The Bioconcentration and Degradation of Nonylphenol and Nonylphenol Polyethoxylates by Chlorella vulgaris
Abstract
:1. Introduction
2. Results and Discussion
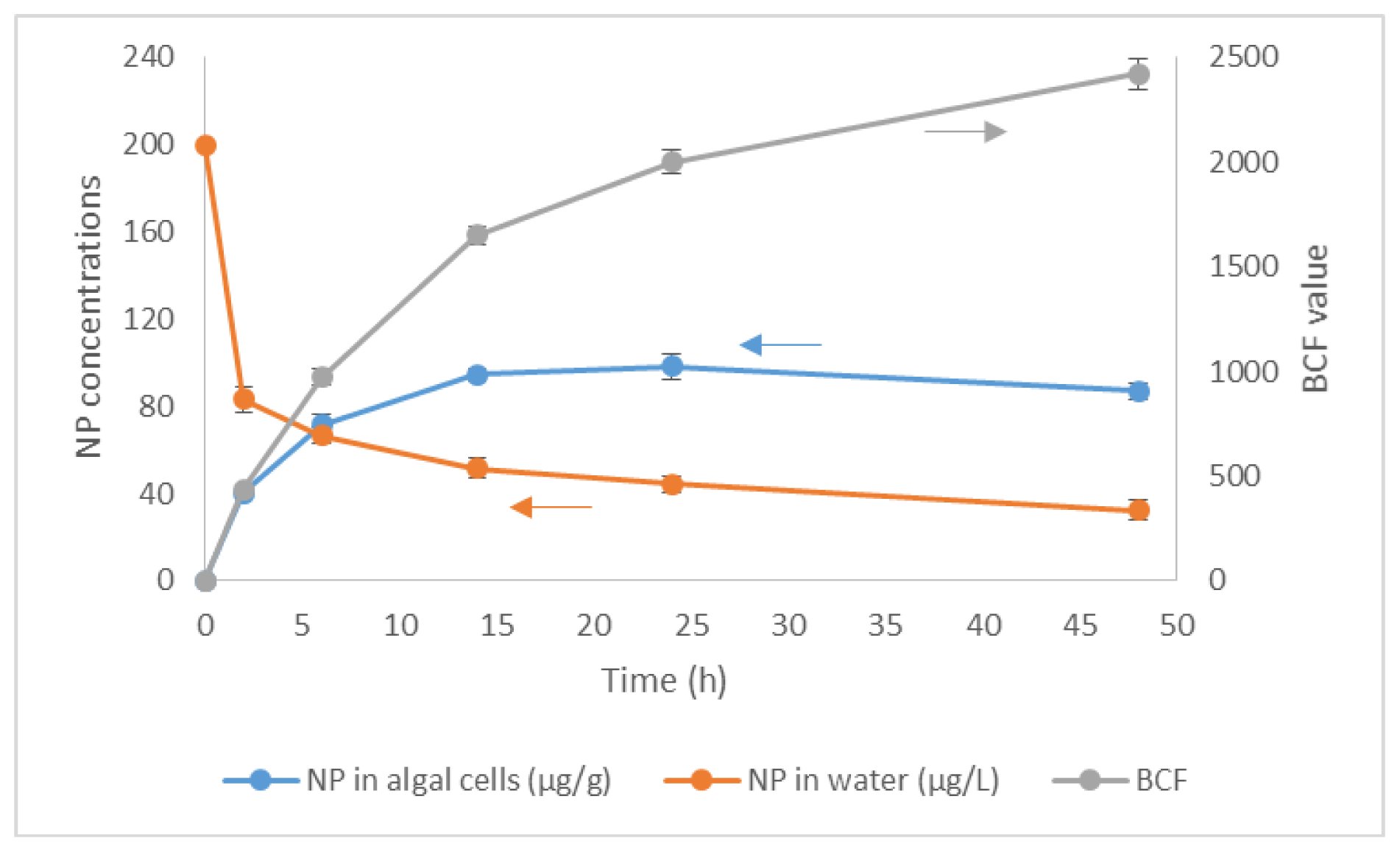
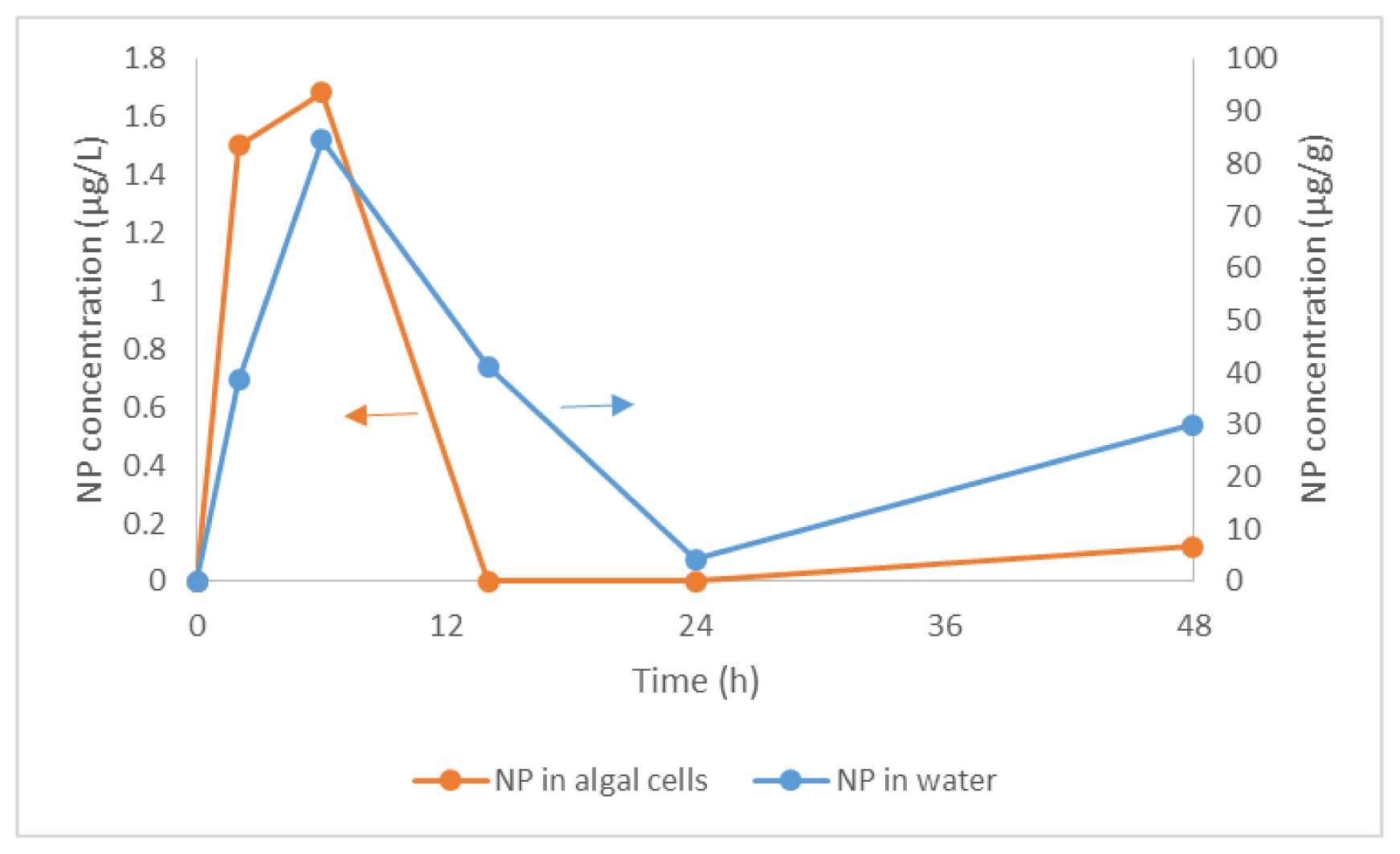
2.1. Bioconcentration and Biodegradation of NP by C. vulgaris
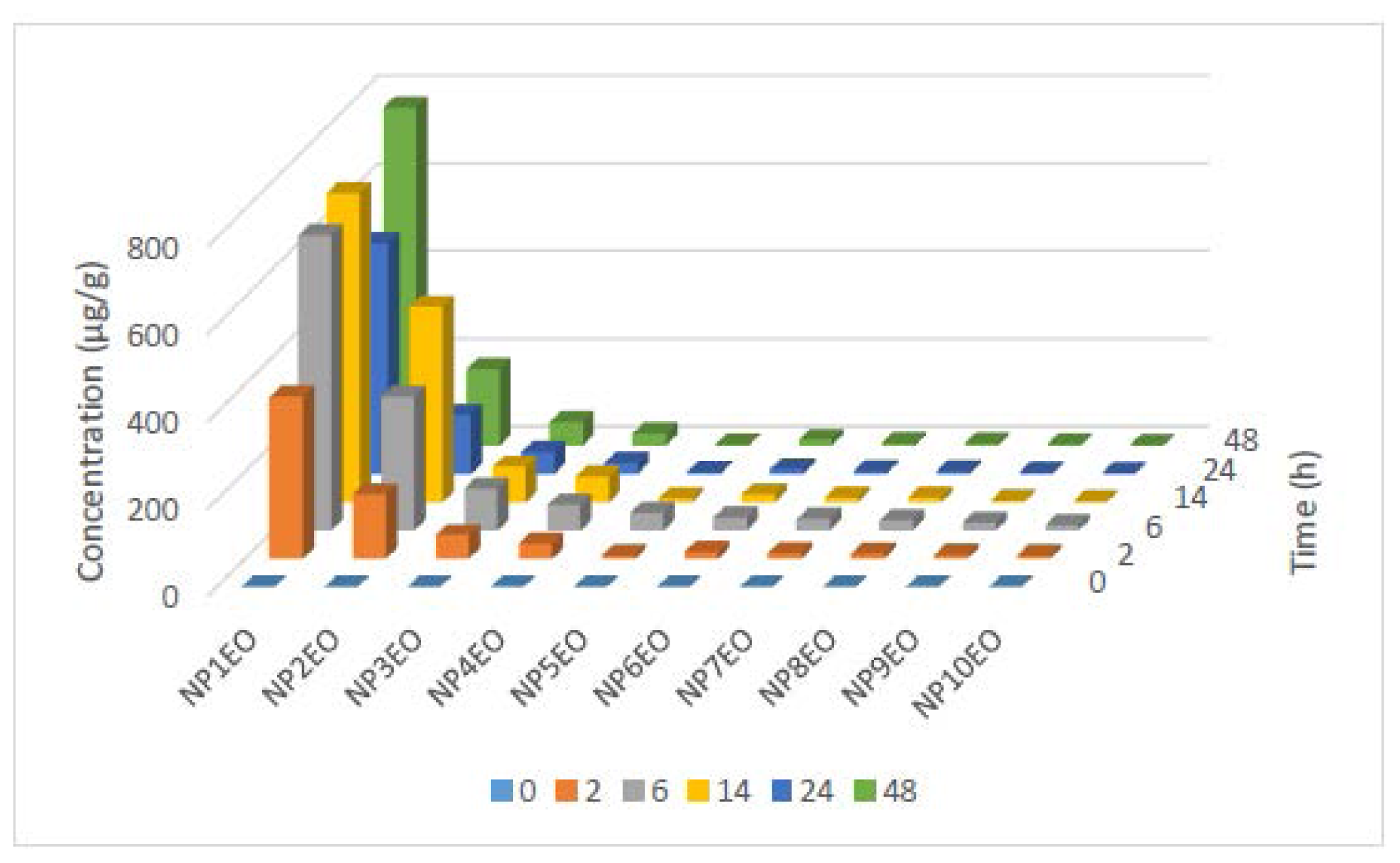
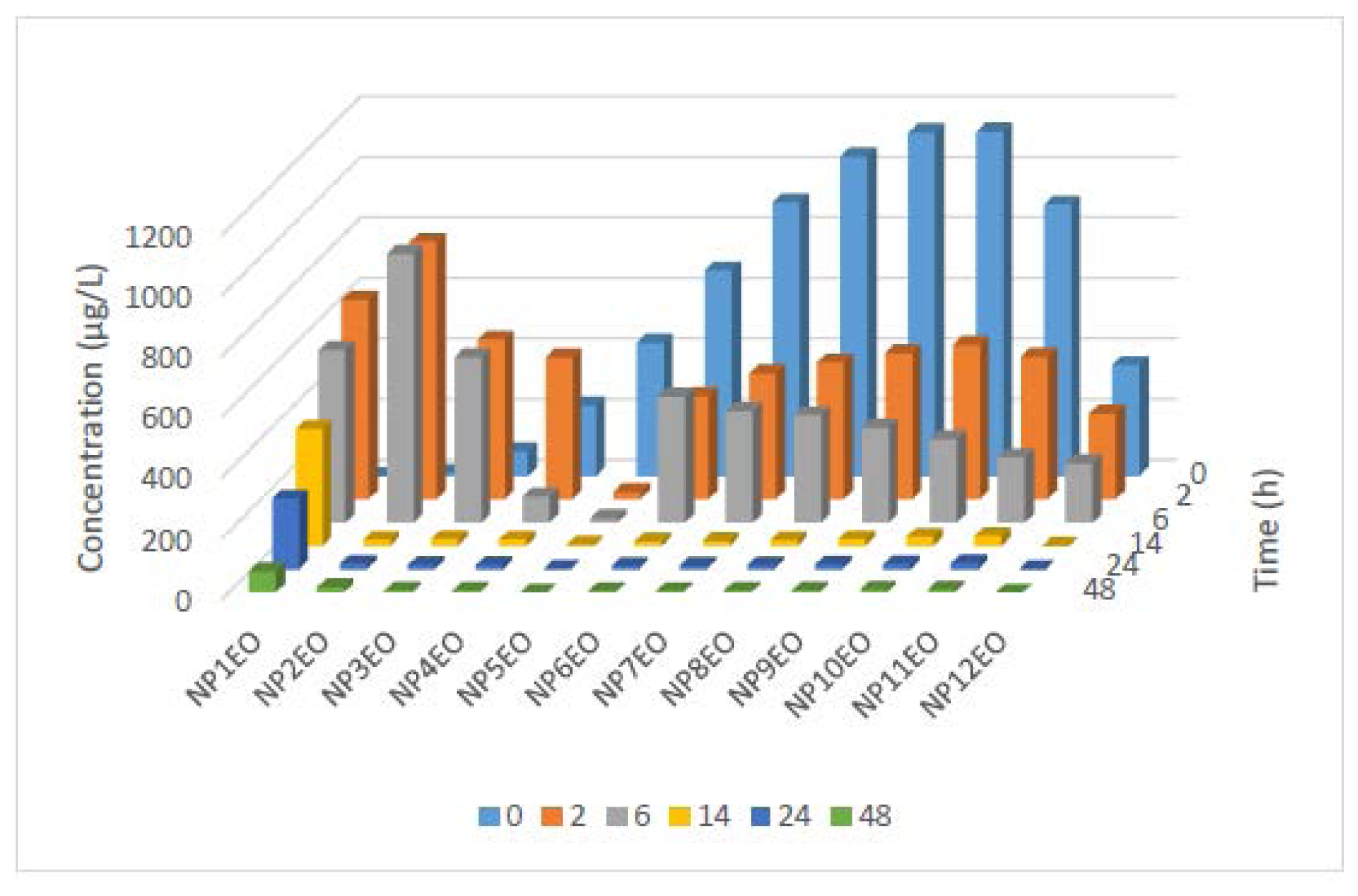
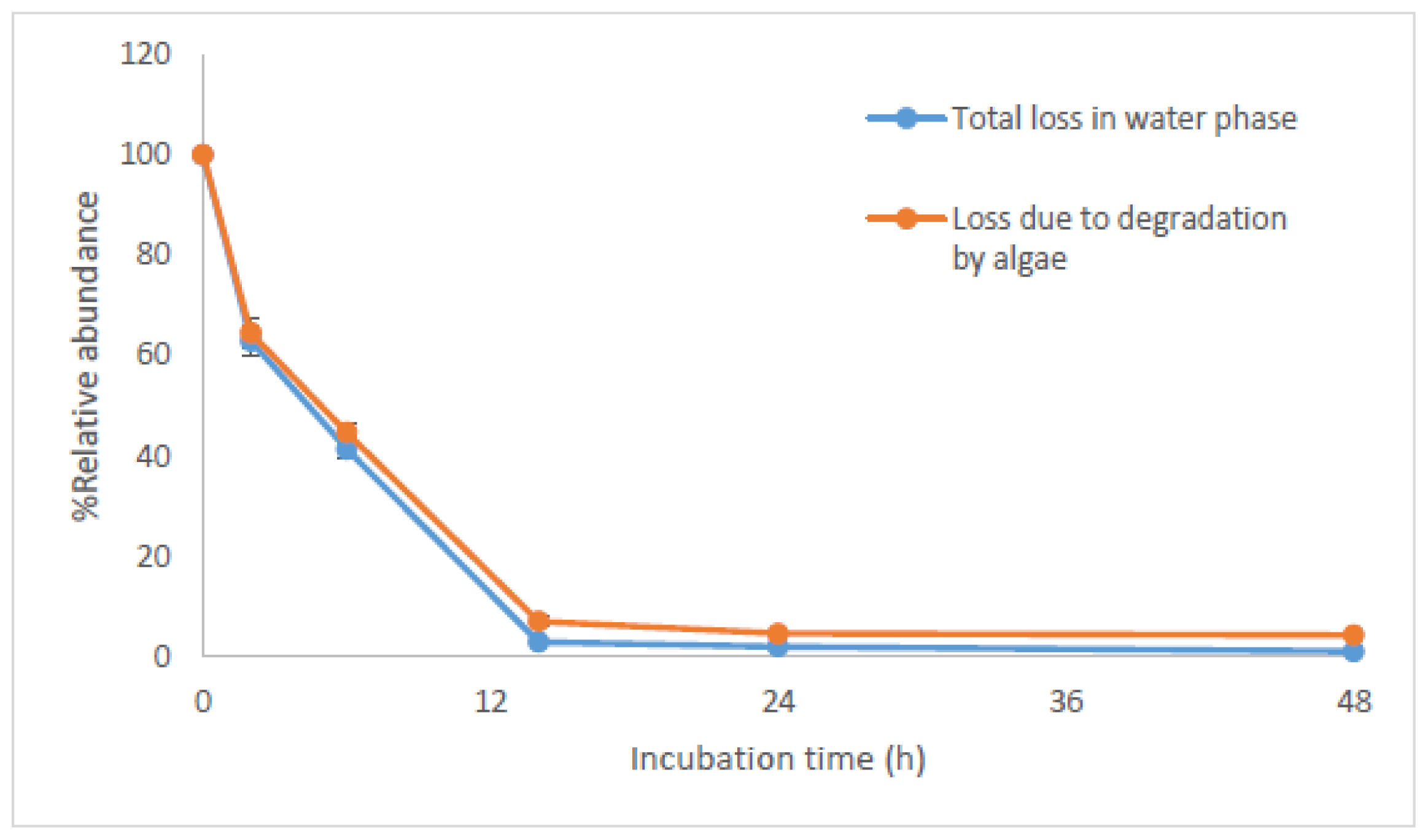
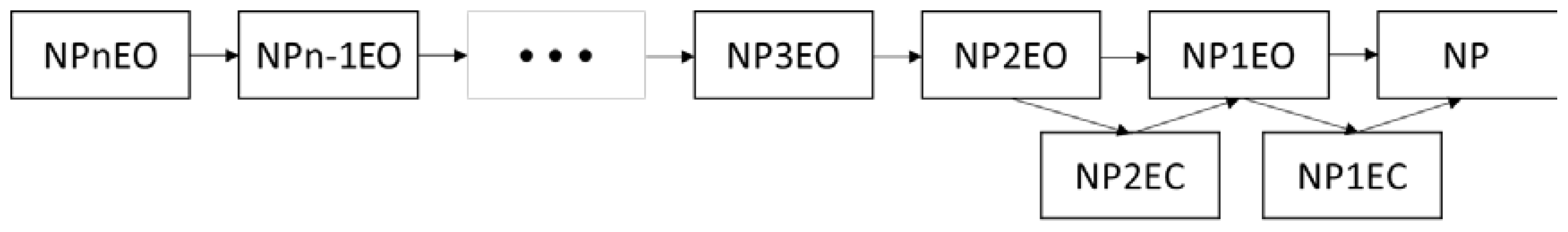
2.2. Bioconcentration and Biodegradation of NPnEOs (n = 1–12) by C. vulgaris
3. Experimental Section
3.1. Chemicals and Algae
3.2. Bioaccumulation and Biodegradation of NP and NPnEOs by C. vulgaris
3.3. Sample Pre-Treatment
3.4. HPLC Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Staples, C.A.; Weeks, J.; Hall, J.F.; Naylor, C.G. Evaluation of aquatic toxicity and bioaccumulation of C8- and C9- alkylphenol ethoxylates. Environ. Toxicol. Chem 1998, 17, 2470–2480. [Google Scholar]
- Bettinetti, R.; Cuccato, D.; Galassi, S.; Provini, A. Toxicity of 4-nonylphenol in spiked sediment to three populations of Chironomus riparius. Chemosphere 2002, 46, 201–207. [Google Scholar]
- Kannan, K.; Keith, T.L.; Naylor, C.G.; Staples, C.A.; Snyder, S.A.; Giesy, J.P. Nonylphenol and nonylphenol ethoxylates in fish, sediment, and water from the Kalamazoo River, Michigan. Arch. Environ. Con. Tox 2003, 44, 77–82. [Google Scholar]
- Ying, G.-G.; Williams, B.; Kookana, R. Environmental fate of alkylphenols and alkylphenol ethoxylates—A review. Environ. Int 2002, 28, 215–226. [Google Scholar]
- Vazquez-Duhalt, R.; Marquez-Rocha, F.; Ponce, E.; Licea, A.F.; Viana, M.T. Nonylphenol, an integrated vision of a pollutant. Appl. Ecol. Env. Res 2005, 4, 1–25. [Google Scholar]
- Soares, A.; Guieysse, B.; Jefferson, B.; Cartmell, E.; Lester, J. Nonylphenol in the environment: A critical review on occurrence, fate, toxicity and treatment in wastewaters. Environ. Int 2008, 34, 1033. [Google Scholar]
- Fytianos, K.; Pegiadou, S.; Raikos, N.; Eleftheriadis, I.; Tsoukali, H. Determination of non-ionic surfactants (polyethoxylated-nonylphenols) by HPLC in waste waters. Chemosphere 1997, 35, 1423–1429. [Google Scholar]
- Ahel, M.; Schaffner, C.; Giger, W. Behaviour of alkylphenol polyethoxylate surfactants in the aquatic environment—III. Occurrence and elimination of their persistent metabolites during infiltration of river water to groundwater. Water Res 1996, 30, 37–46. [Google Scholar]
- Staples, C.A.; Naylor, C.G.; Williams, J.B.; Gledhill, W.E. Ultimate biodegradation of alkylphenol ethoxylate surfactants and their biodegradation intermediates. Environ. Toxicol. Chem 2001, 20, 2450–2455. [Google Scholar]
- Venkatesan, A.K.; Halden, R.U. National inventory of alkylphenol ethoxylate compounds in US sewage sludges and chemical fate in outdoor soil mesocosms. Environ. Pollut 2013, 174, 189–193. [Google Scholar]
- Field, J.A.; Reed, R.L. Nonylphenol polyethoxy carboxylate metabolites of nonionic surfactants in US paper mill effluents, municipal sewage treatment plant effluents, and river waters. Environ. Sci. Technol 1996, 30, 3544–3550. [Google Scholar]
- Potter, T.L.; Simmons, K.; Wu, J.; Sanchez-Olvera, M.; Kostecki, P.; Calabrese, E. Static die-away of a nonylphenol ethoxylate surfactant in estuarine water samples. Environ. Sci. Technol 1999, 33, 113–118. [Google Scholar]
- Lussier, S.M.; Champlin, D.; LiVolsi, J.; Poucher, S.; Pruell, R.J. Acute toxicity of para-nonylphenol to saltwater animals. Environ. Toxicol. Chem 2000, 19, 617–621. [Google Scholar]
- Wang, X.; Han, X.; Hou, Y.; Yao, G.; Wang, Y. Effect of nonylphenol on apoptosis of Sertoli cells in vitro. Bull. Environ. Contam. Tox. 2003, 70, 0898–0904. [Google Scholar]
- Correa-Reyes, G.; Viana, M.T.; Marquez-Rocha, F.J.; Licea, A.F.; Ponce, E.; Vazquez-Duhalt, R. Nonylphenol algal bioaccumulation and its effect through the trophic chain. Chemosphere 2007, 68, 662–670. [Google Scholar]
- Teneyck, M.C.; Markee, T.P. Toxicity of nonylphenol, nonylphenol monoethoxylate, and nonylphenol diethoxylate and mixtures of these compounds to Pimephales promelas (fathead minnow) and Ceriodaphnia dubia. Arch. Environ. Con. Tox 2007, 53, 599–606. [Google Scholar]
- Lee, P.-C.; Lee, W. In vivo estrogenic action of nonylphenol in immature female rats. Bull. Environ. Contam. Tox 1996, 57, 341–348. [Google Scholar]
- Goto, R.; Kubota, T.; Ibuki, Y.; Kaji, K.; Goto, A. Degradation of nonylphenol polyethoxylates by ultraviolet B irradiation and effects of their products on mammalian cultured cells. Toxicology 2004, 202, 237–247. [Google Scholar]
- Zumbado, M.; Boada, L.D.; Torres, S.; Monterde, J.G.; Díaz-Chico, B.N.; Afonso, J.L.; Cabrera, J.J.; Blanco, A. Evaluation of acute hepatotoxic effects exerted by environmental estrogens nonylphenol and 4-octylphenol in immature male rats. Toxicology 2002, 175, 49–62. [Google Scholar]
- Nair-Menon, J.U.; Campbell, G.T.; Blake, C.A. Toxic effects of octylphenol on cultured rat and murine splenocytes. Toxicol. Appl. Pharm 1996, 139, 437–444. [Google Scholar]
- European Commission, Directive 2003/53/EC of the European Parliament and of the Council of 18 June 2003 amending for the 26th time Council Directive 76/769/EEC relating to restrictions on the marketing and use of certain dangerous substances and preparations (nonylphenol, nonylphenol ethoxylate and cement). In Official Journal of the European Union; Publications Office of the European Union: Luxembourg City, Luxembourg, 2003; Volume 178, pp. 24–178.
- Nonylphenol (NP) and Nonylphenol Ethoxylates (NPEs) Action Plan. Available online: http://www.epa.gov/oppt/existingchemicals/pubs/actionplans/RIN2070-ZA09_NP-NPEs%20Action%20Plan_Final_2010-08-09.pdf (accessed on 8 September 2010).
- Stachel, B.; Ehrhorn, U.; Heemken, O.-P.; Lepom, P.; Reincke, H.; Sawal, G.; Theobald, N. Xenoestrogens in the River Elbe and its tributaries. Environ. Pollut 2003, 124, 497–507. [Google Scholar]
- Tsuda, T.; Suga, K.; Kaneda, E.; Ohsuga, M. 4-Nonylphenol, 4-nonylphenol mono-and diethoxylates, and other 4-alkylphenols in water and shellfish from rivers flowing into Lake Biwa. Bull. Environ. Contam. Tox 2002, 68, 126–131. [Google Scholar]
- Keith, T.L.; Snyder, S.A.; Naylor, C.G.; Staples, C.A.; Summer, C.; Kannan, K.; Giesy, J.P. Identification and quantitation of nonylphenol ethoxylates and nonylphenol in fish tissues from Michigan. Environ. Sci. Technol 2001, 35, 10–13. [Google Scholar]
- Ferrara, F.; Fabietti, F.; Delise, M.; Bocca, A.P.; Funari, E. Alkylphenolic compounds in edible molluscs of the Adriatic Sea (Italy). Environ. Sci. Technol 2001, 35, 3109–3112. [Google Scholar]
- Jin, X.; Jiang, G.; Huang, G.; Liu, J.; Zhou, Q. Determination of 4-tert-octylphenol, 4-nonylphenol and bisphenol A in surface waters from the Haihe River in Tianjin by gas chromatography–mass spectrometry with selected ion monitoring. Chemosphere 2004, 56, 1113–1119. [Google Scholar]
- Zgoła-Grześkowiak, A. Dispersive liquid–liquid microextraction applied to isolation and concentration of alkylphenols and their short-chained ethoxylates in water samples. J. Chromatogr. A 2010, 1217, 1761–1766. [Google Scholar]
- Mao, Z.; Zheng, X.; Zhang, Y.; Tao, X.; Li, Y.; Wang, W. Occurrence and biodegradation of nonylphenol in the environment. Int. J. Mol. Sci 2012, 13, 491–505. [Google Scholar]
- Xu, J.; Wang, P.; Guo, W.; Dong, J.; Wang, L.; Dai, S. Seasonal and spatial distribution of nonylphenol in Lanzhou Reach of Yellow River in China. Chemosphere 2006, 65, 1445–1451. [Google Scholar]
- Vega Morales, T.; Torres Padrón, M.; Sosa Ferrera, Z.; Santana Rodríguez, J. Determination of alkylphenol ethoxylates and their degradation products in liquid and solid samples. TrAC-Trend. Anal. Chem 2009, 28, 1186–1200. [Google Scholar]
- Solé, M.; de Alda, M.J.L.; Castillo, M.; Porte, C.; Ladegaard-Pedersen, K.; Barceló, D. Estrogenicity determination in sewage treatment plants and surface waters from the Catalonian area (NE Spain). Environ. Sci. Technol 2000, 34, 5076–5083. [Google Scholar]
- Nakada, N.; Tanishima, T.; Shinohara, H.; Kiri, K.; Takada, H. Pharmaceutical chemicals and endocrine disrupters in municipal wastewater in Tokyo and their removal during activated sludge treatment. Water Res 2006, 40, 3297–3303. [Google Scholar]
- Klecka, G.M.; Naylor, C.G.; Staples, C.A.; Losey, B. Occurrence of nonylphenol ethoxylates and their metabolites in municipal wastewater treatment plants and receiving waters. Water Environ. Res 2010, 82, 447–454. [Google Scholar]
- Fan, Z.; Hu, J.; An, W.; Yang, M. Detection and occurrence of chlorinated byproducts of bisphenol a, nonylphenol, and estrogens in drinking water of china: Comparison to the parent compounds. Environ. Sci. Technol 2013, 47, 10841–10850. [Google Scholar]
- Spehar, R.L.; Brooke, L.T.; Markee, T.P.; Kahl, M.D. Comparative toxicity and bioconcentration of nonylphenol in freshwater organisms. Environ. Toxicol. Chem 2010, 29, 2104–2111. [Google Scholar]
- Liu, Y.; Tam, N.F.; Guan, Y.; Yasojima, M.; Zhou, J.; Gao, B. Acute toxicity of nonylphenols and bisphenol A to the embryonic development of the abalone Haliotis diversicolor supertexta. Ecotoxicology 2011, 20, 1233–1245. [Google Scholar]
- Preuss, T.G.; Telscher, M.; Ratte, H.T. Life stage-dependent bioconcentration of a nonylphenol isomer in Daphnia magna. Environ. Pollut 2008, 156, 1211–1217. [Google Scholar]
- Shan, J.; Wang, T.; Li, C.; Klumpp, E.; Ji, R. Bioaccumulation and bound-residue formation of a branched 4-nonylphenol isomer in the geophagous earthworm Metaphire guillelmi in a rice paddy soil. Environ. Sci. Technol 2010, 44, 4558–4563. [Google Scholar]
- Riva, C.; Porte, C.; Binelli, A.; Provini, A. Evaluation of 4-nonylphenol in vivo exposure in Dreissena polymorpha: Bioaccumulation, steroid levels and oxidative stress. Comp. Biochem. Phys. C 2010, 152, 175–181. [Google Scholar]
- Yuan, H.X.; Xu, X.; Sima, Y.H.; Xu, S.Q. Reproductive toxicity effects of 4-nonylphenol with known endocrine disrupting effects and induction of vitellogenin gene expression in silkworm Bombyx mori. Chemosphere 2013, 93, 263–268. [Google Scholar]
- Yuan, S.; Yu, C.; Chang, B. Biodegradation of nonylphenol in river sediment. Environ. Pollut 2004, 127, 425–430. [Google Scholar]
- Chang, B.; Yu, C.; Yuan, S. Degradation of nonylphenol by anaerobic microorganisms from river sediment. Chemosphere 2004, 55, 493–500. [Google Scholar]
- Ahel, M.; McEvoy, J.; Giger, W. Bioaccumulation of the lipophilic metabolites of nonionic surfactants in freshwater organisms. Environ. Pollut 1993, 79, 243–248. [Google Scholar]
- Routledge, E.J.; Sumpter, J.P. Estrogenic activity of surfactants and some of their degradation products assessed using a recombinant yeast screen. Environ. Toxicol. Chem 1996, 15, 241–248. [Google Scholar]
- Sun, H.; Gu, X. Comprehensive toxicity study of nonylphenol and short-chain nonylphenol polyethoxylates on Daphnia magna. Bull. Environ. Contam. Tox 2005, 75, 677–683. [Google Scholar]
- Gao, Q.; Tam, N. Growth, photosynthesis and antioxidant responses of two microalgal species, Chlorella vulgaris and Selenastrum capricornutum, to nonylphenol stress. Chemosphere 2011, 82, 346–354. [Google Scholar]
- Kumaran, S.S.; Kavitha, C.; Ramesh, M.; Grummt, T. Toxicity studies of nonylphenol and octylphenol: hormonal, hematological and biochemical effects in Clarias gariepinus. J. Appl. Toxicol 2011, 31, 752–761. [Google Scholar]
- Semple, K.T.; Cain, R.B.; Schmidt, S. Biodegradation of aromatic compounds by microalgae. Fems Microbiol. Lett 1999, 170, 291–300. [Google Scholar]
- Guolan, H.; Hongwen, S.; Li, C.L. Study on the physiology and degradation of dye with immobilized algae. Artif. Cell. Blood Sub 2000, 28, 347–363. [Google Scholar]
- Huang, G.; Sun, H.; Song, Z. Interactions between dibutyl phthalate and aquatic organisms. Bull. Environ. Contam. Tox 1999, 63, 759–765. [Google Scholar]
- Meylan, W.M.; Howard, P.H.; Boethling, R.S.; Aronson, D.; Printup, H.; Gouchie, S. Improved method for estimating bioconcentration/bioaccumulation factor from octanol/water partition coefficient. Environ. Toxicol. Chem 1999, 18, 664–672. [Google Scholar]
- Schwarzenbach, R.P.; Gschwend, P.M.; Imboden, D.M. Environmental organic chemistry, 2nd ed; John Wiley & Sons, Inc: Hoboken, NJ, USA, 2005; p. 599. [Google Scholar]
- Snyder, S.A.; Keith, T.L.; Pierens, S.L.; Snyder, E.M.; Giesy, J.P. Bioconcentration of nonylphenol in fathead minnows (Pimephales promelas). Chemosphere 2001, 44, 1697–1702. [Google Scholar]
- Ekelund, R.; Bergman, A.; Granmo, A.; Berggren, M. Bioaccumulation of 4-nonylphenol in marine animals—A re-evaluation. Environ. Pollut 1990, 64, 107–120. [Google Scholar]
- Chang, B.; Chiang, F.; Yuan, S. Anaerobic degradation of nonylphenol in sludge. Chemosphere 2005, 59, 1415–1420. [Google Scholar]
- Chang, B.; Lu, Z.; Yuan, S. Anaerobic degradation of nonylphenol in subtropical mangrove sediments. J. Hazard. Mater 2009, 165, 162–167. [Google Scholar]
- Gabriel, F.L.; Heidlberger, A.; Rentsch, D.; Giger, W.; Guenther, K.; Kohler, H.-P.E. A novel metabolic pathway for degradation of 4-nonylphenol environmental contaminants by Sphingomonas xenophaga Bayram ipso-hydroxylation and intramolecular rearrangement. J. Biol. Chem 2005, 280, 15526–15533. [Google Scholar]
- Huang, K.; Shigeta, K.; Kunieda, H. Phase behavior of polyoxyethylene dodecyl ether-water systems. In Trends in Colloid and Interface Science XII; Steinkopff: Dresden, Germany, 1998; Volume 110, pp. 171–174. [Google Scholar]
- Kunieda, H.; Umizu, G.; Aramaki, K. Effect of mixing oils on the hexagonal liquid crystalline structures. J. Phys. Chem. B 2000, 104, 2005–2011. [Google Scholar]
- Kunieda, H.; Shigeta, K.; Ozawa, K.; Suzuki, M. Self-organizing structures in poly (oxyethylene) oleyl ether-water system. J. Phys. Chem. B 1997, 101, 7952–7957. [Google Scholar]
- Jonkers, N.; Knepper, T.P.; De Voogt, P. Aerobic biodegradation studies of nonylphenol ethoxylates in river water using liquid chromatography-electrospray tandem mass spectrometry. Environ. Sci. Technol 2001, 35, 335–340. [Google Scholar]
- Ahel, M.; Giger, W.; Koch, M. Behaviour of alkylphenol polyethoxylate surfactants in the aquatic environment—I. Occurrence and transformation in sewage treatment. Water Res 1994, 28, 1131–1142. [Google Scholar]
- Karahan, Ö.; Olmez-Hanci, T.; Arslan-Alaton, I.; Orhon, D. Modelling biodegradation of nonylphenol ethoxylate in acclimated and non-acclimated microbial cultures. Bioresour. Technol 2010, 101, 8058–8066. [Google Scholar]
- Nichols, H.; Bold, H. Growth media—Freshwater. In Handbook of Physiological Methods; Stein, J.R., Ed.; Cambridge University Press: London, UK, 1973; pp. 7–24. [Google Scholar]
- Hou, S.; Sun, H.; Gao, Y. Sorption of small metabolites of nonylphenol polyethoxylates in single and complex systems on aquatic suspended particulate matter. Chemosphere 2006, 63, 31–38. [Google Scholar]


| Homologue | BCF at different time (h) | ||||
|---|---|---|---|---|---|
| 2 | 6 | 14 | 24 | 48 | |
| NP1EO | 564 | 1186 | 1857 | 2271 | 11,028 |
| NP2EO | 174 | 345 | 22,350 | 7500 | 10,294 |
| NP3EO | 103 | 176 | 3727 | 3571 | 6750 |
| NP4EO | 75 | 682 | 2950 | 1821 | 4000 |
| NP5EO | 343 | 2783 | 2204 | 3357 | 7065 |
| NP6EO | 49 | 72 | 1200 | 1100 | 2416 |
| NP7EO | 32 | 71 | 725 | 585 | 1270 |
| NP8EO | 27 | 65 | 579 | 485 | 874 |
| NP9EO | 19 | 61 | 204 | 232 | 459 |
| NP10EO | 15 | 48 | 86 | 136 | 171 |
| Total NPnEOs | 155 | 448 | 6563 | 5406 | 13,831 |
| Homologue | Removal rate of NPnEO homologue in water phase (%) | Degradation rate of NPnEO homologue in algal culture (%) |
|---|---|---|
| NP1EO | −1356 * | - |
| NP2EO | −1.59 * | - |
| NP3EO | 89.6 | 88.0 |
| NP4EO | 96.9 | 97.7 |
| NP5EO | 99.9 | 88.7 |
| NP6EO | 99.1 | 98.6 |
| NP7EO | 99.4 | 99.0 |
| NP8EO | 99.3 | 98.8 |
| NP9EO | 99.3 | 98.8 |
| NP10EO | 99.1 | 98.6 |
| NP11EO | 98.8 | 98.7 |
| NP12EO | 100 | 100 |
| Time (min) | n-hexane/isopropanol (98/2, v/v) | isopropanol/water (98/2, v/v) |
|---|---|---|
| 0 | 5 | 95 |
| 30 | 50 | 50 |
| 35 | 70 | 30 |
| 40 | 65 | 35 |
| 45 | 50 | 50 |
| 55 | 5 | 95 |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Sun, H.-W.; Hu, H.-W.; Wang, L.; Yang, Y.; Huang, G.-L. The Bioconcentration and Degradation of Nonylphenol and Nonylphenol Polyethoxylates by Chlorella vulgaris. Int. J. Mol. Sci. 2014, 15, 1255-1270. https://doi.org/10.3390/ijms15011255
Sun H-W, Hu H-W, Wang L, Yang Y, Huang G-L. The Bioconcentration and Degradation of Nonylphenol and Nonylphenol Polyethoxylates by Chlorella vulgaris. International Journal of Molecular Sciences. 2014; 15(1):1255-1270. https://doi.org/10.3390/ijms15011255
Chicago/Turabian StyleSun, Hong-Wen, Hong-Wei Hu, Lei Wang, Ying Yang, and Guo-Lan Huang. 2014. "The Bioconcentration and Degradation of Nonylphenol and Nonylphenol Polyethoxylates by Chlorella vulgaris" International Journal of Molecular Sciences 15, no. 1: 1255-1270. https://doi.org/10.3390/ijms15011255
APA StyleSun, H.-W., Hu, H.-W., Wang, L., Yang, Y., & Huang, G.-L. (2014). The Bioconcentration and Degradation of Nonylphenol and Nonylphenol Polyethoxylates by Chlorella vulgaris. International Journal of Molecular Sciences, 15(1), 1255-1270. https://doi.org/10.3390/ijms15011255






