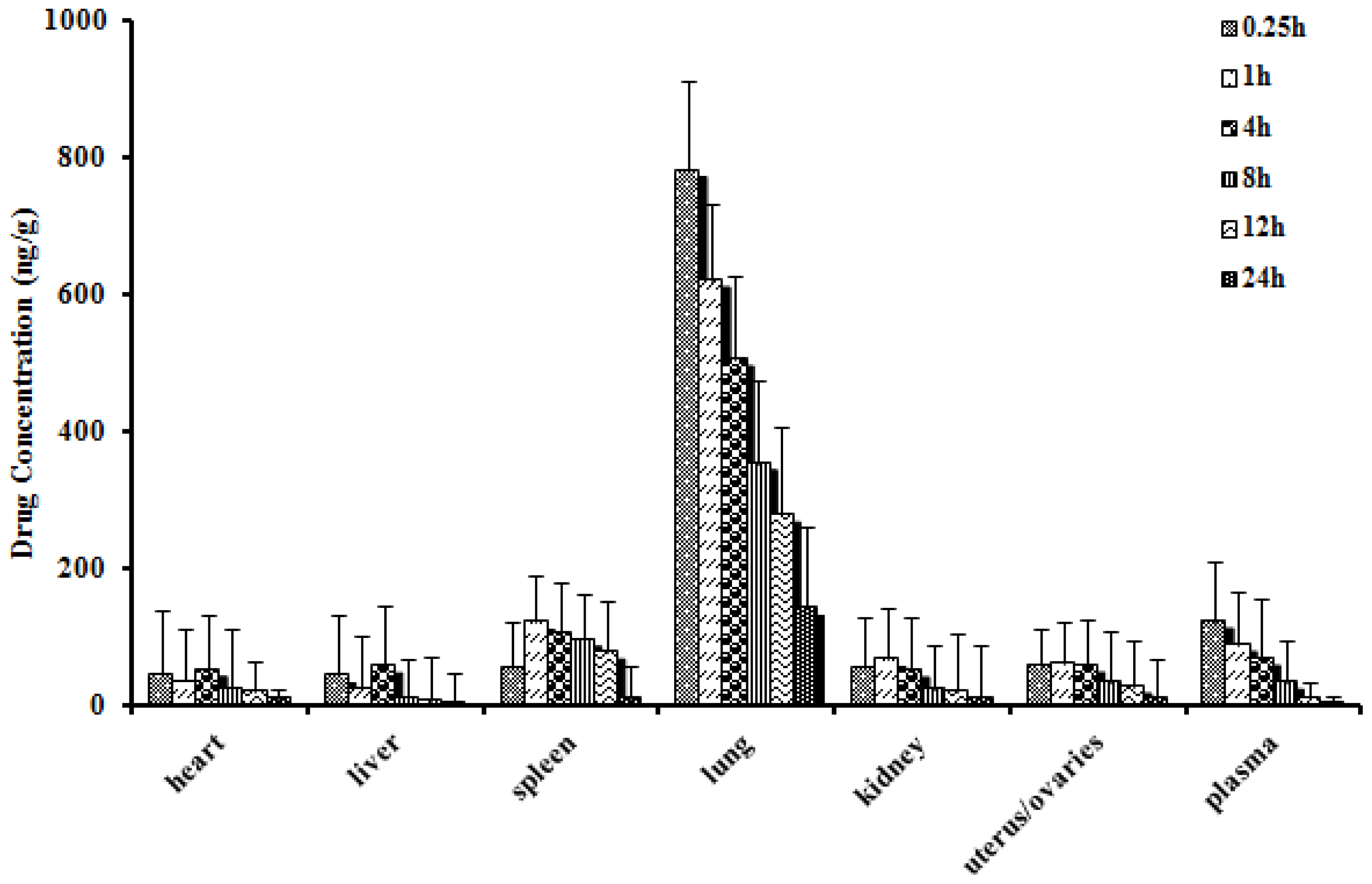
2.4. Biodistribution Studies
3. Materials and Methods
3.1. Materials
3.2. Preparation of the Microspheres
3.3. Morphological Characterization and Particle Sizing
3.4. Encapsulation Efficiency
3.5. Characterization of Microspheres
3.6. In Vitro Drug Release Studies
3.7. Pharmacokinetics Evaluation
3.8. Biodistribution Studies
3.9. LC-MS/MS Analysis
3.10. Statistical Analysis
4. Conclusions
Conflicts of Interest
References
- Herbst, R.S.; Khuri, R. Mode of action of docetaxel a basis for combination with novel anticancer agents. Cancer Treat. Rev. 2003, 29, 407–415. [Google Scholar]
- Gelmon, K. The taxoids: Paclitaxel and docetaxel. Lancet 1994, 344, 1267–1272. [Google Scholar]
- Yared, J.A.; Tkaczuk, K.H. Update on taxane development: New analogs and new formulations. Drug Des. Dev. Ther. 2012, 6, 371–384. [Google Scholar]
- Horwitz, S.B. Mechanismof action of taxol. Trends Pharmacol. Sci. 1992, 13, 134–136. [Google Scholar]
- Lavelle, F.; Bissery, M.C.; Combeau, C.; Riou, J.F.; Vrignaud, P.; Andre, S. Preclinical evaluation of docetaxel (Taxotere). Semin. Oncol. 1995, 22, 3–16. [Google Scholar]
- Chu, C.Y.; Yang, C.H.; Yang, C.Y.; Hsiao, G.H.; Chiu, H.C. Fixed erythrodysaesthesia plaque due to intravenous injection of docetaxel. Br. J. Dermatol. 2000, 142, 808–811. [Google Scholar]
- Gelderblom, H.; Verweij, J.; Nooter, K.; Sparreboom, A. Cremophor EL: The drawbacks and advantages of vehicle selection for drug formulation. Eur. J. Cancer 2001, 37, 1590–1598. [Google Scholar]
- Immordino, M.L.; Brusa, P.; Arpicco, S.; Stella, B.; Dosio, F.; Cattel, L. Preparation characterization cytotoxicity and pharmacokinetics of liposomes containing docetaxel. J. Control. Release 2003, 91, 417–429. [Google Scholar]
- Alexopoulos, A.; Karamouzis, M.V.; Stavrinides, H.; Ardavanis, A.; Kandilis, K.; Stavrakakis, J.; Georganta, C.; Rigatos, G. Phase II study of pegylated liposomal doxorubicin (caelyx) and docetaxel as first-line treatment in metastatic breast cancer. Ann. Oncol. 2004, 15, 891–895. [Google Scholar]
- Musumeci, T.; Ventura, C.A.; Giannone, I.; Ruozi, B.; Montenegro, L.; Pignatello, R.; Puglisi, G. PLA/PLGA nanoparticles for sustained release of docetaxel. Int. J. Pharm. 2006, 325, 172–179. [Google Scholar]
- Yang, Y.; Wang, J.; Zhang, X.; Lu, W.; Zhang, Q. A novel mixed micelle gel with thermo-sensitive property for the local delivery of docetaxel. J. Control. Release 2009, 135, 175–182. [Google Scholar]
- Edlund, U.; Albertsson, A.C. Degradable polymer microspheres for controlled drug delivery. Adv. Polym. Sci. 2002, 157, 67–112. [Google Scholar]
- Zolnik, B.S.; Burgess, D.J. Evaluation of in vivo-in vitro release of dexamethasone from PLGA microspheres. J. Control. Release 2008, 127, 137–145. [Google Scholar]
- Morita, T.; Sakamura, Y.; Horikiri, Y.; Suzuki, T.; Yoshino, H. Evaluation of in vivo release characteristics of proteined-loaded biodegradable microspheres in rats and severe combined immunodeficiency disease mice. J. Control. Release 2001, 73, 213–221. [Google Scholar]
- Artursson, P.; Lindmark, T.; Davis, S.S.; Illum, L. Effect of chitosan on the permeability of monolayers of intestinal epithelial cells (Caco-2). Pharm. Res. 1994, 11, 1358–1361. [Google Scholar]
- Islam, M.A.; Firdous, J.; Choi, Y.J.; Yun, C.H.; Cho, C.S. Design and application of chitosan microspheres as oral and nasal vaccine carriers: An updated review. Int. J. Nanomed. 2012, 7, 6077–6093. [Google Scholar]
- Jameela, S.R.; Kumary, T.V.; Lal, A.V.; Jayakrishnan, A. Progesterone-loaded chitosan microspheres: A long acting biodegradable controlled delivery system. J. Control. Release 1998, 52, 17–24. [Google Scholar]
- Jiang, H.L.; Park, I.K.; Shin, N.R.; Kang, S.G.; Yoo, H.S.; Kim, S.I.; Suh, S.B.; Akaike, T.; Cho, C.S. In vitro study of the immune stimulating activity of an atrophic [correction of athrophic] rhinitis vaccine associated to chitosan microspheres. Eur. J. Pharm. Biopharm. 2004, 58, 471–476. [Google Scholar]
- Orienti, I.; Aiedeh, K.; Gianasi, E. Chitosan-indomethacin conjugates: Effect of different substituents on the polysaccharide molecule on drug release. Arch. Pharm. Pharm. Med. Chem 1996, 329, 245–250. [Google Scholar]
- Mitra, A.; Dey, B. Chitosan microspheres in novel drug delivery systems. Indian J. Pharm. Sci. 2011, 73, 355–366. [Google Scholar]
- Bogataj, M.; Mrhar, A.; Grabnar, I.; Rajtman, Z.; Bukovec, P.; Srcic, S.; Urleb, U. The influence of magnesium stearate on the characteristics of mucoadhesive microspheres. J. Microencapsul. 2000, 17, 499–508. [Google Scholar]
- He, P.; Davis, S.S.; Illum, L. Sustained release chitosan microspheres prepared by novel spray drying methods. J. Microencapsul. 1999, 16, 343–355. [Google Scholar]
- Li, X.; Wei, K.; Luo, Y.; Gao, X.; Zhao, N.; Guo, W.S. Preparation and in vitro release of Docetaxel loaded PLGA/nHA microspheres. China J. Modern Med. 2009, 19, 2946–2950. [Google Scholar]
- Higuchi, W.I. The analysis of data on the medicament release from ointments. J. Pharm. Sci. 1962, 51, 802–804. [Google Scholar]
- Casettari, L.; Castagnino, E.; Stolnik, S.; Lewis, A.; Howdle, S.M.; Illum, L. Surface characterisation of bioadhesive PLGA/chitosan microparticles produced by supercritical fluid technology. Pharm. Res 2011, 28, 1668–1682. [Google Scholar]
- Pavanetto, F.; Perugini, P.; Conti, B.; Modena, T.; Genta, I. Evaluation of process parameters involved in chitosan microsphere preparation by the O/W/O multiple emulsion method. J. Microencapsul 1996, 13, 679–688. [Google Scholar]
- Thanoo, B.C.; Sunny, M.C.; Jayakrishnan, A. Cross-linked chitosan microspheres: Preparation and evaluation as a matrix for the controlled release of pharmaceuticals. J. Pharm. Pharmacol 1992, 44, 283–286. [Google Scholar]
- Kim, S.Y.; Lee, Y.M. Taxol-loaded block copolymer nanospheres composed of methoxy poly(ethylene glycol) and poly(epsilon-caprolactone) as novel anticancer drug carriers. Biomaterials 2001, 22, 1697–1704. [Google Scholar]
- Shi, B.; Fang, C.; You, M.X. Stealth MePEG-PCL micelles: Effects of polymer composition on micelle physicochemical characteristics in vitro drug release in vivo pharmacokinetics in rats and biodistribution in S180 tumor bearing mice. Colloid Polym. Sci 2005, 283, 954–967. [Google Scholar]



| Time (month) | 4 °C | 25 °C | ||
|---|---|---|---|---|
| Drug remaining (%) | Surface morphology | Drug remaining (%) | Surface morphology | |
| Initial | 99.6 | spherical | 101.3 | spherical |
| 1 | 100.1 | spherical | 101.7 | spherical |
| 2 | 101.2 | spherical | 99.7 | spherical |
| 3 | 99.2 | spherical | 98.9 | spherical |
| Parameter | Formulations | |
|---|---|---|
| Injection | Microspheres | |
| t1/2α (h) | 0.22 ± 0.1 | 0.86 ± 0.3 * |
| t1/2β (h) | 4.9 ± 1.3 | 7.9 ± 3.4 * |
| Cmax (ng/mL) | 1425.6 ± 154.6 | 1239.5 ± 136.3 |
| AUC0–t (ng·h/mL) | 2234.6 ± 234.1 | 2555.6 ± 309.2 |
| AUC0–∞ (ng·h/mL) | 2635.8 ± 253.2 | 2875.3 ± 339.6 |
| MRT (h) | 12.8 ± 4.6 | 21.6 ± 3.9 * |
| CL (L/h) | 25.8 ± 6.5 | 16.7 ± 4.2 * |
| Formulation | Heart | Liver | Spleen | Lung | Kidney | Uterus/ovaries | Plasma |
|---|---|---|---|---|---|---|---|
| DTX injection (ng·h/g) | 1332.8 | 2671.6 | 1979.4 | 1772.9 | 6754.8 | 3139.8 | 1494.4 |
| DTX-loaded microspheres (ng·h/g) | 613.1 | 376.3 | 1699.4 | 7720.6 | 651.9 | 776.1 | 698.5 |
| Ratio a | 0.46 | 0.14 * | 0.86 | 4.35 * | 0.10 * | 0.25 * | 0.47 |
| Parameter | Value |
|---|---|
| Scan type | MRM (multiple reaction monitoring) |
| Ion polarity | Positive |
| Fragmentor voltage, V | 115 |
| Nebulizer pressure, psi | 35 |
| Drying gas temperature, °C | 340 |
| Dry gas flow, L/min | 32 |
| Dwell time per transition, ms | 220 |
| Resolution | Unit |
| Collision energy, eV | 30 |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, H.; Xu, Y.; Zhou, X. Docetaxel-Loaded Chitosan Microspheres as a Lung Targeted Drug Delivery System: In Vitro and in Vivo Evaluation. Int. J. Mol. Sci. 2014, 15, 3519-3532. https://doi.org/10.3390/ijms15033519
Wang H, Xu Y, Zhou X. Docetaxel-Loaded Chitosan Microspheres as a Lung Targeted Drug Delivery System: In Vitro and in Vivo Evaluation. International Journal of Molecular Sciences. 2014; 15(3):3519-3532. https://doi.org/10.3390/ijms15033519
Chicago/Turabian StyleWang, Hao, Yongdong Xu, and Xiao Zhou. 2014. "Docetaxel-Loaded Chitosan Microspheres as a Lung Targeted Drug Delivery System: In Vitro and in Vivo Evaluation" International Journal of Molecular Sciences 15, no. 3: 3519-3532. https://doi.org/10.3390/ijms15033519




