Protein A Detection Based on Quantum Dots-Antibody Bioprobe Using Fluorescence Coupled Capillary Electrophoresis
Abstract
:1. Introduction
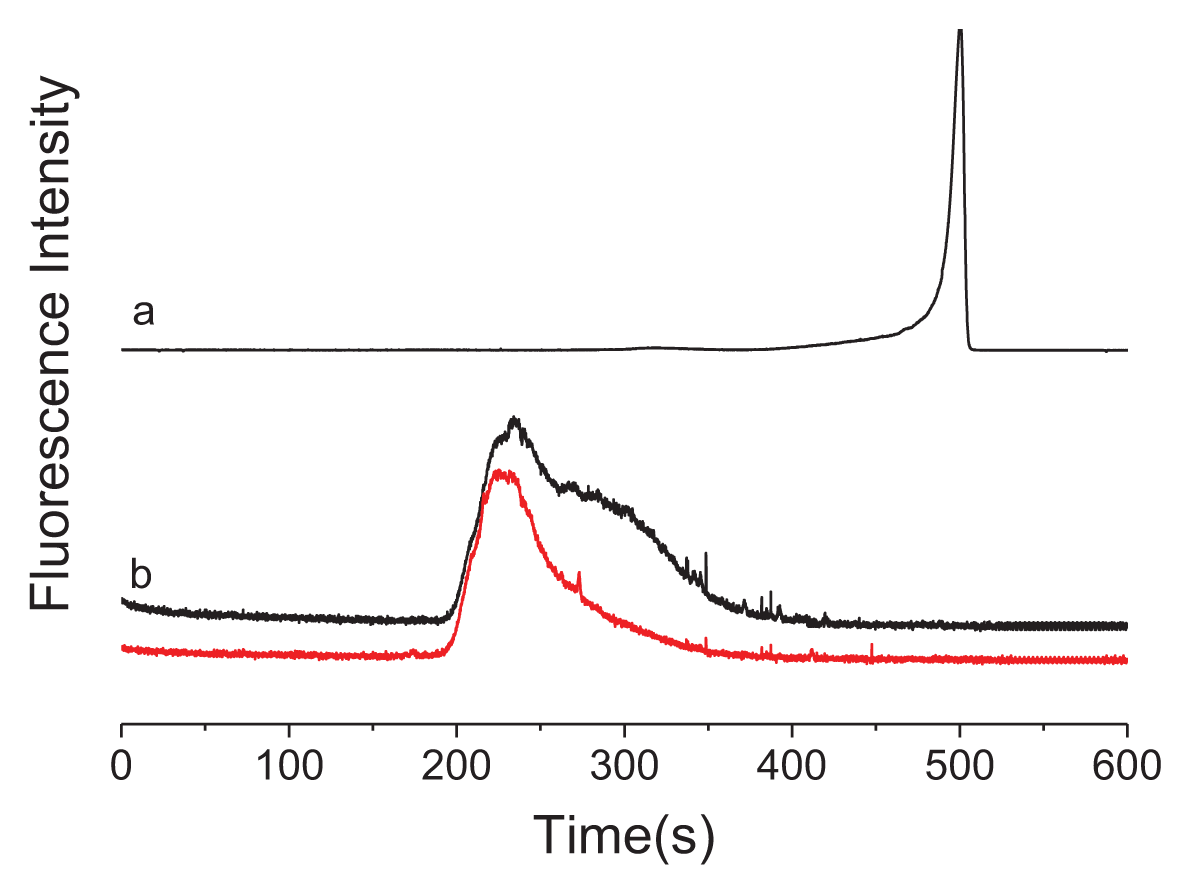
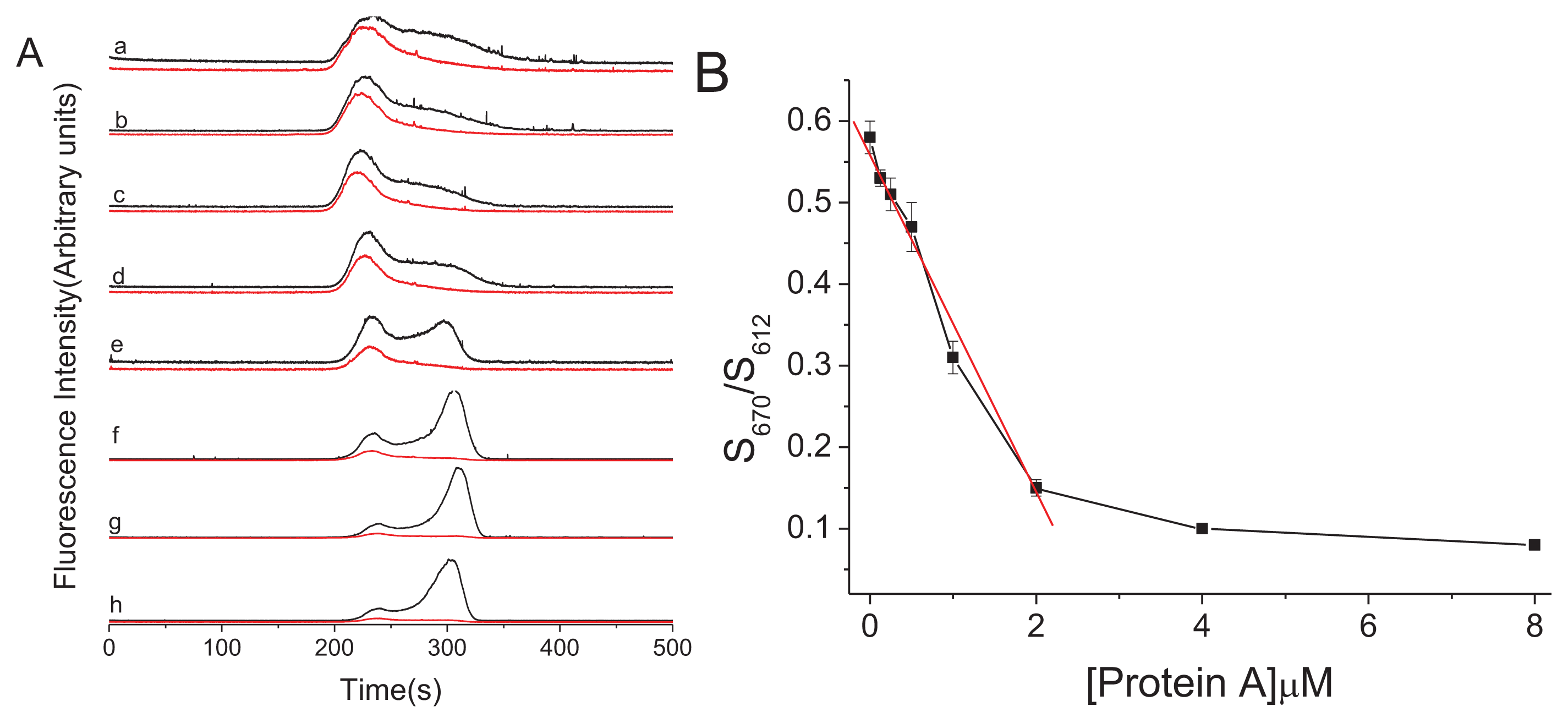
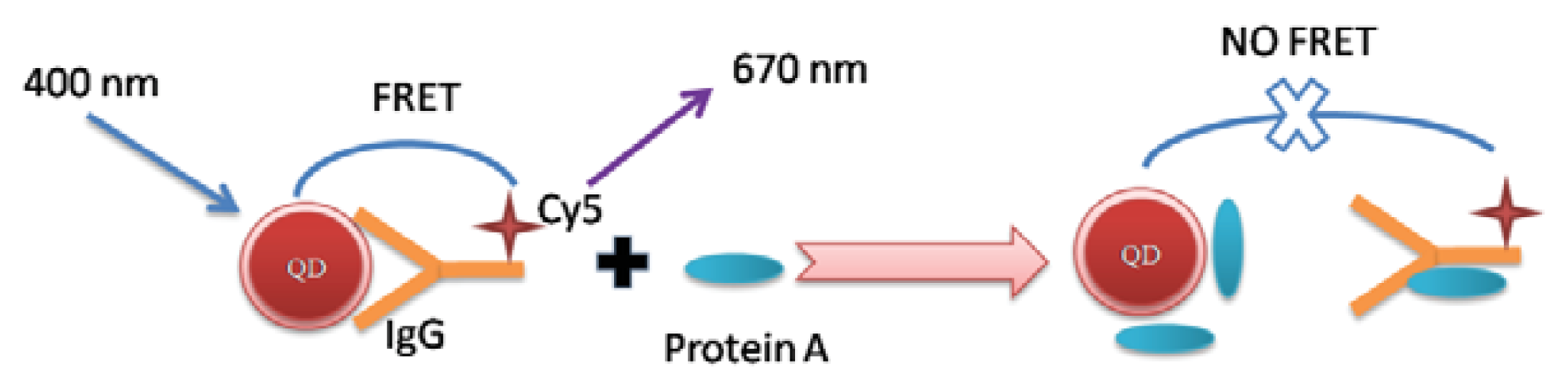
2. Results and Discussion
3. Experimental Section
3.1. Materials and Instruments
3.2. Preparation of GSH Stabilized QDs
3.3. Preparation of Cy5-NHS
3.4. Preparation of IgG-Cy5
3.5. Preparation of QDs-IgG-Cy5 Conjugates
3.6. Procedure of Capillary Electrophoresis
4. Conclusions
Acknowledgments
Conflicts of Interest
- Author ContributionsJ.W. and P.J. designed the research; Y.B., J.L., P.G. and J.L. performed the research; C.W. and W.H. analyzed data; and J.W. and P.J. wrote the paper.
References
- Bruchez, M.; Moronne, M.; Gin, P.; Weiss, S.; Alivisatos, A.P. Semiconductor nanocrystals as fluorescent biological labels. Science 1998, 281, 2013–2016. [Google Scholar]
- Medintz, I.L.; Uyeda, H.T.; Goldman, E.R.; Mattoussi, H. Quantum dot bioconjugates for imaging, labeling and sensing. Nat. Mater 2005, 4, 435–446. [Google Scholar]
- Bailey, R.E.; Smith, A.M.; Nie, S. Quantum dots in biology and medicine. Physica 2004, 25, 1–12. [Google Scholar]
- Michalet, X.; Pinaud, F.F.; Bentolila, L.A.; Tsay, J.M.; Doose, S.; Li, J.J.; Sundaresan, G.; Wu, A.M.; Gambhir, S.S.; Weiss, S. Quantum dots for live cells, in vivo imaging, and diagnostics. Science 2005, 307, 538–544. [Google Scholar]
- Wang, S.P.; Mamedova, N.; Kotov, N.A.; Chen, W.; Studer, J. Antigen/antibody immunocomplex from CdTe nanoparticle bioconjugates. Nano Lett 2002, 2, 817–822. [Google Scholar]
- Sapsford, K.E.; Pons, T.; Medintz, I.L.; Higashiya, S.; Brunel, F.M.; Dawson, P.E.; Mattoussi, H. Kinetics of metal-affinity driven self-assembly between proteins or peptides and CdSe–ZnS quantum dots. J. Phys. Chem 2007, 111, 11528–11538. [Google Scholar]
- Dennis, A.M.; Bao, G. Quantum dot fluorescent protein pairs as novel fluorescence resonance energy transfer probes. Nano Lett 2008, 8, 1439–1445. [Google Scholar]
- Goldman, E.R.; Balighian, E.D.; Kuno, M.K.; Labrenz, S.; Tran, P.T.; Anderson, G.P.; Mauro, J.M.; Mattoussi, H. Luminescent quantum dot-adaptor protein–antibody conjugates for use in fluoroimmunoassays. Phys. Status Solidi 2002, 229, 407–414. [Google Scholar]
- Mattoussi, H.; Mauro, J.M.; Goldman, E.R.; Green, T.M.; Anderson, G.P.; Sundar, V.C.; Bawendi, M.G. Bioconjugation of highly luminescent colloidal CdSe–ZnS quantum dots with an engineered two-domain recombinant protein. Phys. Status Solidi 2001, 224, 277–283. [Google Scholar]
- Li, Y.; Wang, J.; Zhang, H.; Yang, J.; Guan, L.; Chen, H.; Luo, Q.; Zhao, Y. High-sensitivity quantum dot-based fluorescence resonance energy transfer bioanalysis by capillary electrophoresis. Biosens. Bioelectron 2010, 25, 1283–1289. [Google Scholar]
- Tang, B.; Cao, L.H.; Xu, K.H.; Zhuo, L.H.; Ge, J.H.; Li, Q.F.; Yu, L.J. A new nanobiosensor for glucose with high sensitivity and selectivity in serum based on fluorescence resonance energy transfer (FRET) between CdTe quantum dots and Au nanoparticles. Chem. Eur. J 2008, 14, 3637–3644. [Google Scholar]
- Kim, Y.P.; Park, S.; Oh, E.; Oh, Y.H.; Kim, H.S. On-chip detection of protein glycosylation based on energy transfer between nanoparticles. Biosens. Bioelectron 2009, 24, 1189–1194. [Google Scholar]
- Willard, D.M.; Carillo, L.L.; Jung, J.; Orden, A.V. CdSe–ZnS quantum dots as resonance energy transfer donors in a model protein-protein binding assay. Nano Lett 2001, 1, 469–474. [Google Scholar]
- Zhang, C.Y.; Johnson, L.W. Quantum-dot-based nanosensor for RRE IIB RNA-Rev peptide interaction assay. J. Am. Chem. Soc 2006, 128, 5324–5325. [Google Scholar]
- Wang, J.; Jiang, P.; Gao, L.; Yu, Y.; Lu, Y.; Qiu, L.; Wang, C.; Xia, J. Unique self-assembly properties of a bridge-shaped protein dimer with quantum dots. J. Nanopart. Res 15.
- Wang, J.; Jin, X.; Liu, J.; Khosla, C.; Xia, J. Resolving multiple protein-peptide binding events: Implication for HLA-DQ2 mediated antigen presentation in celiac disease. Chem. Asian J 2012, 7, 992–999. [Google Scholar]
- Wang, J.; Xia, J. Capillary electrophoretic studies on displacement and proteolytic cleavage of surface bound oligohistidine peptide on quantum dots. Anal. Chim. Acta 2012, 709, 120–127. [Google Scholar]
- Chang, Y.; Cai, C.; Li, L.; Miao, J.; Ucakturk, E.; Li, G.; Ly, M.; Linhardt, R.J. Ultrasensitive detection and quantification of acidic disaccharides using capillary electrophoresis and quantum dot-based fluorescence resonance energy transfer. Anal. Chem 2013, 85, 9356–9362. [Google Scholar]
- Pholphana, N.; Rangkadilok, N.; Thongnest, S.; Ruchirawat, S.; Ruchirawat, M.; Satayavivad, J. Determination and variation of three active diterpenoids in Andrographis paniculata (Burm. f.) Nees. Anal. Biochem 2004, 333, 365–371. [Google Scholar]
- Hardy, F.; Djavadi-Ohaniance, L.; Goldberg, M.E. Measurement of antibody/antigen association rate constants in solution by a method based on the enzyme-linked immunosorbent assay. J. Immunol. Methods 1997, 200, 155–159. [Google Scholar]
- Pierce, M.M.; Raman, C.S.; Nall, B.T. Isothermal titration calorimetry of protein-protein interactions. Methods 1999, 19, 213–221. [Google Scholar]
- Zhao, J.; Gao, T.; Yan, Y.; Chen, G.; Li, G. Probing into the interaction of β-amyloid peptides with bilayer lipid membrane by electrochemical techniques. Electrochem. Commun 2013, 30, 26–28. [Google Scholar]
- Wang, J.; Xia, J. Preferential binding of a novel polyhistidine peptide dendrimer ligand on quantum dots probed by capillary electrophoresis. Anal. Chem 2011, 83, 6323–6329. [Google Scholar]
- Krizkova, S.; Ryvolova, M.; Hynek, D.; Eckschlager, T.; Hodek, P.; Masarik, M.; Adam, V.; Kizek, R. Immunoextraction of zinc proteins from human plasma using chicken yolk antibodies immobilized onto paramagnetic particles and their electrophoretic analysis. Electrophoresis 2012, 33, 1824–1832. [Google Scholar]
- Freeman, R.; Finder, T.; Gill, R.; Willner, I. Probing protein kinase (CK2) and alkaline phosphatase with CdSe/ZnS quantum dots. Nano Lett 2010, 10, 2192–2196. [Google Scholar]
- Yu, W.W.; Qu, L.; Guo, W.; Peng, X. Experimental determination of the extinction coefficient of CdTe, CdSe, and CdS nanocrystals. Chem. Mater 2003, 15, 2854–2860. [Google Scholar]
- Mujumdar, R.B.; Ernst, L.A.; Mujumdar, S.R.; Lewis, C.J.; Waggoner, A.S. Cyanine dye labeling reagents: Sulfoindocyanine succinimidyl esters. Bioconjug. Chem 1993, 4, 105–111. [Google Scholar]

© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Qiu, L.; Bi, Y.; Wang, C.; Li, J.; Guo, P.; Li, J.; He, W.; Wang, J.; Jiang, P. Protein A Detection Based on Quantum Dots-Antibody Bioprobe Using Fluorescence Coupled Capillary Electrophoresis. Int. J. Mol. Sci. 2014, 15, 1804-1811. https://doi.org/10.3390/ijms15021804
Qiu L, Bi Y, Wang C, Li J, Guo P, Li J, He W, Wang J, Jiang P. Protein A Detection Based on Quantum Dots-Antibody Bioprobe Using Fluorescence Coupled Capillary Electrophoresis. International Journal of Molecular Sciences. 2014; 15(2):1804-1811. https://doi.org/10.3390/ijms15021804
Chicago/Turabian StyleQiu, Lin, Yanhua Bi, Cheli Wang, Jingyan Li, Peilin Guo, Jinchen Li, Weijiang He, Jianhao Wang, and Pengju Jiang. 2014. "Protein A Detection Based on Quantum Dots-Antibody Bioprobe Using Fluorescence Coupled Capillary Electrophoresis" International Journal of Molecular Sciences 15, no. 2: 1804-1811. https://doi.org/10.3390/ijms15021804
APA StyleQiu, L., Bi, Y., Wang, C., Li, J., Guo, P., Li, J., He, W., Wang, J., & Jiang, P. (2014). Protein A Detection Based on Quantum Dots-Antibody Bioprobe Using Fluorescence Coupled Capillary Electrophoresis. International Journal of Molecular Sciences, 15(2), 1804-1811. https://doi.org/10.3390/ijms15021804




