Analysis of the rs10046 Polymorphism of Aromatase (CYP19) in Premenopausal Onset of Human Breast Cancer
Abstract
:1. Introduction
2. Results and Discussion
2.1. CYP19 rs10046 SNP and Breast Cancer Risk
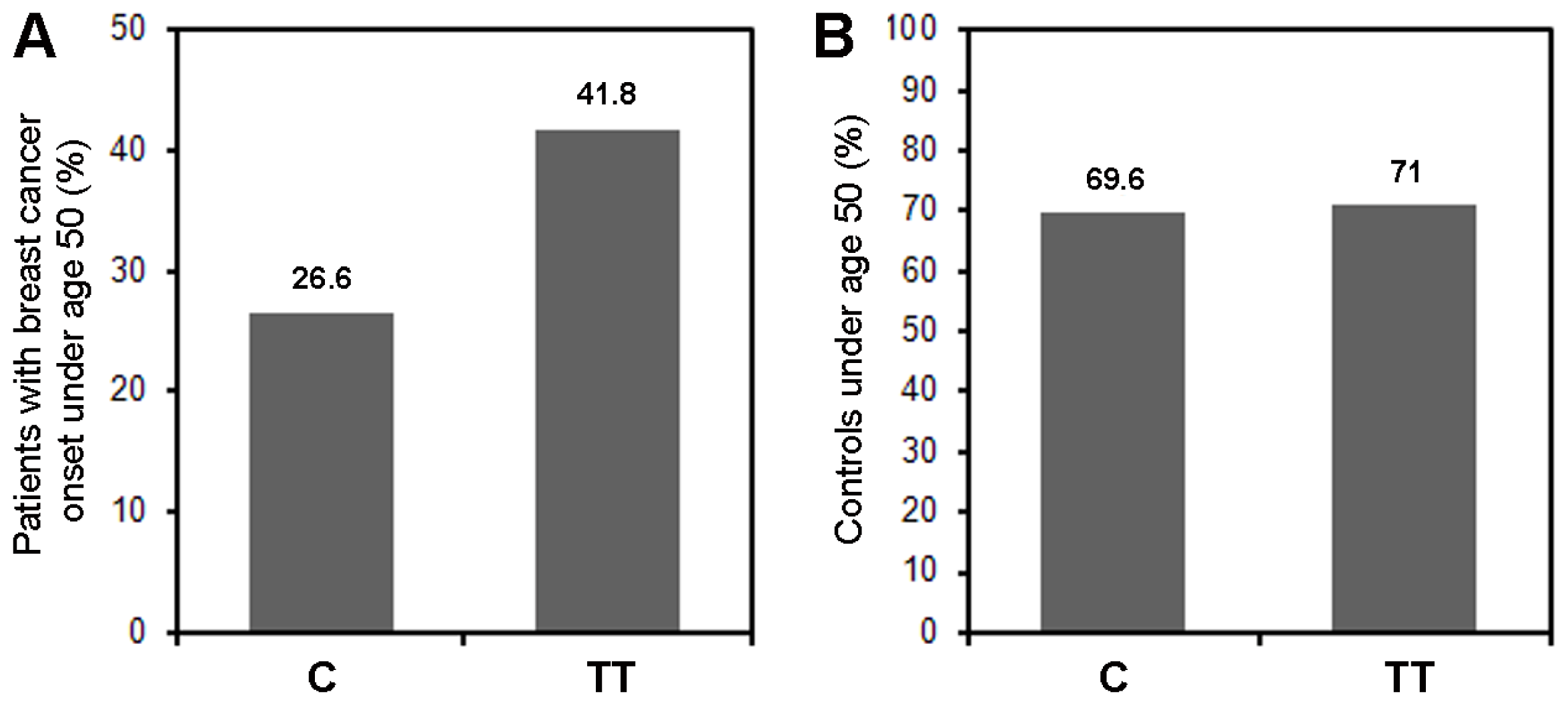
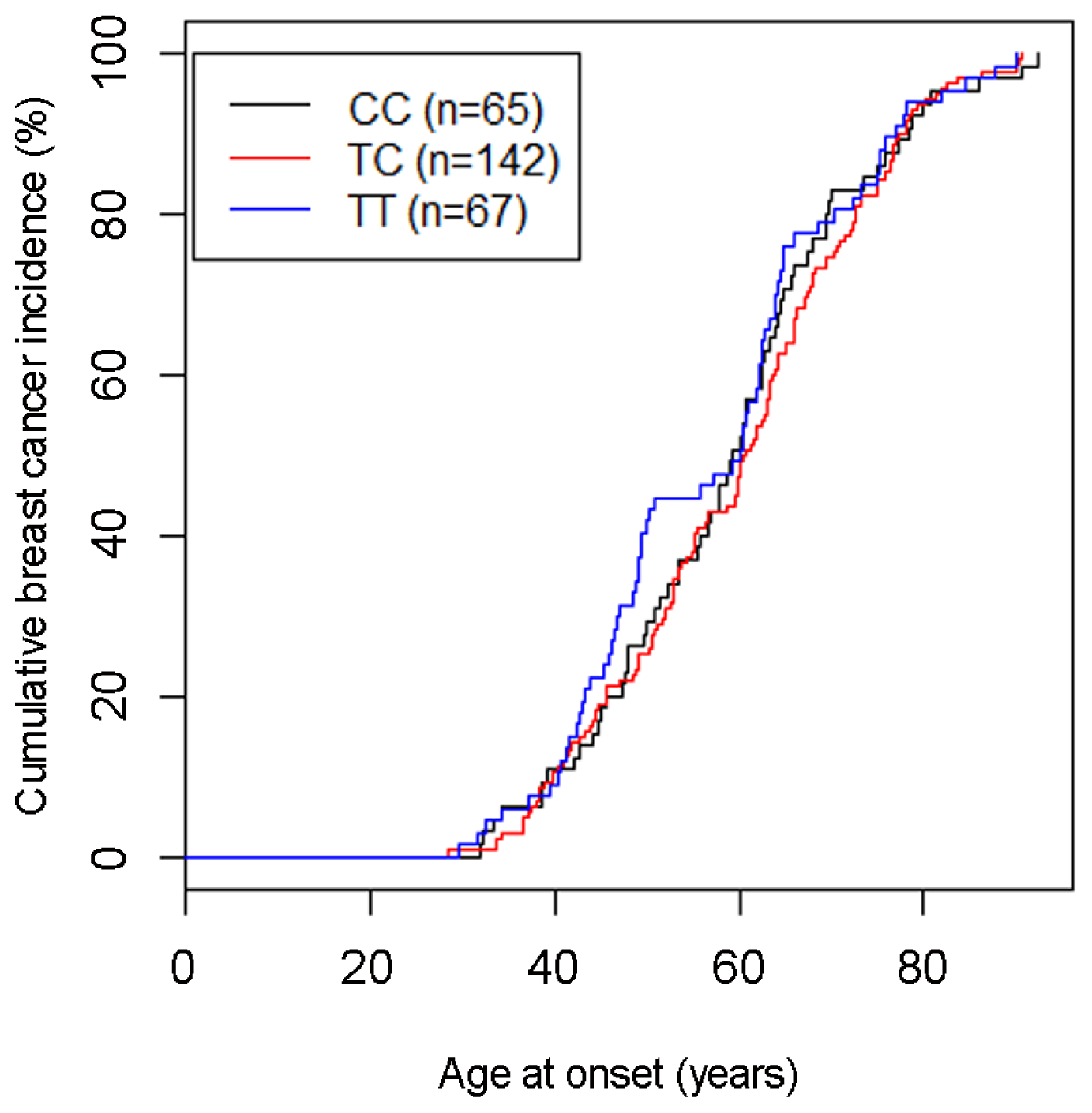
2.1.1. SNP rs10046 and Age at Breast Cancer Onset
2.2. Discussion
3. Experimental Section
3.1. Study Population
3.2. DNA Isolation and Genotyping
3.3. Statistical Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Germain, D. Estrogen carcinogenesis in breast cancer. Endocrinol. Metab. Clin. N. Am 2011, 40, 473–484. [Google Scholar]
- Bulun, S.E.; Zeitoun, K.; Sasano, H.; Simpson, E.R. Aromatase in aging women. Semin. Reprod. Endocrinol 1999, 17, 349–358. [Google Scholar]
- Corbin, C.J.; Graham-Lorence, S.; McPhaul, M.; Mason, J.I.; Mendelson, C.R.; Simpson, E.R. Isolation of a full-length cDNA insert encoding human aromatase system cytochrome P-450 and its expression in nonsteroidogenic cells. Proc. Natl. Acad. Sci. USA 1988, 85, 8948–8952. [Google Scholar]
- Bulun, S.E.; Sebastian, S.; Takayama, K.; Suzuki, T.; Sasano, H.; Shozu, M. The human CYP19 (aromatase P450) gene: Update on physiologic roles and genomic organization of promoters. J. Steroid. Biochem. Mol. Biol 2003, 86, 219–224. [Google Scholar]
- Toda, K.; Terashima, M.; Kawamoto, T.; Sumimoto, H.; Yokoyama, Y.; Kuribayashi, I.; Mitsuuchi, Y.; Maeda, T.; Yamamoto, Y.; Sagara, Y.; et al. Structural and functional characterization of human aromatase P-450 gene. Eur. J. Biochem 1990, 193, 559–565. [Google Scholar]
- Sebastian, S.; Bulun, S.E. A highly complex organization of the regulatory region of the human CYP19 (aromatase) gene revealed by the Human Genome Project. J. Clin. Endocrinol. Metab 2001, 86, 4600–4602. [Google Scholar]
- Yue, W.; Wang, J.P.; Hamilton, C.J.; Demers, L.M.; Santen, R.J. In situ aromatization enhances breast tumor estradiol levels and cellular proliferation. Cancer Res 1998, 58, 927–932. [Google Scholar]
- Brueggemeier, R.W.; Hackett, J.C.; Diaz-Cruz, E.S. Aromatase inhibitors in the treatment of breast cancer. Endocr. Rev 2005, 26, 331–345. [Google Scholar]
- Fasching, P.A.; Loehberg, C.R.; Strissel, P.L.; Lux, M.P.; Bani, M.R.; Schrauder, M.; Geiler, S.; Ringleff, K.; Oeser, S.; Weihbrecht, S.; et al. Single nucleotide polymorphisms of the aromatase gene (CYP19A1), HER2/neu status, and prognosis in breast cancer patients. Breast Cancer Res. Treat 2008, 112, 89–98. [Google Scholar]
- Huang, C.S.; Kuo, S.H.; Lien, H.C.; Yang, S.Y.; You, S.L.; Shen, C.Y.; Lin, C.H.; Lu, Y.S.; Chang, K.J. The CYP19 TTTA repeat polymorphism is related to the prognosis of premenopausal stage I-II and operable stage III breast cancers. Oncologist 2008, 13, 751–760. [Google Scholar]
- Long, J.R.; Kataoka, N.; Shu, X.O.; Wen, W.; Gao, Y.T.; Cai, Q.; Zheng, W. Genetic polymorphisms of the CYP19A1 gene and breast cancer survival. Cancer Epidemiol. Biomark. Prev 2006, 15, 2115–2122. [Google Scholar]
- Dunning, A.M.; Dowsett, M.; Healey, C.S.; Tee, L.; Luben, R.N.; Folkerd, E.; Novik, K.L.; Kelemen, L.; Ogata, S.; Pharoah, P.D.; et al. Polymorphisms associated with circulating sex hormone levels in postmenopausal women. J. Natl. Cancer Inst 2004, 96, 936–945. [Google Scholar]
- Tworoger, S.S.; Chubak, J.; Aiello, E.J.; Ulrich, C.M.; Atkinson, C.; Potter, J.D.; Yasui, Y.; Stapleton, P.L.; Lampe, J.W.; Farin, F.M.; et al. Association of CYP17, CYP19, CYP1B1, and COMT polymorphisms with serum and urinary sex hormone concentrations in postmenopausal women. Cancer Epidemiol. Biomark. Prev 2004, 13, 94–101. [Google Scholar]
- Haiman, C.A.; Dossus, L.; Setiawan, V.W.; Stram, D.O.; Dunning, A.M.; Thomas, G.; Thun, M.J.; Albanes, D.; Altshuler, D.; Ardanaz, E.; et al. Genetic variation at the CYP19A1 locus predicts circulating estrogen levels but not breast cancer risk in postmenopausal women. Cancer Res 2007, 67, 1893–1897. [Google Scholar]
- Wang, L.; Ellsworth, K.A.; Moon, I.; Pelleymounter, L.L.; Eckloff, B.W.; Martin, Y.N.; Fridley, B.L.; Jenkins, G.D.; Batzler, A.; Suman, V.J.; et al. Functional genetic polymorphisms in the aromatase gene CYP19 vary the response of breast cancer patients to neoadjuvant therapy with aromatase inhibitors. Cancer Res 2010, 70, 319–328. [Google Scholar]
- Lunardi, G.; Piccioli, P.; Bruzzi, P.; Notaro, R.; Lastraioli, S.; Serra, M.; Marroni, P.; Bighin, C.; Mansutti, M.; Puglisi, F.; et al. Plasma estrone sulfate concentrations and genetic variation at the CYP19A1 locus in postmenopausal women with early breast cancer treated with letrozole. Breast Cancer Res. Treat 2013, 137, 167–174. [Google Scholar]
- Park, I.H.; Lee, Y.S.; Lee, K.S.; Kim, S.Y.; Hong, S.H.; Jeong, J.; Lee, H.; Ro, J.; Nam, B.H. Single nucleotide polymorphisms of CYP19A1 predict clinical outcomes and adverse events associated with letrozole in patients with metastatic breast cancer. Cancer Chemother. Pharmacol 2011, 68, 1263–1271. [Google Scholar]
- Ferraldeschi, R.; Arnedos, M.; Hadfield, K.D.; A’Hern, R.; Drury, S.; Wardley, A.; Howell, A.; Evans, D.G.; Roberts, S.A.; Smith, I.; et al. Polymorphisms of CYP19A1 and response to aromatase inhibitors in metastatic breast cancer patients. Breast Cancer Res. Treat 2012, 133, 1191–1198. [Google Scholar]
- Chen, C.; Sakoda, L.C.; Doherty, J.A.; Loomis, M.M.; Fish, S.; Ray, R.M.; Lin, M.G.; Fan, W.; Zhao, L.P.; Gao, D.L.; et al. Genetic variation in CYP19A1 and risk of breast cancer and fibrocystic breast conditions among women in Shanghai, China. Cancer Epidemiol. Biomark. Prev 2008, 17, 3457–3466. [Google Scholar]
- Ma, X.; Qi, X.; Chen, C.; Lin, H.; Xiong, H.; Li, Y.; Jiang, J. Association between CYP19 polymorphisms and breast cancer risk: Results from 10,592 cases and 11,720 controls. Breast Cancer Res. Treat 2010, 122, 495–501. [Google Scholar]
- Cai, Q.; Kataoka, N.; Li, C.; Wen, W.; Smith, J.R.; Gao, Y.T.; Shu, X.O.; Zheng, W. Haplotype analyses of CYP19A1 gene variants and breast cancer risk: Results from the Shanghai breast cancer study. Cancer Epidemiol. Biomarkers Prev 2008, 17, 27–32. [Google Scholar]
- Kristensen, V.N.; Harada, N.; Yoshimura, N.; Haraldsen, E.; Lonning, P.E.; Erikstein, B.; Karesen, R.; Kristensen, T.; Borresen-Dale, A.L. Genetic variants of CYP19 (aromatase) and breast cancer risk. Oncogene 2000, 19, 1329–1333. [Google Scholar]
- Haiman, C.A.; Hankinson, S.E.; Spiegelman, D.; Brown, M.; Hunter, D.J. No association between a single nucleotide polymorphism in CYP19 and breast cancer risk. Cancer Epidemiol. Biomark. Prev 2002, 11, 215–216. [Google Scholar]
- Ralph, D.A.; Zhao, L.P.; Aston, C.E.; Manjeshwar, S.; Pugh, T.W.; DeFreese, D.C.; Gramling, B.A.; Shimasaki, C.D.; Jupe, E.R. Age-specific association of steroid hormone pathway gene polymorphisms with breast cancer risk. Cancer 2007, 109, 1940–1948. [Google Scholar]
- Zhang, L.; Gu, L.; Qian, B.; Hao, X.; Zhang, W.; Wei, Q.; Chen, K. Association of genetic polymorphisms of ER-alpha and the estradiol-synthesizing enzyme genes CYP17 and CYP19 with breast cancer risk in Chinese women. Breast Cancer Res. Treat 2009, 114, 327–338. [Google Scholar]
- Pineda, B.; Garcia-Perez, M.A.; Cano, A.; Lluch, A.; Eroles, P. Associations between aromatase CYP19 rs10046 polymorphism and breast cancer risk: From a case-control to a meta-analysis of 20,098 subjects. PLoS One 2013, 8, e53902. [Google Scholar]
- Buzdar, A.U.; Vergote, I.; Sainsbury, R. The impact of hormone receptor status on the clinical efficacy of the new-generation aromatase inhibitors: A review of data from first-line metastatic disease trials in postmenopausal women. Breast J 2004, 10, 211–217. [Google Scholar]
- Thomas, H.V.; Key, T.J.; Allen, D.S.; Moore, J.W.; Dowsett, M.; Fentiman, I.S.; Wang, D.Y. A prospective study of endogenous serum hormone concentrations and breast cancer risk in post-menopausal women on the island of Guernsey. Br. J. Cancer 1997, 76, 401–405. [Google Scholar]
- Colomer, R.; Monzo, M.; Tusquets, I.; Rifa, J.; Baena, J.M.; Barnadas, A.; Calvo, L.; Carabantes, F.; Crespo, C.; Munoz, M.; et al. A single-nucleotide polymorphism in the aromatase gene is associated with the efficacy of the aromatase inhibitor letrozole in advanced breast carcinoma. Clin. Cancer Res 2008, 14, 811–816. [Google Scholar]
- Proestling, K.; Hebar, A.; Pruckner, N.; Marton, E.; Vinatzer, U.; Schreiber, M. The Pro Allele of the p53 codon 72 polymorphism is associated with decreased intratumoral expression of BAX and p21, and increased breast cancer risk. PLoS One 2012, 7, e47325. [Google Scholar]
- Taubenschuß, E.; Marton, E.; Mogg, M.; Frech, B.; Ehart, L.; Muin, D.; Schreiber, M. The L10P polymorphism and serum levels of transforming growth factor β1 in human breast cancer. Int. J. Mol. Sci 2013, 14, 15376–15385. [Google Scholar]
- The R Development Core Team, R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2009.
- Bender, R.; Lange, S. Adjusting for multiple testing—When and how? J. Clin. Epidemiol 2001, 54, 343–349. [Google Scholar]
| Total | CC | TC | TT | p-value | ||
|---|---|---|---|---|---|---|
| All subjects | 527 | 120 (22.8%) | 278 (52.8%) | 129 (24.5%) | ||
| Patients | 274 | 65 (23.7%) | 142 (51.8%) | 67 (24.5%) | ||
| Controls | 253 | 55 (21.7%) | 136 (53.8%) | 62 (24.5%) | ||
| Patient subgroups | ||||||
| Age (years) | <50 | 83 | 19 (22.9%) | 36 (43.4%) | 28 (33.7%) | 0.053 |
| ≥50 | 191 | 46 (24.1%) | 106 (55.5%) | 39 (20.4%) | ||
| Menopausal status | pre | 63 | 13 (20.6%) | 29 (46.0%) | 21 (33.3%) | 0.249 |
| post | 176 | 40 (22.7%) | 96 (54.5%) | 40 (22.7%) | ||
| na | 35 | 12 (34.3%) | 17 (48.6%) | 6 (17.1%) | ||
| Tumor size | pT1 | 136 | 28 (20.6%) | 72 (52.9%) | 36 (26.5%) | 0.339 |
| pT2–4 | 67 | 20 (29.9%) | 32 (47.8%) | 15 (22.4%) | ||
| other, na | 71 | 17 (23.9%) | 38 (53.5%) | 16 (22.5%) | ||
| Tumor type | ductal | 153 | 41 (26.8%) | 76 (49.7%) | 36 (23.5%) | 0.996 |
| lobular | 48 | 13 (27.1%) | 24 (50.0%) | 11 (22.9%) | ||
| other, na | 73 | 11 (15.1%) | 42 (57.5%) | 20 (27.4%) | ||
| Stage | 0 or I | 117 | 27 (23.1%) | 67 (57.3%) | 23 (19.7%) | 0.193 |
| II–IV | 93 | 25 (26.9%) | 42 (45.2%) | 26 (28.0%) | ||
| other, na | 64 | 13 (20.3%) | 33 (51.6%) | 18 (28.1%) | ||
| Grade | pG1–2 | 161 | 39 (24.2%) | 82 (50.9%) | 40 (24.8%) | 0.982 |
| pG3 | 92 | 23 (25.0%) | 47 (51.1%) | 22 (23.9%) | ||
| na | 21 | 3 (14.3%) | 13 (61.9%) | 5 (23.8%) | ||
| Lymph node status | pN0 | 148 | 38 (25.7%) | 82 (55.4%) | 28 (18.9%) | 0.181 |
| pN+ | 55 | 13 (23.6%) | 25 (45.5%) | 17 (30.9%) | ||
| na | 69 | 15 (21.7%) | 30 (43.5%) | 24 (34.8%) | ||
| ER status | pos | 202 | 48 (23.8%) | 100 (49.5%) | 54 (26.7%) | 0.690 |
| neg | 60 | 14 (23.3%) | 33 (55.0%) | 13 (21.7%) | ||
| na | 12 | 3 (25.0%) | 9 (75.0%) | 0 (0.0%) | ||
| PR status | pos | 142 | 31 (21.8%) | 76 (53.5%) | 35 (24.6%) | 0.604 |
| neg | 120 | 31 (25.8%) | 57 (47.5%) | 32 (26.7%) | ||
| na | 12 | 3 (25.0%) | 9 (75.0%) | 0 (0.0%) | ||
| HER2 status | pos | 54 | 7 (13.0%) | 31 (57.4%) | 16 (29.6%) | 0.119 |
| neg | 205 | 54 (26.3%) | 101 (49.3%) | 50 (24.4%) | ||
| na | 15 | 4 (26.7%) | 10 (66.7%) | 1 (6.7%) | ||
| Genotypes | OR | 95% CI | p-value |
|---|---|---|---|
| CC vs. TT | 1.09 | 0.66–1.80 | 0.752 |
| CC vs. TC | 1.13 | 0.74–1.74 | 0.549 |
| TC vs. TT | 0.97 | 0.64–1.47 | 0.873 |
| CC + TC vs. TT | 1.00 | 0.67–1.49 | 0.989 |
| CC vs. TC + TT | 1.12 | 0.74–1.68 | 0.587 |
| C vs. T | 1.04 | 0.81–1.34 | 0.734 |
| Subgroup | No. of cases (%) | CC vs. TT | TC vs. TT | C vs. T | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| OR | 95% CI | p-value | OR | 95% CI | p-value | OR | 95% CI | p-value | ||||
| Age (years) 1 | <50 | 83 | 30.3% | 0.76 | (0.39–1.52) | 0.44 | 0.59 | (0.33–1.04) | 0.06 | 0.85 | (0.59–1.21) | 0.36 |
| ≥50 | 191 | 69.7% | 1.33 | (0.76–2.34) | 0.32 | 1.24 | (0.77–2.00) | 0.37 | 1.14 | (0.87–1.48) | 0.34 | |
| Menopausal status | pre | 63 | 26.4% | 0.70 | (0.32–1.52) | 0.38 | 0.63 | (0.33–1.19) | 0.16 | 0.81 | (0.54–1.21) | 0.31 |
| post | 176 | 73.6% | 1.13 | (0.64–1.99) | 0.72 | 1.09 | (0.68–1.76) | 0.76 | 1.06 | (0.80–1.41) | 0.68 | |
| Tumor type | ductal | 153 | 76.1% | 1.28 | (0.72–2.28) | 0.42 | 0.96 | (0.59–1.58) | 0.85 | 1.13 | (0.85–1.52) | 0.39 |
| lobular | 48 | 23.9% | 1.33 | (0.55–3.22) | 0.58 | 0.99 | (0.46–2.16) | 0.92 | 1.16 | (0.74–1.82) | 0.52 | |
| Tumor size | pT1 | 136 | 67.0% | 0.88 | (0.47–1.62) | 0.70 | 0.91 | (0.55–1.50) | 0.75 | 0.93 | (0.69–1.27) | 0.67 |
| pT2–4 | 67 | 33.0% | 1.50 | (0.70–3.22) | 0.29 | 0.97 | (0.49–1.93) | 0.93 | 1.24 | (0.84–1.84) | 0.28 | |
| Stage | 0 or I | 117 | 55.7% | 1.32 | (0.68–2.57) | 0.45 | 1.33 | (0.76–2.33) | 0.30 | 1.15 | (0.83–1.59) | 0.41 |
| II–IV | 93 | 44.3% | 1.08 | (0.56–2.09) | 0.80 | 0.74 | (0.41–1.31) | 0.27 | 1.04 | (0.74–1.46) | 0.84 | |
| Grade | pG1–2 | 161 | 63.6% | 1.10 | (0.62–1.95) | 0.72 | 0.93 | (0.58–1.51) | 0.76 | 1.05 | (0.79–1.39) | 0.76 |
| pG3 | 92 | 36.4% | 1.18 | (0.59–2.35) | 0.66 | 0.97 | (0.54–1.75) | 0.94 | 1.09 | (0.77–1.54) | 0.64 | |
| Nodal status | pN0 | 148 | 72.9% | 1.53 | (0.83–2.83) | 0.19 | 1.34 | (0.79–2.27) | 0.27 | 1.21 | (0.91–1.61) | 0.20 |
| pN+ | 55 | 27.1% | 0.86 | (0.38–1.93) | 0.76 | 0.67 | (0.34–1.33) | 0.24 | 0.91 | (0.60–1.39) | 0.66 | |
| ER status | pos | 202 | 77.1% | 1.00 | (0.59–1.71) | 0.95 | 0.84 | (0.54–1.32) | 0.46 | 1.00 | (0.76–1.30) | 0.98 |
| neg | 60 | 22.9% | 1.21 | (0.53–2.81) | 0.60 | 1.16 | (0.57–2.35) | 0.66 | 1.10 | (0.73–1.67) | 0.65 | |
| PR status | pos | 142 | 54.2% | 1.00 | (0.55–1.83) | 0.94 | 0.99 | (0.60–1.63) | 0.95 | 1.00 | (0.74–1.35) | 0.99 |
| neg | 120 | 45.8% | 1.09 | (0.59–2.02) | 0.82 | 0.81 | (0.48–1.38) | 0.46 | 1.04 | (0.76–1.42) | 0.80 | |
| HER2 status | pos | 54 | 20.8% | 0.49 | (0.19–1.29) | 0.14 | 0.88 | (0.45–1.73) | 0.67 | 0.73 | (0.47–1.14) | 0.17 |
| neg | 205 | 79.2% | 1.22 | (0.72–2.07) | 0.46 | 0.92 | (0.59–1.45) | 0.69 | 1.10 | (0.85–1.44) | 0.47 | |
| Ki67 status | >10% | 105 | 48.2% | 1.08 | (0.22–2.13) | 0.80 | 1.10 | (0.63–1.93) | 0.72 | 1.04 | (0.74–1.46) | 0.82 |
| ≤10% | 113 | 51.8% | 1.21 | (0.65–2.25) | 0.58 | 0.83 | (0.48–1.43) | 0.53 | 1.10 | (0.80–1.51) | 0.56 | |
| p53 status | pos | 58 | 22.7% | 0.65 | (0.29–1.49) | 0.36 | 0.67 | (0.35–1.29) | 0.20 | 0.79 | (0.52–1.20) | 0.27 |
| neg | 197 | 77.3% | 1.18 | (0.68–2.03) | 0.54 | 1.02 | (0.64–1.62) | 0.95 | 1.08 | (0.83–1.42) | 0.56 | |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zins, K.; Mogg, M.; Schneeberger, C.; Abraham, D.; Schreiber, M. Analysis of the rs10046 Polymorphism of Aromatase (CYP19) in Premenopausal Onset of Human Breast Cancer. Int. J. Mol. Sci. 2014, 15, 712-724. https://doi.org/10.3390/ijms15010712
Zins K, Mogg M, Schneeberger C, Abraham D, Schreiber M. Analysis of the rs10046 Polymorphism of Aromatase (CYP19) in Premenopausal Onset of Human Breast Cancer. International Journal of Molecular Sciences. 2014; 15(1):712-724. https://doi.org/10.3390/ijms15010712
Chicago/Turabian StyleZins, Karin, Maurice Mogg, Christian Schneeberger, Dietmar Abraham, and Martin Schreiber. 2014. "Analysis of the rs10046 Polymorphism of Aromatase (CYP19) in Premenopausal Onset of Human Breast Cancer" International Journal of Molecular Sciences 15, no. 1: 712-724. https://doi.org/10.3390/ijms15010712






