TMPRSS4 as a Poor Prognostic Factor for Triple-Negative Breast Cancer
Abstract
:1. Introduction
2. Results and Discussion
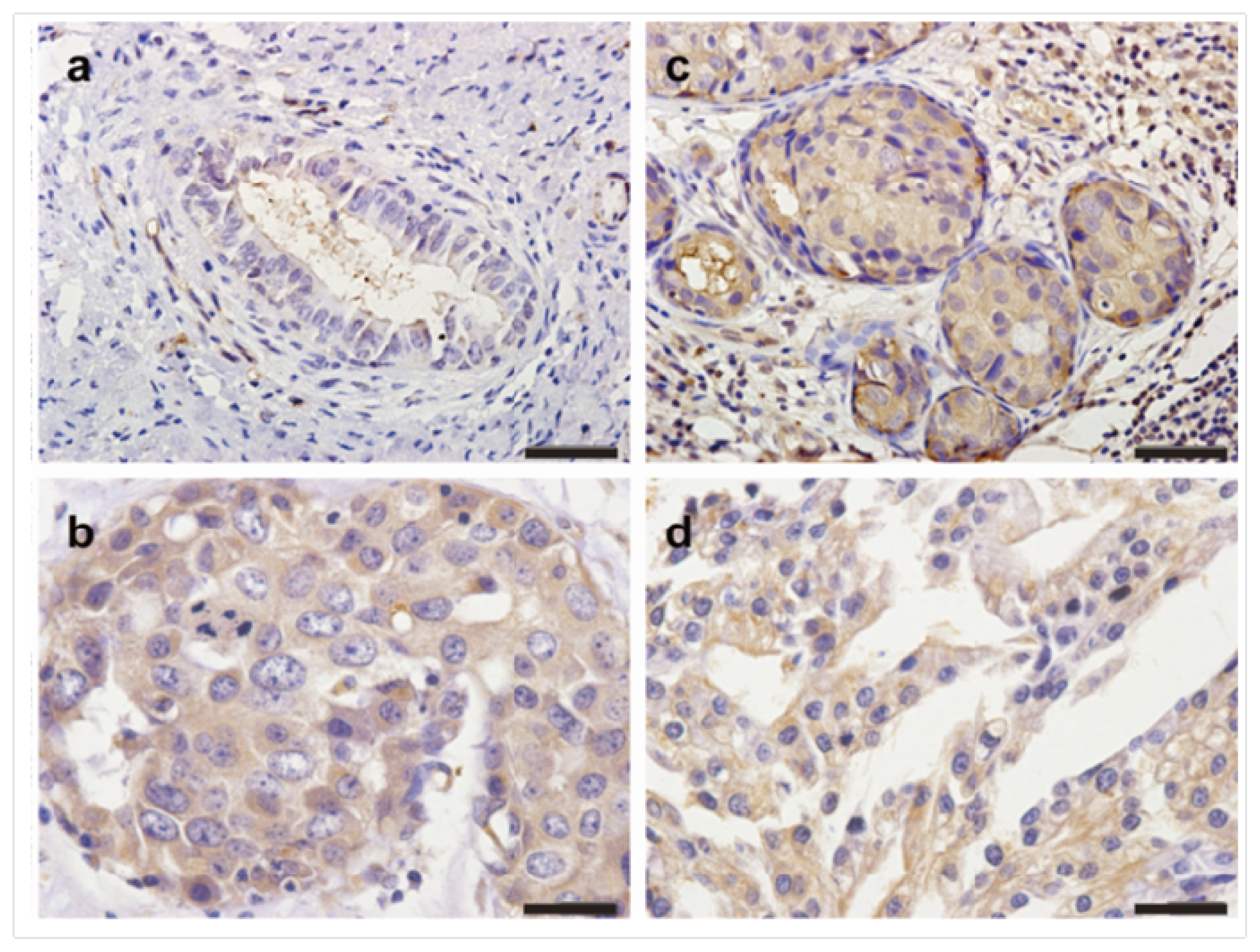
2.1. The Expression Profiles of TMPRSS4 in Breast Cancer Tissues
2.2. Correlation of TMPRSS4 Expression in TNBC with Clinicopathological Characteristics
2.3. Survival Analysis Correlation of TMPRSS4 Expression in TNBC with Clinicopathological Characteristics
3. Experimental Section
3.1. Patients
3.2. Immunohistochemical Staining
3.3. Scoring
3.4. Statistical Analysis
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Jemal, A.; Bray, F.; Center, M.M.; Ferlay, J.; Ward, E.; Forman, D. Global cancer statistics. CA 2011, 61, 69–90. [Google Scholar]
- Zhao, S.; Ma, W.; Zhang, M.; Tang, D.; Shi, Q.; Xu, S.; Zhang, X.; Liu, Y.; Song, Y.; Liu, L.; et al. High expression of CD147 and MMP-9 is correlated with poor prognosis of triple-negative breast cancer (TNBC) patients. Med. Oncol 2013, 30, 335. [Google Scholar]
- Nielsen, T.O.; Hsu, F.D.; Jensen, K.; Cheang, M.; Karaca, G.; Hu, Z.; Hernandez-Boussard, T.; Livasy, C.; Cowan, D.; Dressler, L.; et al. Immunohistochemical and clinical characterization of the basal-like subtype of invasive breast carcinoma. Clin. Cancer Res 2004, 10, 5367–5374. [Google Scholar]
- Sorlie, T.; Tibshirani, R.; Parker, J.; Hastie, T.; Marron, J.S.; Nobel, A.; Deng, S.; Johnsen, H.; Pesich, R.; Geisler, S.; et al. Repeated observation of breast tumor subtypes in independent gene expression data sets. Proc. Natl. Acad. Sci. USA 2003, 100, 8418–8423. [Google Scholar]
- Carey, L.A.; Perou, C.M.; Livasy, C.A.; Dressler, L.G.; Cowan, D.; Conway, K.; Karaca, G.; Troester, M.A.; Tse, C.K.; Edmiston, S.; et al. Race, breast cancer subtypes, and survival in the Carolina Breast Cancer Study. JAMA 2006, 295, 2492–2502. [Google Scholar]
- Dent, R.; Hanna, W.M.; Trudeau, M.; Rawlinson, E.; Sun, P.; Narod, S.A. Pattern of metastatic spread in triple-negative breast cancer. Breast Cancer Res. Treat 2009, 115, 423–428. [Google Scholar]
- Rodriguez-Pinilla, S.M.; Sarrio, D.; Honrado, E.; Hardisson, D.; Calero, F.; Benitez, J.; Palacios, J. Prognostic significance of basal-like phenotype and fascin expression in node-negative invasive breast carcinomas. Clin. Cancer Res 2006, 12, 1533–1539. [Google Scholar]
- Hooper, J.D.; Clements, J.A.; Quigley, J.P.; Antalis, T.M. Type II transmembrane serine proteases. Insights into an emerging class of cell surface proteolytic enzymes. J. Biol. Chem 2001, 276, 857–860. [Google Scholar]
- Netzel-Arnett, S.; Hooper, J.D.; Szabo, R.; Madison, E.L.; Quigley, J.P.; Bugge, T.H.; Antalis, T.M. Membrane anchored serine proteases: A rapidly expanding group of cell surface proteolytic enzymes with potential roles in cancer. Cancer Metastasis Rev 2003, 22, 237–258. [Google Scholar]
- Szabo, R.; Wu, Q.; Dickson, R.B.; Netzel-Arnett, S.; Antalis, T.M.; Bugge, T.H. Type II transmembrane serine proteases. Thromb. Haemost 2003, 90, 185–193. [Google Scholar]
- Giancotti, F.G.; Ruoslahti, E. Integrin signaling. Science 1999, 285, 1028–1032. [Google Scholar]
- Kim, S.; Kang, H.Y.; Nam, E.H.; Choi, M.S.; Zhao, X.F.; Hong, C.S.; Lee, J.W.; Lee, J.H.; Park, Y.K. TMPRSS4 induces invasion and epithelial-mesenchymal transition through upregulation of integrin alpha5 and its signaling pathways. Carcinogenesis 2010, 31, 597–606. [Google Scholar]
- Nguyen, T.H.; Weber, W.; Havari, E.; Connors, T.; Bagley, R.G.; McLaren, R.; Nambiar, P.R.; Madden, S.L.; Teicher, B.A.; Roberts, B.; et al. Expression of TMPRSS4 in non-small cell lung cancer and its modulation by hypoxia. Inter. J. Oncol 2012, 41, 829–838. [Google Scholar]
- Jung, H.; Lee, K.P.; Park, S.J.; Park, J.H.; Jang, Y.S.; Choi, S.Y.; Jung, J.G.; Jo, K.; Park, D.Y.; Yoon, J.H.; et al. TMPRSS4 promotes invasion, migration and metastasis of human tumor cells by facilitating an epithelial-mesenchymal transition. Oncogene 2008, 27, 2635–2647. [Google Scholar]
- Benaud, C.M.; Oberst, M.; Dickson, R.B.; Lin, C.Y. Deregulated activation of matriptase in breast cancer cells. Clin. Exp. Metastasis 2002, 19, 639–649. [Google Scholar]
- Larzabal, L.; Nguewa, P.A.; Pio, R.; Blanco, D.; Sanchez, B.; Rodriguez, M.J.; Pajares, M.J.; Catena, R.; Montuenga, L.M.; Calvo, A. Overexpression of TMPRSS4 in non-small cell lung cancer is associated with poor prognosis in patients with squamous histology. Br. J. Cancer 2011, 105, 1608–1614. [Google Scholar]
- Liedtke, C.; Mazouni, C.; Hess, K.R.; Andre, F.; Tordai, A.; Mejia, J.A.; Symmans, W.F.; Gonzalez-Angulo, A.M.; Hennessy, B.; Green, M.; et al. Response to neoadjuvant therapy and long-term survival in patients with triple-negative breast cancer. J. Clin. Oncol 2008, 26, 1275–1281. [Google Scholar]
- Gaedcke, J.; Traub, F.; Milde, S.; Wilkens, L.; Stan, A.; Ostertag, H.; Christgen, M.; von Wasielewski, R.; Kreipe, H.H. Predominance of the basal type and HER-2/neu type in brain metastasis from breast cancer. Mod. Pathol 2007, 20, 864–870. [Google Scholar]
- Greene, F.L. American Joint Committee on Cancer, American Cancer Society, AJCC Cancer Staging Manual, 6th ed; Springer-Verlag: New York, NY, USA, 2002; p. 421. [Google Scholar]

| Parameters | Total | TNBC (n = 72) | Non-TNBC (n = 109) | p-value |
|---|---|---|---|---|
| Age (years) | ||||
| ≤50 | 67 | 28 (38.9%) | 39 (35.8%) | 0.6715 |
| >50 | 114 | 44 (61.1%) | 70 (64.2%) | |
| Menopausal status | ||||
| Premenopausal | 93 | 40 (55.6%) | 53 (48.6%) | 0.3611 |
| Postmenopausal | 88 | 32 (44.4%) | 56 (51.4%) | |
| Histological subtype | ||||
| Ductal | 157 | 69 (95.8%) | 88 (80.7%) | 0.003 |
| Lobular | 24 | 3 (4.2%) | 21 (19.3%) | |
| LN metastasis | ||||
| Negative | 135 | 41 (56.9%) | 84 (77.1%) | 0.004 |
| Positive | 46 | 31 (43.1%) | 25 (22.9%) | |
| Tumor size | ||||
| ≤2 cm | 63 | 27 (37.5%) | 26 (23.9%) | 0.048 |
| >2 cm | 118 | 45 (62.5%) | 83 (76.1%) | |
| Grade | ||||
| I, II | 100 | 38 (52.8%) | 62 (56.9%) | 0.5969 |
| III | 81 | 34 (47.1%) | 47 (43.1%) | |
| TMPRSS4 | ||||
| Low expression | 68 | 19 (26.4%) | 49 (45.0%) | 0.012 |
| High expression | 113 | 53 (73.6%) | 60 (55.0%) | |
| Parameters | Low TMPRSS4 expression (n = 19) | High TMPRSS4 expression (n = 53) | χ2 value | p-value |
|---|---|---|---|---|
| Age (years) | ||||
| ≤50 | 8 (42.1%) | 20 (37.7%) | 0.112 | 0.737 |
| >50 | 11 (57.9%) | 33 (62.3%) | ||
| Menopausal status | ||||
| Premenopausal | 13 (68.4%) | 27 (50.9%) | 1.730 | 0.188 |
| Postmenopausal | 6 (31.6%) | 26 (49.1%) | ||
| Histological subtype | ||||
| Ductal | 18 (94.7%) | 51 (96.2%) | 0.078 | 0.378 |
| Lobular | 1 (5.3%) | 2 (3.8%) | ||
| LN metastasis | ||||
| Negative | 16 (84.2%) | 25 (47.2%) | 7.827 | 0.005 |
| Positive | 3 (15.8%) | 28 (52.8%) | ||
| Tumor size | ||||
| ≤2 cm | 12 (54.8%) | 15 (45.2%) | 7.250 | 0.007 |
| >2 cm | 7 (26.9%) | 38 (73.1%) | ||
| Grade | ||||
| I, II | 14 (73.7%) | 24 (45.3%) | 4.527 | 0.033 |
| III | 5 (26.3%) | 29 (54.7%) | ||
| Variables | Univariate | Multivariate | ||||
|---|---|---|---|---|---|---|
| HR | 95% CI | p-value | HR | 95% CI | p-value | |
| OS | ||||||
| Age (≤50 vs. >50 years) | 1.395 | 0.531–2.856 | 0.534 | |||
| Menopausal status (Pre- vs. Post-) | 1.112 | 0.495–2.141 | 0.707 | |||
| Histological subtype (Ductal vs. Lobular) | 0.937 | 0.644–1.920 | 0.832 | |||
| Lymph node metastasis (Negative vs. Positive) | 5.007 | 2.022–9.637 | 0.012 | 4.003 | 1.985–8.023 | 0.020 |
| Tumor size (≤2 cm vs. >2 cm) | 2.811 | 1.007–5.841 | 0.046 | 2.120 | 1.023–4.111 | 0.138 |
| Grade (I, II vs. III) | 3.223 | 1.021–4.888 | 0.029 | 2.744 | 1.011–4.746 | 0.109 |
| TMPRSS4 expression (Low vs. High) | 3.041 | 1.417–8.036 | 0.032 | 3.009 | 1.419–6.322 | 0.037 |
| DFS | ||||||
| Age (≤50 vs. >50 years) | 1.266 | 0.614–2.563 | 0.661 | |||
| Menopausal status (Pre- vs. Post-) | 1.104 | 0.799–2.251 | 0.721 | |||
| Histological subtype (Ductal vs. Lobular) | 0.916 | 0.647–1.985 | 0.840 | |||
| Lymph node metastasis (Negative vs. Positive) | 4.230 | 1.811–6.829 | 0.018 | 3.734 | 1.666–7.140 | 0.026 |
| Tumor size (≤2 cm vs. >2 cm) | 2.763 | 1.519–6.988 | 0.049 | 2.005 | 1.112–3.814 | 0.151 |
| Grade (I, II vs. III) | 2.851 | 1.003–4.771 | 0.044 | 2.612 | 1.001–4.320 | 0.085 |
| TMPRSS4 expression (Low vs. High) | 2.997 | 1.221–6.470 | 0.040 | 2.836 | 1.302–6.121 | 0.045 |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Cheng, D.; Kong, H.; Li, Y. TMPRSS4 as a Poor Prognostic Factor for Triple-Negative Breast Cancer. Int. J. Mol. Sci. 2013, 14, 14659-14668. https://doi.org/10.3390/ijms140714659
Cheng D, Kong H, Li Y. TMPRSS4 as a Poor Prognostic Factor for Triple-Negative Breast Cancer. International Journal of Molecular Sciences. 2013; 14(7):14659-14668. https://doi.org/10.3390/ijms140714659
Chicago/Turabian StyleCheng, Daye, Hong Kong, and Yunhui Li. 2013. "TMPRSS4 as a Poor Prognostic Factor for Triple-Negative Breast Cancer" International Journal of Molecular Sciences 14, no. 7: 14659-14668. https://doi.org/10.3390/ijms140714659




