Dual Delivery of BMP-2 and bFGF from a New Nano-Composite Scaffold, Loaded with Vascular Stents for Large-Size Mandibular Defect Regeneration
Abstract
:1. Introduction
2. Results and Discussion
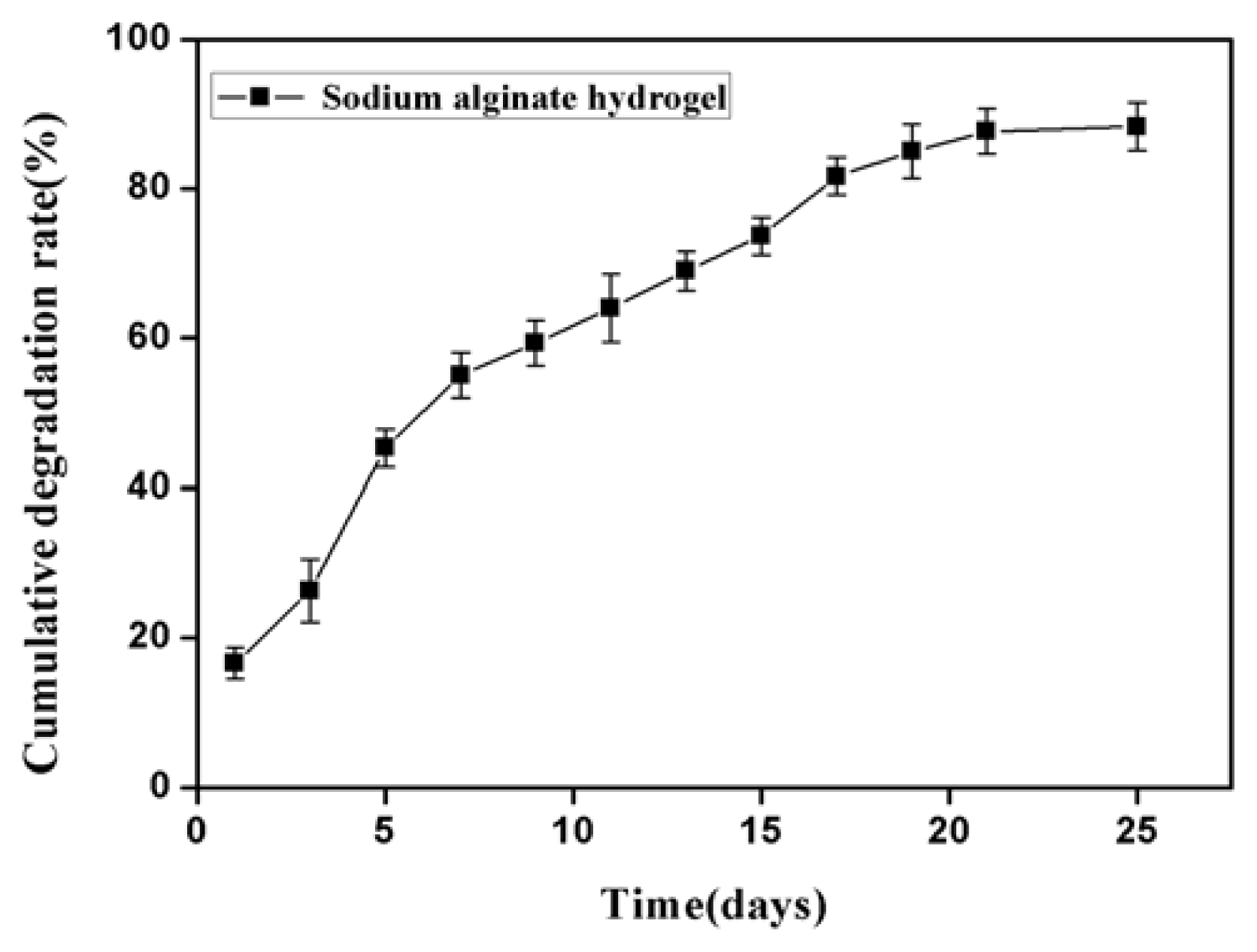
2.1. Properties of Sodium Alginate Hydrogel
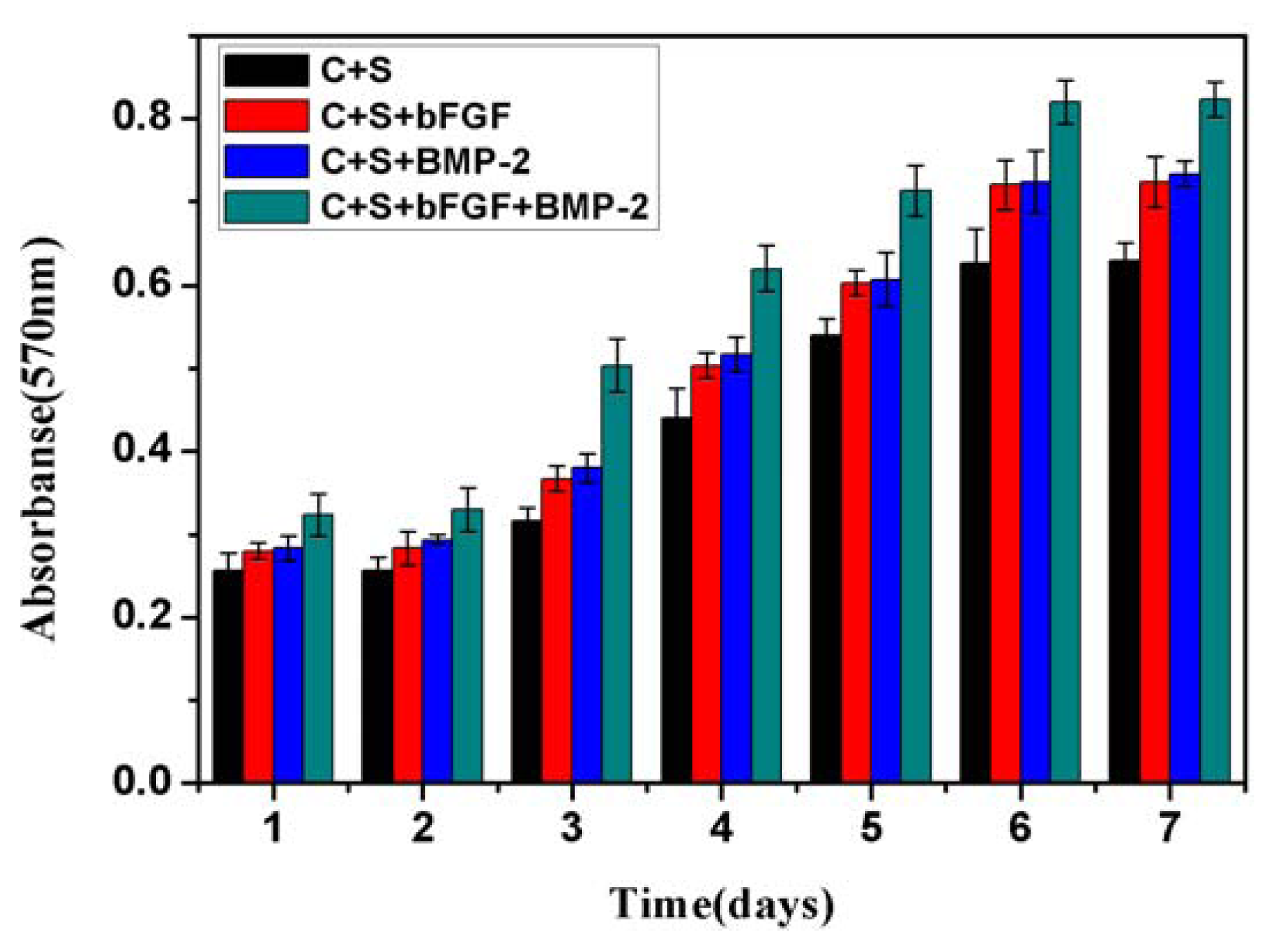
2.2. Bioactivity and Biocompatibility in Vitro
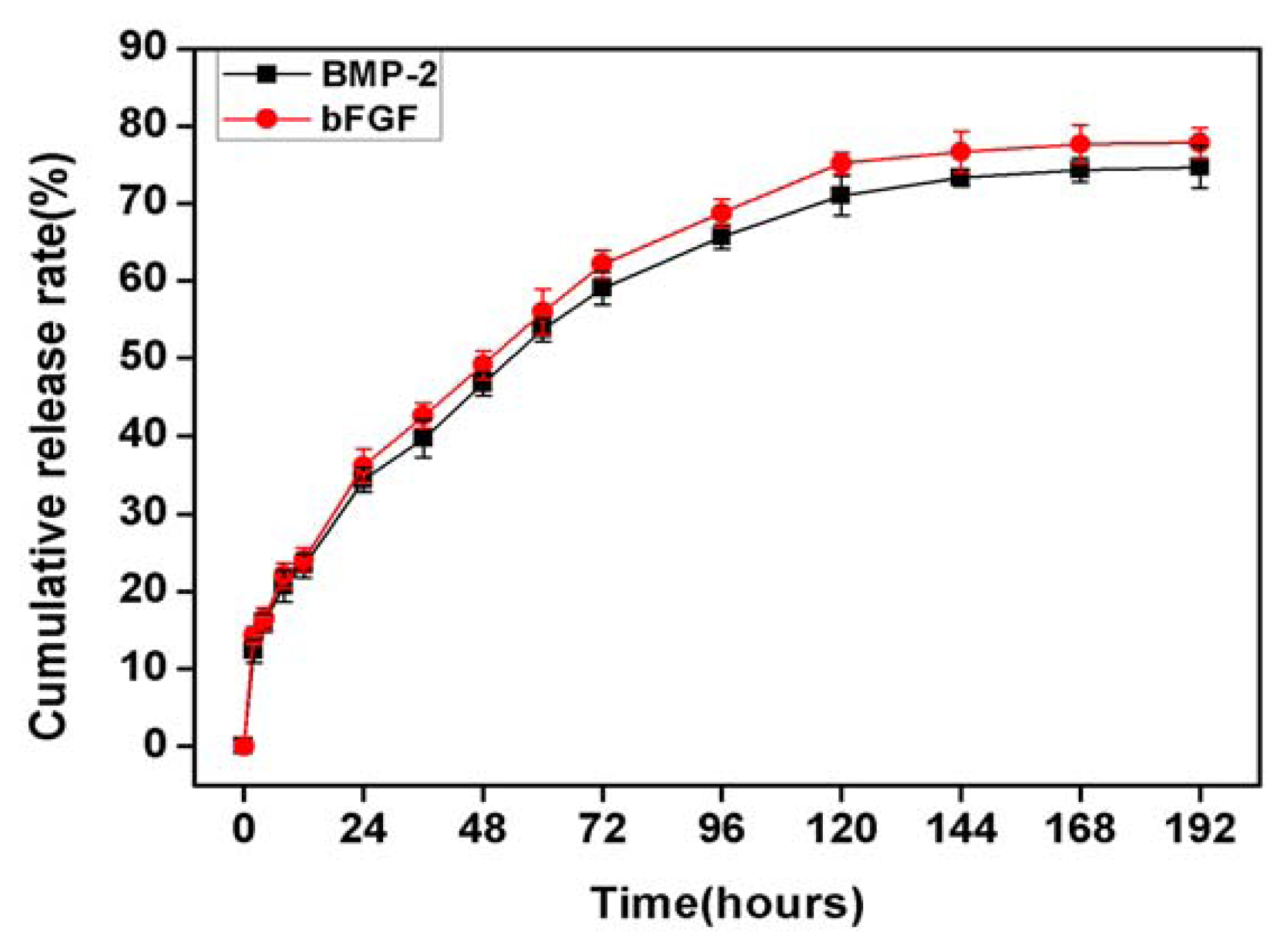
2.3. In Vitro Release of bFGF and BMP-2 from Sodium Alginate Hydrogel and Nano-Composite Scaffolds (PLGA/PCL/nHA) Loaded with Vascular Stents (PLCL/Col/nHA)
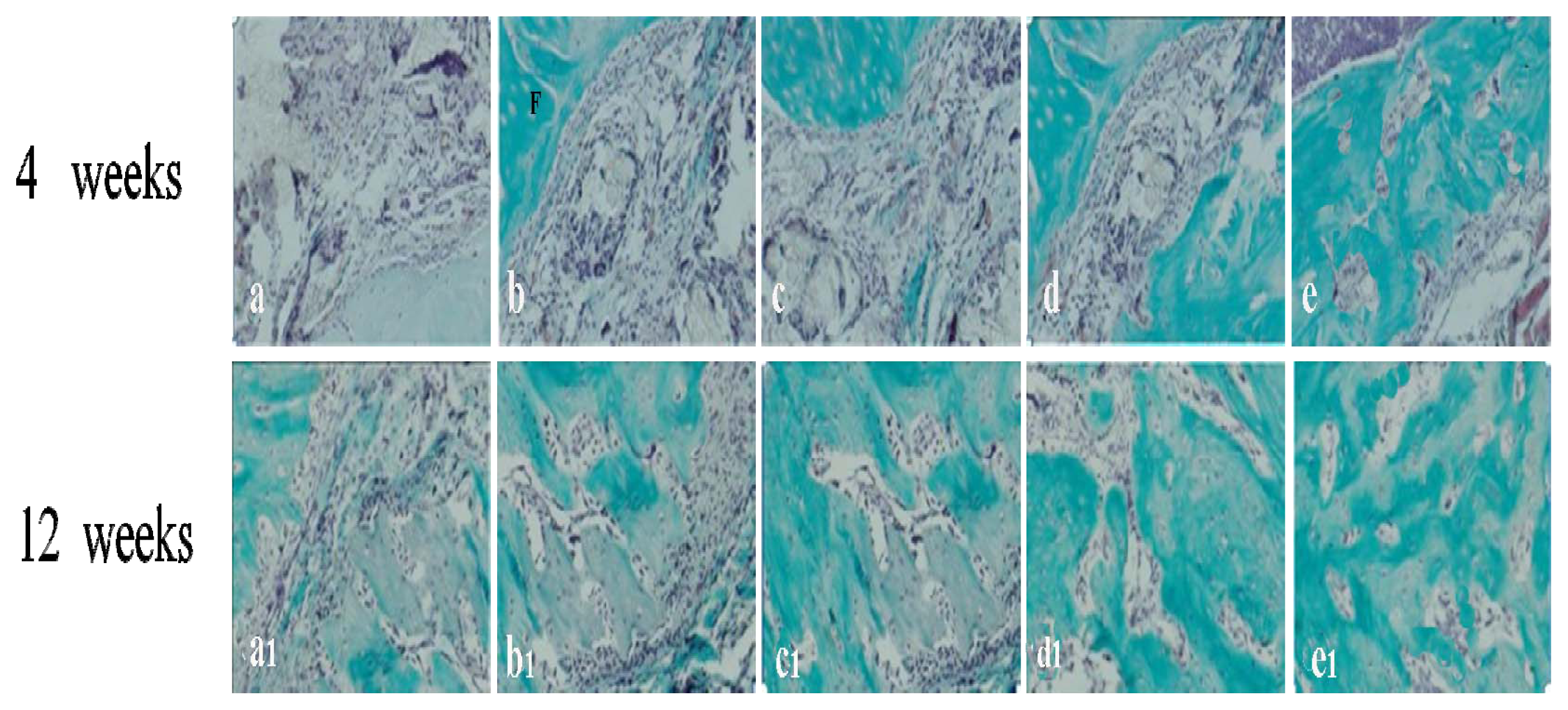
2.4. Histological Analysis
2.5. Radiographic Observations
3. Experimental Section
3.1. BMSC Culture and Seeding
3.2. Preparation and Performance Testing of Sodium Alginate Hydrogel
3.2.1. Hydrogel Preparation
3.2.2. Protein-Loaded Release Study
3.3. Fabrication of BMP-2 and bFGF-Loaded Scaffold
3.4. Creation and Reconstruction of Large Bone Defect
3.4.1. Radiographic Observations
3.4.2. Histological Analysis
3.5. Statistical Analysis
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Hadlock, T.A.; Vacanti, J.P.; Cheney, M.L. Tissue engineering in facial plastic and reconstructive surgery. Facial Plast Surg 1998, 14, 197–203. [Google Scholar]
- Veron, C.; Chanavaz, M.; Ferri, J.; Donazzan, M.; Hildebrand, H.F. A panorama of current materials for osseous application in maxillofacial surgery and oral implantolog (in French). Rev. Stomatol. Chir. Maxillofac 1995, 96, 274–281. [Google Scholar]
- Hoexter, D.L. Bone regeneration graft materials. J. Oral Implantol 2002, 28, 290–294. [Google Scholar]
- Dinopoulos, H.; Dimitriou, R.; Giannoudis, P.V. Bone graft substitutes: What are the options? Surgeon 2012, 10, 230–239. [Google Scholar]
- Dai, K.R.; Xu, X.L.; Tang, T.T.; Zhu, Z.A.; Yu, C.F.; Lou, J.R.; Zhang, X.L. Repairing of goat tibial bone defects with BMP-2 gene-modified tissue-engineered bone. Calcif. Tissue Int 2005, 77, 55–61. [Google Scholar]
- Argintar, E.; Edwards, S.; Delahay, J. Bone morphogenetic proteins in orthopaedic trauma surgery. Injury 2011, 42, 730–734. [Google Scholar]
- Yun, Y.R.; Won, J.E.; Jeon, E.; Lee, S.; Kang, W.; Jo, H.; Jang, J.H.; Shin, U.S.; Kim, H.W. Fibroblast growth factors: Biology, function, and application for tissue regeneration. J. Tissue Eng 2010, 2010, 218142. [Google Scholar]
- Young, S.; Patel, Z.S.; Kretlow, J.D.; Murphy, M.B.; Mountziaris, P.M.; Baggett, L.S.; Ueda, H.; Tabata, Y.; Jansen, J.A.; Wong, M.; et al. Dose effect of dual delivery of vascular endothelial growth factor and bone morphogenetic protein-2 on bone regeneration in a rat critical-size defect model. Tissue Eng. A 2009, 15, 2347–2362. [Google Scholar]
- Zhang, W.; Tsurushima, H.; Oyane, A.; Yazaki, Y.; Sogo, Y.; Ito, A.; Matsumura, A. BMP-2 gene-fibronectin-apatite composite layer enhances bone formation. J. Biomed. Sci 2011, 18, 62. [Google Scholar] [Green Version]
- Rosen, V. BMP and BMP inhibitors in bone. Ann. N. Y. Acad. Sci 2006, 1068, 19–25. [Google Scholar]
- Ishikawa, H.; Kitoh, H.; Sugiura, F.; Ishiguro, N. The effect of recombinant human bone morphogenetic protein-2 on the osteogenic potential of rat mesenchymal stem cells after several passages. Acta Orthop 2007, 78, 285–292. [Google Scholar]
- Solchaga, L.A.; Penick, K.; Goldberg, V.M.; Caplan, A.I.; Welter, J.F. Fibroblast growth factor-2 enhances proliferation and delays loss of chondrogenic potential in human adult bone-marrow-derived mesenchymal stem cells. Tissue Eng. A 2010, 16, 1009–1019. [Google Scholar]
- Simmons, H.A.; Raisz, L.G. Effects of acid and basic fibroblast growth factor and heparin on resorption of cultured fetal rat long bones. J. Bone Miner. Res 1991, 6, 1301–1305. [Google Scholar]
- Draenert, G.F.; Draenert, K.; Tischer, T. Dose-dependent osteoinductive effects of bFGF in rabbits. Growth Factors 2009, 27, 419–424. [Google Scholar]
- Schnettler, R.; Alt, V.; Dingeldein, E.; Pfefferle, H.J.; Kilian, O.; Meyer, C.; Heiss, C.; Wenisch, S. Bone ingrowth in bFGF-coated hydroxyapatite ceramic implants. Biomaterials 2003, 24, 4603–4608. [Google Scholar]
- Zou, D.; Zhang, Z.; He, J.; Zhang, K.; Ye, D.; Han, W.; Zhou, J.; Wang, Y.; Li, Q.; Liu, X.; et al. Blood vessel formation in the tissue-engineered bone with the constitutively active form of HIF-1α mediated BMSCs. Biomaterials 2012, 33, 2097–2108. [Google Scholar]
- Wang, H.; Zou, Q.; Boerman, O.C.; Nijhuis, A.W.; Jansen, J.A.; Li, Y.; Leeuwenburgh, S.C. Combined delivery of BMP-2 and bFGF from nanostructured colloidal gelatin gels and its effect on bone regeneration in vivo. J. Control. Release 2013, 166, 172–181. [Google Scholar]
- Fujimura, K.; Bessho, K.; Okubo, Y.; Kusumoto, K.; Segami, N.; Iizuka, T. The effect of fibroblast growth factor-2 on the osteoinductive activity of recombinant human bone morphogenetic protein-2 in rat muscle. Arch. Oral Biol 2002, 47, 577–584. [Google Scholar]
- Tanaka, E.; Ishino, Y.; Sasaki, A.; Hasegawa, T.; Watanabe, M.; Dalla-Bona, D.A.; Yamano, E.; van Eijden, T.M.; Tanne, K. Fibroblast growth factor-2 augments recombinant human bone morphogenetic protein-2-induced osteoinductive activity. Ann. Biomed. Eng 2006, 34, 717–725. [Google Scholar]
- Scheller, E.L.; Krebsbach, P.H.; Kohn, D.H. Tissue engineering: state of the art in oral rehabilitation. J. Oral Rehabil 2009, 36, 368–389. [Google Scholar]
- Karageorgiou, V.; Kaplan, D. Porosity of 3D biomaterial scaffolds and osteogenesis. Biomaterials 2005, 26, 5474–5491. [Google Scholar]
- Liu, X.; Ma, P.X. Polymeric scaffolds for bone tissue engineering. Ann. Biomed. Eng 2004, 32, 477–486. [Google Scholar]
- Saranya, N.; Saravanan, S.; Moorthi, A.; Ramyakrishna, B.; Selvamurugan, N. Enhanced osteoblast adhesion on polymeric nano-scaffolds for bone tissue engineering. J. Biomed. Nanotechnol 2011, 7, 238–244. [Google Scholar]
- Xu, H.; Su, J.; Sun, J.; Ren, T. Preparation and characterization of new nano-composite scaffolds loaded with vascular stents. Int. J. Mol. Sci 2012, 13, 3366–3381. [Google Scholar]
- Makadia, H.K.; Siegel, S.J. Polylactic-co-glycolic acid (PLGA) as biodegradable controlled drug delivery carrier. Polymers 2011, 3, 1377–1397. [Google Scholar]
- Chen, F.M.; Zhang, M.; Wu, Z.F. Toward delivery of multiple growth factors in tissue engineering. Biomaterials 2010, 31, 6279–6308. [Google Scholar]
- Kretlow, J.D.; Young, S.; Klouda, L.; Wong, M.; Mikos, A.G. Toward delivery of multiple growth factors in tissue engineering. Adv. Mater 2009, 21, 3368–3393. [Google Scholar]
- Yu, L.; Ding, J. Injectable hydrogels as unique biomedical materials. Chem. Soc. Rev 2008, 37, 1473–1481. [Google Scholar]
- Amini, A.A.; Nair, L.S. Injectable hydrogels for bone and cartilage repair. Biomed Mater 2012, 7, 024105. [Google Scholar]
- Cui, Z.; Lee, B.H.; Pauken, C.; Vernon, B.L. Degradation, cytotoxicity, and biocompatibility of NIPAAm-based thermosensitive, injectable, and bioresorbable polymer hydrogels. J. Biomed. Mater. Res. A 2011, 98, 159–166. [Google Scholar]
- Wang, Q.; Jamal, S.; Detamore, M.S.; Berkland, C. PLGA-chitosan/PLGA-alginate nanoparticle blends as biodegradable colloidal gels for seeding human umbilical cord mesenchymal stem cells. J. Biomed. Mater. Res. A 2011, 96, 520–527. [Google Scholar]
- Wang, Q.; Wang, L.M.; Detamore, M.S.; Berkland, C. Biodegradable colloidal gels as moldable tissue engineering scaffolds. Adv. Mater 2008, 20, 236–239. [Google Scholar]
- Wang, Q.; Wang, J.X.; Lu, Q.H.; Detamore, M.S.; Berkland, C. Injectable PLGA based colloidal gels for zero-order dexamethasone release in cranial defects. Biomaterials 2010, 31, 4980–4986. [Google Scholar]
- Kong, H.J.; Smith, M.K.; Mooney, D.J. Designing alginate hydrogels to maintain viability of immobilized cells. Biomaterials 2003, 24, 4023–4029. [Google Scholar]
- Zhou, H.; Xu, H.H. The fast release of stem cells from alginate-fibrin microbeads in injectable scaffolds for bone tissue engineering. Biomaterials 2011, 32, 7503–7513. [Google Scholar]
- Zhao, L.; Tang, M.; Weir, M.D.; Detamore, M.S.; Xu, H.H. Osteogenic media and rhBMP-2-induced differentiation of umbilical cord mesenchymal stem cells encapsulated in alginate microbeads and integrated in an injectable calcium phosphate-chitosan fibrous scaffold. Tissue Eng. A 2011, 17, 969–979. [Google Scholar]
- Basmanav, F.B.; Kose, G.T.; Hasirci, V. Sequential growth factor delivery from complexed microspheres for bone tissue engineering. Biomaterials 2008, 29, 4195–4204. [Google Scholar]
- Yilgor, P.; Hasirci, N.; Hasirci, V. Sequential BMP-2/BMP-7 delivery from polyester nanocapsules. J. Biomed. Mater. Res. A 2010, 93, 528–536. [Google Scholar]
- Nelson, D.M.; Ma, Z.; Leeson, C.E.; Wagner, W.R. Extended and sequential delivery of protein from injectable thermoresponsive hydrogels. J. Biomed. Mater. Res. A 2012, 100, 776–785. [Google Scholar]
- Wang, L.; Huang, Y.; Pan, K.; Jiang, X.; Liu, C. Osteogenic responses to different concentrations/ratios of BMP-2 and bFGF in bone formation. Ann. Biomed. Eng 2010, 38, 77–87. [Google Scholar]
- Wei, G.; Jin, Q.; Giannobile, W.V.; Ma, P.X. Nano-fibrous scaffold for controlled delivery of recombinant human PDGF-BB. J. Control. Release 2006, 112, 103–110. [Google Scholar]
- Niu, X.; Feng, Q.; Wang, M.; Guo, X.; Zheng, Q. Porous nano-HA/collagen/PLLA scaffold containing chitosan microspheres for controlled delivery of synthetic peptide derived from BMP-2. J. Control. Release 2009, 134, 111–117. [Google Scholar]
- Yamamoto, M.; Ikada, Y.; Tabata, Y. Controlled release of growth factors based on biodegradation of gelatin hydrogel. J. Biomater. Sci 2001, 12, 77–88. [Google Scholar]
- Li, B.; Yoshii, T.; Hafeman, A.E.; Nyman, J.S.; Wenke, J.C.; Guelcher, S.A. The effects of rhBMP-2 released from biodegradable polyurethane/microsphere composite scaffolds on new bone formation in rat femora. Biomaterials 2009, 30, 6768–6779. [Google Scholar]
- Zhang, J.C.; Lu, H.Y.; Lv, G.Y.; Mo, A.C.; Yan, Y.G.; Huang, C. The repair of critical-size defects with porous hydroxyapatite/polyamide nanocomposite: An experimental study in rabbit mandibles. Int. J. Oral Maxillofac. Surg 2010, 39, 469–477. [Google Scholar]
- Guo, J.; Meng, Z.S.; Chen, G.; Xie, D.; Chen, Y.L.; Wang, H.; Tang, W.; Liu, L.; Jing, W.; Long, J.; et al. Restoration of critical-size defects in the rabbit mandible using porous nanohydroxyapatite-polyamide scaffolds. Tissue Eng. A 2012, 18, 1239–1252. [Google Scholar]
- Westermeier, R. Sensitive, quantitative, and fast modifications for Coomassie Blue staining of polyacrylamide gels. Proteomics 2006, 6, 61–64. [Google Scholar]





© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Su, J.; Xu, H.; Sun, J.; Gong, X.; Zhao, H. Dual Delivery of BMP-2 and bFGF from a New Nano-Composite Scaffold, Loaded with Vascular Stents for Large-Size Mandibular Defect Regeneration. Int. J. Mol. Sci. 2013, 14, 12714-12728. https://doi.org/10.3390/ijms140612714
Su J, Xu H, Sun J, Gong X, Zhao H. Dual Delivery of BMP-2 and bFGF from a New Nano-Composite Scaffold, Loaded with Vascular Stents for Large-Size Mandibular Defect Regeneration. International Journal of Molecular Sciences. 2013; 14(6):12714-12728. https://doi.org/10.3390/ijms140612714
Chicago/Turabian StyleSu, Jiansheng, Hongzhen Xu, Jun Sun, Xue Gong, and Hang Zhao. 2013. "Dual Delivery of BMP-2 and bFGF from a New Nano-Composite Scaffold, Loaded with Vascular Stents for Large-Size Mandibular Defect Regeneration" International Journal of Molecular Sciences 14, no. 6: 12714-12728. https://doi.org/10.3390/ijms140612714



