Gender-Associated Genomic Differences in Colorectal Cancer: Clinical Insight from Feminization of Male Cancer Cells
Abstract
:1. Introduction
2. Results
2.1. Demographic/Pathologic Data
| Characteristics | Males No. (%) | Females No. (%) | Total No. (%) |
|---|---|---|---|
| No. Of Patients | 57 (49.1) | 59 (50.9) | 116 (100) |
| Mean Age in Years | 63 | 65.4 | – |
| Age | |||
| 31–40 years | 1 (1.8) | 4 (6.8) | 5 (4.3) |
| 41–50 years | 8 (14.0) | 8 (13.6) | 16 (13.8) |
| 51–60 years | 15 (26.3) | 11 (18.6) | 26 (22.4) |
| 61–70 years | 14 (24.5) | 9 (15.2) | 23 (19.8) |
| 71–80 years | 18 (31.6) | 17 (28.8) | 35 (30.2) |
| >81 years | 1 (1.8) | 10 (17.0) | 11 (9.5) |
| Tumor Site | |||
| Right side of colon | 16 (28.1) | 12 (20.3) | 28 (24.2) |
| Left side of colon | 18 (31.6) | 29 (49.2) * | 47 (40.5) |
| Rectum | 11 (19.3) | 12 (20.3) | 23 (19.8) |
| Unspecified | 12 (21.0) | 6 (10.2) | 18 (15.5) |
| Histological Grade | |||
| Well-differentiated | 4 (7.0) | 9 (15.2) | 13 (11.2) |
| Moderately-differentiated | 44 (77.2) | 40 (67.8) | 84 (72.4) |
| Poorly-differentiated | 6 (10.5) | 5 (8.5) | 11 (9.5) |
| Unspecified | 3 (5.3) | 5 (8.5) | 8 (6.9) |
| Dukes’ Stage | |||
| Stage B | 48 (84.2) | 48 (81.4) | 96 (82.8) |
| Stage C | 8 (14.0) | 10 (16.9) | 18 (15.5) |
| Stage D | 1 (1.8) | 1 (1.7) | 2 (1.7) |
| Microsatellite Status—BRAF | |||
| MSS—wild type BRAF | 41 (72.0) | 46 (77.9) | 87 (75.0) |
| MSS—mutated BRAF | 2 (3.5) | 1 (1.7) | 3 (2.6) |
| MSI—wild type BRAF | 6 (10.5) | 4 (6.8) | 10 (8.6) |
| MSI—mutated BRAF | 4 (7.0) | 4 (6.8) | 8 (6.9) |
| Unspecified | 4 (7.0) | 4 (6.8) | 8 (6.9) |
| Follow-Up | |||
| Relapse, local or metastatic | 7 (12.3) | 17 (28.8) ** | 24 (20.7) |
| Relapse-free | 35 (61.4) | 28 (47.5) | 63 (54.3) |
| Unknown | 15 (26.3) | 14 (23.7) | 29 (25.0) |
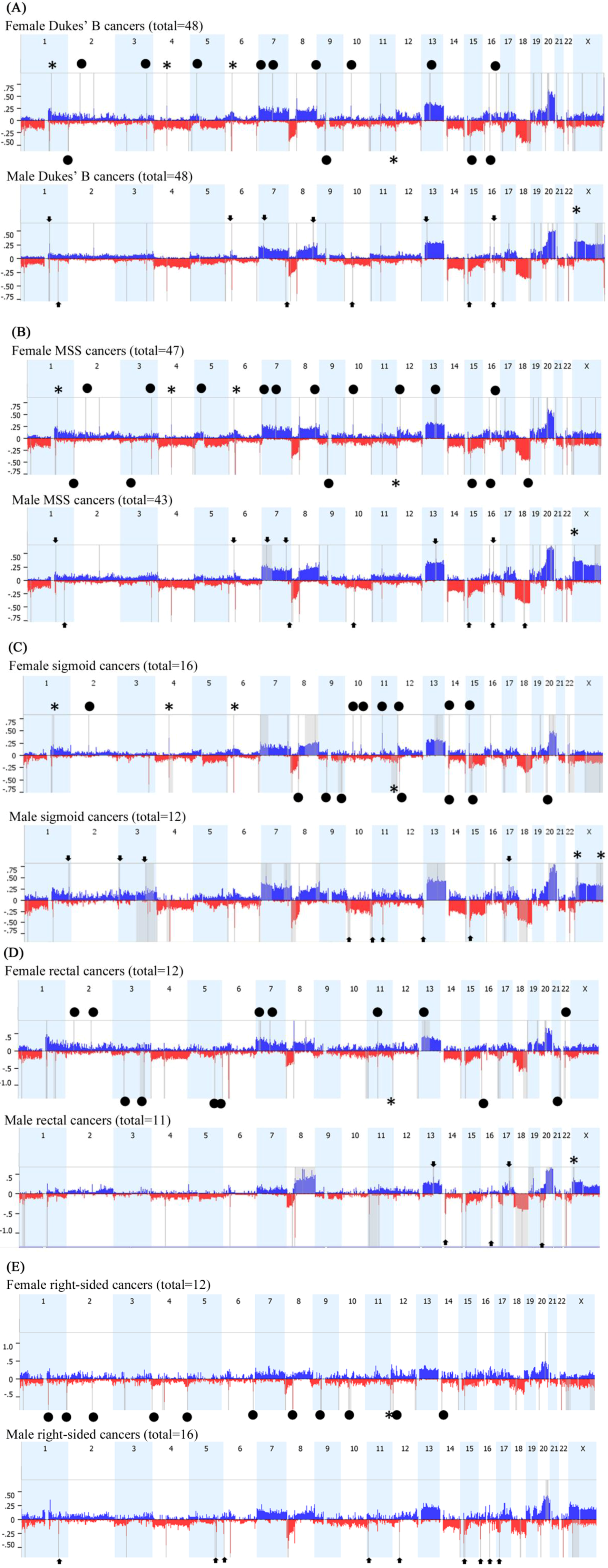
2.2. Array Comparative Genomic Hybridization (aCGH)

| Locus | CNA | Candidate Genes | Females % | Males % | p-Value |
|---|---|---|---|---|---|
| 1q21.2 | Gain | LINC00568, AF289612, AK023606, MIR4257, TSRC1, ADAMTSL4 | 50.8 | 17.5 | <0.0001 |
| 1q21.2–1q21.3 | Gain | U78576, PIP5K1A, BC007833, PSMD4, ZNF687, AK023105, PIK4CB, PI4KB, AJ011123, BC040300, RFX5, AK023875, SELENBP1, PSMB4, BX537561, POGZ, CGN, MIR554, TUFT1, AY358610 | 46.7 | 12.3 | <0.0001 |
| 4q13.2 | Gain | AF064819, UGT2B17 | 33.9 | 1.75 | <0.0001 |
| 6p21.31 | Gain | FKBP5, MIR5690 | 37.3 | 5.3 | <0.0001 |
| 6p21.1 | Gain | TBCC, AK096036, KIAA0240, GLTSCR1L, RPL7L1, C6orf226, LOC441150, PTCRA, CNPY3, TNRC5 | 35.6 | 3.5 | <0.0001 |
| 11q25 | Loss | FLJ45436, AY358331, NTM, BC050716, HNT | 47.45 | 14 | 0.0001 |
| 16p11.2 | Gain | QPRT, C16orf54, BC029149, ZG16, KIF22, BC004352, CR590954, AF489858, MAZ, L01420, BC041629, AK074572, PRRT2, LOC112476, AK092265, C16orf53, PAGR1, MVP, AK131349, CDIPT, LOC440356, BC000567, SEZ6L2, AJ245822, ASPHD1, LOC253982, KCTD13, TMEM219, LOC124446, TAOK2 | 54.2 | 20.8 | 0.0003 |
2.3. Feminization of Colorectal Cancer Cells in Males


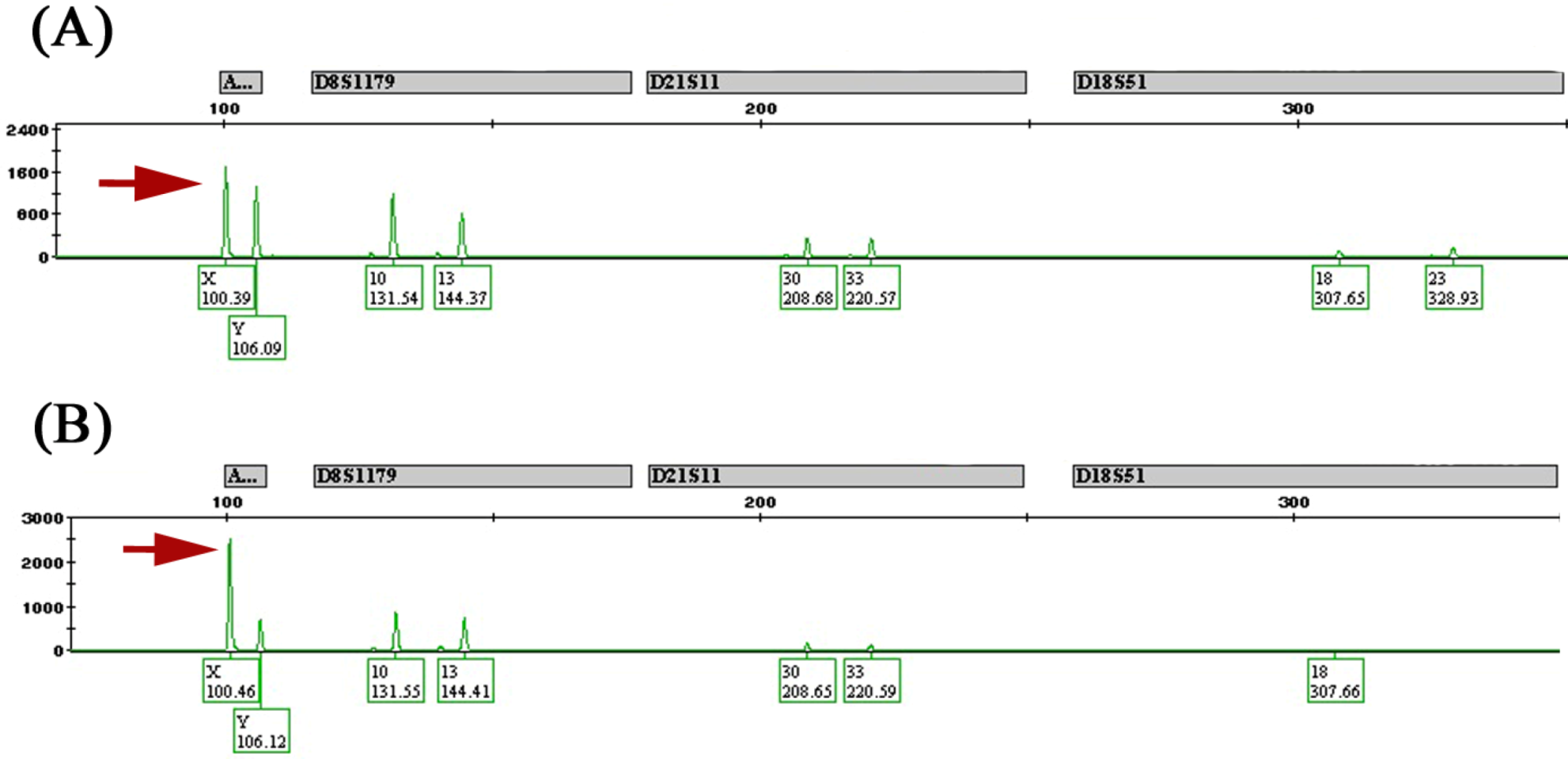
| Locus | CNA | Candidate Genes | Females % | Males % | p-Value |
|---|---|---|---|---|---|
| Xp22.32–p22.31 | Gain | NLGN4X, AK125309, MIR4770, VCX3A, HDHD1A, HDHD1, MIR4767, STS, VCX, PNPLA4, MIR651 | 9.3 | 42.3 | <0.01 |
| Xp22.31–p22.2 | Gain | VCX-C, VCX3B, KAL1, FAM9A, AK097501, FAM9B, AY459291, TBL1X, GPR143, X83543, SHROOM2, APXL, CR749271, LOC100288814, KIAA1280, WWC3, BC035601, CLCN4, MID1, HCCS, AMELX, ARHGAP6, MSL3L1, MSL3, PDZK10, FRMPD4, PRPS2, TLR7, TLR8, AY358296, TLR8-AS1, TMSB4X, FAM9C, LOC100093698, ATXN3L, LOC100133123 | 10.7 | 45.8 | <0.01 |
| Xp22.12–p22.11 | Gain | SH3KBP1, AL833278, CXorf23, AK094661, LOC729609, MIR23C, AK098768, MAP7D2, BC089400, FLJ14503, EIF1AX, SCARNA9L, RPS6KA3, CNKSR2, RP11-450P7.3, KLHL34, SMPX, BC036465, YY2, MBTPS2, SMS, PHEX, PHEX-AS1, ZNF645, LOC100873065, DDX53 | 12.5 | 46.2 | <0.01 |
| Xp22.11–p21.2 | Gain | PDK3, BC045634, PCYT1B, SCARNA23, POLA, POLA1, ARX, MAGEB18, MGC33889, MAGEB6, MAGEB5, VENTXP1, SMEK3P, AK131475, AK057304, DCAF8L2, MAGEB10, DCAF8L1, IL1RAPL1, MAGEB2, MAGEB3, MAGEB4, MAGEB1, NR0B1 | 9.4 | 44.8 | <0.01 |
| Xp21.1–p11.4 | Gain | DMD, MIR3915, BC036103, FAM47A, TMEM47, FAM47B, MAGEB16, CXorf22, RP13-11B7.1, CHDC2, CXorf30, FAM47C, LOC442444, AK125992, FTH1P18, PRRG1, AK130368, LANCL3, XK, CYBB, TCTE1L, DYNLT3, CXorf27, SYTL5, SRPX, RPGR, OTC, TSPAN7, TM4SF2, MID1IP1, LOC286442, AY316592, BCOR, ATP6AP2, BC010395, MPC1L, CXorf38, BC025334, AL832829, CRSP2, MED14 | 11.4 | 45.1 | <0.01 |
| Xq21.1–q21.31 | Gain | APOOL, SATL1, ZNF711, ZNF6, BC067294, AK128541, AK026445, AK025039, POF1B, AF309774, BC017500, FLJ38564, CHM, BC032237, DACH2, AK022715, KLHL4, AB051474, LAMR1P15, CPXCR1 | 9.1 | 42.6 | <0.01 |
| Xq22.3 | Gain | MID2, TEX13B, VSIG1, PSMD10, ATG4A, APG4A, AK054927, COL4A6 | 11.9 | 42.6 | 0.001 |
| Xq13.1 | Gain | EDA, MIR676, DGAT2L4, AWAT2, OTUD6A, HSHIN6, IGBP1 | 4.2 | 35.1 | 0.001 |
| Xq22.1–q22.2 | Gain | BEX4, BEXL1, TCEAL8, TCEAL5, BEX2, TCEAL7, WBP5 | 13.5 | 43.8 | 0.002 |
| Xp11.22 | Gain | LOC401589, SNORA11D, SNORA11E, AF329733, MAGED4, MAGED4B, XAGE2B, XAGE1A, XAGE1C, XAGE1D, XAGE1E, XAGE1, BC009538, XAGE1A, XAGE1B, XAGE2, XAGE1D, XAGE1B, SSX8, SSX7, SSX2, SSX2B, SPANXN5, SPANX-N5, XAGE5 | 8.5 | 39.6 | 0.004 |
| Xq21.1 | Gain | BX649166, HDX, CXorf43 | 11.2 | 41.6 | 0.008 |
| Xp22.12 | Gain | AK131412, MAP3K15 | 16.95 | 47.4 | 0.010 |
| Xq22.1 | Gain | RAB40AL, BEX1 | 10.2 | 40.4 | 0.010 |
| Xq13.1 | Gain | KIF4A, GDPD2, DLG3, AB033058 | 14.7 | 45.6 | 0.016 |
| Xq22.3 | Gain | MUM1L1, AK056478 | 8.5 | 38.6 | 0.019 |
| Xq23 | Gain | AMOT | 10.2 | 40.4 | 0.020 |
| Xq26.2 | Gain | AL832725, PHF6, HPRT1 | 17.3 | 48.2 | 0.022 |
| Xq22.3 | Gain | AK001040, CXorf57, BC070110, FLJ10178, AK024253, RNF128 | 11.9 | 42.1 | 0.023 |
| Xq23 | Gain | SLC6A14 | 12.3 | 44.3 | 0.028 |
| Xp11.23 | Gain | WDR45, GPKOW, MAGIX, FLJ21687, PLP2, PRICKLE3, LMO6, SYP, SSX1 | 18.97 | 49.7 | 0.030 |
| Xq22.2 | Gain | RAB40A | 11.4 | 42.3 | 0.030 |
| Xq27.1 | Gain | SRD5A1P1, F9 | 11.9 | 42.1 | 0.040 |
| Xq28 | Gain | AFF2, FMR2 | 11.9 | 42.1 | 0.042 |
| Yp11.31–p11.2 | Loss | SRY, RPS4Y1, ZFY, LINC00278, TGIF2LY, PCDH11Y, TTTY23, TTTY23B, TSPY1, TSPY2, TTTY1B, TTTY1, TTTY2B, TTTY2, TTTY21, TTTY7B, TTTY8B, TTTY8 | – | 67.89 | <0.01 |
| Yp11.2 | Loss | PRKY, TTTY16, TTTY12 | – | 54.6 | <0.01 |
| Yq11.1–q11.221 | Loss | GYG2P1, TTTY15, USP9Y, DDX3Y, UTY, BC071744, TMSB4Y, VCY, VCY1B, BC032567, NLGN4Y, NLGN4Y-AS1 | – | 57.4 | <0.01 |
| Yq11.222–q11.223 | Loss | TTTY9A, HSFY2, NCRNA00185, CYorf14, CD24, TTTY14, BCORL2, BCORP1, CYorf15A, TXLNG2P, BC035312, CYorf15B, D87072, KDM5D, SMCY, TTTY10, EIF1AY, RPS4Y2, U88898, RBMY2EP, AK026367, RBMY1A1, BC070298, RBMY1F, RBMY1B, TTTY13, RBMY1D, RBMY1E, RBMY1A1, TTTY6, PRY, PRY2, AY597808, TTTY6B, RBMY1F, RBMY1J, RBMY1A1, BC047768, TTTY5, RBMY2FP, TTTY6 | – | 60.01 | <0.01 |
2.4. Clinicopathological Significance
| Characteristics | Feminized Male CRC (Total = 26) No. (%) | Non-Feminized Male CRC (Total = 31) No. (%) | Total Males (Total = 57) No. (%) | p-Value |
|---|---|---|---|---|
| MSS—wild type BRAF | 24 (92.3) | 17 (56.7) | 41 (72.0) | 0.0026 * |
| Wild-type BRAF | 26 (100) | 23 (74.2) | 49 (86.0) | 0.0088 * |
| MMR proficient | 24 (92.3) | 19 (61.2) | 43 (75.4) | 0.0107 * |
3. Discussion
4. Materials and Methods
4.1. Patients and Tissue Samples
4.2. Array Comparative Genomic Hybridization (aCGH)
4.3. Fluorescence in Situ Hybridization (FISH)
4.4. Multiplex Ligation-Dependent Probe Amplification (MLPA)
4.5. Allelotyping
4.6. Clinicopathological Correlation and Statistical Analysis
5. Conclusions
Supplementary Materials
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Markowitz, S.D.; Bertagnolli, M.M. Molecular origins of cancer: Molecular basis of colorectal cancer. N. Engl. J. Med. 2009, 361, 2449–2460. [Google Scholar]
- Ried, T.; Knutzen, R.; Steinbeck, R.; Blegen, H.; Schröck, E.; Heselmeyer, K.; du Manoir, S.; Auer, G. Comparative genomic hybridization reveals a specific pattern of chromosomal gains and losses during the genesis of colorectal tumors. Genes Chromosomes Cancer 1996, 15, 234–245. [Google Scholar]
- Bomme, L.; Bardi, G.; Pandis, N.; Fenger, C.; Kronborg, O.; Heim, S. Clonal karyotypic abnormalities in colorectal adenomas: Clues to the early genetic events in the adenoma-carcinoma sequence. Genes Chromosomes Cancer 1994, 10, 190–196. [Google Scholar]
- Bardi, G.; Sukhikh, T.; Pandis, N.; Fenger, C.; Kronborg, O.; Heim, S. Karyotypic characterization of colorectal adenocarcinomas. Genes Chromosomes Cancer 1995, 12, 97–109. [Google Scholar]
- Al-Mulla, F.; Keith, W.N.; Pickford, I.R.; Going, J.J.; Birnie, G.D. Comparative genomic hybridization analysis of primary colorectal carcinomas and their synchronous metastases. Genes Chromosomes Cancer 1999, 24, 306–314. [Google Scholar]
- De Angelis, P.M.; Clausen, O.P.; Schjolberg, A.; Stokke, T. Chromosomal gains and losses in primary colorectal carcinomas detected by CGH and their associations with tumour DNA ploidy, genotypes and phenotypes. Br. J. Cancer 1999, 80, 526–535. [Google Scholar]
- He, Q.J.; Zeng, W.F.; Sham, J.S.; Xie, D.; Yang, X.W.; Lin, H.L.; Zhan, W.H.; Lin, F.; Zeng, S.D.; Nie, D.; et al. Recurrent genetic alterations in 26 colorectal carcinomas and 21 adenomas from Chinese patients. Cancer Genet. Cytogenet. 2003, 144, 112–118. [Google Scholar]
- Al-Mulla, F.; Behbehani, A.I.; Bitar, M.S.; Varadharaj, G.; Going, J.J. Genetic profiling of stage I and II colorectal cancer may predict metastatic relapse. Mod. Pathol. 2006, 19, 648–658. [Google Scholar]
- Tsafrir, D.; Bacolod, M.; Selvanayagam, Z.; Tsafrir, I.; Shia, J.; Zeng, Z.; Liu, H.; Krier, C.; Stengel, R.F.; Barany, F.; et al. Relationship of gene expression and chromosomal abnormalities in colorectal cancer. Cancer Res. 2006, 66, 2129–2137. [Google Scholar]
- Dorak, M.T.; Karpuzoglu, E. Gender differences in cancer susceptibility: An inadequately addressed issue. Front. Genet. 2012, 3, 268. [Google Scholar]
- Dutrillaux, B.; Muleris, M.; Seureau, M.G. Imbalance of sex chromosomes, with gain of early-replicating X, in human solid tumors. Int. J. Cancer 1986, 38, 475–479. [Google Scholar]
- Muleris, M.; Dutrillaux, A.M.; Salmon, R.J.; Dutrillaux, B. Sex chromosomes in a series of 79 colorectal cancers: Replication pattern, numerical, and structural changes. Genes Chromosomes Cancer 1990, 1, 221–227. [Google Scholar]
- Unotoro, J.; Kamiyama, H.; Ishido, Y.; Yaginuma, Y.; Kasamaki, S.; Sakamoto, K.; Oota, A.; Ishibashi, Y.; Kamano, T. Analysis of the relationship between sex and chromosomal aberrations in colorectal cancer by comparative genomic hybridization. J. Int. Med. Res. 2006, 34, 397–405. [Google Scholar]
- Brim, H.; Lee, E.; Abu-Asab, M.S.; Chaouchi, M.; Razjouyan, H.; Namin, H.; Goel, A.; Schäffer, A.A.; Ashktorab, H. Genomic aberrations in an African American colorectal cancer cohort reveals a MSI-specific profile and chromosome X amplification in male patients. PLoS One 2012, 7, e40392. [Google Scholar]
- Diskin, S.J.; Eck, T.; Greshock, J.; Mosse, Y.P.; Naylor, T.; Stoeckert, C.J., Jr.; Weber, B.L.; Maris, J.M.; Grant, G.R. STAC: A method for testing the significance of DNA copy number aberrations across multiple array-CGH experiments. Genome Res. 2006, 16, 1149–1158. [Google Scholar]
- Beroukhim, R.; Getz, G.; Nghiemphu, L.; Barretina, J.; Hsueh, T.; Linhart, D.; Vivanco, I.; Lee, J.C.; Huang, J.H.; Alexander, S.; et al. Assessing the significance of chromosomal aberrations in cancer: Methodology and application to glioma. Proc. Natl. Acad. Sci. USA 2007, 104, 20007–20012. [Google Scholar]
- Rim, S.H.; Seeff, L.; Ahmed, F.; King, J.B.; Coughlin, S.S. Colorectal cancer incidence in the United States, 1999–2004: An updated analysis of data from the National Program of Cancer Registries and the Surveillance, Epidemiology, and End Results Program. Cancer 2009, 115, 1967–1976. [Google Scholar]
- Purim, O.; Gordon, N.; Brenner, B. Cancer of the colon and rectum: Potential effects of sex–age interactions on incidence and outcome. Med. Sci. Monit. 2013, 19, 203–209. [Google Scholar]
- DeCosse, J.J.; Ngoi, S.S.; Jacobson, J.S.; Cennerazzo, W.J. Gender and colorectal cancer. Eur. J. Cancer Prev. 1993, 2, 105–115. [Google Scholar]
- McCashland, T.M.; Brand, R.; Lyden, E.; de Garmo, P.; Project, C.R. Gender differences in colorectal polyps and tumors. Am. J. Gastroenterol. 2001, 96, 882–886. [Google Scholar]
- Takada, H.; Ohsawa, T.; Iwamoto, S.; Yoshida, R.; Nakano, M.; Imada, S.; Yoshioka, K.; Okuno, M.; Masuya, Y.; Hasegawa, K.; et al. Changing site distribution of colorectal cancer in Japan. Dis. Colon Rectum 2002, 45, 1249–1254. [Google Scholar]
- Ward, R.; Meagher, A.; Tomlinson, I.; O’Connor, T.; Norrie, M.; Wu, R.; Hawkins, N. Microsatellite instability and the clinicopathological features of sporadic colorectal cancer. Gut 2001, 48, 821–829. [Google Scholar]
- Hawkins, N.; Norrie, M.; Cheong, K.; Mokany, E.; Ku, S.L.; Meagher, A.; O’Connor, T.; Ward, R. CpG island methylation in sporadic colorectal cancers and its relationship to microsatellite instability. Gastroenterology 2002, 122, 1376–1387. [Google Scholar]
- Ogino, S.; Cantor, M.; Kawasaki, T.; Brahmandam, M.; Kirkner, G.J.; Weisenberger, D.J.; Campan, M.; Laird, P.W.; Loda, M.; Fuchs, C.S. CpG island methylator phenotype (CIMP) of colorectal cancer is best characterised by quantitative DNA methylation analysis and prospective cohort studies. Gut 2006, 55, 1000–1006. [Google Scholar]
- Ogino, S.; Goel, A. Molecular classification and correlates in colorectal cancer. J. Mol. Diagn. 2008, 10, 13–27. [Google Scholar]
- Ogino, S.; Kawasaki, T.; Kirkner, G.J.; Loda, M.; Fuchs, C.S. CpG island methylator phenotype-low (CIMP-low) in colorectal cancer: Possible associations with male sex and KRAS mutations. J. Mol. Diagn. 2006, 8, 582–588. [Google Scholar]
- Elsaleh, H.; Joseph, D.; Grieu, F.; Zeps, N.; Spry, N.; Iacopetta, B. Association of tumour site and sex with survival benefit from adjuvant chemotherapy in colorectal cancer. Lancet 2000, 355, 1745–1750. [Google Scholar]
- Kotake, K.; Honjo, S.; Sugihara, K.; Kato, T.; Kodaira, S.; Takahashi, T.; Yasutomi, M.; Muto, T.; Koyama, Y. Changes in colorectal cancer during a 20-year period: An extended report from the multi-institutional registry of large bowel cancer, Japan. Dis. Colon Rectum 2003, 46, S32–43. [Google Scholar]
- Micheli, A.; Ciampichini, R.; Oberaigner, W.; Ciccolallo, L.; de Vries, E.; Izarzugaza, I.; Zambon, P.; Gatta, G.; de Angelis, R. EUROCARE Working Group. The advantage of women in cancer survival: An analysis of EUROCARE-4 data. Eur. J. Cancer 2009, 45, 1017–1027. [Google Scholar]
- Hendifar, A.; Yang, D.; Lenz, F.; Lurje, G.; Pohl, A.; Lenz, C.; Ning, Y.; Zhang, W.; Lenz, H.J. Gender disparities in metastatic colorectal cancer survival. Clin. Cancer Res. 2009, 15, 6391–6397. [Google Scholar]
- Majek, O.; Gondos, A.; Jansen, L.; Emrich, K.; Holleczek, B.; Katalinic, A.; Nennecke, A.; Eberle, A.; Brenner, H. GEKID Cancer Survival Working Group. Sex differences in colorectal cancer survival: Population-based analysis of 164,996 colorectal cancer patients in Germany. PLoS One 2013, 8, e68077. [Google Scholar]
- Koo, J.H.; Jalaludin, B.; Wong, S.K.; Kneebone, A.; Connor, S.J.; Leong, R.W. Improved survival in young women with colorectal cancer. Am. J. Gastroenterol. 2008, 103, 1488–1495. [Google Scholar]
- Koo, J.H.; Leong, R.W. Sex differences in epidemiological, clinical and pathological characteristics of colorectal cancer. J. Gastroenterol. Hepatol. 2010, 25, 33–42. [Google Scholar]
- Ponz de Leon, M.; Sant, M.; Micheli, A.; Sacchetti, C.; di Gregorio, C.; Fante, R.; Zanghieri, G.; Melotti, G.; Gatta, G. Clinical and pathologic prognostic indicators in colorectal cancer. A population-based study. Cancer 1992, 69, 626–635. [Google Scholar]
- Manfredi, S.; Bouvier, A.M.; Lepage, C.; Hatem, C.; Dancourt, V.; Faivre, J. Incidence and patterns of recurrence after resection for cure of colonic cancer in a well defined population. Br. J. Surg. 2006, 93, 1115–1122. [Google Scholar]
- Slattery, M.L.; Potter, J.D.; Curtin, K.; Edwards, S.; Ma, K.N.; Anderson, K.; Schaffer, D.; Samowitz, W.S. Estrogens reduce and withdrawal of estrogens increase risk of microsatellite instability-positive colon cancer. Cancer Res. 2001, 61, 126–130. [Google Scholar]
- Shahabi, S.; He, S.; Kopf, M.; Mariani, M.; Petrini, J.; Scambia, G.; Ferlini, C. Free testosterone drives cancer aggressiveness: Evidence from US population studies. PLoS One 2013, 8, e61955. [Google Scholar]
- Orsted, D.D.; Nordestgaard, B.G.; Bojesen, S.E. Plasma testosterone in the general population, cancer prognosis and cancer risk: A prospective cohort study. Ann. Oncol. 2014, 25, 712–718. [Google Scholar]
- Al-Qallaf, D.A.; Al-Mulla, F. Colorectal Carcinoma in Kuwait: Apoptosis and Its Relation to Clinicopathological Characteristics, p53 Expression and ki-ras Proto-Oncogene Mutations. Ph.D. Thesis, Kuwait University, Kuwait City, Kuwait, 2012. [Google Scholar]
- Terracciano, L.M.; Bernasconi, B.; Ruck, P.; Stallmach, T.; Briner, J.; Sauter, G.; Moch, H.; Vecchione, R.; Pollice, L.; Pettinato, G.; et al. Comparative genomic hybridization analysis of hepatoblastoma reveals high frequency of X-chromosome gains and similarities between epithelial and stromal components. Hum. Pathol. 2003, 34, 864–871. [Google Scholar]
- Visakorpi, T.; Hyytinen, E.; Kallioniemi, A.; Isola, J.; Kallioniemi, O.P. Sensitive detection of chromosome copy number aberrations in prostate cancer by fluorescence in situ hybridization. Am. J. Pathol. 1994, 145, 624–630. [Google Scholar]
- Looijenga, L.H.; Oosterhuis, J.W. Pathogenesis of testicular germ cell tumours. Rev. Reprod. 1999, 4, 90–100. [Google Scholar]
- Spatz, A.; Borg, C.; Feunteun, J. X-chromosome genetics and human cancer. Nat. Rev. Cancer 2004, 4, 617–629. [Google Scholar]
- Muscatelli, F.; Walker, A.P.; de Plaen, E.; Stafford, A.N.; Monaco, A.P. Isolation and characterization of a MAGE gene family in the Xp21.3 region. Proc. Natl. Acad. Sci. USA 1995, 92, 4987–4991. [Google Scholar]
- Dos Santos, N.R.; Torensma, R.; de Vries, T.J.; Schreurs, M.W.; de Bruijn, D.R.; Kater-Baats, E.; Ruiter, D.J.; Adema, G.J.; van Muijen, G.N.; van Kessel, A.G. Heterogeneous expression of the SSX cancer/testis antigens in human melanoma lesions and cell lines. Cancer Res. 2000, 60, 1654–1662. [Google Scholar]
- Simpson, A.J.; Caballero, O.L.; Jungbluth, A.; Chen, Y.T.; Old, L.J. Cancer/testis antigens, gametogenesis and cancer. Nat. Rev. Cancer 2005, 5, 615–625. [Google Scholar]
- Choi, J.; Chang, H. The expression of MAGE and SSX, and correlation of COX2, VEGF, and survivin in colorectal cancer. Anticancer Res. 2012, 32, 559–564. [Google Scholar]
- Bottarelli, L.; Azzoni, C.; Necchi, F.; Lagrasta, C.; Tamburini, E.; D’Adda, T.; Pizzi, S.; Sarli, L.; Rindi, G.; Bordi, C. Sex chromosome alterations associate with tumor progression in sporadic colorectal carcinomas. Clin. Cancer Res. 2007, 13, 4365–4370. [Google Scholar]
- Xiao, X.Y.; Zhou, X.Y.; Yan, G.; Sun, M.H.; Du, X. Chromosomal alteration in Chinese sporadic colorectal carcinomas detected by comparative genomic hybridization. Diagn. Mol. Pathol. 2007, 16, 96–103. [Google Scholar]
- Lothe, R.A.; Peltomäki, P.; Meling, G.I.; Aaltonen, L.A.; Nyström-Lahti, M.; Pylkkänen, L.; Heimdal, K.; Andersen, T.I.; Møller, P.; Rognum, T.O.; et al. Genomic instability in colorectal cancer: Relationship to clinicopathological variables and family history. Cancer Res. 1993, 53, 5849–5852. [Google Scholar]
- Muleris, M.; Dutrillaux, A.M.; Olschwang, S.; Salmon, R.J.; Dutrillaux, B. Predominance of normal karyotype in colorectal tumors from hereditary non-polyposis colorectal cancer patients. Genes Chromosomes Cancer 1995, 14, 223–226. [Google Scholar]
- Goel, A.; Arnold, C.N.; Niedzwiecki, D.; Chang, D.K.; Ricciardiello, L.; Carethers, J.M.; Dowell, J.M.; Wasserman, L.; Compton, C.; Mayer, R.J.; et al. Characterization of sporadic colon cancer by patterns of genomic instability. Cancer Res. 2003, 63, 1608–1614. [Google Scholar]
- Camps, J.; Armengol, G.; del Rey, J.; Lozano, J.J.; Vauhkonen, H.; Prat, E.; Egozcue, J.; Sumoy, L.; Knuutila, S.; Miró, R. Genome-wide differences between microsatellite stable and unstable colorectal tumors. Carcinogenesis 2006, 27, 419–428. [Google Scholar]
- Trautmann, K.; Terdiman, J.P.; French, A.J.; Roydasgupta, R.; Sein, N.; Kakar, S.; Fridlyand, J.; Snijders, A.M.; Albertson, D.G.; Thibodeau, S.N.; et al. Chromosomal instability in microsatellite-unstable and stable colon cancer. Clin. Cancer Res. 2006, 12, 6379–6385. [Google Scholar]
- Hawkins, N.J.; Tomlinson, I.; Meagher, A.; Ward, R.L. Microsatellite-stable diploid carcinoma: A biologically distinct and aggressive subset of sporadic colorectal cancer. Br. J. Cancer 2001, 84, 232–236. [Google Scholar]
- Sinicrope, F.A.; Rego, R.L.; Halling, K.C.; Foster, N.; Sargent, D.J.; La Plant, B.; French, A.J.; Laurie, J.A.; Goldberg, R.M.; Thibodeau, S.N.; et al. Prognostic impact of microsatellite instability and DNA ploidy in human colon carcinoma patients. Gastroenterology 2006, 131, 729–737. [Google Scholar]
- Kazama, Y.; Watanabe, T.; Kanazawa, T.; Tanaka, J.; Tanaka, T.; Nagawa, H. Microsatellite instability in poorly differentiated adenocarcinomas of the colon and rectum: Relationship to clinicopathological features. J. Clin. Pathol. 2007, 60, 701–704. [Google Scholar]
- Watanabe, T.; Kobunai, T.; Yamamoto, Y.; Matsuda, K.; Ishihara, S.; Nozawa, K.; Yamada, H.; Hayama, T.; Inoue, E.; Tamura, J.; et al. Chromosomal instability (CIN) phenotype, CIN high or CIN low, predicts survival for colorectal cancer. J. Clin. Oncol. 2012, 30, 2256–2264. [Google Scholar]
- Al-Mulla, F. Microarray-based CGH and copy number analysis of FFPE samples. Methods Mol. Biol. 2011, 724, 131–145. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ali, R.H.; Marafie, M.J.; Bitar, M.S.; Al-Dousari, F.; Ismael, S.; Haider, H.B.; Al-Ali, W.; Jacob, S.P.; Al-Mulla, F. Gender-Associated Genomic Differences in Colorectal Cancer: Clinical Insight from Feminization of Male Cancer Cells. Int. J. Mol. Sci. 2014, 15, 17344-17365. https://doi.org/10.3390/ijms151017344
Ali RH, Marafie MJ, Bitar MS, Al-Dousari F, Ismael S, Haider HB, Al-Ali W, Jacob SP, Al-Mulla F. Gender-Associated Genomic Differences in Colorectal Cancer: Clinical Insight from Feminization of Male Cancer Cells. International Journal of Molecular Sciences. 2014; 15(10):17344-17365. https://doi.org/10.3390/ijms151017344
Chicago/Turabian StyleAli, Rola H., Makia J. Marafie, Milad S. Bitar, Fahad Al-Dousari, Samar Ismael, Hussain Bin Haider, Waleed Al-Ali, Sindhu P. Jacob, and Fahd Al-Mulla. 2014. "Gender-Associated Genomic Differences in Colorectal Cancer: Clinical Insight from Feminization of Male Cancer Cells" International Journal of Molecular Sciences 15, no. 10: 17344-17365. https://doi.org/10.3390/ijms151017344




