Re-Evaluation of Binding Properties of Recombinant Lymphocyte Receptors NKR-P1A and CD69 to Chemically Synthesized Glycans and Peptides
Abstract
:1. Introduction
2. Results and Discussion
2.1. Results
2.1.1. Protein Expression, Refolding and Purification
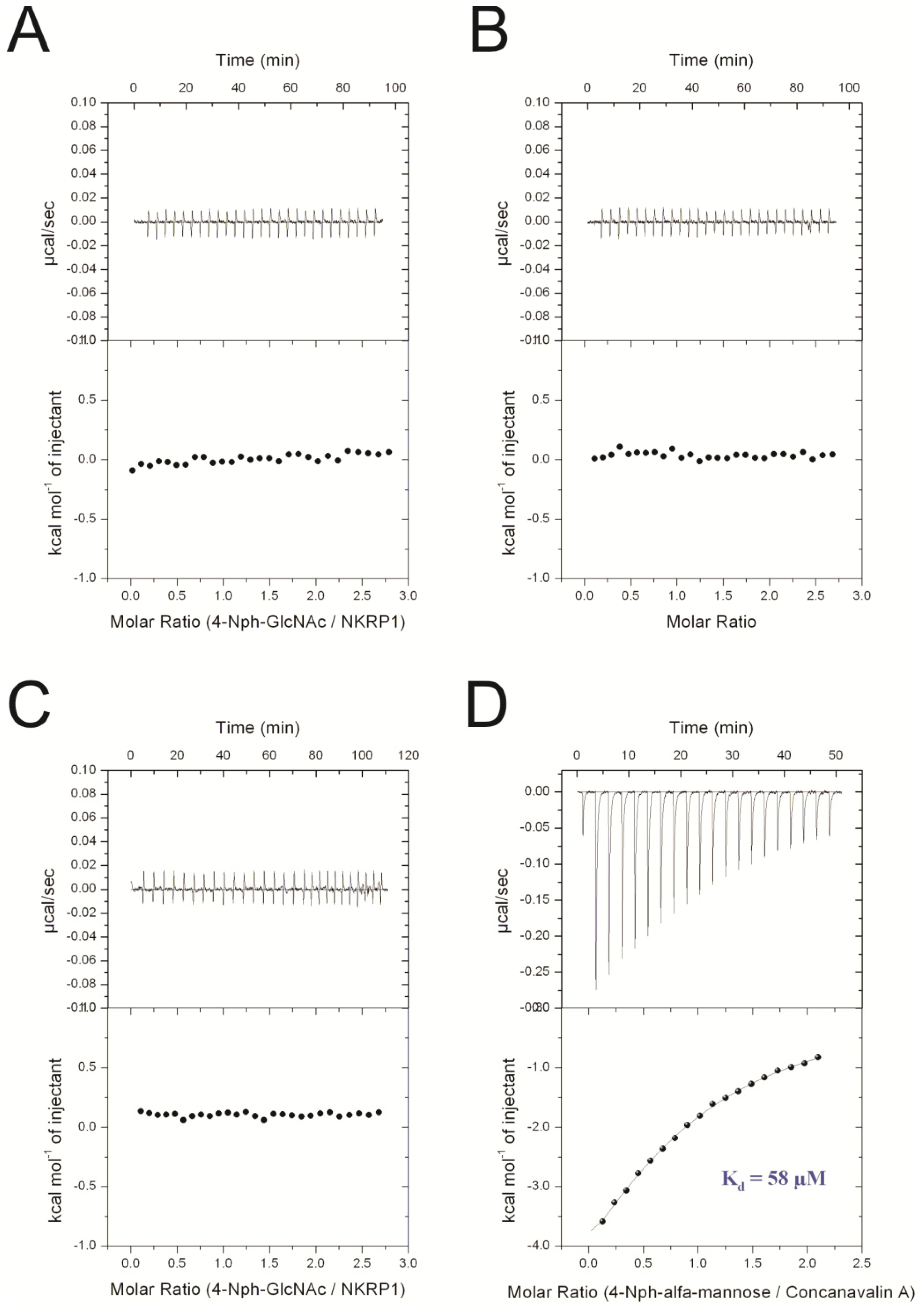
2.1.2. Isothermal Titration Calorimetry Measurements
2.1.3. NMR Titrations
2.2. Discussion
3. Experimental Section
3.1. Protein Expression and Purification
3.2. Calorimetric Experiments
3.3. NMR Titration Experiments
4. Conclusions
Supplementary Information
ijms-15-01271-s001.pdfAcknowledgments
Conflicts of Interest
Abbreviations
| GalNAc | 2-acetamido-2-deoxy-d-galactopyranose |
| GlcNAc | 2-acetamido-2-deoxy- d-glucopyranose |
| pNP-GlcNAc | p-nitrophenyl 2-acetamido-β-d-glucopyranoside |
| ITC | isothermal titration calorimetry |
| ManNAc | 2-acetamido-d-mannopyranose |
| NK | natural killer |
References
- Moretta, A.; Poggi, A.; Pende, D.; Tripodi, G.; Orengo, A.M.; Pella, N.; Augugliaro, R.; Bottino, C.; Ciccone, E.; Moretta, L. CD69-mediated pathway of lymphocyte activation: Anti-CD69 monoclonal antibodies trigger the cytolytic activity of different lymphoid effector cells with the exception of cytolytic T lymphocytes expressing T cell receptor alpha/beta. J. Exp. Med 1991, 174, 1393–1398. [Google Scholar]
- Giorda, R.; Rudert, W.A.; Vavassori, C.; Chambers, W.H.; Hiserodt, J.C.; Trucco, M. NKR-P1, a signal transduction molecule on natural killer cells. Science 1990, 249, 1298–1300. [Google Scholar]
- Bezouska, K.; Vlahas, G.; Horvath, O.; Jinochova, G.; Fiserova, A.; Giorda, R.; Chambers, W.H.; Feizi, T.; Pospisil, M. Rat natural killer cell antigen, NKR-P1, related to C-type animal lectins is a carbohydrate-binding protein. J. Biol. Chem 1994, 269, 16945–16952. [Google Scholar]
- Bezouska, K.; Yuen, C.T.; O’Brien, J.; Childs, R.A.; Chai, W.; Lawson, A.M.; Drbal, K.; Fiserova, A.; Pospisil, M.; Feizi, T. Oligosaccharide ligands for NKR-P1 protein activate NK cells and cytotoxicity. Nature 1994, 372, 150–157. [Google Scholar]
- Bezouska, K.; Nepovim, A.; Horvath, O.; Pospisil, M.; Hamann, J.; Feizi, T. CD 69 antigen of human lymphocytes is a calcium-dependent carbohydrate-binding protein. Biochem. Biophys. Res. Commun 1995, 208, 68–74. [Google Scholar]
- Kogelberg, H.; Montero, E.; Bay, S.; Lawson, A.M.; Feizi, T. Re-evaluation of monosaccharide binding property of recombinant soluble carbohydrate recognition domain of the natural killer cell receptor NKR-P1A. J. Biol. Chem 1999, 274, 30335–30336. [Google Scholar]
- Childs, R.A.; Galustian, C.; Lawson, A.M.; Dougan, G.; Benwell, K.; Frankel, G.; Feizi, T. Recombinant soluble human CD69 dimer produced in Escherichia coli: Re-evaluation of saccharide binding. Biochem. Biophys. Res. Commun 1999, 266, 19–23. [Google Scholar]
- Bezouska, K.; Sklenar, J.; Dvorakova, J.; Havlicek, V.; Pospisil, M.; Thiem, J.; Kren, V. NKR-P1A protein, an activating receptor of rat natural killer cells, binds to the chitobiose core of uncompletely glycosylated N-linked glycans, and to linear chitooligomers. Biochem. Biophys. Res. Commun 1997, 38, 149–153. [Google Scholar]
- Krist, P.; Herkommerova-Rajnochova, E.; Rauvolfova, J.; Semenuk, T.; Vavruskova, P.; Pavlicek, J.; Bezouska, K.; Petrus, L.; Kren, V. Toward an optimal oligosaccharide ligand for rat natural killer cell activation receptor NKR-P1. Biochem. Biophys. Res. Commun 2001, 287, 11–20. [Google Scholar]
- Sedmera, P.; Prikrylova, V.; Bezouska, K.; Rajnochova, E.; Thiem, J.; Kren, V. Preparation of ManNAc containing chitooligomers by isomerisation and their binding to NKR-P1 protein. J. Carbohydr. Chem 1998, 17, 1351–1357. [Google Scholar]
- Kren, V.; Dvorakova, J.; Gambert, U.; Sedmera, P.; Havlicek, V.; Thiem, J.; Bezouska, K. β-Glucosylation of chitooligomers by galactosyltransferase. Carbohydr. Res 1997, 305, 517–523. [Google Scholar]
- Bezouska, K.; Kren, V.; Kieburg, C.; Lindhorst, T.K. GlcNAc-terminated glycodendrimers form defined precipitates with the soluble dimeric receptor of rat natural killer cells, sNKR-P1A. FEBS Lett 1998, 26, 243–247. [Google Scholar]
- Pospisil, M.; Vannucci, L.; Fiserova, A.; Krausova, K.; Horvath, O.; Kren, V.; Mosca, F.; Lindhorst, T.K.; Sadalapure, K.; Bezouska, K. Glycodendrimeric ligands of c-type lectin receptors as therapeutic agents in experimental cancer. Prog. Basic Clin. Immunol 2001, 495, 343–347. [Google Scholar]
- Krist, P.; Vannucci, L.; Kuzma, M.; Man, P.; Sadalapure, K.; Patel, A.; Bezouska, K.; Pospisil, M.; Petrus, L.; Lindhorst, T.K.; et al. Fluorescent labelled thiourea-bridged glycodendrons. ChemBioChem 2004, 5, 445–452. [Google Scholar]
- Vannucci, L.; Fiserova, A.; Sadalapure, K.; Lindhorst, T.K.; Kuldova, M.; Rossmann, P.; Horvath, O.; Kren, V.; Krist, P.; Bezouska, K.; et al. Effects of N-acetyl-glucosamine-coated glycodendrimers as biological modulators in the B16F10 melanoma model in vivo. Int. J. Oncol. 2003, 23, 285–296. [Google Scholar]
- Pavlicek, J.; Sopko, B.; Ettrich, R.; Kopecky, V.; Baumruk, V.; Man, P.; Havlicek, V.; Vrbacky, M.; Martinkova, L.; Kren, V.; et al. Molecular characterization of binding of calcium and carbohydrates by an early activation antigen of lymphocytes CD69. Biochemistry 2003, 42, 9295–9306. [Google Scholar]
- Natarajan, K.; Sawicki, M.W.; Margulies, D.H.; Mariuzza, R.A. Crystal structure of human CD69: A C-type lectin-like activation marker of hematopoietic cells. Biochemistry 2000, 39, 14779–14786. [Google Scholar]
- Kavan, D.; Kubickova, M.; Bily, J.; Vanek, O.; Hofbauerova, K.; Mrazek, H.; Rozbesky, D.; Bojarova, P.; Kren, V.; Zidek, L.; et al. Cooperation between subunits is essential for high-affinity binding of N-acetyl-D-hexosamines to dimeric soluble and dimeric cellular forms of human CD69. Biochemistry 2010, 49, 4060–4067. [Google Scholar]
- Kovalova, A.; Ledvina, M.; Saman, D.; Zyka, D.; Kubickova, M.; Zidek, L.; Sklenar, V.; Pompach, P.; Kavan, D.; Bily, J.; et al. Synthetic N-acetyl-D-glucosamine based fully branched tetrasaccharide, a mimetic of the endogenous ligand for CD69, activates CD69+ killer lymphocytes upon dimerization via a hydrophilic flexible linker. J. Med. Chem 2010, 53, 4050–4065. [Google Scholar]
- Attolino, E.; Bonaccorsi, F.; Catelani, G.; D’Andrea, F.; Krenek, K.; Bezouska, K.; Kren, V. Improved preparation of β-D-ManNAc-(1→4)-D -Glc and β-D-TalNAc-(1→4)-D-Glc disaccharides and evaluation of their activating properties on the natural killer cells NKR-P1 and CD69 receptors. J. Carbohydr. Chem 2008, 27, 156–171. [Google Scholar]
- Catelani, G.; D’Andrea, F.; Griselli, A.; Guazzelli, L.; Nemcova, P.; Bezouska, K.; Krenek, K.; Kren, V. Deoxynojirimycin and its hexosaminyl derivatives bind to natural killer cell receptors rNKR-P1A and hCD69. Bioorg. Med. Chem. Lett 2010, 20, 4645–4648. [Google Scholar]
- Bojarova, P.; Krenek, K.; Kuzma, M.; Petraskova, L.; Bezouska, K.; Namdjou, D.J.; Elling, L.; Kren, V. N-acetylhexosamine triad in one molecule: Chemoenzymatic introduction of 2-acetamido- 2-deoxy-beta-D-galactopyranosyluronic acid residue into a complex oligosaccharide. J. Mol. Catal. B Enzym 2008, 50, 69–73. [Google Scholar]
- Fialova, P.; Namdjou, D.J.; Ettrich, R.; Prikrylova, V.; Rauvolfova, J.; Krenek, K.; Kuzma, M.; Elling, L.; Bezouska, K.; Kren, V. Combined application of galactose oxidase and β-N-acetylhexosaminidase in the synthesis of complex immunoactive N-acetyl-D-galactosaminides. Adv. Synth. Catal 2005, 347, 997–1006. [Google Scholar]
- Bojarova, P.; Slamova, K.; Krenek, K.; Gazak, R.; Kulik, N.; Ettrich, R.; Pelantova, H.; Kuzma, M.; Riva, S.; Adamek, D.; et al. Charged hexosaminides as new substrates for β-N-acetylhexosaminidase-catalyzed synthesis of immunomodulatory disaccharides. Adv. Synth. Catal 2011, 353, 2409–2420. [Google Scholar]
- Bojarova, P.; Krenek, K.; Wetjen, K.; Adamiak, K.; Pelantova, H.; Bezouska, K.; Elling, L.; Kren, V. Synthesis of LacdiNAc-terminated glycoconjugates by mutant galactosyltransferase—A way to new glycodrugs and materials. Glycobiology 2009, 19, 509–517. [Google Scholar]
- Drozdova, A.; Bojarova, P.; Krenek, K.; Weignerova, L.; Henssen, B.; Elling, L.; Christensen, H.; Jensen, H.H.; Pelantova, H.; Kuzma, M.; et al. Enzymatic synthesis of dimeric glycomimetic ligands of NK cell activation receptors. Carbohydr. Res 2011, 346, 1599–1609. [Google Scholar]
- Bezouska, K.; Snajdrova, R.; Krenek, K.; Vancurova, M.; Kadek, A.; Adamek, D.; Lhotak, P.; Kavan, D.; Hofbauerova, K.; Man, P.; et al. Carboxylated calixarenes bind strongly to CD69 and protect CD69(+) killer cells from suicidal cell death induced by tumor cell surface ligands. Bioorg. Med. Chem 2010, 18, 1434–1440. [Google Scholar]
- Slamova, K.; Marhol, P.; Bezouska, K.; Lindkvist, L.; Hansen, S.G.; Kren, V.; Jensen, H.H. Synthesis and biological activity of glycosyl-1H-1,2,3-triazoles. Bioorg. Med. Chem. Lett 2010, 20, 4263–4265. [Google Scholar]
- Renaudet, O.; Krenek, K.; Bossu, I.; Dumy, P.; Kadek, A.; Adamek, D.; Vanek, O.; Kavan, D.; Gazak, R.; Sulc, M.; et al. Synthesis of multivalent glycoconjugates containing the immunoactive LELTE peptide: Effect of glycosylation on cellular activation and natural killing by human peripheral blood mononuclear cells. J. Am. Chem. Soc 2010, 132, 6800–6808. [Google Scholar]
- Bezouska, K.; Pavlicek, J.; Sopko, B.; Kren, V.; Fiserová, A.; Pospisil, M.; Novak, P.; Havlicek, V. Interaction of CD69, the earliest activation antigen of lymphocytes, with calcium and saccharides in three dimensions. Scand. J. Imunnol 2001, 54, 26. [Google Scholar]
- Bezouska, K.; Kubinkova, Z.; Stribny, J.; Volfova, B.; Pompach, P.; Kuzma, M.; Sirova, M.; Rihova, B. Dimerization of an immunoactivating peptide derived from mycobacterial hsp65 using N-hydroxysuccinimide based bifunctional reagents is critical for its antitumor properties. Bioconjug. Chem 2012, 23, 2032–2041. [Google Scholar]
- Vanek, O.; Nalezkova, M.; Kavan, D.; Borovickova, I.; Pompach, P.; Novak, P.; Kumar, V.; Vannucci, L.; Hudecek, J.; Hofbauerova, K.; et al. Soluble recombinant CD69 receptors optimized to have an exceptional physical and chemical stability display prolonged circulation and remain intact in the blood of mice. FEBS J 2008, 275, 5589–5606. [Google Scholar]
- Kveberg, L.; Dai, K.; Westgaard, I.H.; Daws, M.R.; Fossum, S.; Naper, C.; Vaage, J.T. Two major groups of rat NKR-P1 receptors can be distinguished based on chromosomal localization, phylogenetic analysis and Clr ligand binding. Eur. J. Imunnol 2009, 39, 541–551. [Google Scholar]
- Hartmann, J.; Tran, T.V.; Kaudeer, J.; Oberle, K.; Herrmann, J.; Quagliano, I.; Abel, T.; Cohnen, A.; Gatterdam, V.; Jacobs, A.; et al. The stalk domain and the glycosylation status of the activating natural killer cell receptor NKp30 are important for ligand binding. J. Biol. Chem 2012, 287, 31527–31539. [Google Scholar]
- Rozbesky, D.; Kavan, D.; Chmelik, J.; Novak, P.; Vanek, O.; Bezouska, K. High-level expression of soluble form of mouse natural killer cell receptor NKR-P1C(B6) in Escherichia coli. Protein Expr. Purif 2011, 77, 178–184. [Google Scholar]
- Report of the Joint Ethical Committee of the Institute of Microbiology, Prague and Charles University in Prague. English Translation Can Be Found in the Supplementary Material. Available online: http://www.biomed.cas.cz/mbu/doc/VyjadreniEK.PDF (accessed on 15 January 2014).
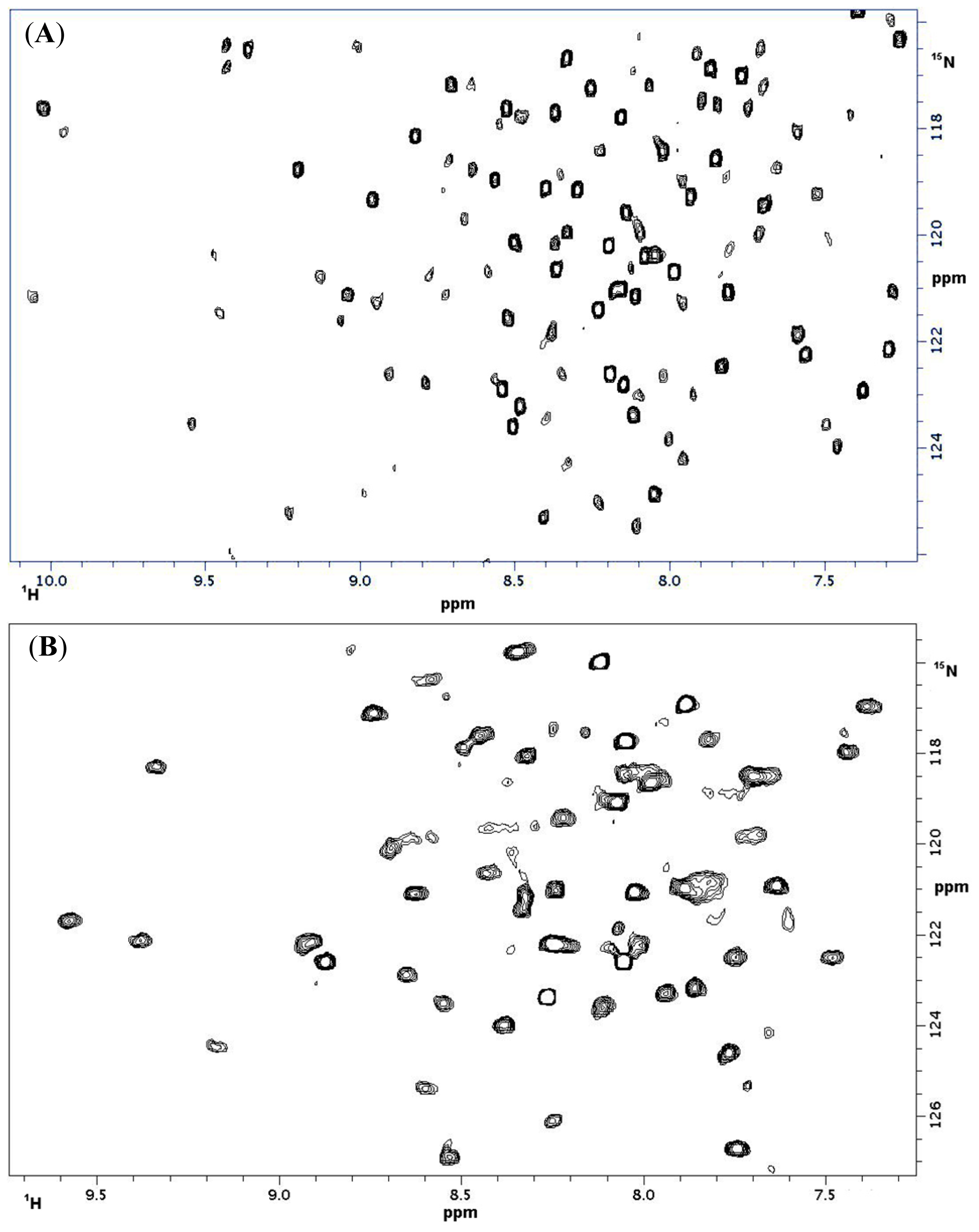
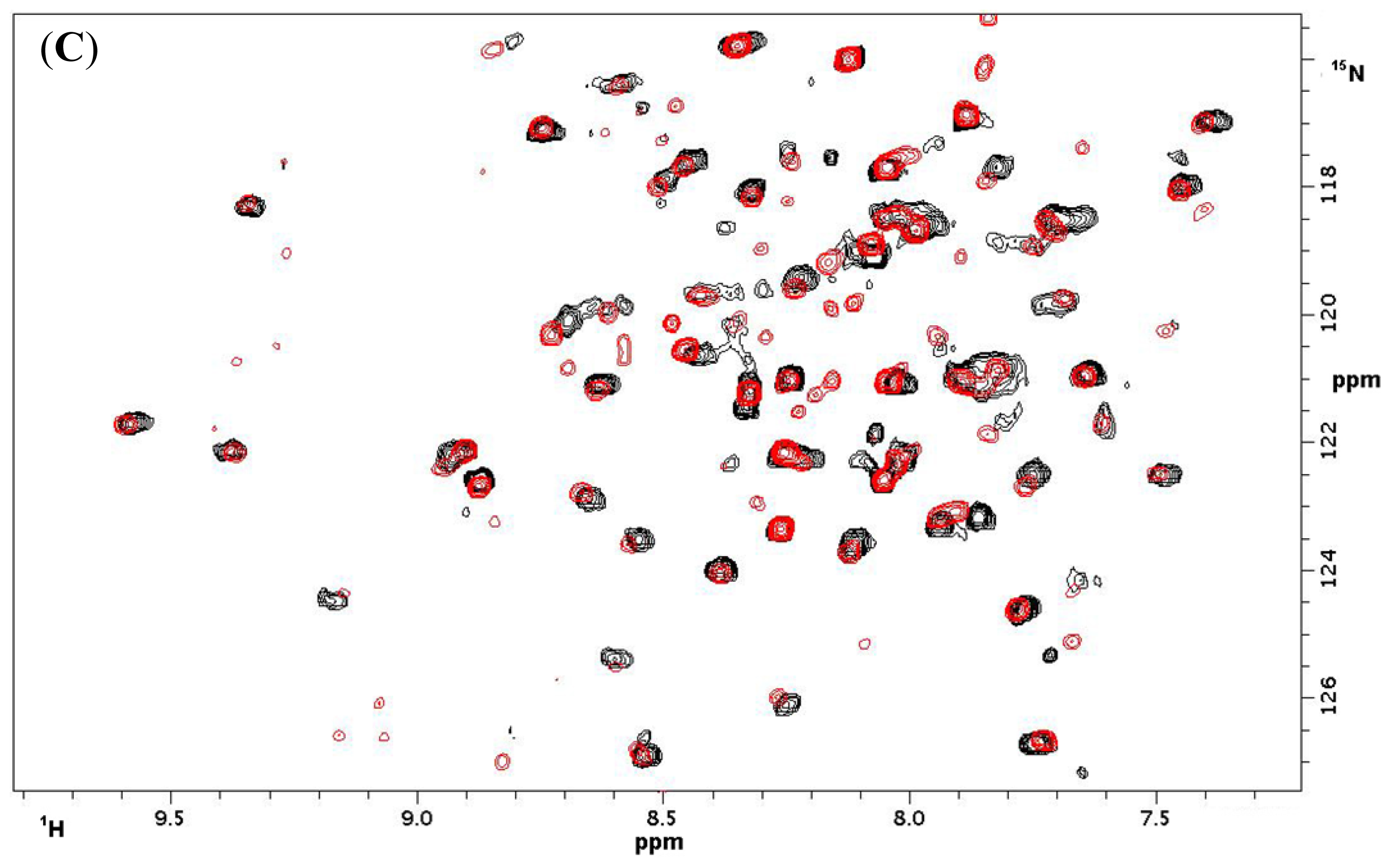
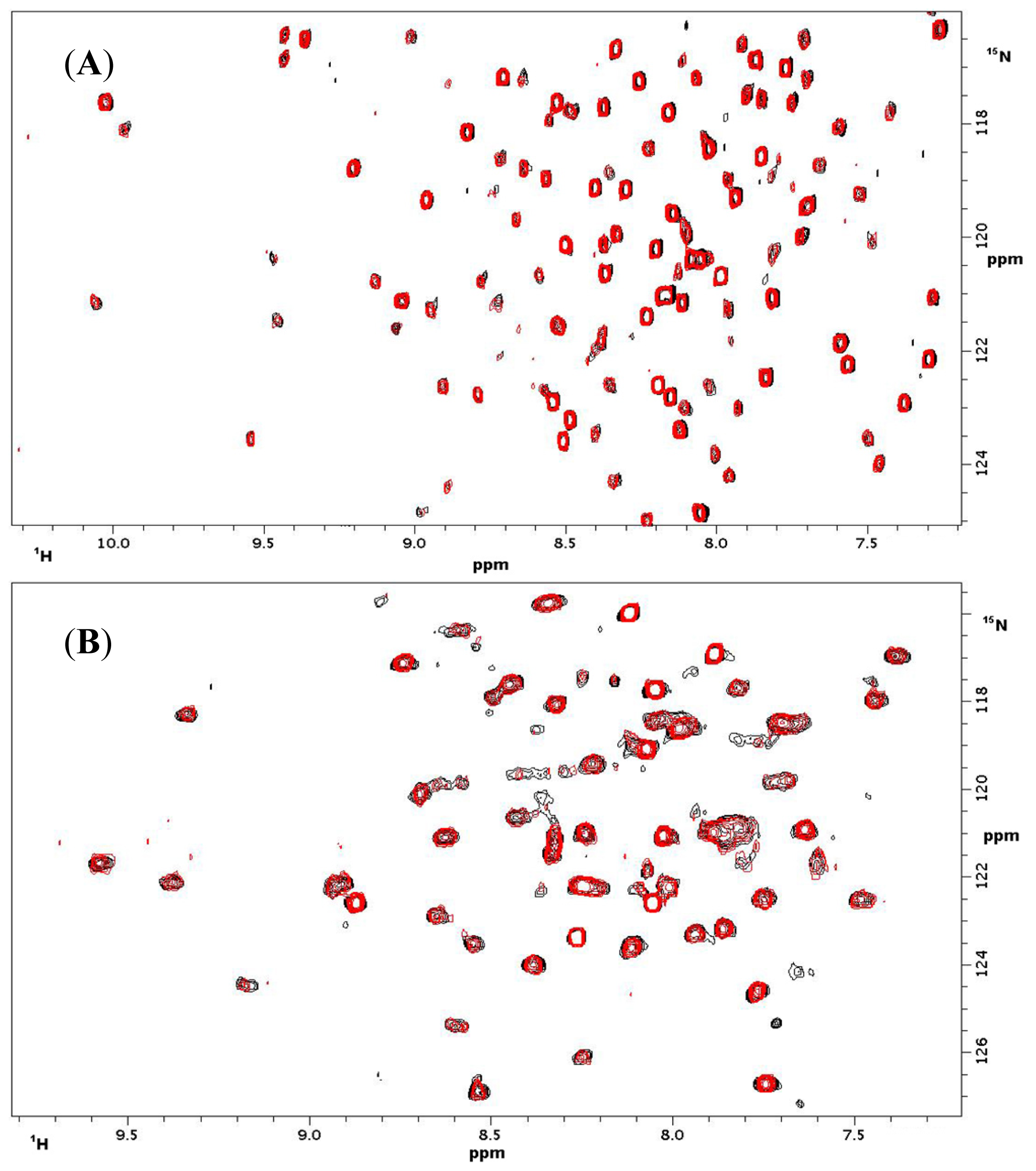
| CD 69 | CD 69 + L | NKR-P1A | NKR-P1A + L | ||||
|---|---|---|---|---|---|---|---|
| 1H (ppm) | 15N (ppm) | 1H (ppm) | 15N (ppm) | 1H (ppm) | 15N (ppm) | 1H (ppm) | 15N (ppm) |
| 9.5772 | 121.684 | 9.5784 | 121.648 | 10.0616 | 121.160 | 10.0579 | 121.1426 |
| 9.3354 | 118.332 | 9.3341 | 118.269 | 9.2078 | 118.775 | 9.2070 | 118.772 |
| 8.5472 | 123.488 | 8.5516 | 123.538 | 7.0786 | 117.172 | 7.0773 | 117.179 |
| 7.7638 | 124.592 | 7.7651 | 124.581 | 8.0078 | 123.829 | 8.0068 | 123.820 |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Rozbeský, D.; Krejzová, J.; Křenek, K.; Prchal, J.; Hrabal, R.; Košek, M.; Weignerová, L.; Fiore, M.; Dumy, P.; Renaudet, O.; et al. Re-Evaluation of Binding Properties of Recombinant Lymphocyte Receptors NKR-P1A and CD69 to Chemically Synthesized Glycans and Peptides. Int. J. Mol. Sci. 2014, 15, 1271-1283. https://doi.org/10.3390/ijms15011271
Rozbeský D, Krejzová J, Křenek K, Prchal J, Hrabal R, Košek M, Weignerová L, Fiore M, Dumy P, Renaudet O, et al. Re-Evaluation of Binding Properties of Recombinant Lymphocyte Receptors NKR-P1A and CD69 to Chemically Synthesized Glycans and Peptides. International Journal of Molecular Sciences. 2014; 15(1):1271-1283. https://doi.org/10.3390/ijms15011271
Chicago/Turabian StyleRozbeský, Daniel, Jana Krejzová, Karel Křenek, Jan Prchal, Richard Hrabal, Milan Košek, Lenka Weignerová, Michele Fiore, Pascal Dumy, Olivier Renaudet, and et al. 2014. "Re-Evaluation of Binding Properties of Recombinant Lymphocyte Receptors NKR-P1A and CD69 to Chemically Synthesized Glycans and Peptides" International Journal of Molecular Sciences 15, no. 1: 1271-1283. https://doi.org/10.3390/ijms15011271
APA StyleRozbeský, D., Krejzová, J., Křenek, K., Prchal, J., Hrabal, R., Košek, M., Weignerová, L., Fiore, M., Dumy, P., Renaudet, O., & Křen, V. (2014). Re-Evaluation of Binding Properties of Recombinant Lymphocyte Receptors NKR-P1A and CD69 to Chemically Synthesized Glycans and Peptides. International Journal of Molecular Sciences, 15(1), 1271-1283. https://doi.org/10.3390/ijms15011271






