Modulation of Phytoalexin Biosynthesis in Engineered Plants for Disease Resistance
Abstract
:1. The Concept of Phytoalexins
2. Genetic Manipulation of Phytoalexin Production and Disease Resistance Addressing Gain- or Loss-of-Function Genetic Approaches
3. Genetic Manipulation of Phytoalexin Glucosylation and Disease Resistance
4. Modulation of Phytoalexin Accumulation through Engineering of Plant Hormones
5. Phytoalexin Accumulation in the Upregulation of Defense-Related Marker and Elicitor Genes
6. Phytoalexin Engineering: Where Plant and Human Interests do not Meet
7. Conclusions
Conflict of Interest
References
- Müller, K.O.; Börger, H. Experimentelle untersuchungen über die Phytophthora resistenz der Kartoffel. Arbeit. Biol. Reichsant Land Forstwirtsch 1940, 23, 189–231. [Google Scholar]
- Deverall, B.J. Introduction. In Phytoalexins; Bailey, J.A., Mansfield, J.W., Eds.; Blackie: Glasgow and London, UK, 1982; pp. 1–20. [Google Scholar]
- Mansfield, J.W.; Bailey, J.A. Phytoalexins: Current Problems and Future Prospects. In Phytoalexins; Bailey, J.A., Mansfield, J.W., Eds.; Blackie: Glasgow and London, UK, 1982; pp. 319–323. [Google Scholar]
- Ahuja, I.; Kissen, R.; Bones, A.M. Phytoalexins in defense against pathogens. Trends Plant Sci 2012, 17, 73–90. [Google Scholar]
- Thomma, B.P.H.J.; Nelissen, I.; Eggermont, K.; Broekaert, W.F. Deficiency in phytoalexin production causes enhanced susceptibilty of Arabidopsis thaliana to the fungus Alternaria brassicola. Plant J 1999, 19, 163–171. [Google Scholar]
- VanWees, S.C.; Chang, H.S.; Zhu, T.; Glazebrook, J. Characterization of the early response of Arabidopsis to Alternaria brassicicola infection using expression profiling. Plant Physiol 2003, 132, 606–617. [Google Scholar]
- Rowe, H.C.; Walley, J.W.; Corwin, J.; Chan, E.K.F.; Dehesh, K.; Kliebenstein, D.J. Deficiencies in jasmonate-mediated plant defense reveal quantitative variation in Botrytis cinerea pathogenesis. PLoS Pathog 2010, 6, e1000861. [Google Scholar]
- Nawrath, C.; Métraux, J.P. Salicylic acid induction-deficient mutants of Arabidopsis express PR-2 and PR-5 and accumulate high levels of camalexin after pathogen inoculation. Plant Cell 1999, 11, 1393–1404. [Google Scholar]
- Roetschi, A.; Si-Ammour, A.; Belbahri, L.; Mauch, F.; Mauch-Mani, B. Characterization of an Arabidopsis-Phytophthora pathosystem: Resistance requires a functional PAD2 gene and is independent of salicylic acid, ethylene and jasmonic acid signalling. Plant J 2001, 28, 293–305. [Google Scholar]
- Denby, K.J.; Jason, L.J.M.; Murray, S.L.; Last, R.L. ups1, an Arabidopsis thaliana camalexin accumulation mutant defective in multiple defence signalling pathways. Plant J 2005, 41, 673–684. [Google Scholar]
- Heck, S.; Grau, T.; Buchala, A.; Métraux, J.P.; Nawrath, C. Genetic evidence that expression of NahG modifies defence pathways independent of salicylic acid biosynthesis in the Arabidopsis-Pseudomonas syringae pv. tomato interaction. Plant J 2003, 36, 342–352. [Google Scholar]
- Robert-Seilaniantz, A.; MacLean, D.; Jikumaru, Y.; Hill, L.; Yamaguchi, S.; Kamiya, Y.; Jones, J.D.G. The microRNA miR393 re-directs secondary metabolite biosynthesis away from camalexin and towards glucosinates. Plant J 2011, 67, 218–231. [Google Scholar]
- Ren, D.; Liu, Y.; Yang, K.Y.; Han, L.; Mao, G.; Glazebrook, J.; Zhang, S. A fungal-responsive MAPK cascade regulates phytoalexin biosynthesis in Arabidopsis. Proc. Natl Acad. Sci. USA 2008, 105, 5638–5643. [Google Scholar]
- Pandey, S.P.; Rocarro, M.; Schön, M.; Logemann, E.; Somssich, I.E. Transcriptional reprogramming regulated by WRKY18 and WRKY40 facilitates powdery mildew infection of Arabidopsis. Plant J 2010, 64, 912–923. [Google Scholar]
- Smith, D.A. Toxicity of Phytoalexins. In Phytoalexins; Bailey, J.A., Mansfield, J.W., Eds.; Blackie: Glasgow and London, UK, 1982; pp. 218–252. [Google Scholar]
- Rathmell, W.G.; Smith, D.A. Lack of activity of selected isoflavonoid phytoalexins as protectant fungicides. Pestic. Sci 1980, 11, 568–572. [Google Scholar]
- Pezet, R.; Pont, V. Ultrastructural observations of pterostilbene fungitoxicity in dormant conidia of Botrytis cinerea Pers. J. Phytopathol 1990, 129, 29–30. [Google Scholar]
- Adrian, M.; Jeandet, P.; Veneau, J.; Weston, L.A.; Bessis, R. Biological activity of resveratrol, a stilbenic compound from grapevines, against Botrytis cinerea, the causal agent for gray mold. J. Chem. Ecol 1997, 23, 1689–1702. [Google Scholar]
- Adrian, M.; Jeandet, P. Effects of resveratrol on the ultrastructure of Botrytis cinerea conidia and biological significance in plant/pathogen interactions. Fitoterapia 2012, 83, 1345–1350. [Google Scholar]
- Shlezinger, N.; Minz, A.; Gur, Y.; Hatam, I.; Dagdas, Y.F.; Talbot, N.J.; Sharon, A. Anti-apoptotic machinery protects the necrotrophic fungus Botrytis cinerea from host-induced apoptotic-like cell death during plant infection. PLoS Pathog 2011, 7, e1002185. [Google Scholar]
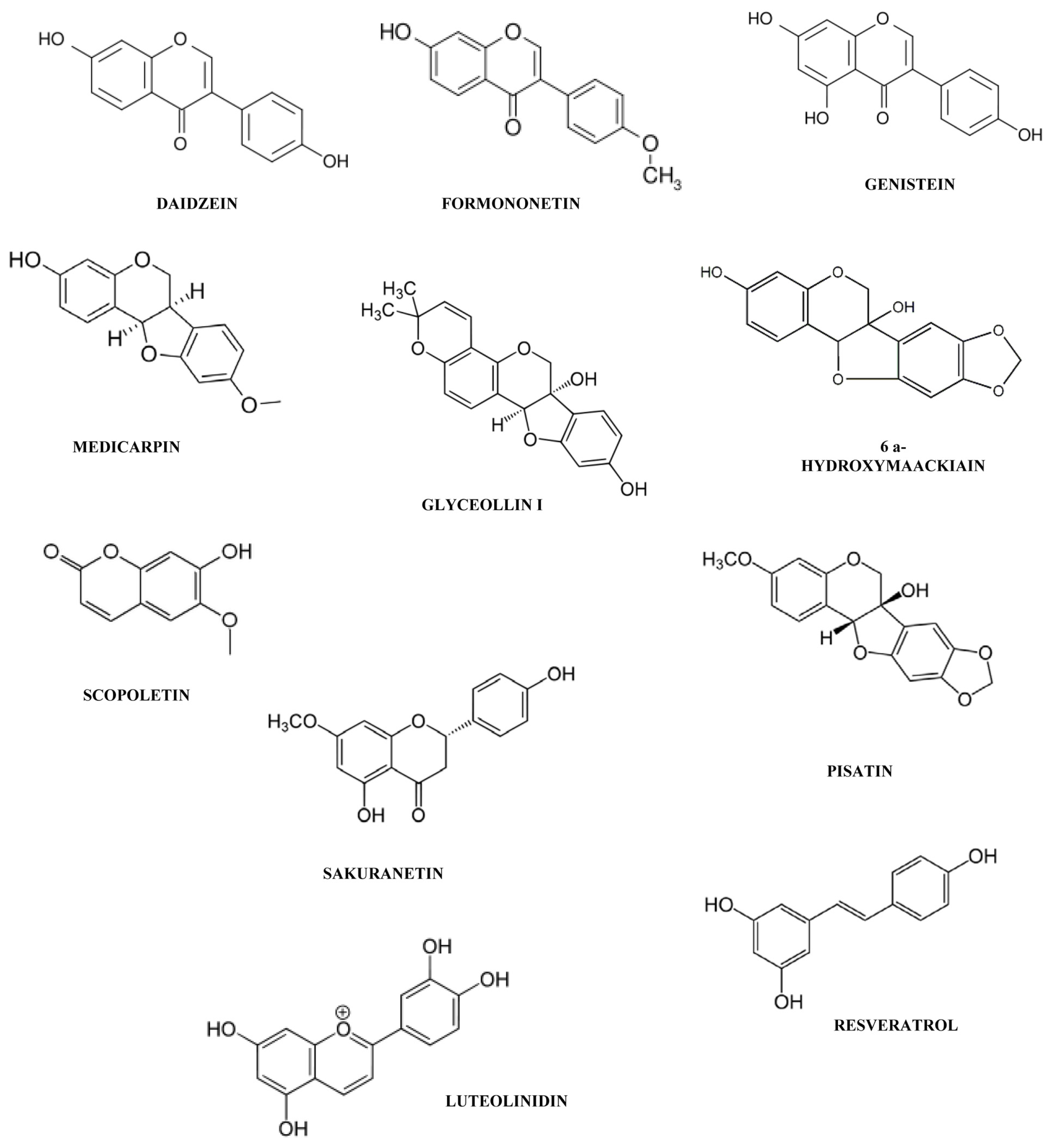
- Ingham, J.L. Phytoalexins from the Leguminosae. In Phytoalexins; Bailey, J.A., Mansfield, J.W., Eds.; Blackie: Glasgow and London, UK, 1982; pp. 21–80. [Google Scholar]
- Kuc, J. Phytoalexins from the Solanaceae. In Phytoalexins; Bailey, J.A., Mansfield, J.W., Eds.; Blackie: Glasgow and London, UK, 1982; pp. 81–105. [Google Scholar]
- Coxon, D.T. Phytoalexins from other Plant Families. In Phytoalexins; Bailey, J.A., Mansfield, J.W., Eds.; Blackie: Glasgow and London, UK, 1982; pp. 106–132. [Google Scholar]
- Keen, N.T.; Zaki, A.; Sims, J.J. Biosynthesis of hydoxyphaseollin and related isoflavanoids in disease-resistant soybean hypocotyls. Phytochemistry 1972, 11, 1031–1039. [Google Scholar]
- Markham, K.R.; Ingham, J.L. Tectorigenin, a phytoalexin from Centrosema haitiense and other Centrosema species. Z. Naturforsch. C 1980, 35, 919–922. [Google Scholar]
- Ingham, J.L. Induced isoflavanoids from fungus-infected stems of pigeon pea (Cajanus cajan). Z. Naturforsch. C 1976, 31, 504–508. [Google Scholar]
- Ingham, J.L.; Keen, N.T.; Hymowitz, T. A new isoflavone phytoalexin from fungus-inoculated stems of Glycine wightii. Phytochemistry 1977, 16, 1943–1946. [Google Scholar]
- Ingham, J.L. Flavonoid and isoflavonoid compounds from leaves of sainfoin (Onobrychis viciifolia). Z. Naturforsch. C 1978, 33, 146–148. [Google Scholar]
- Ingham, J.L. Phytoalexins from hyacinth bean (Lablab niger). Z. Naturforsch. C 1977, 32, 1018–1020. [Google Scholar]
- Hargreaves, J.A.; Mansfield, J.W.; Coxon, D.T. Identification of medicarpin as a phytoalexin in the broad bean plant (Vicia faba L.). Nature 1976, 262, 318–319. [Google Scholar]
- Ingham, J.L. Phytolexin production by species of the genus Caragana. Z. Naturforsch. C 1979, 34, 293–295. [Google Scholar]
- Gnanamanickam, S.S. Isolation of isoflavonoid phytoalexins from seeds of Phaseolus vulgaris. Experientia 1979, 35, 323. [Google Scholar]
- Burden, R.S.; Bailey, J.A. Structure of the phytoalexin from soybean. Phytochemistry 1975, 14, 1389–1390. [Google Scholar]
- Bonde, M.R.; Millar, R.L.; Ingham, J.L. Induction and identification of sativan and vestitol as two phytoalexins from Lotus cornicolatus. Phytochemistry 1973, 12, 2957–2959. [Google Scholar]
- Lyon, F.M.; Wood, R.K.S. Production of phaseollin, coumestrol and related compounds in bean leaves inoculated with Pseudomonas spp. Physiol. Plant Pathol 1975, 6, 117–124. [Google Scholar]
- Mansfield, J.W.; Porter, A.E.A.; Smallman, R.V. Dihydrowyerone derivatives as components of the furanoacetylene phytoalexin response of tissues of Vicia faba. Phytochemistry 1980, 19, 1057–1061. [Google Scholar]
- Hargreaves, J.A.; Mansfield, J.W.; Coxon, D.T. Conversion of wyerone to wyerol by Botrytis cinerea and B. fabae in vitro. Phytochemistry 1976, 15, 651–653. [Google Scholar]
- Ingham, J.L. 3,5,4′-trihydroxystilbene as a phytoalexin from groundnuts (Arachis hypogea). Phytochemistry 1976, 15, 1791–1793. [Google Scholar]
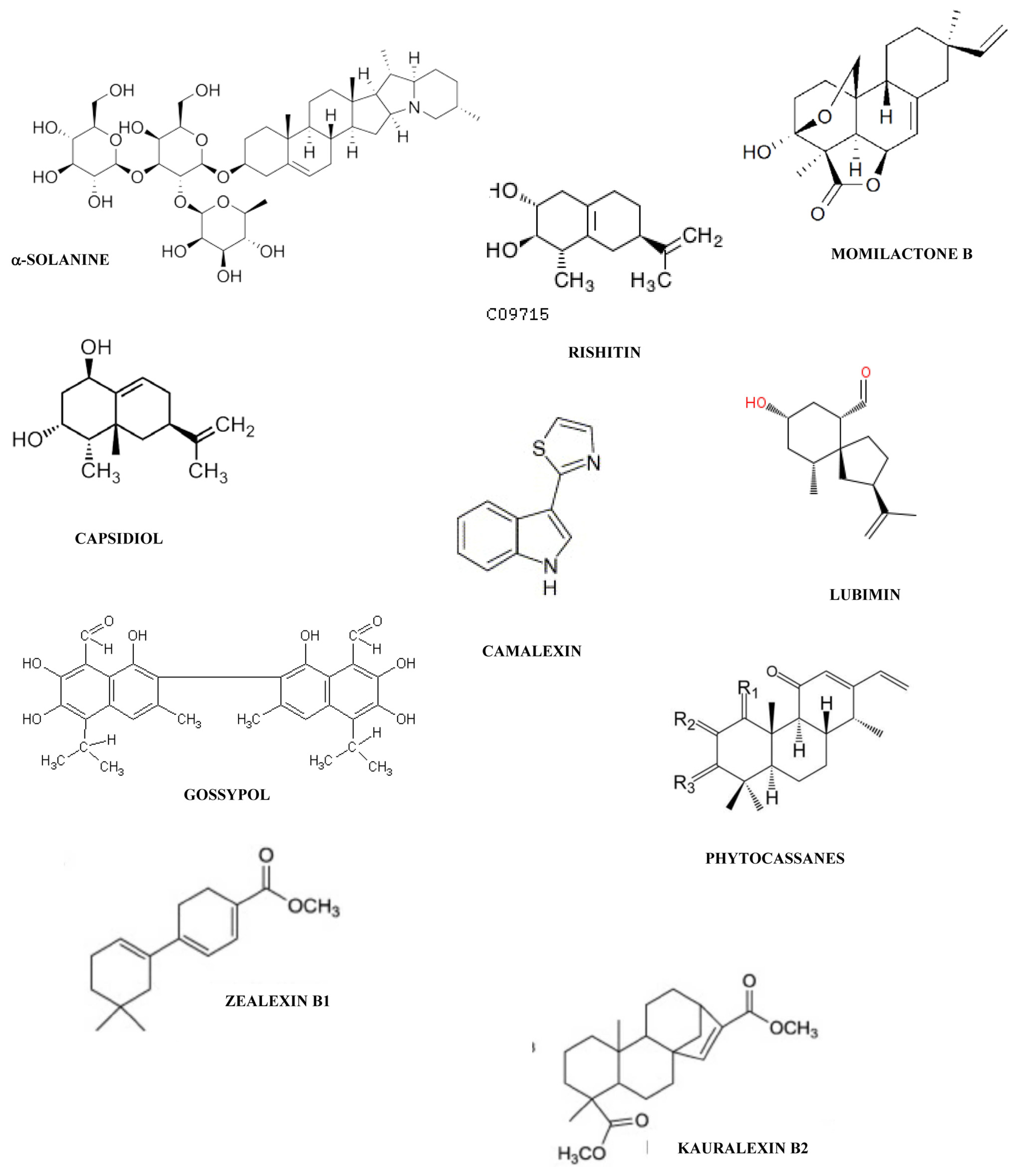
- Kuc, J. A biochemical study of the resistance of potato tuber to attack by various fungi. Phytopathology 1957, 47, 676–680. [Google Scholar]
- Locci, R.; Kuc, J. Steroid glycoalkaloids as compounds produced by potato tuber slices under stress. Phytopathology 1967, 57, 1272–1273. [Google Scholar]
- Tomiyama, K.; Sakuma, T.; Ishizaka, N.; Sato, N.; Takasugi, M.; Katsui, T. A new antifungal substance isolated from potato tuber tissue infected by pathogens. Phytopathology 1968, 58, 115–116. [Google Scholar]
- Katsui, N.; Murai, A.; Takasugi, M.; Imaizumi, K.; Masamune, T. The structure of rishitin, a new anfifungal compound from diseased potato tubers. J. Chem. Soc. Chem. Comm. 1968, 43–44. [Google Scholar]
- Katsui, N.; Matsunaga, A.; Masamune, T. The structure of lubimin, oxylubimin, antifungal metabolites from diseased potato tubers. Tetrahedron Lett 1974, 15, 4483–4486. [Google Scholar]
- Birnbaum, G.I.; Stoessl, A.; Grover, S.H.; Stothers, J.B. The complete stereostructure of capsidiol, X-ray analysis and 13C nuclear magnetic resonance of eremophilane derivatives having trans-vicinal methyl groups. Can. J. Chem 1974, 52, 993–1005. [Google Scholar]
- Reuveni, M.; Cohen, Y. Growth retardation and changes in phenolic compounds, with special reference to scopoletin, in mildewed and ethylene-treated tobacco plants. Physiol. Plant Pathol 1978, 12, 179–189. [Google Scholar]
- Cartwright, D.W.; Langcake, P.; Pryce, R.J.; Leworthy, D.P.; Ride, J.P. Isolation and characterization of two phytoalexins from rice as momilactones A and B. Phytochemistry 1981, 20, 535–537. [Google Scholar]
- Kodama, O.; Miyakawa, J.; Akatsuka, T.; Kiyosawa, S. Sakuranetin, a flavanone phytoalexin from ultraviolet-irradiated rice leaves. Phytochemistry 1992, 31, 3807–3809. [Google Scholar]
- Umemura, K.; Ogawa, N.; Shimura, M.; Koga, J.; Usami, H.; Kono, T. Possible role of phytocassane, rice phytoalexin, in disease resistance of rice against the blast fungus Magnaporthe grisae. Biosci. Biotechnol. Biochem 2003, 67, 899–902. [Google Scholar]
- Schmelz, E.A.; Kaplan, F.; Huffaker, A.; Dafoe, N.J.; Vaughan, M.M.; Ni, X.; Rocca, J.R.; Alborn, H.T.; Teal, P.E.A. Identity, regulation, and activity of inducible diterpenoid phytoalexins in maize. Proc. Natl. Acad. Sci. USA 2011, 108, 5455–5460. [Google Scholar]
- Huffaker, A.; Kaplan, F.; Vaughan, M.M.; Dafoe, N.J.; Ni, X.; Rocca, J.R.; Alborn, H.T.; Teal, P.E.A.; Schmelz, E.A. Novel acidic sesquiterpenoids constitute a dominant class of pathogen-induced phytoalexins in maize. Plant Physiol 2011, 156, 2082–2097. [Google Scholar]
- Nicholson, F.; Kollipara, S.S.; Vincent, J.R.; Lyons, P.C.; Cadena-Gomez, G. Phytoalexin synthesis by the sorghum mesocotyl in response to infection by pathogenic and nonpathogenic fungi. Proc. Natl. Acad. Sci. USA 1987, 84, 5520–5524. [Google Scholar]
- Lo, S.C.; de Verdier, K.; Nicholson, R. Accumulation of 3-deoxyanthocyanidin phytoalexins and resistance to Colletotrichum sublineolum in sorghum. Physiol. Mol. Plant Pathol 1999, 55, 263–273. [Google Scholar]
- Ejike, C.E.C.C.; Gong, M.; Udenigwe, C.C. Phytoalexins from the Poaceae: Biosynthesis, function and prospects in food preservation. Food Res. Int 2013, 52, 167–177. [Google Scholar]
- Bell, A.A. Formation of gossypol in infected or chemically irritated tissues of Gossypium species. Phytopathology 1967, 57, 759–764. [Google Scholar]
- Browne, L.M.; Conn, K.L.; Ayert, W.A.; Tewari, J.P. The camalexins: New phytoalexins produced in the leaves of Camelia sativa (Cruciferae). Tetrahedron 1991, 47, 3909–3914. [Google Scholar]
- Langcake, P.; Pryce, R.J. The production of resveratrol by Vitis vinifera and other members of the Vitaceae as a response to infection or injury. Physiol. Plant Pathol 1976, 9, 77–86. [Google Scholar]
- Pérez-Clemente, R.M.; Vives, V.; Zandalinas, S.I.; Lopez-Climent, M.F.; Munoz, V.; Gomez-Cadenas, A. Biotechnological approaches to study plant responses to stress. BioMed. Res. Int. 2013. [Google Scholar] [CrossRef]
- Grosskinsky, D.K.; van der Graaff, E.; Roitsch, T. Phytoalexin transgenics in crop protection-Fairy tale with a happy end? Plant Sci 2012, 195, 54–70. [Google Scholar]
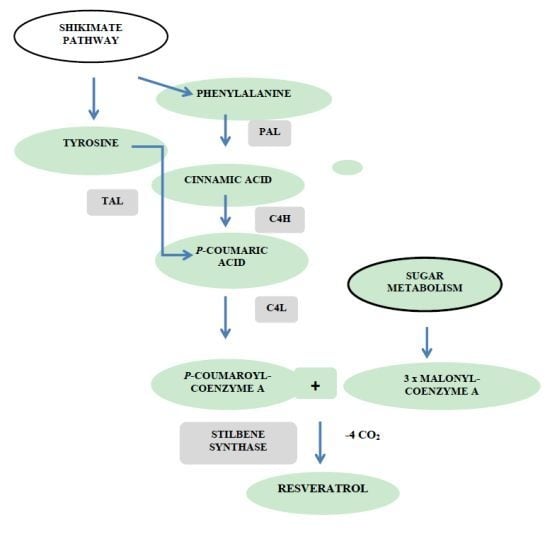
- Halls, C.; Yu, O. Potential for metabolic engineering of resveratrol biosynthesis. Trends Biotechnol 2008, 26, 77–81. [Google Scholar]
- Delaunois, B.; Cordelier, S.; Conreux, A.; Clément, C.; Jeandet, P. Molecular engineering of resveratrol in plants. Plant Biotechnol. J 2009, 7, 2–12. [Google Scholar]
- Donnez, D.; Jeandet, P.; Clément, C.; Courot, E. Bioproduction of resveratrol and stilbene derivatives by plant cells and microorganisms. Trends Biotechnol 2009, 27, 706–713. [Google Scholar]
- Jeandet, P.; Delaunois, B.; Conreux, A.; Donnez, D.; Nuzzo, V.; Cordelier, S.; Clément, C.; Courot, E. Biosynthesis, metabolism, molecular engineering and biological functions of stilbene phytoalexins in plants. BioFactors 2010, 36, 331–341. [Google Scholar]
- Jeandet, P.; Delaunois, B.; Aziz, A.; Donnez, D.; Vasserot, Y.; Cordelier, S.; Courot, E. Metabolic engineering of yeast and plants for the production of the biologically active hydroxystilbene, resveratrol. J. Biomed. Biotechnol. 2012. [Google Scholar] [CrossRef]
- Jeandet, P.; Vasserot, Y.; Chastang, T.; Courot, E. Engineering microbial cells for the biosynthesis of natural compounds of pharmaceutical significance. BioMed. Res. Int. 2013. [Google Scholar] [CrossRef]
- Hain, R.; Reif, H.J.; Krause, E.; Langebartels, R.; Kindl, H.; Vornam, B.; Wiese, W.; Schmelzer, E.; Schreier, P.; Stöcker, R.; Stenzel, K. Disease resistance results from foreign phytoalexin expression in a novel plant. Nature 1993, 361, 153–156. [Google Scholar]
- Hipskind, J.D.; Paiva, N.L. Constitutive accumulation of a resveratrol glucoside in transgenic alfalfa increases resistance to Phoma medicaginis. Mol. Plant-Microbe Interact 2000, 13, 551–562. [Google Scholar]
- Coutos-Thévenot, P.; Poinssot, B.; Bonomelli, A.; Yean, H.; Breda, C.; Buffard, D.; Esnault, R.; Hain, R.; Boulay, M. In vitro tolerance to Botrytis cinerea of grapevine 41B rootstock in transgenic plants expressing the stilbene synthase Vst 1 gene under the control of a pathogen-inducible PR 10 promoter. J. Exp. Bot 2001, 52, 901–910. [Google Scholar]
- Stark-Lorenzen, P.; Nelke, B.; Hänbler, G.; Mühlbach, H.P.; Thomzik, J.E. Transfer of a grapevine stilbene synthase gene to rice (Oryza sativa L.). Plant Cell Rep 1997, 16, 668–673. [Google Scholar]
- Leckband, G.; Lörz, H. Transformation and expression of a stilbene synthase gene of Vitis vinifera L. in barley and wheat for increased fungal resistance. Theor. Appl. Genet 1998, 96, 1004–1012. [Google Scholar]
- Liang, H.; Zheng, J.; Shuange, J.I.A.; Wang, D.; Ouyang, J.; Li, J.; Li, L.; Tian, W.; Jia, X.; Duan, X.; Sheng, B.; Hain, R. A transgenic wheat with a stilbene synthase gene resistant to powdery mildew obtained by biolistic method. Chin. Sci. Bull 2000, 45, 634–638. [Google Scholar]
- Thomzik, J.E.; Stenzel, K.; Stöcker, R.; Schreier, P.H.; Hain, R.; Stahl, D.J. Synthesis of a grapevine phytoalexin in transgenic tomatoes (Lycopersicon esculentum Mill.) conditions resistance against Phytophthora infestans. Physiol. Mol. Plant Pathol 1997, 51, 265–278. [Google Scholar]
- Serazetdinova, L.; Oldach, K.; Lörz, H. Expression of transgenic stilbene synthases in wheat causes the accumulation of unknown stilbene derivatives with antifungal activity. J. Plant Physiol 2005, 162, 985–1002. [Google Scholar]
- Zhu, Y.J.; Agbayani, R.; Jazckson, M.C.; Tang, C.S.; Moore, P.M. Expression of the grapevine stilbene synthase gene VST1 in papaya provides increased resistance against diseaes caused by Phytophthora palmivora. Planta 2004, 12, 807–812. [Google Scholar]
- Lim, J.D.; Yun, S.J.; Chung, I.M.; Yu, C.Y. Resveratrol synthase transgene expression and accumulation of resveratrol glycoside in Rehmannia glutinosa. Mol. Breed 2005, 16, 219–233. [Google Scholar]
- Liu, Z.Y.; Zhuang, C.X.; Sheng, S.J. Overexpression of a resveratrol synthase gene (PcRs) from Polygonum cuspidatum in transgenic Arabidopsis causes the accumulation of trans-piceid with antifungal activity. Plant Cell Rep 2011, 30, 2027–2036. [Google Scholar]
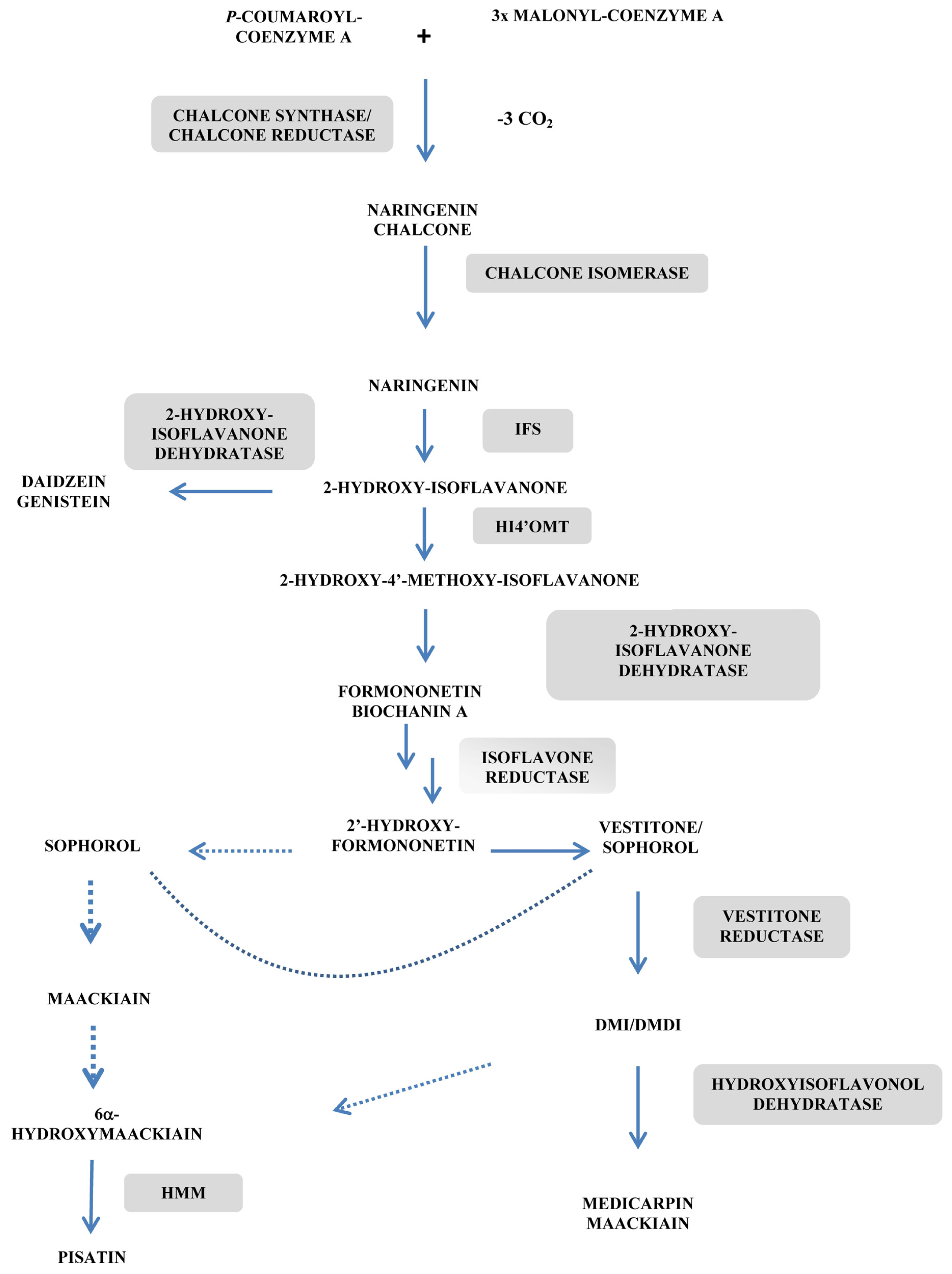
- Deavours, B.E.; Dixon, R.A. Metabolic engineering of isoflavonoid biosynthesis in alfalfa. Plant Physiol 2005, 138, 2245–2259. [Google Scholar]
- Kaimoyo, E.; VanEtten, H.D. Inactivation of pea genes by RNAi supports the involvement of two similar O-methyltransferases in the biosynthesis of (+)-pisatin and of chiral intermediates with a configuration opposite that found in (+)-pisatin. Phytochemistry 2008, 69, 76–87. [Google Scholar]
- Giorcelli, A.; Sparvoli, F.; Mattivi, F.; Tava, A.; Balestrazzi, A.; Vrhovsek, U.; Calligari, P.; Bollini, R.; Confalonieri, M. Expression of the stilbene synthase (StSy) gene from grapevine in transgenic white poplar results in high accumulation of the antioxidant resveratrol glucosides. Transgenic Res 2004, 13, 203–214. [Google Scholar]
- Seppänen, S.K.; Syrjälä, L.; von Weissenberg, K.; Teeri, T.H.; Paajanen, L.; Pappinen, A. Antifungal activity of stilbenes in vitro bioassays and in transgenic Populus expressing a gene encoding pinosylvin synthase. Plant Cell Rep 2004, 22, 584–593. [Google Scholar]
- Kobayashi, S.; Ding, C.K.; Nakamura, Y.; Nakajima, I.; Matsumoto, R. Kiwifruits (Actinidia deliciosa) transformed with a Vitis stilbene synthase gene produce piceid (resveratrol-glucoside). Plant Cell Rep 2000, 19, 904–910. [Google Scholar]
- Fischer, R.; Budde, I.; Hain, R. Stilbene synthase gene expression causes changes in flower colour and male sterility in tobacco. Plant J 1997, 11, 489–498. [Google Scholar]
- Höfig, K.P.; Möller, R.; Donaldson, L.; Putterill, J.; Walter, C. Towards male sterility in Pinus radiata—A stilbene synthase approach to genetically engineer nuclear male sterility. Plant Biotechnol. J 2006, 4, 333–343. [Google Scholar]
- He, X.Z.; Dixon, R.A. Genetic manipulation of isoflavone 7-O-methyltransferase enhances biosynthesis of 4′-O-methylated isoflavonoid phytoalexins and disease resistance in alfalfa. Plant Cell 2000, 12, 1689–1702. [Google Scholar]
- Blount, J.W.; Dixon, R.A.; Paiva, N.L. Stress responses in alfalfa (Medicago sativa L.) XVI. Antifungal activity of medicarpin and its biosynthetic precursors; implications for the genetic manipulation of stress metabolites. Physiol. Mol. Plant Pathol 1993, 41, 333–349. [Google Scholar]
- Wu, Q.; VanEtten, H.D. Introduction of plant and fungal genes into pea (Pisum sativum L.) hairy roots reduces their ability to produce pisatin and affects their response to a fungal pathogen. Mol. Plant-Microbe Interact 2004, 17, 798–804. [Google Scholar]
- Graham, T.L.; Graham, M.Y.; Subramanian, S.; Yu, O. RNAi silencing of genes for elicitation or biosynthesis of 5-deoxyisoflavonoids suppresses race-specific resistance and hypersensitive cell death in Phytophthora sojae infected tissues. Plant Physiol 2007, 144, 728–740. [Google Scholar]
- Ibraheem, F.; Gaffoor, I.; Chopra, S. Flavonoid phytoalexin-dependent resistance to anthracnose leaf blight requires a functional yellow seed1 in Sorghum bicolor. Genetics 2010, 184, 915–926. [Google Scholar]
- Shih, C.H.; Chu, I.K.; Yip, W.K.; Lo, C.S. Differential expression of two flavonoid 3’-hydroxylase cDNAs involved in biosynthesis of anthocyanin pigments and 3-deoxyanthocyanidin phytoalexins in sorghum. Plant Cell Physiol 2006, 47, 1412–1419. [Google Scholar]
- Sharma, M.; Chai, C.; Morohashi, K.; Grotewold, E.; Snook, M.E.; Chopra, S. Expression of flavonoid 3′-hydroxylase is controlled by P1, the regulator of 3-deoxyflavonoid biosynthesis in maize. BMC Plant Biol 2012, 12, 196. [Google Scholar]
- Boddu, J.; Svabek, C.; Ibraheem, F.; Jones, A.D.; Chopra, S. Characterization of a deletion allele of a sorghum Myb gene, yellow seed1 showing loss of 3-deoxyflavonoids. Plant Sci 2005, 169, 542–552. [Google Scholar]
- Boddu, J.; Jiang, C.; Sangar, V.; Olson, T.; Peterson, T.; Chopra, S. Comparative structural and functional characterization of sorghum and maize duplications containing orthologous myb transcription regulators of 3-deoxyflavonoid biosynthesis. Plant Mol. Biol 2006, 60, 185–199. [Google Scholar]
- Glazebrook, J.; Ausubel, F.M. Isolation of phytoalexin-deficient mutants of Arabidopsis thaliana and characterization of their interactions with bacterial pathogens. Proc. Natl. Acad. Sci. USA 1994, 91, 8955–8959. [Google Scholar]
- Glazebrook, J.; Zook, M.; Mert, F.; Kagan, I.; Rogers, E.E.; Crute, I.; Holub, E.; Hammerschmidt, R.; Ausubel, F.M. Phytoalexin-deficient mutants of Arabidopsis reveal that PAD4 encodes a regulatory factor and that four PAD genes contribute to downy mildew resistance. Genetics 1997, 146, 381–392. [Google Scholar]
- Reuber, T.L.; Plotnikova, J.M.; Dewdnev, J.; Rogers, E.E.; Wood, W.; Ausubel, F.M. Correlation of defense gene induction defects with powdery mildew susceptibility in Arabidopsis enhanced disease susceptibility mutants. Plant J 1998, 16, 473–485. [Google Scholar]
- Ahl-Goy, P.; Signer, H.; Reist, R.; Aichholz, R.; Blum, B.; Schmidt, E.; Kessmann, H. Accumulation of scopoletin is associated with the high disease resistance of the hybrid Nicotiana tabacum × Nicotiana debneyi. Planta 1993, 191, 200–206. [Google Scholar]
- Gnonlonfin, G.J.B.; Sanni, A.; Brimer, L. Scopoletin—A coumarin phytoalexin with medicinal properties. Crit. Rev. Plant Sci 2012, 31, 47–56. [Google Scholar]
- Li, Y.; Baldauf, S.; Lim, E.K.; Bowles, J.D. Phylogenetic analysis of the UDP-glycosyltransferase multigene family of Arabidopsis thaliana. J. Biol. Chem 2001, 276, 4338–4343. [Google Scholar]
- Chong, J.; Baltz, R.; Schmitt, C.; Beffa, R.; Fritig, B.; Saindrenan, P. Downregulation of a pathogen-responsive tobacco UDP-Glc:phenylpropanoid glucosyltransferase reduces scopoletin glucoside accumulation, enhances oxidative stress, and weakens virus resistance. Plant Cell 2002, 14, 1093–1107. [Google Scholar]
- Gachon, C.; Baltz, R.; Saindrenan, P. Over-expression of a scopoletin glucosyltransferase in Nicotiana tabacum leads to precocious lesion formation during the hypersensitive response to tobacco mosaic virus but does not affect virus resistance. Plant Mol. Biol 2004, 54, 137–146. [Google Scholar]
- Grosskinsky, D.K.; Naseem, M.; Abdelmoshem, U.A.; Plickert, N.; Engelke, T.; Griebel, T.; Zeier, J.; Novak, O.; Strand, M.; Pfeifhofer, H.; et al. Cytokinins mediate resistance against Pseudomonas syringae in tobacco through increased antimicrobial phytoalexin synthesis independent of salicylic acid signaling. Plant Physiol 2011, 157, 815–830. [Google Scholar]
- Mauch-Mani, B.; Mauch, F. The role of abscissic acid in plant-pathogen interactions. Curr. Opin. Plant Biol 2005, 8, 409–414. [Google Scholar]
- Jones, J.D.; Dangles, J.L. The plant immune system. Nature 2006, 444, 323–329. [Google Scholar]
- Robert-Seilaniantz, A.; Navarro, L.; Bari, R.; Jones, J.D. Pathological hormone imbalances. Curr. Opin. Plant Biol 2007, 10, 372–379. [Google Scholar]
- Asselbergh, B.; de Vleesschauwer, D.; Höfte, M. Global switches and fine-tuning-ABA modulates plant pathogen defense. Mol. Plant-Microbe Interact 2008, 21, 709–719. [Google Scholar]
- Lopez, M.A.; Bannenberg, G.; Castresana, C. Controlling hormone signaling is a plant and pathogen challenge for growth and survival. Curr. Opin. Plant Biol 2008, 11, 420–427. [Google Scholar]
- Spoel, S.H.; Dong, X. Making sense of hormone crosstalk during plant immune responses. Cell Host Microbe 2008, 3, 348–351. [Google Scholar]
- Bari, R.; Jones, J.D. Role of plant hormones in plant defence responses. Plant Mol. Biol. 2009, 69, 473–488. [Google Scholar]
- Verhage, A.; van Wees, S.C.M.; Pieterse, C.M.J. Plant immunity: It’s the hormones talking, but what do they say? Plant Physiol 2010, 154, 536–540. [Google Scholar]
- Erb, M.; Meldau, S.; Howe, G.A. Role of phytohormones in insect-specific plant reactions. Trends Plant Sci 2012, 17, 250–259. [Google Scholar]
- Navarro, L.; Dunoyer, P.; Jay, F.; Arnold, B.; Dharmasiri, N.; Estelle, M.; Voinnet, O.; Jones, J.D.G. A plant miRNA contributes to antibacterial resistance by repressing auxin signaling. Science 2006, 312, 436–439. [Google Scholar]
- Domingo, C.; Andrès, F.; Tharreau, D.; Iglesias, D.J.; Talon, M. Constitutive expression of OsGH3.1 reduces auxin content and enhances defense responses and resistance to a fungal pathogen in rice. Mol. Plant-Microbe Interact 2009, 22, 201–210. [Google Scholar]
- Mayda, E.; Marqués, M.C.; Conejero, V.; Vera, P. Expression of a pathogen-induced gene can be mimicked by auxin insensitivity. Mol. Plant-Microbe Interact 2000, 13, 23–31. [Google Scholar]
- Padmanabhan, M.S.; Goregaoker, S.P.; Golem, S.; Shiferaw, H.; Culver, J.N. Interaction of the tobacco mosaic virus replicase protein with the Aux/IAA protein PAP1/IAA26 is associated with disease development. J. Virol 2005, 79, 2549–2558. [Google Scholar]
- Padmanabhan, M.S.; Shiferaw, H.; Culver, J.N. The tobacco mosaic virus replicase protein disrupts the localization and function of interacting Aux/IAA proteins. Mol. Plant-Microbe Interact 2006, 19, 864–873. [Google Scholar]
- Ding, X.; Cao, Y.; Huang, L.; Zhao, J.; Xu, C.; Li, X.; Wang, S. Activation of the indole-3-acetic acid-amido synthetase GH3-8 suppresses expansin expression and promotes salicylate- and jasmonate- independent basal immunity in rice. Plant Cell 2008, 20, 228–240. [Google Scholar]
- Iriti, M.; Faro, F. Chemical diversity and defence metabolism: How plant cope with pathogens and ozone pollution. Int. J. Mol. Sci 2009, 10, 3371–3399. [Google Scholar]
- Ristaino, J.B.; Duniway, J.M. Effect of preinoculation and postinoculation water-stress on the severity of Phytophthora root-rot in processing tomatoes. Plant Dis 1989, 73, 349–352. [Google Scholar]
- Audenaert, K.; de Meyer, G.B.; Höfte, M.M. Abscisic acid determines basal susceptibility of tomato to Botrytis cinerea and suppresses salicylic acid-dependent signaling mechanisms. Plant Physiol 2002, 128, 491–501. [Google Scholar]
- Asselbergh, B.; Achuo, A.E.; Höfte, M.; van Breusegem, F. Abscisic acid deficiency leads to rapid activation of tomato defence responses upon infection with Erwinia chrysanthemi. Mol. Plant Pathol 2008, 9, 11–24. [Google Scholar]
- Balazs, E.; Gaborjanyi, R.; Kiraly, Z. Leaf senescence and increased virus susceptibility in tobacco: The effect of abscisic acid. Physiol. Mol. Plant Pathol 1973, 3, 341–346. [Google Scholar]
- Rezzonico, E.; Flury, N.; Meins, F., Jr; Beffa, R. transcriptional down-regulation by abscisic acid of pathogenesis-related beta-1,3-glucanase genes in tobacco cell cultures. Plant Physiol. 1998, 117, 585–592. [Google Scholar]
- Goosens, J.F.V.; Vendrig, J.C. effects of abscissic acid, cytokinins, and light on isoflavonoid phytoalexin accumulation in Phaseolus vulgaris. Planta 1982, 154, 441–446. [Google Scholar]
- Ward, E.W.; Cahill, D.M.; Bhattacharyya, M.K. Abscisic acid suppression of phenylalanine ammonia-lyase activity and mRNA, and resistance of soybeans to Phytophthora megasperma f.s.p. glycinea. Plant Physiol 1989, 91, 23–27. [Google Scholar]
- Mohr, P.; Cahill, D.M. Relative roles of glyceollin, lignin and the hypersensitive response and the influence of ABA in compatible and incompatible interactions of soybeans with Phytophthora sojae. Physiol. Mol. Plant Pathol 2001, 58, 31–41. [Google Scholar]
- Henfling, J.W.D.M.; Bostock, R.M.; Kuc, J. Effect of abscisic acid on rishitin and lubimin accumulation and resistance to Phytophthora infestans and Cladosporium cucumerinum in potato tuber tissue slices. Phytopathology 1980, 70, 1074–1078. [Google Scholar]
- Mialoundama, A.S.; Heintz, D.; Debayle, D.; Rahier, A.; Camara, B.; Bouvier, F. Abscisic acid negatively regulates elicitor-induced synthesis of capsidiol in wild tobacco. Plant Physiol 2009, 150, 1556–1566. [Google Scholar]
- Thomas, S.G.; Phillips, A.L.; Hedden, P. Molecular cloning and functional expression of gibberellin 2-oxidases, multifunctional enzymes involved in gibberellin deactivation. Proc. Natl. Acad. Sci. USA 1999, 96, 4698–4703. [Google Scholar]
- Yang, D.L.; Li, Q.; Deng, Y.W.; Lou, Y.G.; Wang, M.Y.; Zhou, G.X.; Zhang, Y.Y.; He, Z.H. Altered disease development in the eui mutants and Eui overexpressors indicates that gibberellins negatively regulate rice basal disease resistance. Mol. Plant 2008, 1, 528–537. [Google Scholar]
- Sakakibara, H. Cytokinins: Activity, biosynthesis, and translocation. Annu. Rev. Plant Biol 2006, 57, 431–449. [Google Scholar]
- Sano, H.; Seo, S.; Koizumi, N.; Niki, T.; Iwamura, H.; Ohashi, Y. Regulation by cytokinins of endogenous levels of jasmonic and salicylic acids in mechanically wounded tobacco plants. Plant Cell Physiol 1996, 37, 762–769. [Google Scholar]
- Pogany, M.; Koehl, J.; Heiser, I.; Elstner, E.; Barna, B. Juvenility of tobacco induced by cytokinin gene introduction decreases susceptibility to Tobacco necrosis virus and confers tolerance to oxidative stress. Physiol. Mol. Plant Pathol 2004, 65, 39–47. [Google Scholar]
- Barna, B.; Smigocki, A.C.; Baker, J.C. Transgenic production of cytokinin suppresses bacterially induced hypersensitive response symptoms and increases antioxidative enzyme levels in Nicotiana spp. Phytopathology 2008, 98, 1242–1247. [Google Scholar]
- Choi, J.; Huh, S.U.; Kojima, M.; Sakakibara, H.; Paek, K.H.; Hwang, I. The cytokinin-activated transcription factor A RR2 promotes plant immunity via TGA3/NPR1-dependent salicylic acid signaling in Arabidopsis. Dev. Cell 2010, 19, 284–295. [Google Scholar]
- Boller, T.; Felix, G. A renaissance of elicitors: Perception of microbe-associated molecular patterns and danger signals by pattern-recognition receptors. Annu. Rev. Plant Biol 2009, 60, 379–406. [Google Scholar]
- Dodds, P.N.; Rathjen, J.P. Plant mmunity: Towards an integrated view of plant-pathogen interactions. Nat. Rev. Genet 2010, 11, 539–548. [Google Scholar]
- Newmann, M.A.; Sundelin, T.; Nielsen, J.T.; Erbs, G. MAMP (microbe-associated molecular pattern) triggered immunity in plants. Front. Plant Sci 2013, 4, 139. [Google Scholar]
- Pitzschke, A.; Schikora, A.; Hirt, H. MAPK cascade signaling networks in plant defence. Curr. Opin. Plant Biol 2009, 12, 421–426. [Google Scholar]
- Garcia-Brugger, A.; Lamotte, O.; Vandelle, E.; Bourque, S.; Lecourieux, D.; Poinssot, B.; Wendehenne, D.; Pugin, A. Early signaling events induced by elicitors of plant defenses. Mol. Plant-Microbe Interact 2006, 19, 711–724. [Google Scholar]
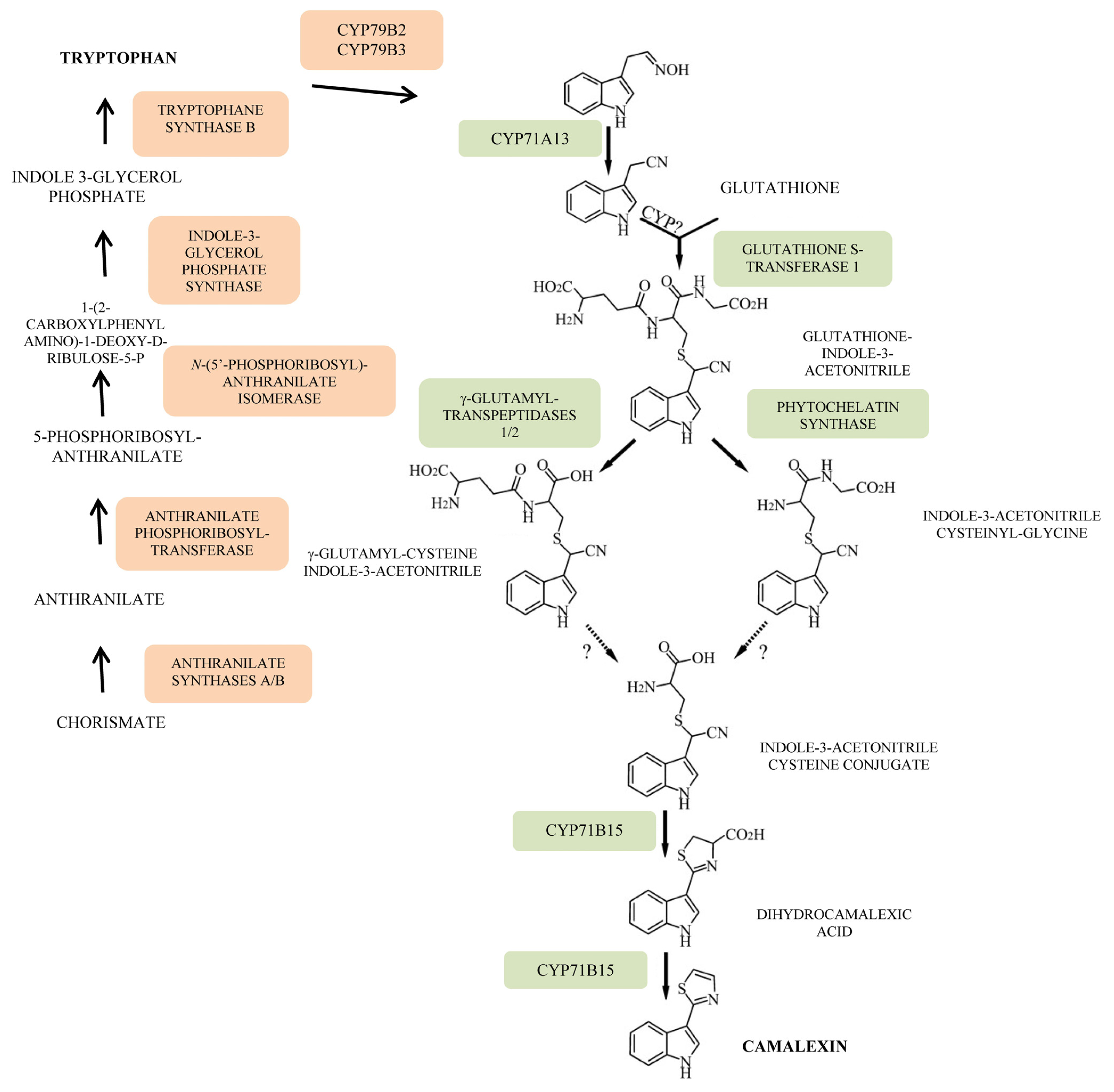
- Zhao, J.; Last, R.L. Coordinate regulation of the tryptophan biosynthetic pathway and indolic phytoalexin accumulation in Arabidopsis. Plant Cell 1996, 8, 2235–2244. [Google Scholar]
- Su, T.; Xu, J.; Li, Y.; Lei, L.; Zhao, L.; Yang, H.; Feng, J.; Liu, G.; Ren, D. Glutathione-indole-3-acetonitrile is required for camalexin biosynthesis in Arabidopsis thaliana. Plant Cell 2011, 23, 364–380. [Google Scholar]
- Kim, J.A.; Cho, K.; Singh, R.; Jung, Y.H.; Jeong, S.H.; Kim, S.H.; Lee, J.E.; Cho, Y.S.; Agrawal, G.K.; Rakwal, R.; et al. Rice OsACDR1 (Oryza sativa accelerated cell death and resistance 1) is a potential positive regulator of fungal disease resistance. Mol. Cells 2009, 28, 431–439. [Google Scholar]
- Grant, M.; Lamb, C. Systemic immunity. Curr. Opin. Plant Biol 2006, 9, 414–420. [Google Scholar]
- Cao, H.; Li, X.; Dong, X. Generation of broad-spectrum disease resistance by overexpression of an essential regulatory gene in systemic acquired resistance. Proc. Natl. Acad. Sci. USA 1998, 95, 6531–6536. [Google Scholar]
- Quilis, J.; Penas, G.; Messeguer, J.; Brugidou, C.; San Segundo, B. The Arabidopsis AtNPR1 inversely modulates defense responses against fungal, bacterial, or viral pathogens while conferring hypersensitivity to abiotic stresses in transgenic rice. Mol. Plant-Microbe Interact 2008, 21, 1215–1231. [Google Scholar]
- Lin, W.C.; Lu, C.F.; Wu, J.W.; Cheng, M.L.; Lin, Y.M.; Yang, N.S.; Black, L.; Green, S.K.; Wang, J.F.; Cheng, C.P. Transgenic tomato plants expressing the Arabidopsis NPR1 gene display enhanced resistance to a spectrum of fungal and bacterial diseases. Transgenic Res 2004, 13, 567–581. [Google Scholar]
- Makandar, R.; Essig, J.S.; Schapaugh, M.A.; Trick, H.N.; Shah, J. Genetically engineered resistance to Fusarium head blight in wheat by expression of Arabidopsis NPR1. Mol. Plant-Microbe Interact 2006, 19, 123–129. [Google Scholar]
- Wally, O.; Jayaraj, J.; Punja, Z.K. Broad-spectrum disease resistance to necrotrophic and biotrophic pathogens in trnsgnic carrots (Daucus carota L.) expressing an Arabidopsis NPR1 gene. Planta 2009, 231, 131–141. [Google Scholar]
- Parkhi, V.; Kumar, V.; Campbell, L.M.; Bell, A.A.; Shah, J.; Rathore, K.S. Resistance against various fungal pathogens and reniform nematode in transgenic cotton plants expressing Arabidopsis NRP1. Transgenic Res 2010, 19, 959–975. [Google Scholar]
- Berken, A. ROPs in the spotlight of plant signal transduction. Cell. Mol. Life Sci 2006, 63, 2446–2459. [Google Scholar]
- Ono, E.; Wong, H.L.; Kawasaki, T.; Hasegawa, M.; Kodama, O.; Shimamoto, K. Essential role of the small GTPase Rac in disease resistance of rice. Proc. Natl. Acad. Sci. USA 2001, 98, 759–764. [Google Scholar]
- Kawano, Y.; Akamatsu, A.; Hayashi, K.; Housen, Y.; Okuda, J.; Yao, A.; Nakashima, A.; Takahashi, H.; Yoshida, H.; Wong, H.L.; et al. Activation of a Rac GTPase by the NLR family disease resistance protein pit plays a critical role in rice innate immunity. Cell Host Microbe 2010, 7, 362–375. [Google Scholar]
- Chen, L.; Shiotani, K.; Togashi, T.; Miki, D.; Aoyama, M.; Wong, H.L.; Kawasaki, T.; Shimamoto, K. Analysis of the Rac/Rop small GTPase family in rice: Expression, subcellular localization and role in disease resistance. Plant Cell Physiol 2010, 51, 85–95. [Google Scholar]
- Sadawa, K.; Hasegawa, M.; Tokuda, L.; Kameyama, J.; Kodama, O.; Kohchi, T.; Yoshida, K.; Shinmyo, A. Enhanced resistance to blast fungus and bacterial blight in transgenic rice constitutively expressing OsSBP, a rice homologue of mamalian selenium-binding proteins. Biosci. Biotechnol. Biochem 2004, 68, 873–880. [Google Scholar]
- Flemetakis, E.; Agalou, A.; Kavroulakis, N.; Dimou, M.; Martsikovskaya, A.; Slater, A.; Spaink, H.P.; Roussis, A.; Katinakis, P. Lotus japonicus gene Ljsbp is highly conserved among plants and animals and encodes a homologue to the mamalian selenium-binding proteins. Mol. Plant-Microbe Interact 2002, 15, 313–322. [Google Scholar]
- Sawada, K.; Tokuda, L.; Shinmyo, A. Characterization of the rice blast fungal elicitor-responsive gene OsSBP encoding a homolog to the mamalian selenium-binding proteins. Plant Biotechnol 2003, 20, 179–183. [Google Scholar]
- Dodd, A.N.; Kudla, J.; Sanders, D. The language of calcium signalling. Annu. Rev. Plant Biol 2010, 61, 593–620. [Google Scholar]
- Reddy, A.S.; Ali, G.S.; Celesnik, H.; Day, I.S. Coping with stresses: Roles of calcium- and calcium/calmodulin-regulated gene expression. Plant Cell 2011, 23, 2010–2032. [Google Scholar]
- Romeis, T.; Ludwig, A.; Martin, R.; Jones, J.D.G. Calcium-dependent protein kinases play an essential role in a plant defence response. EMBO J 2001, 20, 5556–5567. [Google Scholar]
- Luan, S. The CBL-CIPK network in plant calcium signaling. Trends Plant Sci 2009, 14, 37–42. [Google Scholar]
- Kurusu, T.; Hamada, J.; Nokajima, H.; Kitagawa, Y.; Kiyoduka, M.; Takahashi, A.; Hanamata, S.; Ohno, R.; Hayashi, T.; Okada, K.; et al. Regulation of microbe-associated molecular pattern-induced hypersensitive cell death, phytoalexin production, and defense gene expression by calcineurin B-like protein-interacting protein kinases, OsCIPK14/15, in rice cultured cells. Plant Physiol 2010, 153, 678–692. [Google Scholar]
- Okada, K. The biosynthesis of isoprenoids and the mechanisms regulating it in plants. Biosci. Biotechnol. Biochem 2011, 75, 1219–1225. [Google Scholar]
- Rauhut, T.; Luberacki, B.; Seitz, H.U.; Glawischnig, E. Inducible expression of a Nep1-like protein serves as a model trigger system of camalexin biosynthesis. Phytochemistry 2009, 70, 185–189. [Google Scholar]
- Pemberton, C.L.; Salmond, G.P.C. The Nep1-like proteins—A growing family of microbial elicitors of plant necrosis. Mol. Plant Pathol 2004, 5, 353–359. [Google Scholar]
- Qutob, D.; Kemmerling, B.; Brunner, F.; Küfner, L.; Engelhardt, S.; Gust, A.A.; Luberacki, B.; Seitz, H.U.; Stahl, D.; Rauhut, T.; et al. Phytotoxicity and innate immune responses induced by Nep1-like proteins. Plant Cell 2006, 18, 3721–3744. [Google Scholar]
- Mori, M.; Tomita, C.; Sugimoto, K.; Hasegawa, M.; Hayashi, N.; Dubouzet, J.G.; Ochiai, H.; Sekimoto, H.; Hirochika, H.; Kikuchi, S. Isolation and molecular characterization of a Spotted leaf 18 mutant by modified activation-tagging in rice. Plant Mol. Biol 2007, 63, 847–860. [Google Scholar]
- Kachroo, A.; Lapchyk, L.; Fukushige, H.; Hildebrand, D.; Klessig, D.; Kachroo, P. Plastidial fatty acid signaling modulates salicylic acid-and jasmonic acid-mediated defense pathways in the Arabidopsis ssi2 mutant. Plant Cell 2003, 15, 2952–2965. [Google Scholar]
- Kachroo, A.; Venugopal, S.C.; Lapchyk, L.; Falcone, D.; Hildebrand, D.; Kachroo, P. Oleic acid levels regulated by glycerolipid metabolism modulate defense gene expression in Arabidopsis. Proc. Natl. Acad. Sci. USA 2004, 101, 5152–5157. [Google Scholar]
- Matthews, D.; Jones, H.; Gans, P.; Coates, S.; Smith, L.M.J. Toxic secondary metabolite production in genetically modified potatoes in response to stress. J. Agric. Food Chem 2005, 53, 7766–7776. [Google Scholar]
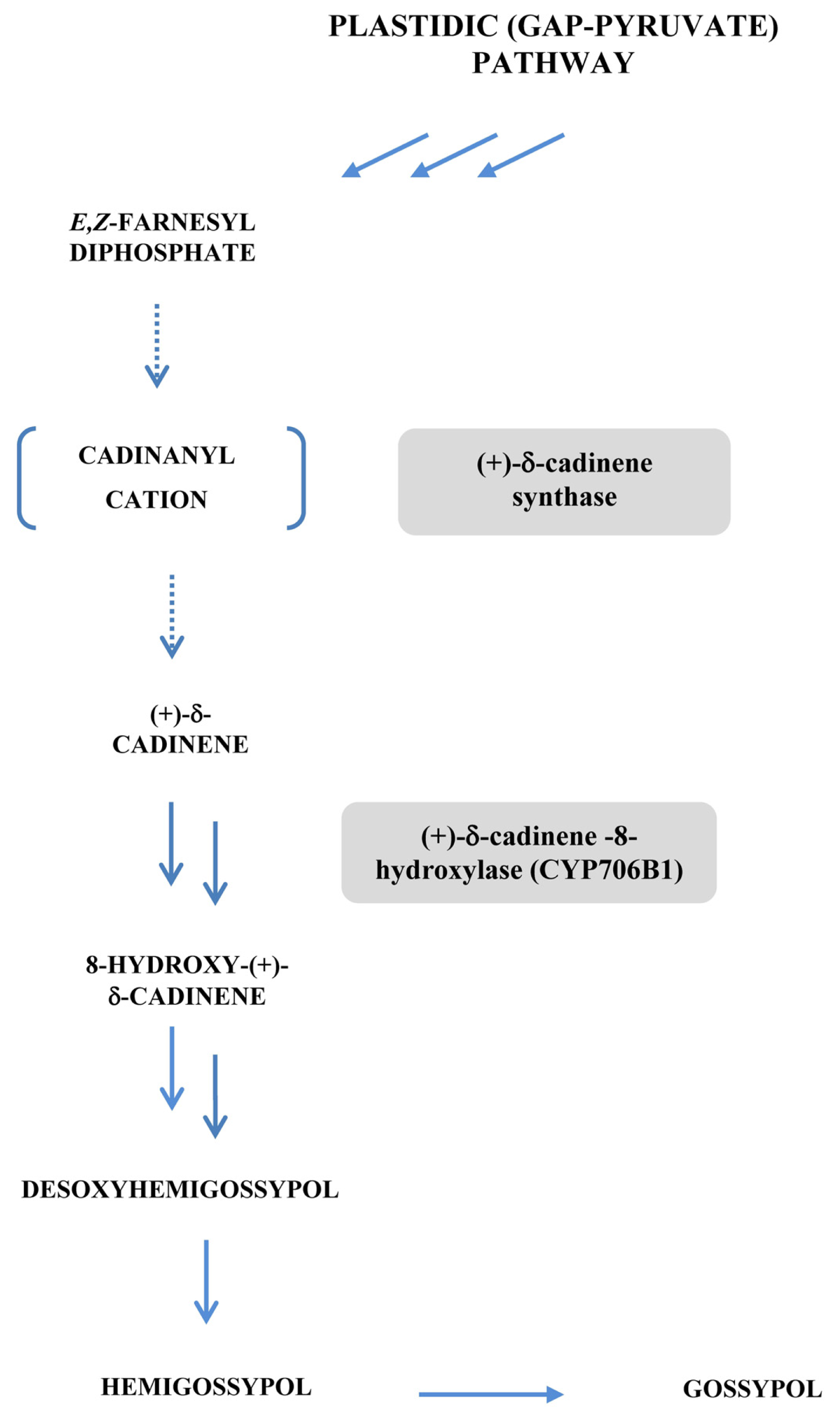
- Benedict, C.R.; Lu, J.L.; Pettigrew, D.W.; Liu, J.; Stipanovic, R.D.; Williams, H.J. The cyclization of farnesyl diphosphate and nerolidyl diphosphate by a purified recombinant δ-cadinene synthase. Plant Physiol 2001, 125, 1754–1765. [Google Scholar]
- Martin, G.S.; Liu, J.; Benedict, C.R.; Stipanovic, R.D.; Magill, W.M. Reduced levels of cadinane sesquiterpenoids in cotton plants expressing antisense (+)-δ-cadinene synthase. Phytochemistry 2003, 62, 31–38. [Google Scholar]
- Chen, X.Y.; Chen, Y.; Heinstein, P.; Davisson, V.J. Cloning, expression, and characterization of (+)-δ-cadinene synthase: A catalyst for cotton phytoalexin biosynthesis. Arch. Biochem. Biophys 1995, 324, 255–266. [Google Scholar]
- Chen, X.Y.; Wang, M.; Chen, Y.; Davisson, V.J.; Heinstein, P. Cloning and heterologous expression, and characterization of a second (+)-δ-cadinene synthase from Gossypium arboreum. J. Nat. Prod 1996, 59, 944–951. [Google Scholar]
- Kalani, R.; Gamboa, D.A.; Calhoun, M.C.; Haq, A.U.; Bailey, C.A. Relative toxicity of cottonseed gossypol enantiomers in broilers. Open Toxicol. J 2010, 4, 26–31. [Google Scholar]
- Townsend, B.J.; Poole, A.; Blake, C.J.; Llewellyn, D.J. Antisense suppression of a (+)-δ-cadinene synthase gene in cotton prevents the induction of this defense response gene during blight infection but not its constitutive expression. Plant Physiol 2005, 138, 516–528. [Google Scholar]
- Benedict, C.R.; Martin, G.S.; Liu, J.; Puckhaber, L.; Magill, C.W. Terpenoid aldehyde formation and lysigenous gland storage sites in cotton: Variant with mature glands but suppressed levels of terpenoid aldehydes. Phytochemistry 2004, 65, 1351–1359. [Google Scholar]
- Sunilkumar, G.; Campbell, L.M.; Pukhaber, L.; Stipanovic, R.D.; Rathore, K.S. Engineering cottonseed for use in human nutrition by tissue-specific reduction of toxic gossypol. Proc. Natl Acad. Sci. USA 2006, 103, 18054–18059. [Google Scholar]
- Sunilkumar, G.; Connell, J.P.; Smith, C.W.; Reddy, A.S.; Rathore, K.S. Cotton α-globulin promoter: Isolation and functional characterization in transgenic cotton, Arabidospsis, and tobacco. Transgenic Res 2002, 11, 347–359. [Google Scholar]
- Rathore, K.S.; Sunilkumar, G.; Campbell, L.M. Cotton plant with seed specific reduction in gossypol. US Patent N° 20070199098 2007. [Google Scholar]

© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Jeandet, P.; Clément, C.; Courot, E.; Cordelier, S. Modulation of Phytoalexin Biosynthesis in Engineered Plants for Disease Resistance. Int. J. Mol. Sci. 2013, 14, 14136-14170. https://doi.org/10.3390/ijms140714136
Jeandet P, Clément C, Courot E, Cordelier S. Modulation of Phytoalexin Biosynthesis in Engineered Plants for Disease Resistance. International Journal of Molecular Sciences. 2013; 14(7):14136-14170. https://doi.org/10.3390/ijms140714136
Chicago/Turabian StyleJeandet, Philippe, Christophe Clément, Eric Courot, and Sylvain Cordelier. 2013. "Modulation of Phytoalexin Biosynthesis in Engineered Plants for Disease Resistance" International Journal of Molecular Sciences 14, no. 7: 14136-14170. https://doi.org/10.3390/ijms140714136