Regulation of miRNA Expression by Low-Level Laser Therapy (LLLT) and Photodynamic Therapy (PDT)
Abstract
:1. Low-Level Laser Therapy (LLLT) and Its Effects on miRNA Expression
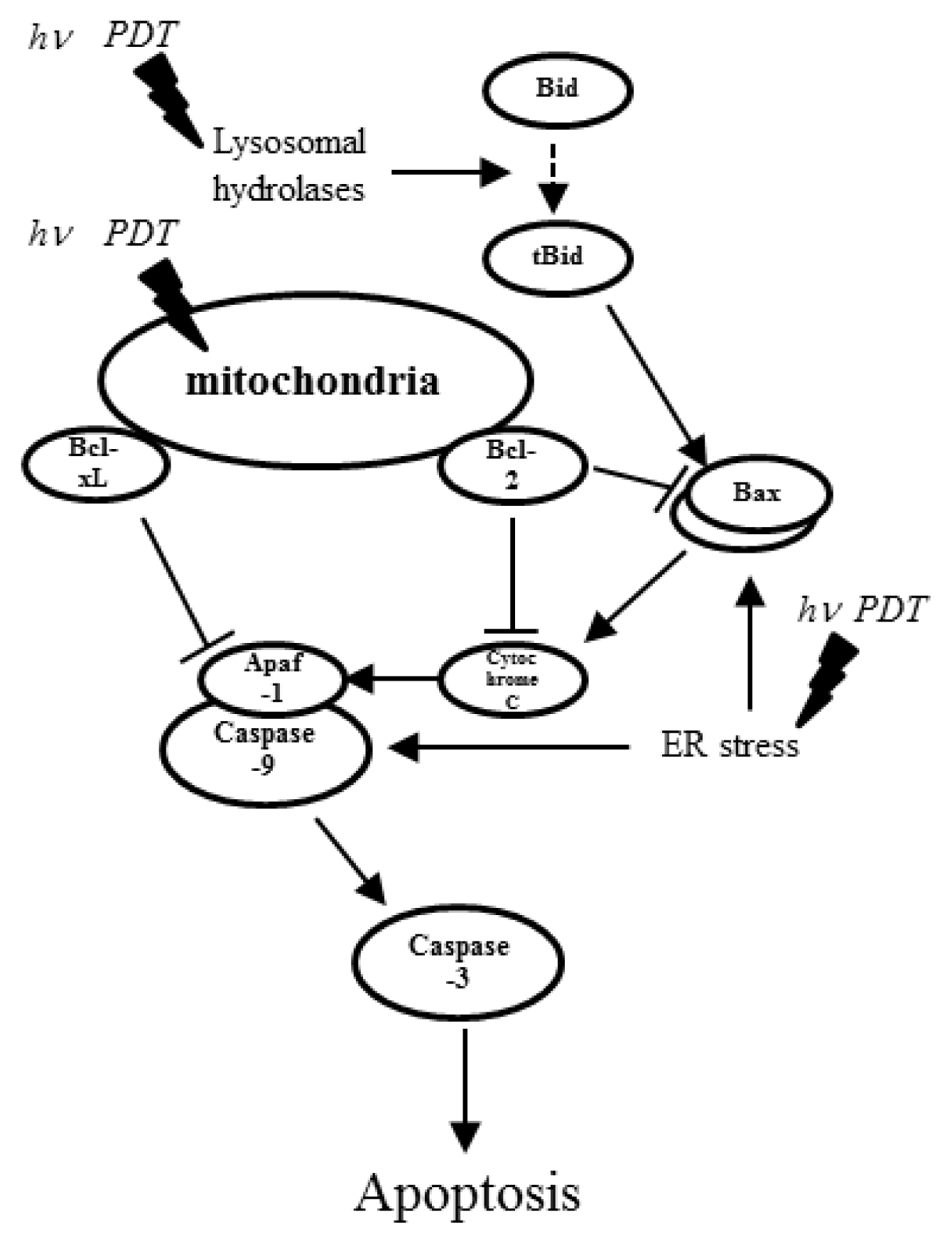
2. Photodynamic Therapy and Its Effects on miRNA Expression
3. Conclusions
Acknowledgments
Conflict of Interest
References
- Maiman, T.H. Stimulated optical radiation in ruby. Nature 1960, 187, 493–494. [Google Scholar]
- Khatri, K.A.; Mahoney, D.L.; McCartney, M.J. Laser scar revision: A review. J. Cosmet. Laser Ther 2011, 13, 54–62. [Google Scholar]
- Chung, S.H.; Mazur, E. Surgical applications of femtosecond lasers. J. Biophotonics 2009, 2, 557–572. [Google Scholar]
- Zhao, Z.; Wu, F. Minimally-invasive thermal ablation of early-stage breast cancer: A systemic review. Eur. J. Surg. Oncol 2010, 36, 1149–1155. [Google Scholar]
- Siribumrungwong, B.; Noorit, P.; Wilasrusmee, C.; Attia, J.; Thakkinstian, A. A systematic review and meta-analysis of randomised controlled trials comparing endovenous ablation and surgical intervention in patients with varicose vein. Eur. J. Vasc. Endovasc. Surg 2012, 44, 214–223. [Google Scholar]
- Vuylsteke, M.E.; Mordon, S.R. Endovenous laser ablation: A review of mechanisms of action. Ann. Vasc. Surg 2012, 26, 424–433. [Google Scholar]
- Vogel, A.; Venugopalan, V. Mechanisms of pulsed laser ablation of biological tissues. Chem. Rev 2003, 103, 577–644. [Google Scholar]
- Casas, A.; di Venosa, G.; Hasan, T.; Al, B. Mechanisms of resistance to photodynamic therapy. Curr. Med. Chem 2011, 18, 2486–2515. [Google Scholar]
- Anand, S.; Ortel, B.J.; Pereira, S.P.; Hasan, T.; Maytin, E.V. Biomodulatory approaches to photodynamic therapy for solid tumors. Cancer Lett 2012, 326, 8–16. [Google Scholar]
- Mester, E.; Szende, B.; Gartner, P. The effect of laser beams on the growth of hair in mice. Radiobiol. Radiother 1968, 9, 621–626. [Google Scholar]
- Mester, E.; Spiry, T.; Szende, B.; Tota, J.G. Effect of laser rays on wound healing. Am. J. Surg 1971, 122, 532–535. [Google Scholar]
- Da Silva, J.P.; da Silva, M.A.; Almeida, A.P.; Lombardi, I., Junior; Matos, A.P. Laser therapy in the tissue repair process: A literature review. Photomed. Laser Surg 2010, 28, 17–21. [Google Scholar]
- Buravlev, E.A.; Zhidkova, T.V.; Vladimirov, Y.A.; Osipov, A.N. Effects of laser and led radiation on mitochondrial respiration in experimental endotoxic shock. Lasers Med. Sci 2012, 28, 785–790. [Google Scholar]
- De Sousa, A.P.; Santos, J.N.; dos Reis, J.A., Jr; Ramos, T.A.; de Souza, J.; Cangussu, M.C.; Pinheiro, A.L. Effect of led phototherapy of three distinct wavelengths on fibroblasts on wound healing: A histological study in a rodent model. Photomed. Laser Surg. 2010, 28, 547–552. [Google Scholar]
- Lev-Tov, H.; Brody, N.; Siegel, D.; Jagdeo, J. Inhibition of fibroblast proliferation in vitro using low-level infrared light-emitting diodes. Dermatol. Surg 2012, 39, 422–425. [Google Scholar]
- Nishioka, M.A.; Pinfildi, C.E.; Sheliga, T.R.; Arias, V.E.; Gomes, H.C.; Ferreira, L.M. Led (660 nm) and laser (670 nm) use on skin flap viability: Angiogenesis and mast cells on transition line. Lasers Med. Sci 2012, 27, 1045–1050. [Google Scholar]
- Pinheiro, A.L.; Soares, L.G.; Cangussu, M.C.; Santos, N.R.; Barbosa, A.F.; Silveira, L., Junior. Effects of led phototherapy on bone defects grafted with mta, bone morphogenetic proteins and guided bone regeneration: A raman spectroscopic study. Lasers Med. Sci 2012, 27, 903–916. [Google Scholar]
- AlGhamdi, K.M.; Kumar, A.; Moussa, N.A. Low-level laser therapy: A useful technique for enhancing the proliferation of various cultured cells. Lasers Med. Sci 2012, 27, 237–249. [Google Scholar]
- Gao, X.; Xing, D. Molecular mechanisms of cell proliferation induced by low power laser irradiation. J. Biomed. Sci 2009, 16, 4. [Google Scholar]
- Tafur, J.; van Wijk, E.P.; van Wijk, R.; Mills, P.J. Biophoton detection and low-intensity light therapy: A potential clinical partnership. Photomed. Laser Surg 2010, 28, 23–30. [Google Scholar]
- Gavish, L.; Perez, L.S.; Reissman, P.; Gertz, S.D. Irradiation with 780 nm diode laser attenuates inflammatory cytokines but upregulates nitric oxide in lipopolysaccharide-stimulated macrophages: Implications for the prevention of aneurysm progression. Lasers Surg. Med 2008, 40, 371–378. [Google Scholar]
- Lindgard, A.; Hulten, L.M.; Svensson, L.; Soussi, B. Irradiation at 634 nm releases nitric oxide from human monocytes. Lasers Med. Sci 2007, 22, 30–36. [Google Scholar]
- Moriyama, Y.; Moriyama, E.H.; Blackmore, K.; Akens, M.K.; Lilge, L. In vivo study of the inflammatory modulating effects of low-level laser therapy on inos expression using bioluminescence imaging. Photochem. Photobiol 2005, 81, 1351–1355. [Google Scholar]
- Moriyama, Y.; Nguyen, J.; Akens, M.; Moriyama, E.H.; Lilge, L. In vivo effects of low level laser therapy on inducible nitric oxide synthase. Lasers Surg. Med 2009, 41, 227–231. [Google Scholar]
- Tuby, H.; Maltz, L.; Oron, U. Modulations of vegf and inos in the rat heart by low level laser therapy are associated with cardioprotection and enhanced angiogenesis. Lasers Surg. Med 2006, 38, 682–688. [Google Scholar]
- Fukuda, T.Y.; Tanji, M.M.; Silva, S.R.; Sato, M.N.; Plapler, H. Infrared low-level diode laser on inflammatory process modulation in mice: Pro- and anti-inflammatory cytokines. Lasers Med. Sci. 2012. [Google Scholar] [CrossRef]
- Oliveira, R.G.; Ferreira, A.P.; Cortes, A.J.; Aarestrup, B.J.; Andrade, L.C.; Aarestrup, F.M. Low-level laser reduces the production of tnf-alpha, ifn-gamma, and il-10 induced by ova. Lasers Med. Sci. 2013. [Google Scholar] [CrossRef]
- Yamaura, M.; Yao, M.; Yaroslavsky, I.; Cohen, R.; Smotrich, M.; Kochevar, I.E. Low level light effects on inflammatory cytokine production by rheumatoid arthritis synoviocytes. Lasers Surg. Med 2009, 41, 282–290. [Google Scholar]
- Aimbire, F.; Ligeiro de Oliveira, A.P.; Albertini, R.; Correa, J.C.; Ladeira de Campos, C.B.; Lyon, J.P.; Silva, J.A., Jr; Costa, M.S. Low level laser therapy (lllt) decreases pulmonary microvascular leakage, neutrophil influx and il-1beta levels in airway and lung from rat subjected to lps-induced inflammation. Inflammation 2008, 31, 189–197. [Google Scholar]
- Safavi, S.M.; Kazemi, B.; Esmaeili, M.; Fallah, A.; Modarresi, A.; Mir, M. Effects of low-level he-ne laser irradiation on the gene expression of il-1beta, tnf-alpha, ifn-gamma, tgf-beta, bfgf, and pdgf in rat’s gingiva. Lasers Med. Sci 2008, 23, 331–335. [Google Scholar]
- Boschi, E.S.; Leite, C.E.; Saciura, V.C.; Caberlon, E.; Lunardelli, A.; Bitencourt, S.; Melo, D.A.; Oliveira, J.R. Anti-inflammatory effects of low-level laser therapy (660 nm) in the early phase in carrageenan-induced pleurisy in rat. Lasers Surg. Med 2008, 40, 500–508. [Google Scholar]
- Shiba, H.; Tsuda, H.; Kajiya, M.; Fujita, T.; Takeda, K.; Hino, T.; Kawaguchi, H.; Kurihara, H. Neodymium-doped yttrium-aluminium-garnet laser irradiation abolishes the increase in interleukin-6 levels caused by peptidoglycan through the p38 mitogen-activated protein kinase pathway in human pulp cells. J. Endod 2009, 35, 373–376. [Google Scholar]
- Houreld, N.N.; Sekhejane, P.R.; Abrahamse, H. Irradiation at 830 nm stimulates nitric oxide production and inhibits pro-inflammatory cytokines in diabetic wounded fibroblast cells. Lasers Surg. Med 2010, 42, 494–502. [Google Scholar]
- Simunovic-Soskic, M.; Pezelj-Ribaric, S.; Brumini, G.; Glazar, I.; Grzic, R.; Miletic, I. Salivary levels of tnf-alpha and il-6 in patients with denture stomatitis before and after laser phototherapy. Photomed. Laser Surg 2010, 28, 189–193. [Google Scholar]
- Fushimi, T.; Inui, S.; Nakajima, T.; Ogasawara, M.; Hosokawa, K.; Itami, S. Green light emitting diodes accelerate wound healing: Characterization of the effect and its molecular basis in vitro and in vivo. Wound Repair Regen 2012, 20, 226–235. [Google Scholar]
- Saygun, I.; Karacay, S.; Serdar, M.; Ural, A.U.; Sencimen, M.; Kurtis, B. Effects of laser irradiation on the release of basic fibroblast growth factor (bfgf), insulin like growth factor-1 (igf-1), and receptor of igf-1 (igfbp3) from gingival fibroblasts. Lasers Med. Sci 2008, 23, 211–215. [Google Scholar]
- Schwartz, F.; Brodie, C.; Appel, E.; Kazimirsky, G.; Shainberg, A. Effect of helium/neon laser irradiation on nerve growth factor synthesis and secretion in skeletal muscle cultures. J. Photochem. Photobiol. B 2002, 66, 195–200. [Google Scholar]
- Yu, W.; Naim, J.O.; Lanzafame, R.J. The effect of laser irradiation on the release of bfgf from 3t3 fibroblasts. Photochem. Photobiol 1994, 59, 167–170. [Google Scholar]
- Hu, W.P.; Wang, J.J.; Yu, C.L.; Lan, C.C.; Chen, G.S.; Yu, H.S. Helium-neon laser irradiation stimulates cell proliferation through photostimulatory effects in mitochondria. J. Invest. Dermatol 2007, 127, 2048–2057. [Google Scholar]
- Lan, C.C.; Wu, C.S.; Chiou, M.H.; Chiang, T.Y.; Yu, H.S. Low-energy helium-neon laser induces melanocyte proliferation via interaction with type iv collagen: Visible light as a therapeutic option for vitiligo. Br. J. Dermatol 2009, 161, 273–280. [Google Scholar]
- Wu, S.; Xing, D.; Gao, X.; Chen, W.R. High fluence low-power laser irradiation induces mitochondrial permeability transition mediated by reactive oxygen species. J. Cell. Physiol 2009, 218, 603–611. [Google Scholar]
- Zungu, I.L.; Hawkins Evans, D.; Abrahamse, H. Mitochondrial responses of normal and injured human skin fibroblasts following low level laser irradiation—An in vitro study. Photochem. Photobiol 2009, 85, 987–996. [Google Scholar]
- Karu, T. Photobiology of low-power laser effects. Health Phys 1989, 56, 691–704. [Google Scholar]
- Karu, T.I. Mitochondrial signaling in mammalian cells activated by red and near-ir radiation. Photochem. Photobiol 2008, 84, 1091–1099. [Google Scholar]
- Tiphlova, O.; Karu, T. Role of primary photoacceptors in low-power laser effects: Action of he-ne laser radiation on bacteriophage t4-escherichia coli interaction. Lasers Surg. Med 1989, 9, 67–69. [Google Scholar]
- Zhang, L.; Xing, D.; Zhu, D.; Chen, Q. Low-power laser irradiation inhibiting abeta25–35-induced pc12 cell apoptosis via pkc activation. Cell. Physiol. Biochem 2008, 22, 215–222. [Google Scholar]
- Aimbire, F.; Santos, F.V.; Albertini, R.; Castro-Faria-Neto, H.C.; Mittmann, J.; Pacheco-Soares, C. Low-level laser therapy decreases levels of lung neutrophils anti-apoptotic factors by a nf-κ dependent mechanism. Int. Immunopharmacol 2008, 8, 603–605. [Google Scholar]
- Kushibiki, T.; Hirasawa, T.; Okawa, S.; Ishihara, M. Blue laser irradiation generates intracellular reactive oxygen species in various types of cells. Photomed. Laser Surg 2013, 31, 95–104. [Google Scholar]
- Lipovsky, A.; Nitzan, Y.; Lubart, R. A possible mechanism for visible light-induced wound healing. Lasers Surg. Med 2008, 40, 509–514. [Google Scholar]
- Ignatieva, N.; Zakharkina, O.; Andreeva, I.; Sobol, E.; Kamensky, V.; Lunin, V. Effects of laser irradiation on collagen organization in chemically induced degenerative annulus fibrosus of lumbar intervertebral disc. Lasers Surg. Med 2008, 40, 422–432. [Google Scholar]
- Silveira, L.B.; Prates, R.A.; Novelli, M.D.; Marigo, H.A.; Garrocho, A.A.; Amorim, J.C.; Sousa, G.R.; Pinotti, M.; Ribeiro, M.S. Investigation of mast cells in human gingiva following low-intensity laser irradiation. Photomed. Laser Surg 2008, 26, 315–321. [Google Scholar]
- Coombe, A.R.; Ho, C.T.; Darendeliler, M.A.; Hunter, N.; Philips, J.R.; Chapple, C.C.; Yum, L.W. The effects of low level laser irradiation on osteoblastic cells. Clin. Orthod. Res 2001, 4, 3–14. [Google Scholar]
- Kushibiki, T.; Awazu, K. Controlling osteogenesis and adipogenesis of mesenchymal stromal cells by regulating a circadian clock protein with laser irradiation. Inter. J. Med. Sci 2008, 5, 319–326. [Google Scholar]
- Kushibiki, T.; Awazu, K. Blue laser irradiation enhances extracellular calcification of primary mesenchymal stem cells. Photomed. Laser Surg 2009, 27, 493–498. [Google Scholar]
- Kushibiki, T.; Tajiri, T.; Ninomiya, Y.; Awazu, K. Chondrogenic mrna expression in prechondrogenic cells after blue laser irradiation. J. Photochem. Photobiol. B 2010, 98, 211–215. [Google Scholar]
- Wang, J.; Huang, W.; Wu, Y.; Hou, J.; Nie, Y.; Gu, H.; Li, J.; Hu, S.; Zhang, H. Microrna-193 pro-proliferation effects for bone mesenchymal stem cells after low-level laser irradiation treatment through inhibitor of growth family, member 5. Stem Cells Dev 2012, 21, 2508–2519. [Google Scholar]
- Gu, X.; Nylander, E.; Coates, P.J.; Nylander, K. Effect of narrow-band ultraviolet b phototherapy on p63 and microrna (mir-21 and mir-125b) expression in psoriatic epidermis. Acta Derm. Venereol 2011, 91, 392–397. [Google Scholar]
- Tome, M.; Lopez-Romero, P.; Albo, C.; Sepulveda, J.C.; Fernandez-Gutierrez, B.; Dopazo, A.; Bernad, A.; Gonzalez, M.A. Mir-335 orchestrates cell proliferation, migration and differentiation in human mesenchymal stem cells. Cell Death Differ 2011, 18, 985–995. [Google Scholar]
- Bouvet-Gerbettaz, S.; Merigo, E.; Rocca, J.P.; Carle, G.F.; Rochet, N. Effects of low-level laser therapy on proliferation and differentiation of murine bone marrow cells into osteoblasts and osteoclasts. Lasers Surg. Med 2009, 41, 291–297. [Google Scholar]
- Da Silva, A.P.; Petri, A.D.; Crippa, G.E.; Stuani, A.S.; Stuani, A.S.; Rosa, A.L.; Stuani, M.B. Effect of low-level laser therapy after rapid maxillary expansion on proliferation and differentiation of osteoblastic cells. Lasers Med. Sci 2012, 27, 777–783. [Google Scholar]
- Ebrahimi, T.; Moslemi, N.; Rokn, A.; Heidari, M.; Nokhbatolfoghahaie, H.; Fekrazad, R. The influence of low-intensity laser therapy on bone healing. J. Dent 2012, 9, 238–248. [Google Scholar]
- Fujimoto, K.; Kiyosaki, T.; Mitsui, N.; Mayahara, K.; Omasa, S.; Suzuki, N.; Shimizu, N. Low-intensity laser irradiation stimulates mineralization via increased bmps in mc3t3-e1 cells. Lasers Surg. Med 2010, 42, 519–526. [Google Scholar]
- Hou, J.F.; Zhang, H.; Yuan, X.; Li, J.; Wei, Y.J.; Hu, S.S. In vitro effects of low-level laser irradiation for bone marrow mesenchymal stem cells: Proliferation, growth factors secretion and myogenic differentiation. Lasers Surg. Med 2008, 40, 726–733. [Google Scholar]
- Kim, H.; Choi, K.; Kweon, O.K.; Kim, W.H. Enhanced wound healing effect of canine adipose-derived mesenchymal stem cells with low-level laser therapy in athymic mice. J. Dermatol. Sci 2012, 68, 149–156. [Google Scholar]
- Lin, F.; Josephs, S.F.; Alexandrescu, D.T.; Ramos, F.; Bogin, V.; Gammill, V.; Dasanu, C.A.; de Necochea-Campion, R.; Patel, A.N.; Carrier, E.; et al. Lasers, stem cells, and copd. J. Transl. Med 2010, 8, 16. [Google Scholar]
- Luo, L.; Sun, Z.; Zhang, L.; Li, X.; Dong, Y.; Liu, T.C. Effects of low-level laser therapy on ros homeostasis and expression of igf-1 and tgf-beta1 in skeletal muscle during the repair process. Lasers Med. Sci 2013, 28, 725–734. [Google Scholar]
- Medrado, A.P.; Soares, A.P.; Santos, E.T.; Reis, S.R.; Andrade, Z.A. Influence of laser photobiomodulation upon connective tissue remodeling during wound healing. J. Photochem. Photobiol. B 2008, 92, 144–152. [Google Scholar]
- Nogueira, G.T.; Mesquita-Ferrari, R.A.; Souza, N.H.; Artilheiro, P.P.; Albertini, R.; Bussadori, S.K.; Fernandes, K.P. Effect of low-level laser therapy on proliferation, differentiation, and adhesion of steroid-treated osteoblasts. Lasers Med. Sci 2012, 27, 1189–1193. [Google Scholar]
- Renno, A.C.; McDonnell, P.A.; Parizotto, N.A.; Laakso, E.L. The effects of laser irradiation on osteoblast and osteosarcoma cell proliferation and differentiation in vitro. Photomed. Laser Surg 2007, 25, 275–280. [Google Scholar]
- Rosa, A.P.; de Sousa, L.G.; Regalo, S.C.; Issa, J.P.; Barbosa, A.P.; Pitol, D.L.; de Oliveira, R.H.; de Vasconcelos, P.B.; Dias, F.J.; Chimello, D.T.; et al. Effects of the combination of low-level laser irradiation and recombinant human bone morphogenetic protein-2 in bone repair. Lasers Med. Sci 2012, 27, 971–977. [Google Scholar]
- Saito, K.; Hashimoto, S.; Jung, H.S.; Shimono, M.; Nakagawa, K. Effect of diode laser on proliferation and differentiation of pc12 cells. Bull. Tokyo Dent. Coll 2011, 52, 95–102. [Google Scholar]
- Soleimani, M.; Abbasnia, E.; Fathi, M.; Sahraei, H.; Fathi, Y.; Kaka, G. The effects of low-level laser irradiation on differentiation and proliferation of human bone marrow mesenchymal stem cells into neurons and osteoblasts—An in vitro study. Lasers Med. Sci 2012, 27, 423–430. [Google Scholar]
- Song, S.; Zhou, F.; Chen, W.R. Low-level laser therapy regulates microglial function through src-mediated signaling pathways: Implications for neurodegenerative diseases. J. Neuroinflammation 2012, 9, 219. [Google Scholar]
- Stein, A.; Benayahu, D.; Maltz, L.; Oron, U. Low-level laser irradiation promotes proliferation and differentiation of human osteoblasts in vitro. Photomed. Laser Surg 2005, 23, 161–166. [Google Scholar]
- Stein, E.; Koehn, J.; Sutter, W.; Wendtlandt, G.; Wanschitz, F.; Thurnher, D.; Baghestanian, M.; Turhani, D. Initial effects of low-level laser therapy on growth and differentiation of human osteoblast-like cells. Wien. Klin. Wochenschr 2008, 120, 112–117. [Google Scholar]
- Dziunycz, P.; Iotzova-Weiss, G.; Eloranta, J.J.; Lauchli, S.; Hafner, J.; French, L.E.; Hofbauer, G.F. Squamous cell carcinoma of the skin shows a distinct microrna profile modulated by uv radiation. J. Invest. Dermatol 2010, 130, 2686–2689. [Google Scholar]
- Glorian, V.; Maillot, G.; Poles, S.; Iacovoni, J.S.; Favre, G.; Vagner, S. Hur-dependent loading of mirna risc to the mrna encoding the ras-related small gtpase rhob controls its translation during uv-induced apoptosis. Cell Death Differ 2011, 18, 1692–1701. [Google Scholar]
- Guo, L.; Huang, Z.X.; Chen, X.W.; Deng, Q.K.; Yan, W.; Zhou, M.J.; Ou, C.S.; Ding, Z.H. Differential expression profiles of micrornas in nih3t3 cells in response to UVB irradiation. Photochem. Photobiol 2009, 85, 765–773. [Google Scholar]
- Pothof, J.; Verkaik, N.S.; van IJcken, W.; Wiemer, E.A.; Ta, V.T.; van der Horst, G.T.; Jaspers, N.G.; van Gent, D.C.; Hoeijmakers, J.H.; Persengiev, S.P. Microrna-mediated gene silencing modulates the uv-induced DNA-damage response. EMBO J 2009, 28, 2090–2099. [Google Scholar]
- Tan, G.; Niu, J.; Shi, Y.; Ouyang, H.; Wu, Z.H. Nf-kappab-dependent microrna-125b up-regulation promotes cell survival by targeting p38alpha upon ultraviolet radiation. J. Biol. Chem 2012, 287, 33036–33047. [Google Scholar]
- Tan, G.; Shi, Y.; Wu, Z.H. Microrna-22 promotes cell survival upon uv radiation by repressing pten. Biochem. Biophys. Res. Commun 2012, 417, 546–551. [Google Scholar]
- Gu, X.; Lundqvist, E.N.; Coates, P.J.; Thurfjell, N.; Wettersand, E.; Nylander, K. Dysregulation of tap63 mrna and protein levels in psoriasis. J. Invest. Dermatol 2006, 126, 137–141. [Google Scholar]
- Okuyama, R.; Ogawa, E.; Nagoshi, H.; Yabuki, M.; Kurihara, A.; Terui, T.; Aiba, S.; Obinata, M.; Tagami, H.; Ikawa, S. P53 homologue, p51/p63, maintains the immaturity of keratinocyte stem cells by inhibiting notch1 activity. Oncogene 2007, 26, 4478–4488. [Google Scholar]
- Shen, C.S.; Tsuda, T.; Fushiki, S.; Mizutani, H.; Yamanishi, K. The expression of p63 during epidermal remodeling in psoriasis. J. Dermatol 2005, 32, 236–242. [Google Scholar]
- Menter, A.; Korman, N.J.; Elmets, C.A.; Feldman, S.R.; Gelfand, J.M.; Gordon, K.B.; Gottlieb, A.; Koo, J.Y.; Lebwohl, M.; Lim, H.W.; et al. Guidelines of care for the management of psoriasis and psoriatic arthritis: Section 5. Guidelines of care for the treatment of psoriasis with phototherapy and photochemotherapy. J. Am. Acad. Dermatol 2010, 62, 114–135. [Google Scholar]
- Ozawa, M.; Ferenczi, K.; Kikuchi, T.; Cardinale, I.; Austin, L.M.; Coven, T.R.; Burack, L.H.; Krueger, J.G. 312-nanometer ultraviolet b light (narrow-band UVB) induces apoptosis of t cells within psoriatic lesions. J. Exp. Med 1999, 189, 711–718. [Google Scholar]
- Schneider, L.A.; Hinrichs, R.; Scharffetter-Kochanek, K. Phototherapy and photochemotherapy. Clin. Dermatol 2008, 26, 464–476. [Google Scholar]
- Celli, J.P.; Spring, B.Q.; Rizvi, I.; Evans, C.L.; Samkoe, K.S.; Verma, S.; Pogue, B.W.; Hasan, T. Imaging and photodynamic therapy: Mechanisms, monitoring, and optimization. Chem. Rev 2010, 110, 2795–2838. [Google Scholar]
- Dolmans, D.E.; Fukumura, D.; Jain, R.K. Photodynamic therapy for cancer. Nat. Rev. Cancer 2003, 3, 380–387. [Google Scholar]
- Verma, S.; Watt, G.M.; Mai, Z.; Hasan, T. Strategies for enhanced photodynamic therapy effects. Photochem. Photobiol 2007, 83, 996–1005. [Google Scholar]
- Buytaert, E.; Dewaele, M.; Agostinis, P. Molecular effectors of multiple cell death pathways initiated by photodynamic therapy. Biochim. Biophys. Acta 2007, 1776, 86–107. [Google Scholar]
- Brown, S.B.; Brown, E.A.; Walker, I. The present and future role of photodynamic therapy in cancer treatment. Lancet Oncol 2004, 5, 497–508. [Google Scholar]
- Dougherty, T.J. An update on photodynamic therapy applications. J. Clin. Laser Med. Surg 2002, 20, 3–7. [Google Scholar]
- Tomioka, Y.; Kushibiki, T.; Awazu, K. Evaluation of oxygen consumption of culture medium and in vitro photodynamic effect of talaporfin sodium in lung tumor cells. Photomed. Laser Surg 2010, 28, 385–390. [Google Scholar]
- Oleinick, N.L.; Morris, R.L.; Belichenko, I. The role of apoptosis in response to photodynamic therapy: What, where, why, and how. Photochem. Photochem. Photobiol. Sci 2002, 1, 1–21. [Google Scholar]
- Danial, N.N.; Korsmeyer, S.J. Cell death: Critical control points. Cell 2004, 116, 205–219. [Google Scholar]
- Ferri, K.F.; Kroemer, G. Organelle-specific initiation of cell death pathways. Nat. Cell Biol 2001, 3, E255–E263. [Google Scholar]
- Hengartner, M.O. The biochemistry of apoptosis. Nature 2000, 407, 770–776. [Google Scholar]
- Piette, J.; Volanti, C.; Vantieghem, A.; Matroule, J.Y.; Habraken, Y.; Agostinis, P. Cell death and growth arrest in response to photodynamic therapy with membrane-bound photosensitizers. Biochem. Pharmacol 2003, 66, 1651–1659. [Google Scholar]
- Helbig, D.; Simon, J.C.; Paasch, U. Photodynamic therapy and the role of heat shock protein 70. Int. J. Hyperthermia 2011, 27, 802–810. [Google Scholar]
- Matroule, J.Y.; Volanti, C.; Piette, J. Nf-kappab in photodynamic therapy: Discrepancies of a master regulator. Photochem. Photobiol 2006, 82, 1241–1246. [Google Scholar]
- Agostinis, P.; Buytaert, E.; Breyssens, H.; Hendrickx, N. Regulatory pathways in photodynamic therapy induced apoptosis. Photochem. Photobiol. Sci 2004, 3, 721–729. [Google Scholar]
- Dewaele, M.; Verfaillie, T.; Martinet, W.; Agostinis, P. Death and survival signals in photodynamic therapy. Methods Mol. Biol 2010, 635, 7–33. [Google Scholar]
- Kessel, D.; Oleinick, N.L. Photodynamic therapy and cell death pathways. Methods Mol. Biol 2010, 635, 35–46. [Google Scholar]
- Chen, Q.; Huang, Z.; Chen, H.; Shapiro, H.; Beckers, J.; Hetzel, F.W. Improvement of tumor response by manipulation of tumor oxygenation during photodynamic therapy. Photochem. Photobiol 2002, 76, 197–203. [Google Scholar]
- Henderson, B.W.; Busch, T.M.; Vaughan, L.A.; Frawley, N.P.; Babich, D.; Sosa, T.A.; Zollo, J.D.; Dee, A.S.; Cooper, M.T.; Bellnier, D.A.; et al. Photofrin photodynamic therapy can significantly deplete or preserve oxygenation in human basal cell carcinomas during treatment, depending on fluence rate. Cancer Res 2000, 60, 525–529. [Google Scholar]
- Sitnik, T.M.; Hampton, J.A.; Henderson, B.W. Reduction of tumour oxygenation during and after photodynamic therapy in vivo: Effects of fluence rate. Br. J. Cancer 1998, 77, 1386–1394. [Google Scholar]
- Engbrecht, B.W.; Menon, C.; Kachur, A.V.; Hahn, S.M.; Fraker, D.L. Photofrin-mediated photodynamic therapy induces vascular occlusion and apoptosis in a human sarcoma xenograft model. Cancer Res 1999, 59, 4334–4342. [Google Scholar]
- Fingar, V.H.; Kik, P.K.; Haydon, P.S.; Cerrito, P.B.; Tseng, M.; Abang, E.; Wieman, T.J. Analysis of acute vascular damage after photodynamic therapy using benzoporphyrin derivative (bpd). Br. J. Cancer 1999, 79, 1702–1708. [Google Scholar]
- Keith, B.; Johnson, R.S.; Simon, M.C. Hif1alpha and hif2alpha: Sibling rivalry in hypoxic tumour growth and progression. Nat. Rev. Cancer 2012, 12, 9–22. [Google Scholar]
- Semenza, G.L. Hypoxia-inducible factors in physiology and medicine. Cell 2012, 148, 399–408. [Google Scholar]
- Forsythe, J.A.; Jiang, B.H.; Iyer, N.V.; Agani, F.; Leung, S.W.; Koos, R.D.; Semenza, G.L. Activation of vascular endothelial growth factor gene transcription by hypoxia-inducible factor 1. Mol. Cell. Biol 1996, 16, 4604–4613. [Google Scholar]
- Takenaga, K. Angiogenic signaling aberrantly induced by tumor hypoxia. Front. Biosci 2011, 16, 31–48. [Google Scholar]
- Deininger, M.H.; Weinschenk, T.; Morgalla, M.H.; Meyermann, R.; Schluesener, H.J. Release of regulators of angiogenesis following hypocrellin-a and -b photodynamic therapy of human brain tumor cells. Biochem. Biophys. Res. Commun 2002, 298, 520–530. [Google Scholar]
- Ferrario, A.; von Tiehl, K.F.; Rucker, N.; Schwarz, M.A.; Gill, P.S.; Gomer, C.J. Antiangiogenic treatment enhances photodynamic therapy responsiveness in a mouse mammary carcinoma. Cancer Res 2000, 60, 4066–4069. [Google Scholar]
- Jiang, F.; Zhang, Z.G.; Katakowski, M.; Robin, A.M.; Faber, M.; Zhang, F.; Chopp, M. Angiogenesis induced by photodynamic therapy in normal rat brains. Photochem. Photobiol 2004, 79, 494–498. [Google Scholar]
- Schmidt-Erfurth, U.; Schlotzer-Schrehard, U.; Cursiefen, C.; Michels, S.; Beckendorf, A.; Naumann, G.O. Influence of photodynamic therapy on expression of vascular endothelial growth factor (vegf), vegf receptor 3, and pigment epithelium-derived factor. Invest. Ophthalmol. Vis. Sci 2003, 44, 4473–4480. [Google Scholar]
- Lu, J.; Getz, G.; Miska, E.A.; Alvarez-Saavedra, E.; Lamb, J.; Peck, D.; Sweet-Cordero, A.; Ebert, B.L.; Mak, R.H.; Ferrando, A.A.; et al. Microrna expression profiles classify human cancers. Nature 2005, 435, 834–838. [Google Scholar]
- Cheng, A.M.; Byrom, M.W.; Shelton, J.; Ford, L.P. Antisense inhibition of human mirnas and indications for an involvement of mirna in cell growth and apoptosis. Nucleic Acids Res 2005, 33, 1290–1297. [Google Scholar]
- Kushibiki, T. Photodynamic therapy induces microrna-210 and -296 expression in HeLa cells. J. Biophotonics 2010, 3, 368–372. [Google Scholar]
- Giannakakis, A.; Sandaltzopoulos, R.; Greshock, J.; Liang, S.; Huang, J.; Hasegawa, K.; Li, C.; O’Brien-Jenkins, A.; Katsaros, D.; Weber, B.L.; et al. Mir-210 links hypoxia with cell cycle regulation and is deleted in human epithelial ovarian cancer. Cancer Biol. Ther 2008, 7, 255–264. [Google Scholar]
- Wurdinger, T.; Tannous, B.A.; Saydam, O.; Skog, J.; Grau, S.; Soutschek, J.; Weissleder, R.; Breakefield, X.O.; Krichevsky, A.M. Mir-296 regulates growth factor receptor overexpression in angiogenic endothelial cells. Cancer Cell 2008, 14, 382–393. [Google Scholar]
- Bach, D.; Fuereder, J.; Karbiener, M.; Scheideler, M.; Ress, A.L.; Neureiter, D.; Kemmerling, R.; Dietze, O.; Wiederstein, M.; Berr, F.; et al. Comprehensive analysis of alterations in the mirnome in response to photodynamic treatment. J. Photochem. Photobiol. B 2013, 120, 74–81. [Google Scholar]
- Sato, M.; Kubota, N.; Inada, E.; Saitoh, I.; Ohtsuka, M.; Nakamura, S.; Sakurai, T.; Watanabe, S. Hela cells consist of two cell types, as evidenced by cytochemical staining for alkaline phosphatase activity: A possible model for cancer stem cell study. Adv. Stem. Cell 2013. [Google Scholar] [CrossRef]

| Upregulation | Downregulation |
|---|---|
| miR-30e * | |
| miR-15b | |
| miR-30b-5p | miR-204 * |
| miR-322 | miR-7a |
| miR-215 | miR-423 |
| miR-449a | miR-678 |
| miR-126 | miR-25 * |
| miR-133b | miR-327 |
| miR-21 * | miR-351 |
| miR-455 | miR-23a |
| miR-759 | miR-667 |
| miR-872 * | miR-770 |
| miR-29b | miR-324-3p |
| miR-192 | miR-30c-2 * |
| miR-219-1-3p | miR-758 |
| miR-301a | miR-320 |
| miR-551b | miR-466c |
| miR-224 | |
| miR-193 |
| Upregulation | Downregulation |
|---|---|
| miR-1290 | miR-1260b |
| miR-634 | miR-720 |
| miR-1246 | miR-1260 |
| miR-1280 |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kushibiki, T.; Hirasawa, T.; Okawa, S.; Ishihara, M. Regulation of miRNA Expression by Low-Level Laser Therapy (LLLT) and Photodynamic Therapy (PDT). Int. J. Mol. Sci. 2013, 14, 13542-13558. https://doi.org/10.3390/ijms140713542
Kushibiki T, Hirasawa T, Okawa S, Ishihara M. Regulation of miRNA Expression by Low-Level Laser Therapy (LLLT) and Photodynamic Therapy (PDT). International Journal of Molecular Sciences. 2013; 14(7):13542-13558. https://doi.org/10.3390/ijms140713542
Chicago/Turabian StyleKushibiki, Toshihiro, Takeshi Hirasawa, Shinpei Okawa, and Miya Ishihara. 2013. "Regulation of miRNA Expression by Low-Level Laser Therapy (LLLT) and Photodynamic Therapy (PDT)" International Journal of Molecular Sciences 14, no. 7: 13542-13558. https://doi.org/10.3390/ijms140713542
APA StyleKushibiki, T., Hirasawa, T., Okawa, S., & Ishihara, M. (2013). Regulation of miRNA Expression by Low-Level Laser Therapy (LLLT) and Photodynamic Therapy (PDT). International Journal of Molecular Sciences, 14(7), 13542-13558. https://doi.org/10.3390/ijms140713542




