Synthesis and Characterization of β-Cyclodextrin Functionalized Ionic Liquid Polymer as a Macroporous Material for the Removal of Phenols and As(V)
Abstract
:1. Introduction
2. Results and Discussion
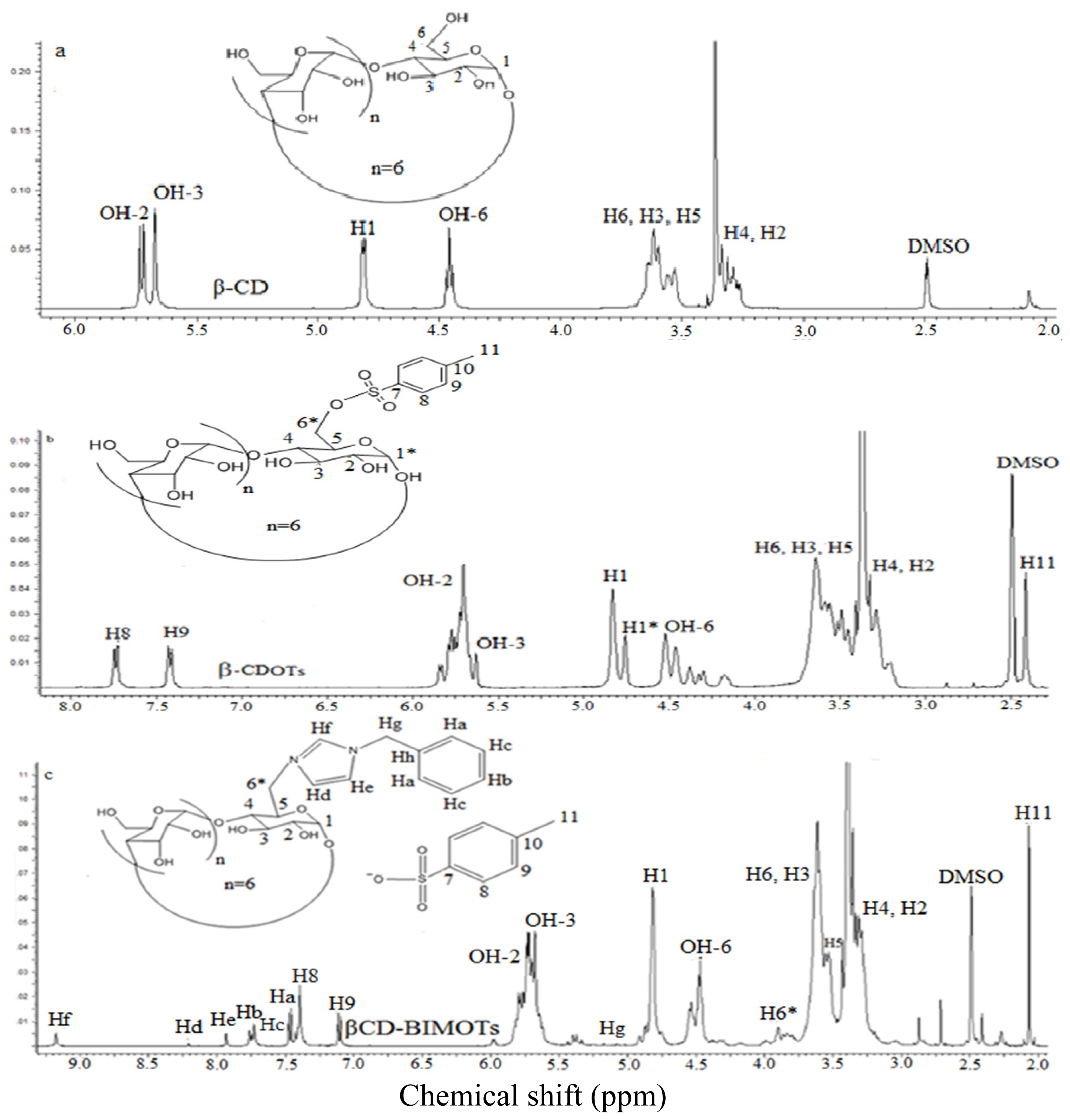
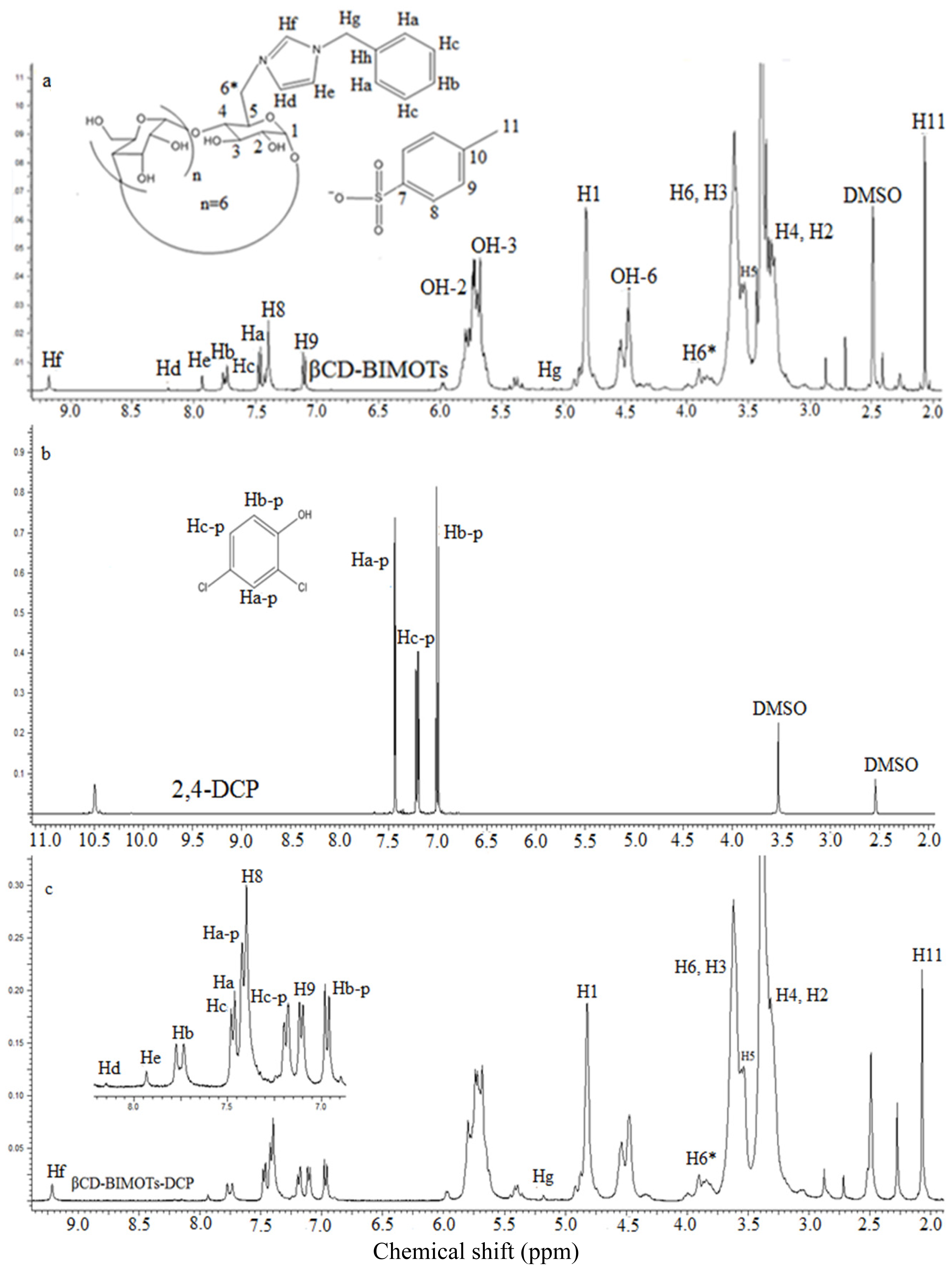
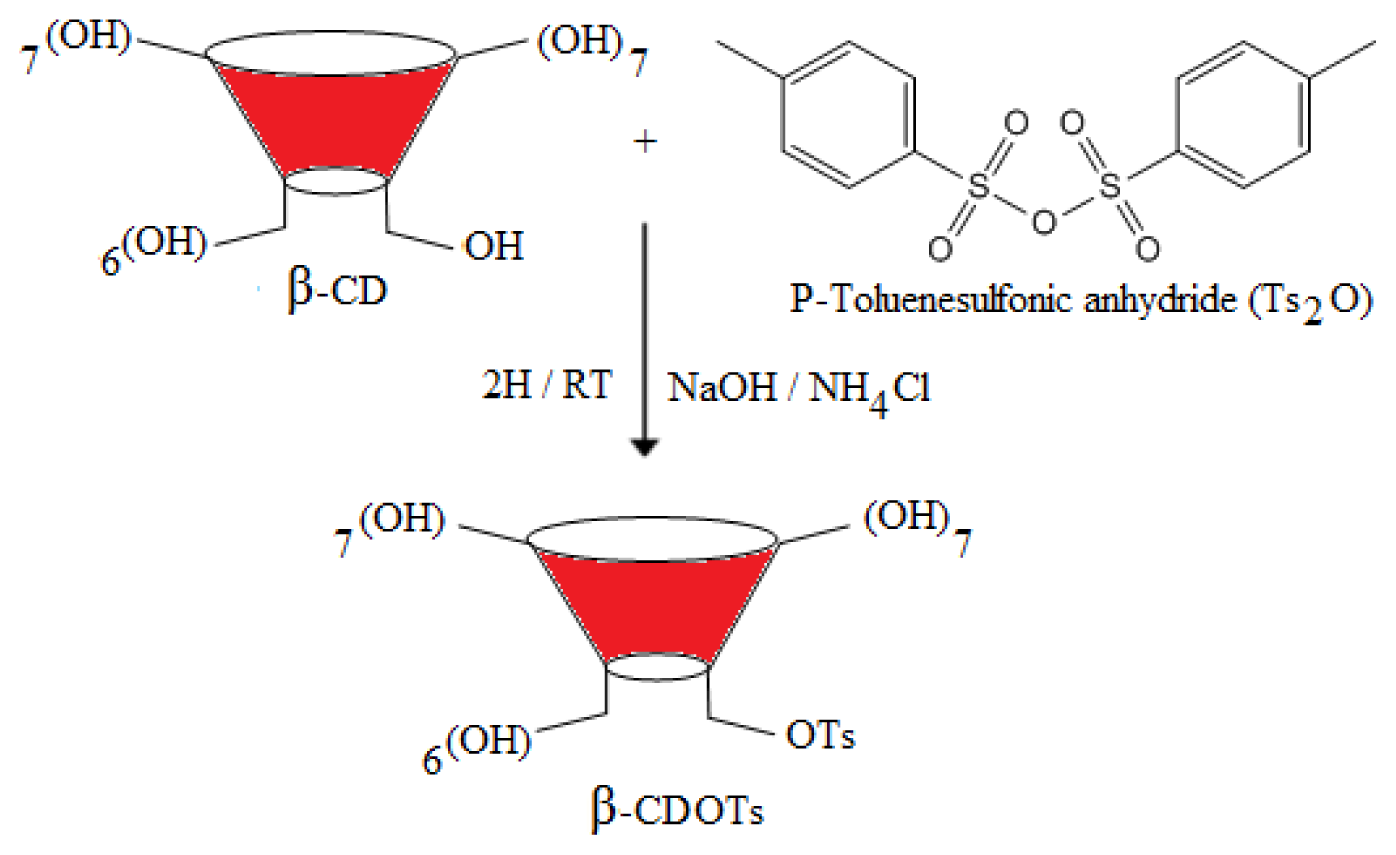
2.1. Preparation of 6-O-Monotosyl-6-deoxy-β-cyclodextrin (β-CDOTs) (1) and Structural Characterization by 1H NMR
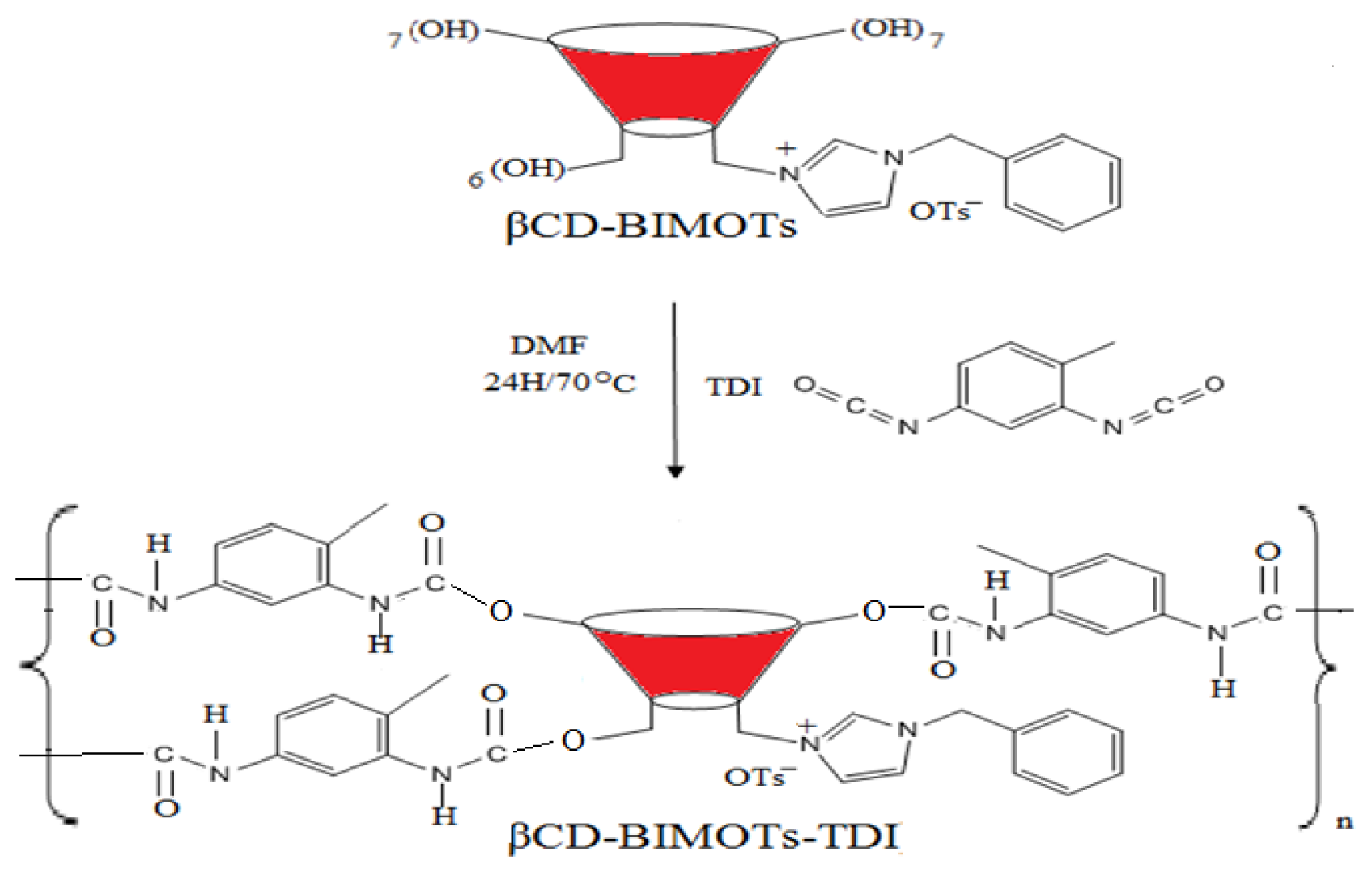
2.2. Preparation of Mono-6-deoxy-6-(3-benzylimidazolium)-β-cyclodextrin (βCD-BIMOTs) (2) and Structural Characterization by 1H NMR
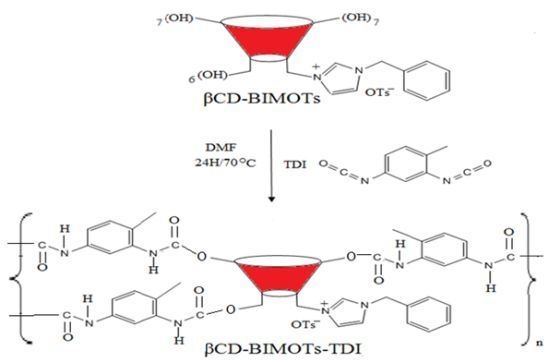
2.3. Preparation of Insoluble β-Cyclodextrin Polymers
2.4. FT-IR Analysis
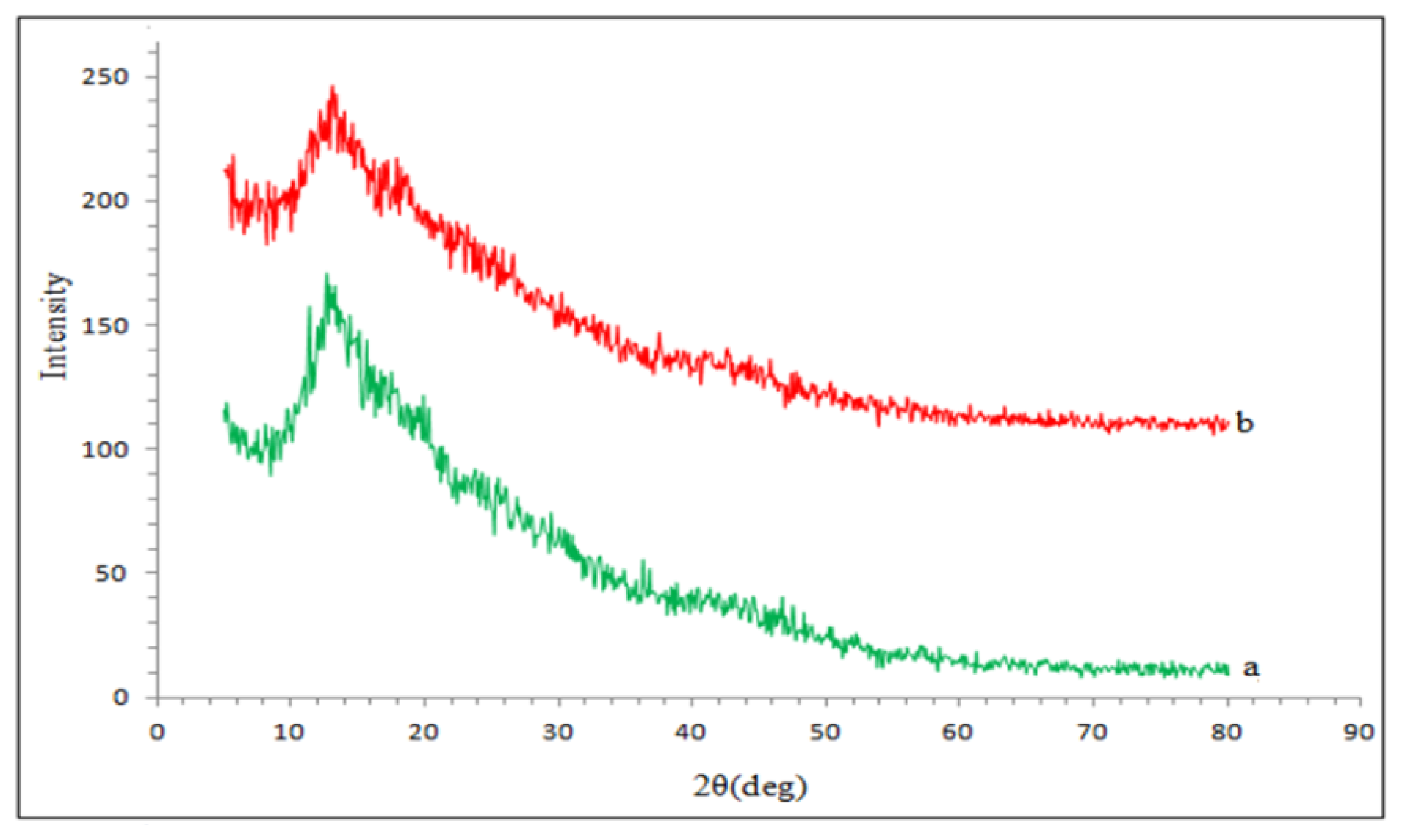
2.5. X-ray Diffraction (XRD) Analysis
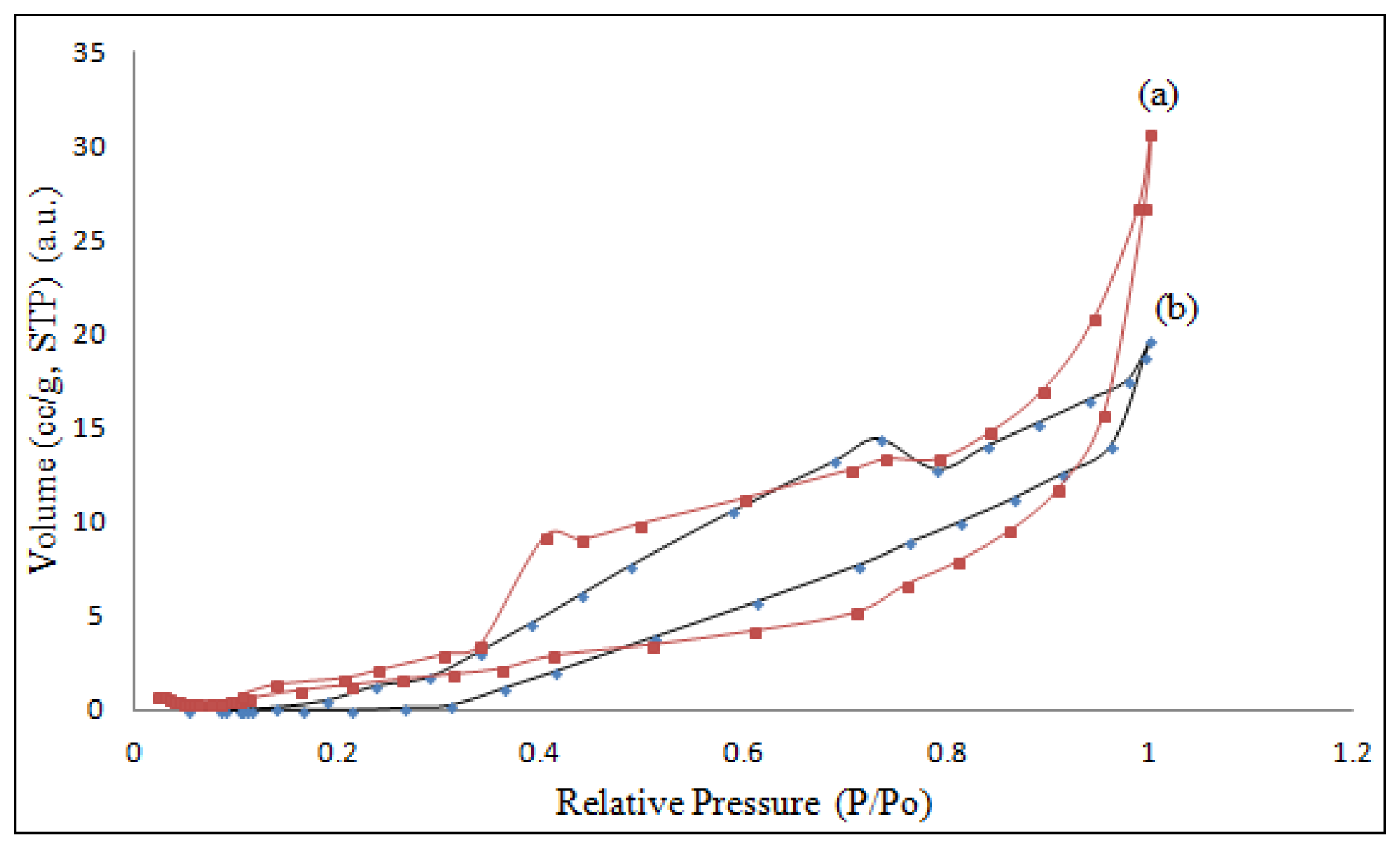
2.6. Surface Morphology Studies
2.7. Scanning Electron Microscope (SEM) Analysis
2.8. Thermal Analysis of the Polymers
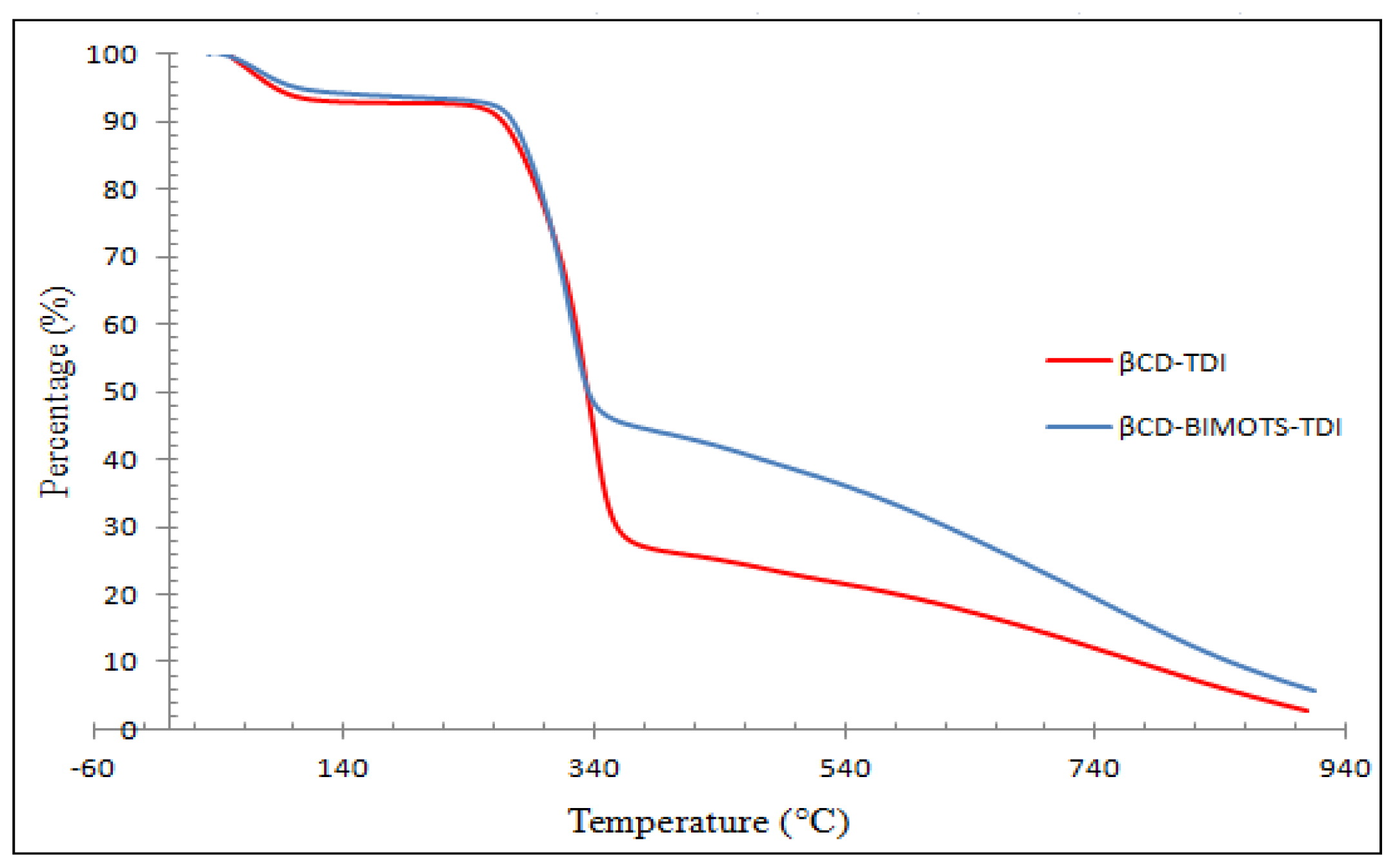
2.8.1. Thermo Gravimetric Analyses (TGA)
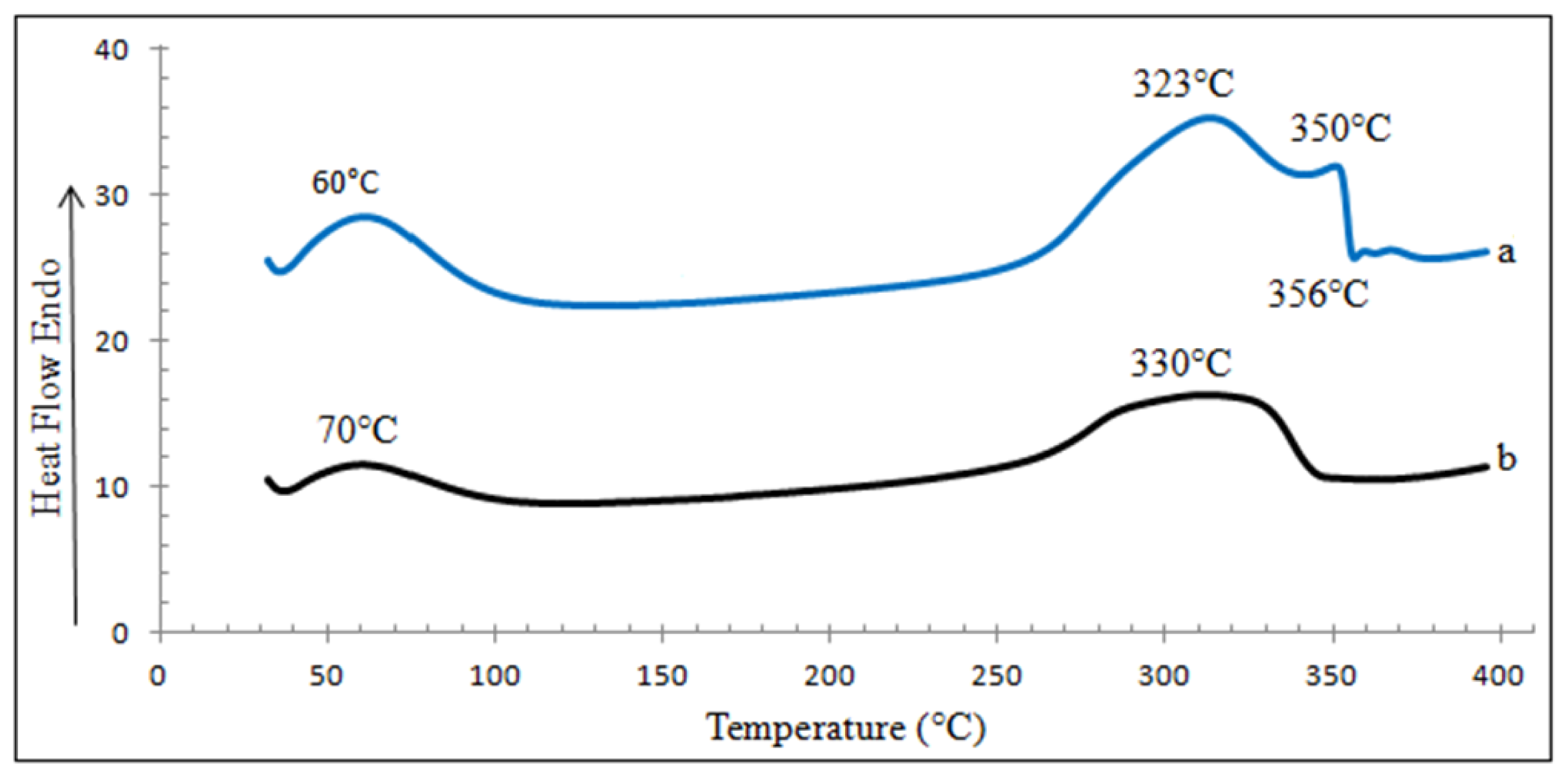
2.8.2. Differential Scanning Calorimetry (DSC)
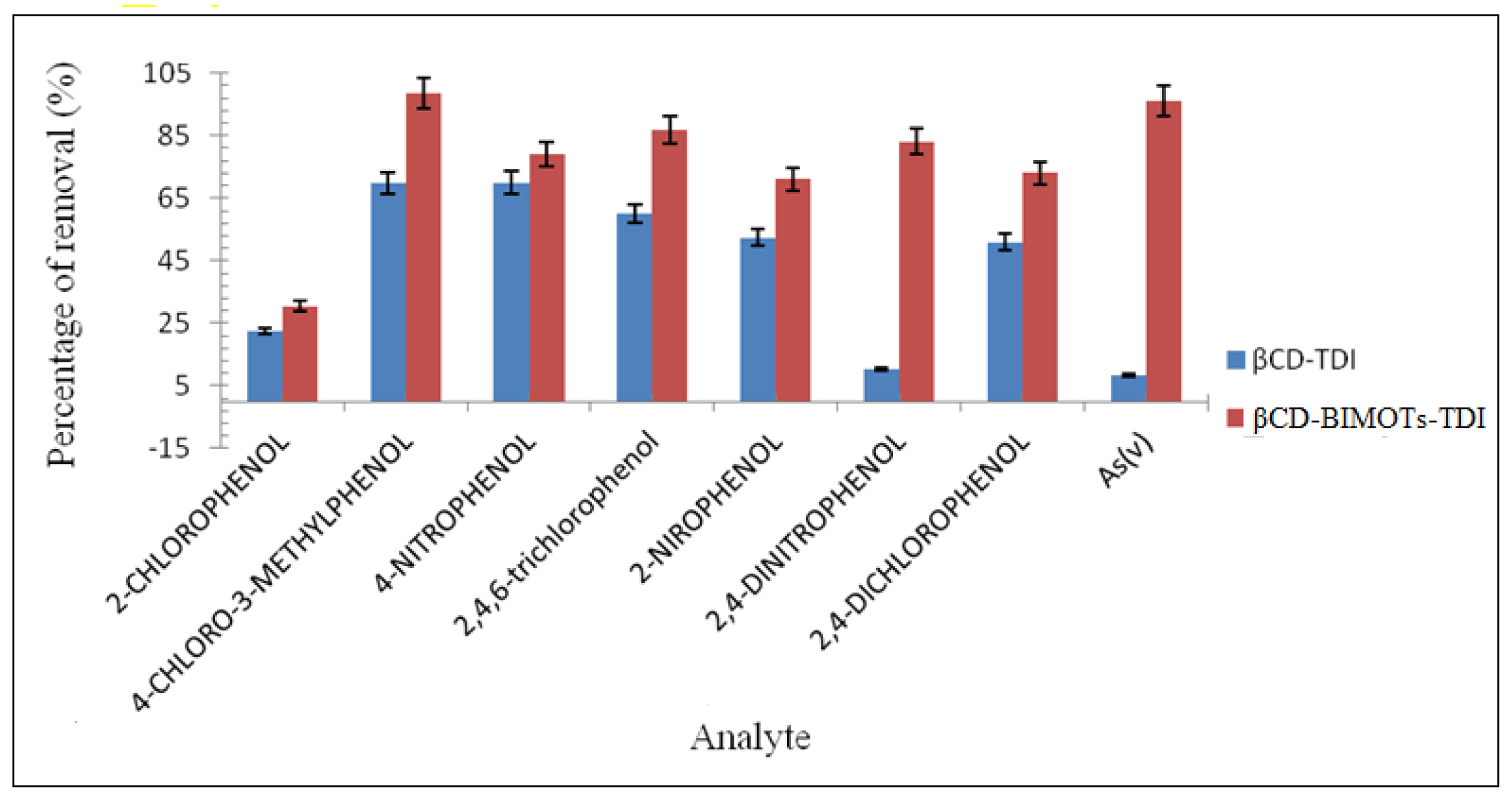
2.9. Sorption Studies
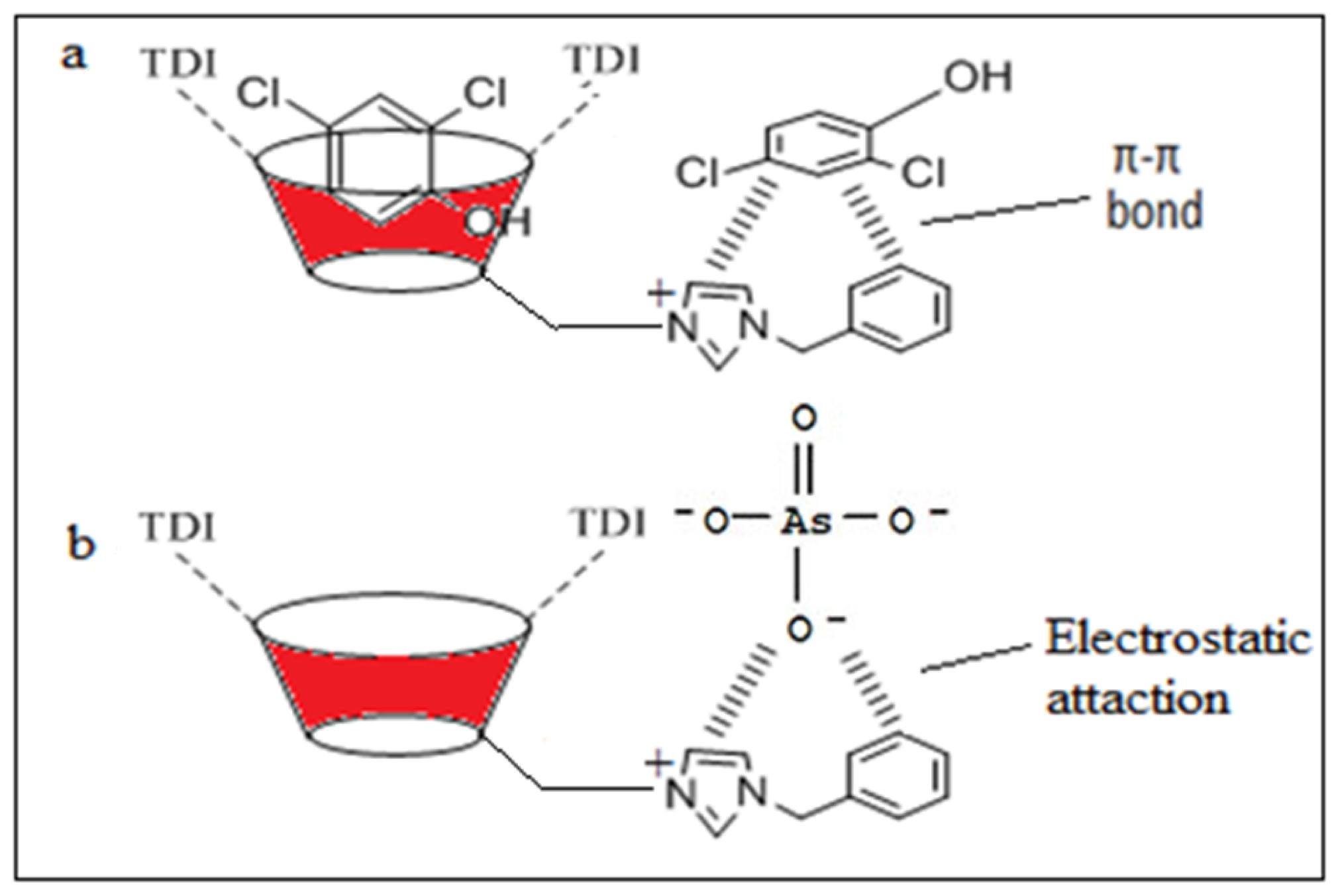
2.10. Adsorption Behavior of βCD-BIMOTs via Inclusion Complex and π–π Interaction
3. Experimental Section
3.1. Materials
3.2. Characterization of the Samples
3.3. Synthesis Method and Characterization
3.3.1. Synthesis of 6-O-Monotosyl-6-deoxy-β-cyclodextrin (β-CDOTs) (1)
3.3.2. Synthesis of Mono-6-deoxy-6-(3-benzylimidazolium)-β-cyclodextrin (βCD-BIMOTs) (2)
3.3.3. Synthesis of Insoluble β-Cyclodextrin Polymers
3.4. Sorption Experiments
4. Conclusions
Supplementary Information
ijms-15-00100-s001.pdfAcknowledgments
Conflicts of Interest
References
- Sun, Z.Y.; Shen, M.X.; Cao, G.P.; Deng, J.; Liu, Y.; Liu, T.; Zhao, L.; Yuan, W.K. Preparation of bimodal porous copolymer containing β-cyclodextrin and its inclusion adsorption behavior. J. App. Polym. Sci 2010, 118, 2176–2185. [Google Scholar]
- Szejtli, J. Past, present, and future of cyclodextrin research. Pure Appl. Chem 2004, 76, 1825–1845. [Google Scholar]
- Pandian, K.; Mohamad, S.; Sarih, N.M.; Ismail, N.A. Synthesis and characterization of the inclusion complex of β-cyclodextrin with azomethine. Int. J. Mol. Sci 2013, 14, 3671–3682. [Google Scholar]
- Mohamad, S.; Surikumaran, H.; Raoov, M.; Marimuthu, T.; Chandrasekaram, K.; Subramaniam, P. Conventional Study on Novel Dicationic Ionic Liquid Inclusion with β-Cyclodextrin. Int. J. Mol. Sci 2011, 12, 6329–6345. [Google Scholar]
- Mohamad, S.; Kamal, S.N.R.M. Study on an inclusion complex of β-cyclodextrin-dithizone-zinc (β-CD-H2Dz-Zn) by spectrophotometry and its analytical application. Asian J. Chem 2013, 25, 1965–1969. [Google Scholar]
- Zhang, J.; Shen, X.; Chen, Q. Separation processes in the presence of cyclodextrins using molecular imprinting technology and ionic liquid cooperating approach. Curr. Org. Chem 2011, 15, 75–85. [Google Scholar]
- Crini, G. Kinetic and equilibrium studies on the removal of cationic dyes from aqueous solution by adsorption onto a cyclodextrin polymer. Dyes Pigments 2008, 77, 415–426. [Google Scholar]
- Chin, Y.P.; Mohamad, S.; Abas, M.R. Removal of parabens from aqueous solution using cyclodextrin cross-linked polymer. Int. J. Mol. Sci 2010, 11, 3459–3471. [Google Scholar]
- Mohamad, S.; Yusof, N.H.M.; Asman, S. The effect of bifunctional isocyanate linker on adsorption of chromium(VI)-diphenylcarbazide complex onto β-cyclodextrin polymer. Asian J. Chem 2013, 25, 2213–2220. [Google Scholar]
- Li, N.; Wei, X.; Mei, Z.; Xiong, X.; Chen, S.; Ye, M.; Ding, S. Synthesis and characterization of a novel polyamidoamine-cyclodextrin crosslinked copolymer. Carbohyd. Res 2011, 346, 1721–1727. [Google Scholar]
- López-de-Dicastillo, C.; Gallur, M.; Catalá, R.; Gavara, R.; Hernandez-Muñoz, P. Immobilization of β-cyclodextrin in ethylene-vinyl alcohol copolymer for active food packaging applications. J. Membr. Sci 2010, 353, 184–191. [Google Scholar] [Green Version]
- Qian, L.; Guan, Y.; Xiao, H. Preparation and characterization of inclusion complexes of a cationic β-cyclodextrin polymer with butylparaben or triclosan. Int. J. Pharm 2008, 357, 244–251. [Google Scholar]
- Subramaniam, P.; Mohamad, S.; Alias, Y. Synthesis and characterization of the inclusion complex of dicationic ionic liquid and cyclodextrin. Int. J. Mol. Sci 2010, 11, 3675–3685. [Google Scholar]
- McEwen, A.B.; Ngo, H.L.; LeCompte, K.; Goldman, J.L. Electrochemical properties of imidazolium salt electrolytes for electrochemical capacitor applications. J. Electrochem. Soc 1999, 146, 1687–1695. [Google Scholar]
- Rahim, N.Y.; Mohamad, S.; Alias, Y.; Sarih, N.M. Extraction behavior of cu(ii) ion from chloride medium to the hydrophobic ionic liquids using 1,10-phenanthroline. Separ. Sci. Technol 2012, 47, 250–255. [Google Scholar]
- Mecerreyes, D. Polymeric ionic liquid: Broadening the properties and applications of polyelectrolytes. Prog. Polym. Sci 2011, 36, 1629–1648. [Google Scholar]
- Qiu, H.; Sawada, T.; Jiang, S.; Ihara, H. Synthesis and chacterization of poly(ionic liquid)-grafted silica hybrid materials through surface radical chain-transfer polymerization and aqueous anion-exchange. Mater. Lett 2010, 64, 1653–1655. [Google Scholar]
- Zhou, Z.; Li, X.; Chen, X.; Hao, X. Synthesis of ionic liquids functionalized β-cyclodextrin-bonded chiral stationary phases and their applications in high-performance liquid chromatography. Anal. Chim. Acta 2010, 678, 208–214. [Google Scholar]
- Ong, T.T.; Tang, W.; Muderawan, W.; Ng, S.C.; Chan, H.S.O. Synthesis and application of single isomer 6-mono(alkylimidazolium)-β-cyclodextrin as chiral selectors in chiral capillary electrophoresis. Electrophoresis 2005, 26, 3839–3848. [Google Scholar]
- Melton, L.D.; Slessor, K.N. Synthesis of monosubstituted cyclohexaamy-loses. Carbohyd. Res 1971, 18, 29–37. [Google Scholar]
- Khan, A.R.; Forgo, P.; Stine, K.J.; D’Souza, V.T. Methods for selective modifications of cyclodextrins. Chem. Rev 1998, 98, 1977–1996. [Google Scholar]
- Zhong, N.; Byun, H.-S.; Bittman, R. An improved synthesis of 6-O-monotosyl-6-deoxy-β-cyclodextrin. Tetrahedron Lett 1998, 39, 2919–2920. [Google Scholar]
- Bhaskar, M.; Aruna, P.; Jeevan, R.G.; Radhakrishnan, G. β-Cyclodextrin-polyurethane polymer as solid-phase extraction material for the analysis of carcinogenic aromatic amines. Anal. Chim. Acta 2004, 509, 39–45. [Google Scholar]
- Zha, F.; Li, S.; Chang, Y. Preparation and adsorption property of chitosan beads bearing β-cyclodextrin cross-linked by 1,6-hexamaethylene diisocyanate. Carbohyd. Polym 2008, 72, 456–461. [Google Scholar]
- Sing, K.S.W.; Everett, D.H.; Haul, R.A.W.; Moscou, L.; Pierotti, R.A.; Rouquerol, J.; Siemieniewska, T. Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity. IUPAC 1985, 57, 603–619. [Google Scholar]
- Schroden, R.C.; Al-Daous, M.; Sokolov, S.; Melde, B.J.; Lytle, J.C.; Stein, A.; Carbajo, M.C.; Fernandez, J.T.; Rodriquez, E.E. Hybrid macroporous materials for heavy metal ion adsorption. J. Mater. Chem 2002, 12, 3261–3267. [Google Scholar]
- Zhang, X.J.; Ma, T.Y.; Yuan, Z.Y. Titania–phosphonate hybrid porous materials: Preparation, photocatalytic activity and heavy metal ion adsorption. J. Mater. Chem 2008, 18, 2003–2010. [Google Scholar]
- Liu, A.M.; Hidajat, K.; Kawi, S.; Zhao, D.Y. A new class of hybrid mesoporous materials with functionalized organic monolayers for selective adsorption of heavy metal ions. Chem. Commun 2000, 1145–1146. [Google Scholar]
- Gokmen, M.T.; Prez, F.E.D. Porous polymer particles—A comprehensive guide to synthesis, characterization, functionalization and applications. Prog. Polym. Sci 2012, 37, 365–405. [Google Scholar] [Green Version]
- Dua, K.F.; Yan, M.; Wanga, Q.Y.; Songa, H. Preparation and characterization of novel macroporous cellulose beads regenerated from ionic liquid for fast chromatography. J. Chromatogr. A 2010, 1217, 1298–1304. [Google Scholar]
- Alahmadi, S.M.; Mohamad, S.; Maah, M.J. Synthesis and characterization of mesoporous silica functionalized with calix[4] arene derivatives. Int. J. Mol. Sci 2012, 13, 13726–13736. [Google Scholar]
- Li, N.; Mei, Z.; Wei, X. Study on sorption of chlorophenols from aqueous solution by an insoluble copolymer containing β-cyclodextrin and polyamidoamine units. Chem. Eng. J 2012, 192, 138–145. [Google Scholar]
- Yoo, K.S.; Lee, S.H. Influence of ionic liquid as a template on preparation of porous n-Al2O3 to DME synthesis from methanol. Bull. Korean Chem. Soc 2010, 31, 1628–1632. [Google Scholar]
- Salipira, K.L.; Krause, R.W.; Mamba, B.B.; Malesfetse, T.J.; Cele, L.M.; Durbach, S.H. Cyclodextrin polyurethanes polymerized with multi-walled carbon nanotubes: Synthesis and chacterization. Mater. Chem. Phys 2008, 111, 218–224. [Google Scholar]
- Abay, I.; Denizli, A.; Biskin, E.; Salih, B. Removal and pre-concentration of phenolic species onto β-cyclodextrin modified poly(hydroxyethylmethacrylate ethyleneglycoldimethacrylate) microbeads. Chemosphere 2005, 61, 1263–1272. [Google Scholar]
- Noble, R.D.; Gin, D.L. Perspective on ionic liquids and ionic liquid membranes. J. Membr. Sci 2011, 369, 1–4. [Google Scholar]
- Hanna, K.; de Brauer, C.; Germain, P. Solubilization of the neutral and charged forms of 2,4,6-trichlorophenol by β-cyclodextrin, methyl-β-cyclodextrin and hydroxypropyl-β-cyclodextrin in water. J. Hazard. Mater 2003, 100, 109–116. [Google Scholar]
- Leyva, E.; Moctezuma, E.; Strouse, J.; GarcÍagaribay, M.A. Spectrometric and 2D NMR studies on the complexation of chlorophenols with cyclodextrins. J. Incl. Phenom. Macrocycl. Chem 2001, 39, 41–46. [Google Scholar]
- Chattopadhyay, P.; Sinha, C.; Pal, D.K. Preparation and properties of a new chelating resin containing imidazolyl azo groups. Fresen. J. Anal. Chem 1997, 357, 368–372. [Google Scholar]
- Li, J.; Ni, X.; Zhou, Z.; Leong, K.W. Preparation and characterization of polypseudorotaxanes based on block-selected inclusion complexation between poly(propylene oxide)-poly(ethyleneoxide)-poly(propylene oxide) triblock copolymers and α-cyclodextrin. J. Am. Chem. Soc 2003, 125, 1788–1795. [Google Scholar]
- Gonil, P.; Sajomsanga, W.; Ruktanonchaia, U.R.; Pimphaa, N.; Sramalaa, I.; Nuchuchuaa, O.; Saesooa, S.; Chaleawlert-umpona, S.; Puttipipatkhachornb, S. Novel quaternized chitosan containing β-cyclodextrin moiety: Synthesis, characterization and antimicrobial activity. Carbohyd. Polym 2011, 83, 905–913. [Google Scholar]
- Mahlambi, M.M.; Malefetse, T.J.; Mamba, B.B.; Krause, R.W. β-Cyclodextrin-ionic liquid polyurethanes for the removal of organic pollutants and heavy metals from water: Synthesis and characterization. J. Polym. Res 2010, 17, 589–600. [Google Scholar]
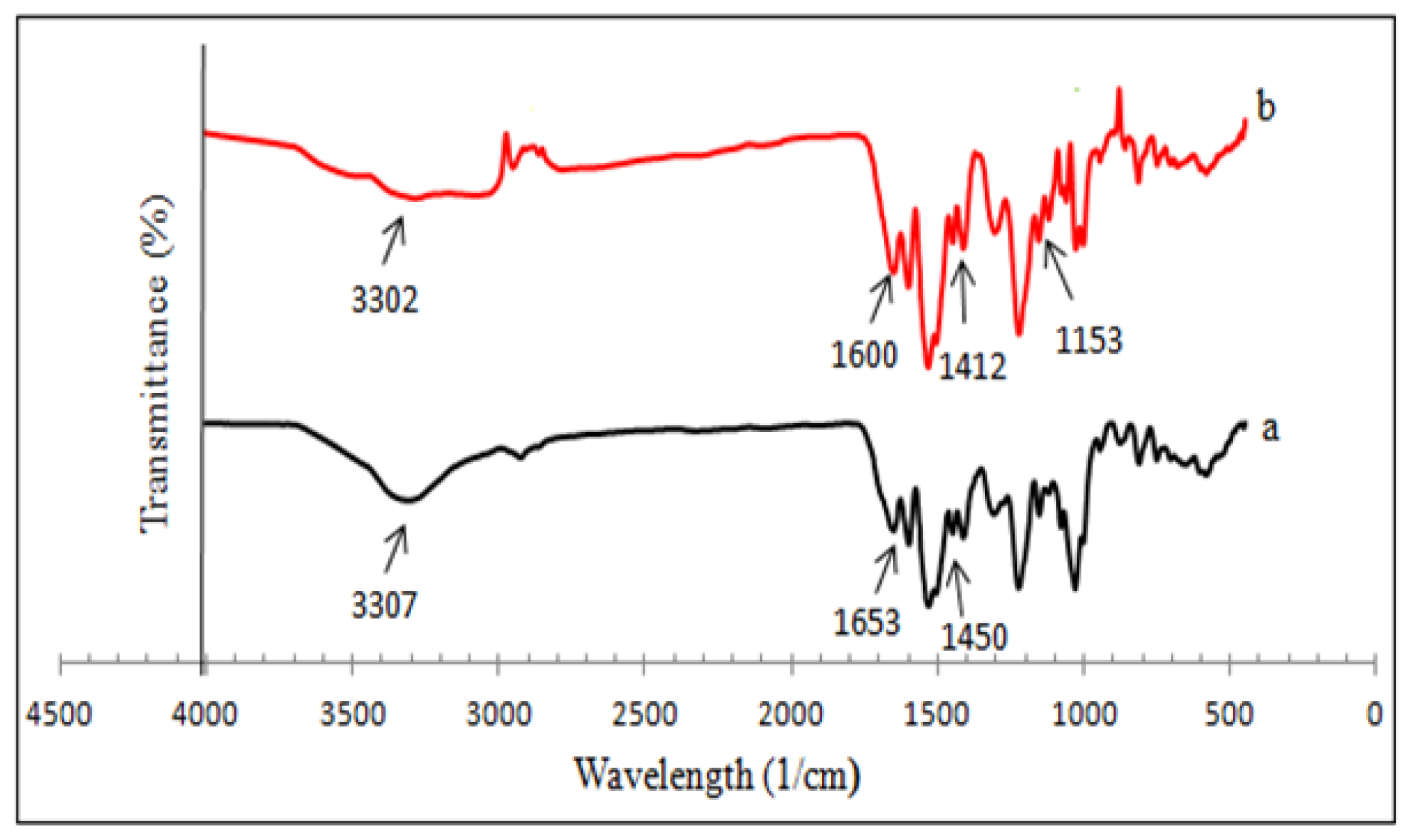



| Samples | Wavelength (cm−1) | Assignments |
|---|---|---|
| βCD-TDI | 3307 | N–H and O–H stretch |
| 2270 | Absence of isocyanate group | |
| 1653, 1534 | NHCO, carbamate linkage | |
| 1450 | Aromatic group in TDI | |
| βCD-BIMOTs-TDI | 3302 | N–H, O–H stretch and imidazole ring |
| 2270 | Absence of isocyanate group | |
| 1600, 1412 | Aromatic group in TDI | |
| 1535, 1651 | NHCO, carbamate linkage | |
| 1153 | C–N stretch |
| Sample | Region (°C) | Weight loss (%) | Assignment |
|---|---|---|---|
| βCD-TDI | 50–140 | 8 | Water loss/Moisture |
| 260–365 | 68 | Carbamate group and β-CD | |
| 365–900 | 26 | β-CD | |
| βCD-BIMOTs-TDI | 32–100 | 6 | Water loss/Moisture |
| 270–357 | 46 | Carbamate group, BIM, β-CD, OTs | |
| 357–915 | 40 | β-CD |
| Proton | βCD-BIMOTs | 2,4-DCP | βCD-BIMOTs-DCP | Changes |
|---|---|---|---|---|
| δ | δ | δ | Δδ | |
| H1 | 4.8330 | 4.8345 | +0.0015 | |
| H2 | 3.3030 | 3.3102 | +0.0072 | |
| H3 | 3.5450 | 3.6231 | +0.0781 | |
| H4 | 3.3268 | 3.3239 | −0.0029 | |
| H5 | 3.3980 | 3.5567 | +0.1587 | |
| H6 | 3.6298 | 3.6359 | −0.0061 | |
| H8 | 7.4175 | 7.4138 | −0.0037 | |
| H9 | 7.1269 | 7.1135 | −0.0134 | |
| H11 | 2.0868 | 2.0834 | +0.0034 | |
| Ha | 7.4597 | 7.4829 | +0.0232 | |
| Hb | 7.7450 | 7.7454 | +0.0004 | |
| Hc | 7.4914 | 7.4957 | +0.0043 | |
| Hd | 8.2100 | 8.1502 | −0.0598 | |
| He | 7.9458 | 7.9480 | +0.0026 | |
| Hf | 9.2031 | 9.2226 | +0.0195 | |
| Hg | 5.1880 | 5.2032 | +0.0152 | |
| Ha-p | 7.4334 | 7.4300 | −0.0034 | |
| Hb-p | 7.0258 | 6.9765 | −0.0493 | |
| Hc-p | 7.2095 | 7.1135 | −0.0960 |
| Parameter | Value |
|---|---|
| RF Power | 1550 watts |
| RF Matching | 1.55 V |
| Reflected Power | 0 W |
| Sample Uptake Time | 30 sec |
| Sample Uptake Rate | 0.4 r sec−1 |
| Chamber temperature | 2 °C |
| Nebuliser | Babington |
| Cones | Ni |
| Coolant Argon Flow Rate | 15 L min−1 |
| Carrier Gas Flow Rate | 1.2 L min−1 |
| Auxiliary gas flow rate | 0.9 L min−1 |
| Water RF/TP Flow Rate | 2.4 L min−1 |
| Water RF/TP Temperature | 20 °C |
| Mode | He |
| Internal standard | 72Ge |
| Integration time (sec per point) | 1 |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Raoov, M.; Mohamad, S.; Abas, M.R. Synthesis and Characterization of β-Cyclodextrin Functionalized Ionic Liquid Polymer as a Macroporous Material for the Removal of Phenols and As(V). Int. J. Mol. Sci. 2014, 15, 100-119. https://doi.org/10.3390/ijms15010100
Raoov M, Mohamad S, Abas MR. Synthesis and Characterization of β-Cyclodextrin Functionalized Ionic Liquid Polymer as a Macroporous Material for the Removal of Phenols and As(V). International Journal of Molecular Sciences. 2014; 15(1):100-119. https://doi.org/10.3390/ijms15010100
Chicago/Turabian StyleRaoov, Muggundha, Sharifah Mohamad, and Mhd Radzi Abas. 2014. "Synthesis and Characterization of β-Cyclodextrin Functionalized Ionic Liquid Polymer as a Macroporous Material for the Removal of Phenols and As(V)" International Journal of Molecular Sciences 15, no. 1: 100-119. https://doi.org/10.3390/ijms15010100
APA StyleRaoov, M., Mohamad, S., & Abas, M. R. (2014). Synthesis and Characterization of β-Cyclodextrin Functionalized Ionic Liquid Polymer as a Macroporous Material for the Removal of Phenols and As(V). International Journal of Molecular Sciences, 15(1), 100-119. https://doi.org/10.3390/ijms15010100