Reconstructing a Flavodoxin Oxidoreductase with Early Amino Acids
Abstract
:1. Introduction
2. Results and Discussion
2.1. Reconstruction of the Flavodoxin Mutant
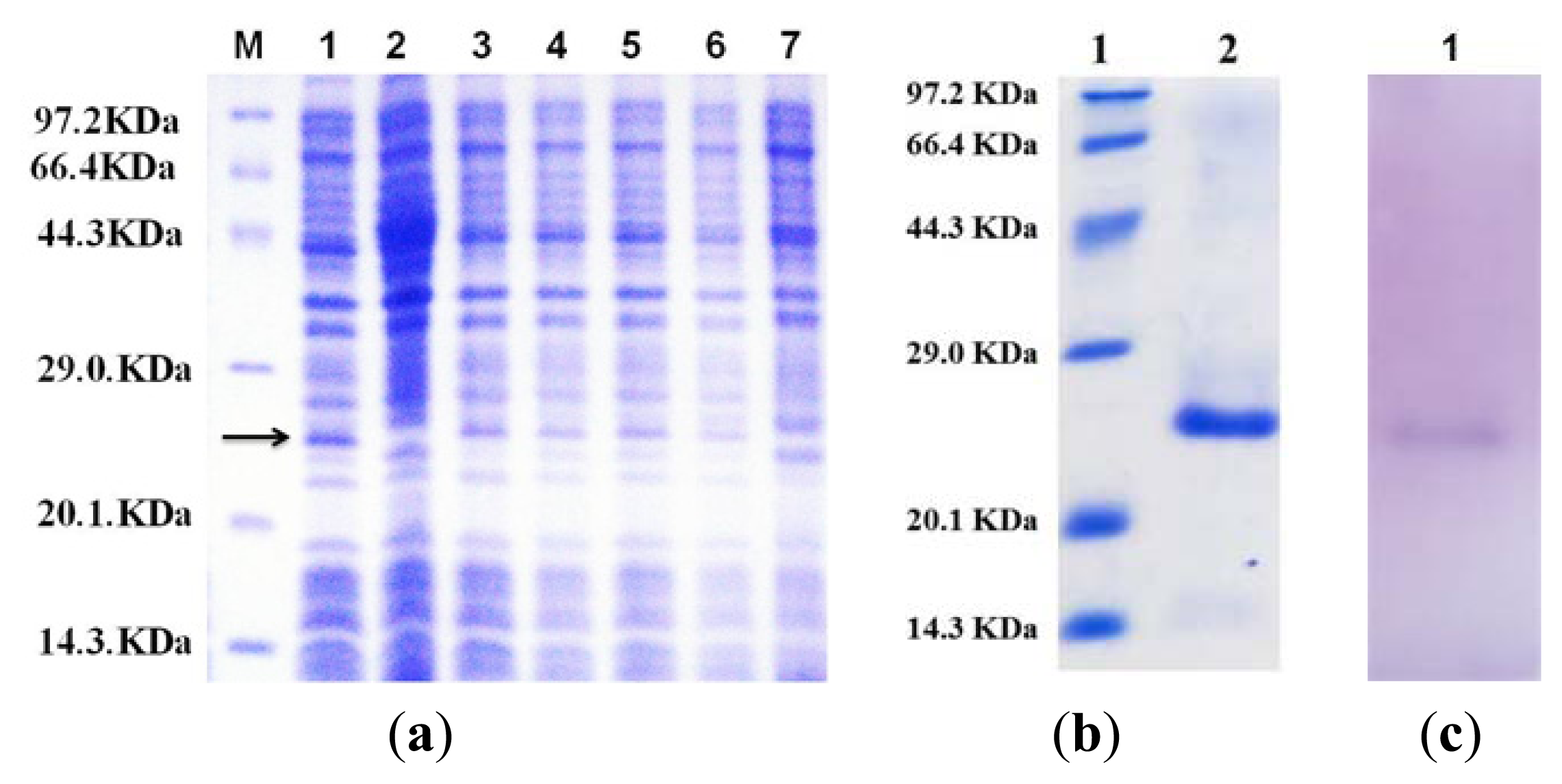
2.2. Expression and Purification of the Flavodoxin Mutant
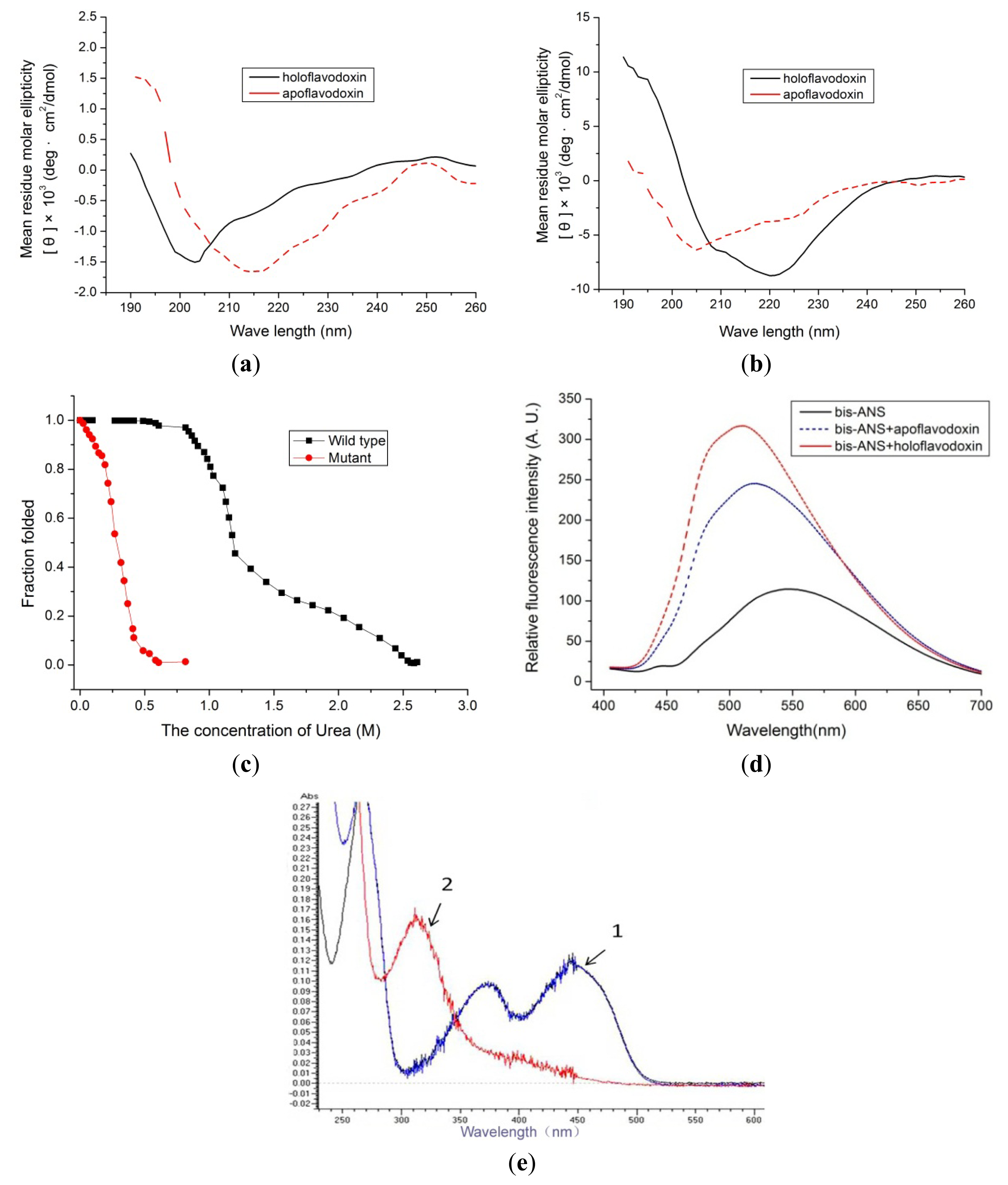
2.3. Spectral Assay
2.4. Redox Potentials and Dissociation Constant of the Mutant Flavodoxin
3. Experimental Section
3.1. Expression and Purification of the Mutant Flavodoxin
3.2. Preparation of Apoflavodoxin and Holoflavodoxin
3.3. Spectral Assay
3.4. Determination of Redox Potentials and Dissociation Constant
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Ji, H.F.; Chen, L.; Zhang, H.Y. Organic cofactors participated more frequently than transition metals in redox reactions of primitive proteins. Bioessays 2008, 30, 766–771. [Google Scholar]
- Walter, K.U.; Vamvaca, K.; Hilvert, D. An active enzyme constructed from a 9-amino acid alphabet. J. Biol. Chem 2005, 280, 37742–37746. [Google Scholar]
- Jumawid, M.T.; Takahashi, T.; Yamazaki, T.; Ashigai, H.; Mihara, H. Selection and structural analysis of de novo proteins from an a3b3 genetic library. Protein Sci 2009, 18, 384–398. [Google Scholar]
- Tanaka, J.; Yanagawa, H.; Doi, N. Comparison of the frequency of functional SH3 domains with different limited sets of amino acids using mRNA display. PLoS One 2011, 6, e18034. [Google Scholar]
- Van Mierlo, C.P.M.; Lijnzaad, P.; Vervoort, J.; Mueller, F.; Berendsen, H.J.; de Vlieg, J. Tertiary structure of two electron reduced Megasphaera elsdenii flavodoxin and some imply-cations, as determined by two-dimensional 1H-NMR and restrained molecular dynamics. Eur. J. Biochem 1990, 194, 185–198. [Google Scholar]
- Jakschitz, T.A.E.; Rode, B.M. Chemical evolution from simple inorganic compounds to chiral peptides. Chem. Soc. Rev 2012, 41, 5484–5489. [Google Scholar]
- Longo, L.M.; Blaber, M. Protein design at the interface of the pre-biotic and biotic worlds. Arch. Biochem. Biophys 2012, 526, 16–21. [Google Scholar]
- Anderson, R.F. Energetics of the one-electron reduction steps of riboflavin, FMN and FAD to their fully reduced forms. Biochim. Biophys. Acta 1983, 722, 158–162. [Google Scholar]
- Mayhew, S.G. The effects of pH and semiquinone formation on the oxidation-reduction potentials of flavin mononucleotide. A reappraisal. Eur. J. Biochem 1999, 265, 698–702. [Google Scholar]
- Geoghegan, S.M.; Mayhew, S.G.; Yalloway, G.N.; Butler, G. Cloning, sequencing and expression of the gene for flavodoxin from Megasphaera elsdenii and the effects of removing the protein negative charge that is closest to N(1) of the bound FMN. Eur. J. Biochem 2000, 267, 4434–4444. [Google Scholar]
- Sancho, J. Flavodoxins: Sequence, folding, binding, function and beyond. Cell Mol. Life Sci 2006, 63, 855–864. [Google Scholar]
- Lostao, A.; Gomez-Moreno, C.; Mayhew, S.G.; Sancho, J. Differential stabilization of the three FMN redox forms by tyrosine 94 and tryptophan 57 in flavodoxin from Anabaena and its influence on the redox potentials. Biochemistry 1997, 36, 14334–14344. [Google Scholar]
- Swenson, R.P.; Krey, G.D. Site-directed mutagenesis of tyrosine-98 in the flavodoxin from Desulfovibrio vulgaris (Hildenborough): Regulation of oxidation-reduction properties of the bound FMN cofactor by aromatic, solvent, and electrostatic interactions. Biochemistry 1994, 33, 8505–8514. [Google Scholar]
- Alagaratnam, S.; van Pouderoyen, G.; Pijning, T. A crystallographic study of Cys69Ala flavodoxin II from Azotobacter vinelandii: Structural determinants of redox potential. Protein Sci 2005, 14, 2284–2295. [Google Scholar]
- Frago, S.; Goni, G.; Herguedas, B. Tuning of the FMN binding and oxido-reduction properties by neighboring side chains in Anabaena flavodoxin. Arch. Biochem. Biophys 2007, 467, 206–217. [Google Scholar]
- Curley, G.P.; Carr, M.C.; Mayhew, S.G.; Voordouw, G. Redox and flavin-binding properties of recombinant flavodoxin from Desulfovibrio vulgaris (Hildenborough). Eur. J. Biochem 1991, 202, 1091–1100. [Google Scholar]
- Mayhew, S.G.; Foust, G.P.; Massey, V. Oxidation-reduction properties of flavodoxin from Peptostreptococcus elsdenii. J. Biol. Chem 1969, 244, 803–810. [Google Scholar]
- Bollen, Y.J.; Nabuurs, S.M.; van Berkel, W.J.; van Mierlo, C.P. Last in, first out: The role of cofactor binding in flavodoxin folding. J. Biol. Chem 2005, 280, 7836–7844. [Google Scholar]
- Davis, B.K. Molecular evolution before the origin of species. Prog. Biophys. Mol. Biol 2002, 79, 77–133. [Google Scholar]
- Freigang, J.; Diederichs, K.; Schafer, K.P.; Welte, W.; Paul, R. Crystal structure of oxidized flavodoxin, an essential protein in Helicobacter pylori. Protein Sci 2002, 11, 253–261. [Google Scholar]

| Amino acid position | 6 | 7 | 16 | 36 | 54 | 57 | 70 | 71 | 86 | 89 | 91 | 96 | 97 | 100 | 120 | 127 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Wild-type | Y | W | M | F | C | M | F | F | F | Y | W | W | M | W | M | C |
| Mutant | V | V | L | V | A | V | L | L | V | V | V | V | L | V | V | V |
| Secondary structure a | s | s | h | s | h | l | h | h | s | s | s | h | h | h | l | s |
| Absorption maxima (nm) | Redox potentials Em,7 (mV) | Kd,ox (μM) | ΔGb,ox (kcal/mol) | Kd,hq (μM) | ΔGb,hq (kcal/mol) | |
|---|---|---|---|---|---|---|
| FMN | 266, 373, 445 a | −207 a | – | – | – | – |
| Wild-type | 272, 378, 446 b | −244 b | 0.00047 b | −12.7 b | 8.1 b | −11.0 b |
| Mutant-type | 274, 376, 450 | −360 | 2.92 | −7.52 | 43,604 | −1.85 |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lu, M.-F.; Ji, H.-F.; Li, T.-X.; Kang, S.-K.; Zhang, Y.-J.; Zheng, J.-F.; Tian, T.; Jia, X.-S.; Lin, X.-M.; Zhang, H.-Y. Reconstructing a Flavodoxin Oxidoreductase with Early Amino Acids. Int. J. Mol. Sci. 2013, 14, 12843-12852. https://doi.org/10.3390/ijms140612843
Lu M-F, Ji H-F, Li T-X, Kang S-K, Zhang Y-J, Zheng J-F, Tian T, Jia X-S, Lin X-M, Zhang H-Y. Reconstructing a Flavodoxin Oxidoreductase with Early Amino Acids. International Journal of Molecular Sciences. 2013; 14(6):12843-12852. https://doi.org/10.3390/ijms140612843
Chicago/Turabian StyleLu, Ming-Feng, Hong-Fang Ji, Ting-Xuan Li, Shou-Kai Kang, Yue-Jie Zhang, Jue-Fei Zheng, Tian Tian, Xi-Shuai Jia, Xing-Ming Lin, and Hong-Yu Zhang. 2013. "Reconstructing a Flavodoxin Oxidoreductase with Early Amino Acids" International Journal of Molecular Sciences 14, no. 6: 12843-12852. https://doi.org/10.3390/ijms140612843





