Metabolomic-Based Study of the Leafy Gall, the Ecological Niche of the Phytopathogen Rhodococcus fascians, as a Potential Source of Bioactive Compounds
Abstract
:1. Introduction
2. Results and Discussion
2.1. The Morphological Features of Plant Material
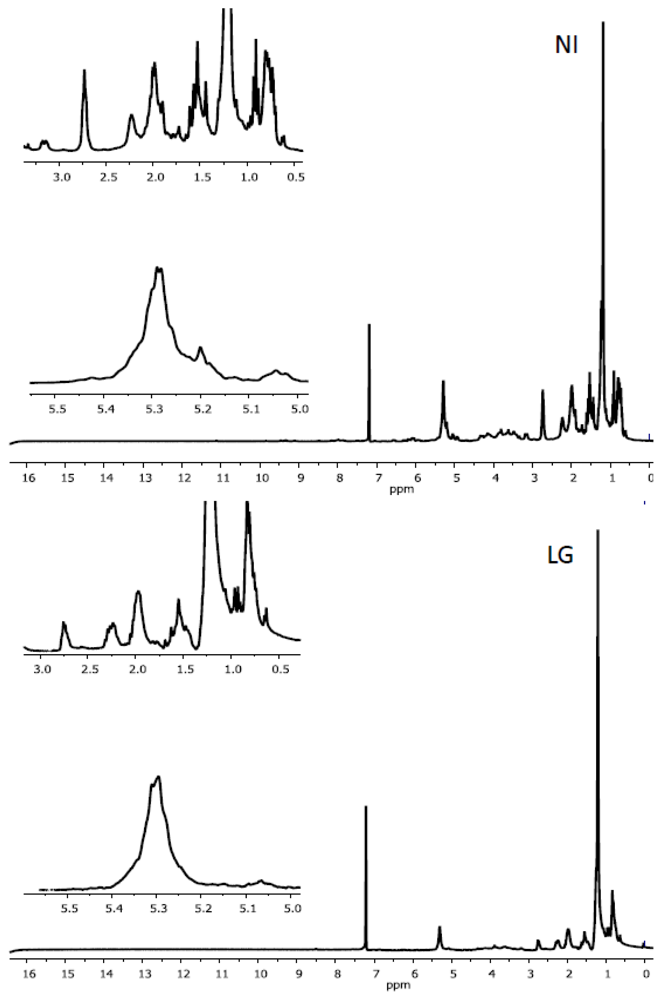
2.2. Characteristics of 1H NMR Spectra of Polar and Non-Polar Tobacco Extracts
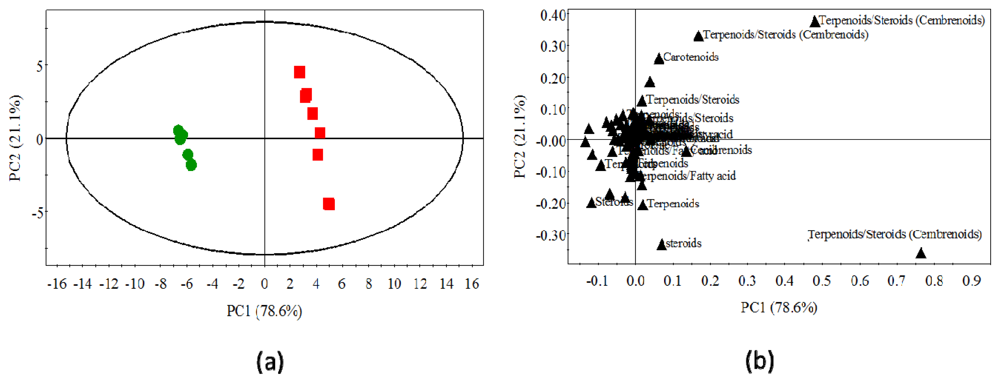
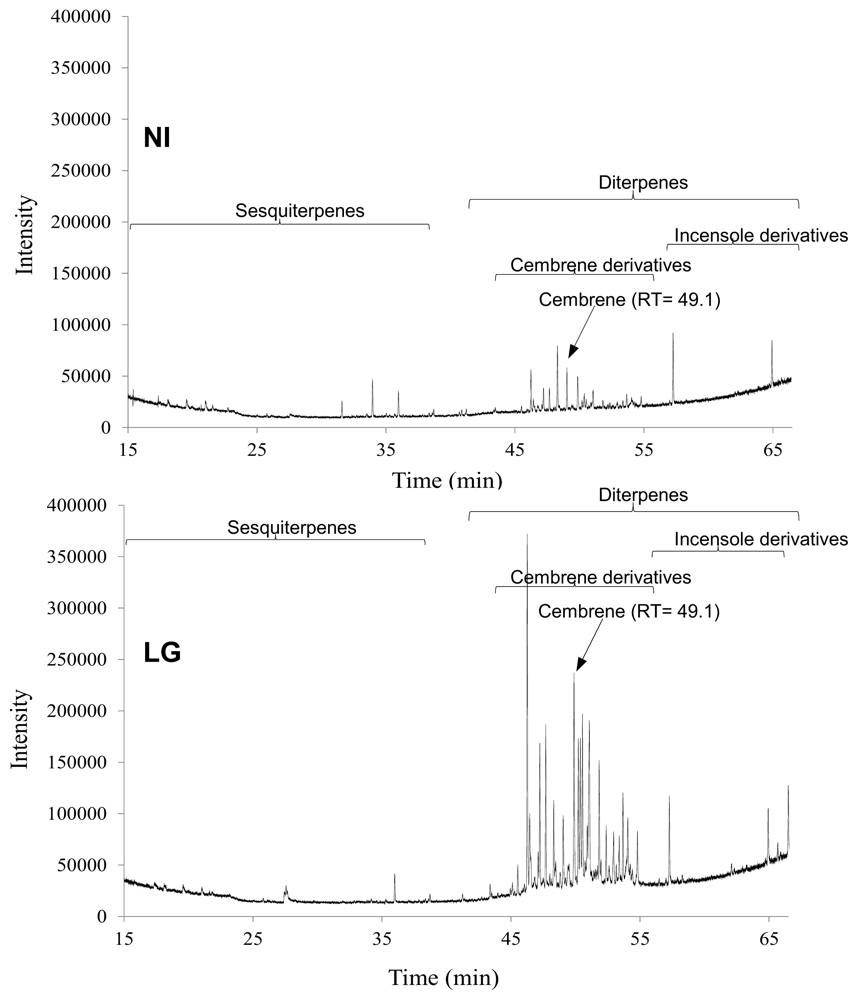
2.3. Statistical Analysis of the Non-Polar Metabolites
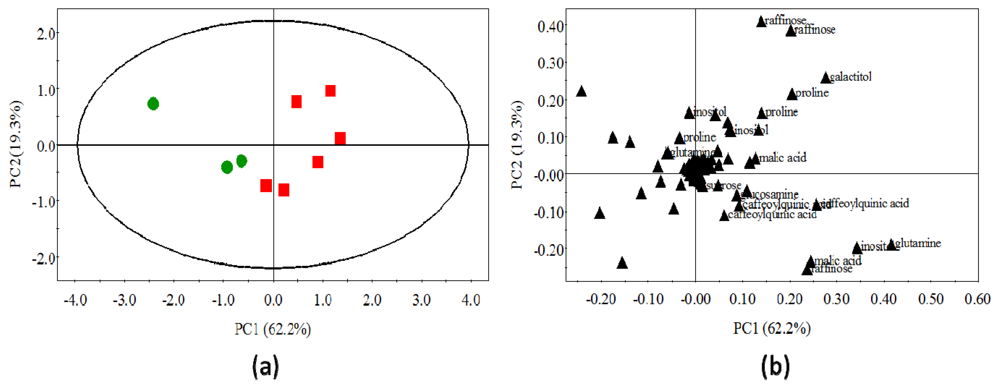
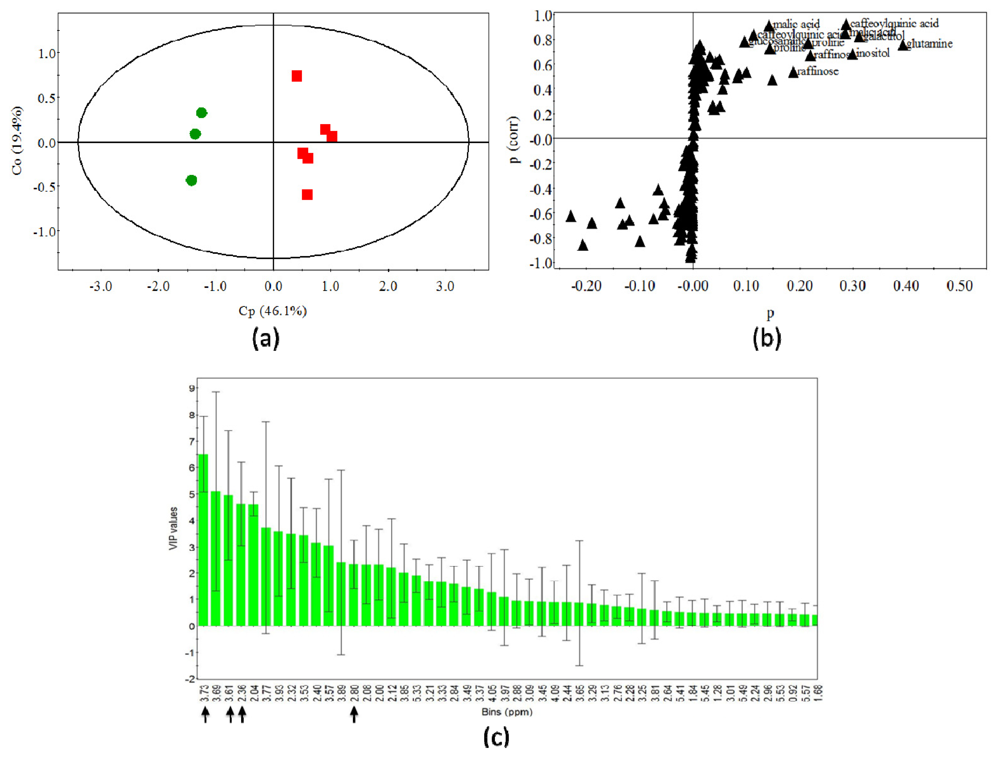
2.4. Statistical Analysis of Polar Metabolites
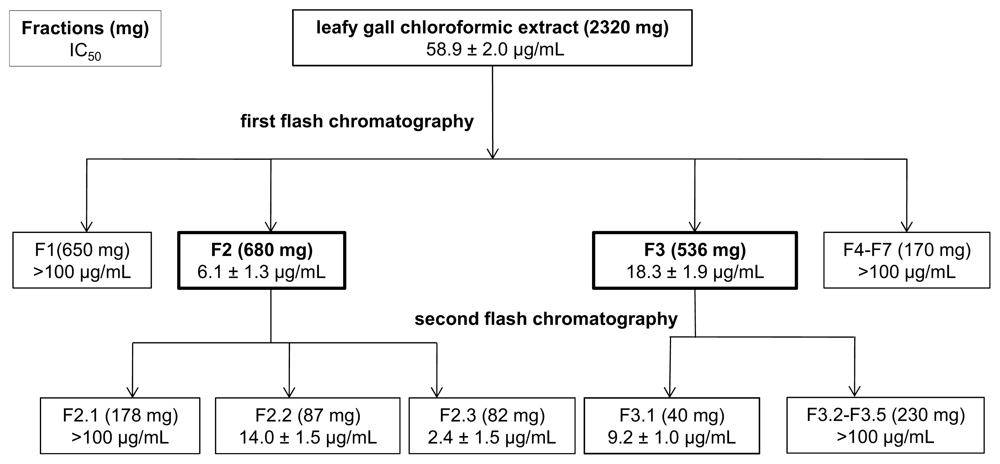
2.5. Leafy Gall (LG) Tissues Contain Potent Compounds That Affect the Proliferation of Different Human Cancer Cell Lines
3. Experimental Section
3.1. Solvents and Chemicals
3.2. Plant Material and Bacterial Infection
3.3. Established Cell Lines
3.4. Extraction of Plant Material
3.4.1. For Nuclear Magnetic Resonance (NMR) Analysis
3.4.2. Chromatgraphic Fractionnaltion of LG and Non-Infected (NI) Extracts for Antiproliferative Activity Measurment
3.5. 1H-NMR-Based Metabolomic Analysis of Tobacco Extracts
3.6. Data Analysis
3.7. Gas Chromatography and Mass Spectrometry GC-MS Analysis
3.8. Antiproliferative Activity by MTT Assay
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Agrios, G.N. Plant PathogensDisease: General Introduction. In Encyclopedia of Microbiology, 3rd ed.; Schaechter, M., Ed.; Elsevier Inc: Oxford, UK, 2009; pp. 613–646. [Google Scholar]
- Hayward, A.; Stone, G. Oak gall wasp communities: Evolution and ecology. Basic Appl. Ecol 2005, 6, 435–443. [Google Scholar]
- Bailey, R.; Schönrogge, K. Host niches and defensive extended phenotypes structure parasitoid wasp communities. PLoS Biol 2009, 7, e1000179. [Google Scholar]
- Barash, I.; Manulis-Sasson, S. Recent evolution of bacterial pathogens: The gall-forming Plantoea Agglomerans case. Annu. Rev. Phytopathol 2009, 47, 133–152. [Google Scholar]
- Stes, E.; Vandeputte, O.M. Successful bacterial coup d’État: How Rhodococcus fascians redirects plant development. Annu. Rev. Phytopathol 2011, 49, 69–86. [Google Scholar]
- Lamien, C.E.; Meda, A. Inhibition of fowlpox virus by an aqueous acetone extract from galls of Guiera senegalensis J.F. Gmel (Combretaceae). J. Ethnopharmacol 2005, 96, 249–253. [Google Scholar]
- Kaur, G.; Athar, M. Quercus infectoria galls possess antioxidant activity and abrogates oxidative stress-induced functional alterations in murine macrophages. Chem. Biol. Interact 2008, 171, 272–282. [Google Scholar]
- Heo, J.; Park, J. Wisteria floribunda gall extract inhibits cell migration in mouse B16F1 melanoma cells by regulating CD44 expression and GTP-RhoA activity. J. Ethnopharmacol 2005, 102, 10–14. [Google Scholar]
- Vandeputte, O.; Oden, S. Biosynthesis of auxin by the Gram-positive phytopathogen Rhodococcus fascians is controlled by compounds specific to infected plants. Appl. Environ. Microbiol 2005, 71, 1169–1177. [Google Scholar]
- Pertry, I.; Václavíková, K. Rhodococcus fascians impacts plant development through the dynamic fas-mediated production of a cytokinin mix. Mol. Plant Microbe Interact 2010, 23, 1164–1174. [Google Scholar]
- Simón-Mateo, C.; Depuydt, S. The phytopathogen Rhodococcus fascians breaks apical dominance and activates axillary meristems by inducing plant genes involved in hormone metabolism. Mol. Plant Pathol 2006, 7, 103–112. [Google Scholar]
- Depuydt, S.; Trenkamp, S. An integrated genomics approach to define niche establishment by Rhodococcus fascians. Plant Physiol 2009, 149, 1366–1386. [Google Scholar]
- Vereecke, D.; Messens, E. Patterns of phenolic compounds in leafy galls of tobacco. Planta 1997, 201, 342–348. [Google Scholar]
- Zotchev, S.B. Marine actinomycetes as an emerging resource for the drug development pipelines. J. Biotechnol 2012, 158, 168–175. [Google Scholar]
- Solecka, J.; Zajko, J. Biologically active secondary metabolites from Actinomycetes. Cent. Eur. J. Biol 2012, 7, 373–390. [Google Scholar]
- Ksouri, R.; Ksouri, W.M. Medicinal halophytes: Potent source of health promoting biomolecules with medical, nutraceutical and food applications. Crit. Rev. Biotechnol 2012, 32, 289–326. [Google Scholar]
- Savoia, D. Plant-derived antimicrobial compounds: Alternatives to antibiotics. Future Microbiol 2012, 7, 979–990. [Google Scholar]
- Zhao, K.; Penttinen, P. The diversity and anti-microbial activity of endophytic Actinomycetes isolated from medicinal plants in Panxi plateau, China. Curr. Microbiol 2011, 62, 182–190. [Google Scholar]
- Nacoulma, A.P.; Compaoré, M. In vitro antioxidant and anti-inflammatory activities of extracts from Nicotiana tabacum L. (Solanaceae) leafy galls induced by Rhodococcus fascians. J. Phytopathol 2012, 160, 617–621. [Google Scholar]
- Kueger, S.; Steinhauser, D. High-resolution plant metabolomics: From mass spectral features to metabolites and from whole-cell analysis to subcellular metabolite distributions. Plant J 2012, 70, 39–50. [Google Scholar]
- Qin, X.; Dai, Y. Metabolic Fingerprinting by 1HNMR for discrimination of the two species used as Radix Bupleuri. Planta Med 2012, 78, 926–933. [Google Scholar]
- Graham, S.F.; Amigues, E. Application of NMR based metabolomics for mapping metabolite variation in European wheat. Metabolomics 2009, 5, 302–306. [Google Scholar]
- Hamm, S.; Lesellier, E. Optimization of headspace solid phase microextraction for gas chromatography/mass spectrometry analysis of widely different volatility and polarity terpenoids in olibanum. J. Chromatogr. A 2003, 1018, 73–83. [Google Scholar]
- Cui, H.; Zhang, S.T. Gene expression profile analysis of tobacco leaf trichomes. BMC Plant Biol 2011, 11, 76–86. [Google Scholar]
- Quaglia, M.; Fabrizi, M. Role of pathogen-induced volatiles in the Nicotiana tabacum-Golovinomyces cichoracearum interaction. Plant Physiol. Biochem 2012, 52, 9–20. [Google Scholar]
- Huang, J.; Cardoza, Y.J. Differential volatile emissions and salicylic acid levels from tobacco plants in response to different strains of Pseudomonas syringae. Planta 2003, 217, 767–775. [Google Scholar]
- Seo, S.; Seto, H. A diterpene as an endogenous signal for the activation of defense responses to infection with tobacco mosaic virus and wounding in tobacco. Plant Cell 2003, 15, 863–873. [Google Scholar]
- Shah, J. Plants under attack: Systemic signals in defense. Curr. Opin. Plant Biol 2009, 12, 459–464. [Google Scholar]
- Wiklund, S.; Johansson, E. Visualization of GC/TOF-MS-based metabolomics data for identification of biochemically interesting compounds using OPLS class models. Anal. Chem 2008, 80, 115–122. [Google Scholar]
- Choi, Y.H.; Kim, H.K. NMR metabolomics to revisit the tobacco mosaic virus infection in Nicotiana tabacum leaves. J. Nat. Prod 2006, 69, 742–748. [Google Scholar]
- Fernie, A.R. The future of metabolic phytochemistry: Larger numbers of metabolites, higher resolution, greater understanding. Phytochemistry 2007, 68, 2861–2880. [Google Scholar]
- Peluffo, L.; Lia, V. Metabolic profiles of sunflower genotypes with contrasting response to Sclerotinia sclerotiorum infection. Phytochemistry 2010, 71, 70–80. [Google Scholar]
- Berger, S.; Sinha, A.K. Plant physiology meets phytopathology: Plant primary metabolism and plant–pathogen interactions. J. Exp. Bot 2007, 58, 4019–4026. [Google Scholar]
- Nouar, H.E.; Vereecke, D. Screening for differential gene expression in Atropa belladonna leafy gall induced following Rhodococcus fascians infection. Eur. J. Plant Pathol 2003, 109, 327–330. [Google Scholar]
- Vandeputte, O.; Oukouomi Lowe, Y. The tobacco Ntann12 gene, encoding an annexin, is induced upon Rhodococcus fascians infection and during leafy gall development. Mol. Plant Pathol 2007, 8, 185–194. [Google Scholar]
- Dixon, R.A. Natural products and plant disease resistance. Nature 2001, 411, 843–847. [Google Scholar]
- Fiamegos, Y.C.; Kastritis, P.L. Antimicrobial and efflux pump inhibitory activity of caffeoylquinic acids from Artemisia absinthium against gram-positive pathogenic bacteria. PLoS One 2011, 6, e18127. [Google Scholar]
- Baraka, H.N.; Khanfar, M.A. Bioactive natural, biocatalytic, and semisynthetic tobacco cembranoids. Planta Med 2011, 77, 467–476. [Google Scholar]
- Rajaonson, S.; Vandeputte, O.M. Virulence quenching with a prenylated isoflavanone renders the Malagasy legume Dalbergia pervillei resistant to Rhodococcus fascians. Environ. Microbiol 2011, 13, 1236–1252. [Google Scholar]
- Debeir, O.; Megalizzi, V. Videomicroscopic extraction of specific information on cell proliferation and migration in vitro. Exp. Cell Res 2008, 314, 2985–2998. [Google Scholar]
- Bury, M.; Girault, A. Ophiobolin A induces paraptosis-like cell death in human glioblastoma cells by decreasing BKCa channel activity. Cell Death Dis 2013, 4. [Google Scholar] [CrossRef]


| Extracts | DF | F | p-value |
|---|---|---|---|
| Polar metabolites | 8 | 0.15 | 0.71 |
| Non-polar metabolites | 12 | 636.3 | 4.37 × 10−11 |
| Multivariate data analysis models | A | n | R2 | Q2 |
|---|---|---|---|---|
| PCA | ||||
| Non-polar metabolites | 3 | 13 | 0.99 | 0.99 |
| Polar metabolites | 6 | 9 | 0.99 | 0.65 |
| OPLS-DA | ||||
| Polar metabolites | 1+4 | 9 | 0.98 | 0.93 |
| Significant polar metabolites | 1H chemical shifts (ppm) | VIP values |
|---|---|---|
| 5,O-caffeoylquinic acid | 3.73 (dd, J = 9.7 Hz, 3.5 Hz, H-4) | 6.48 |
| Inositol | 3.61 (t, J = 9.7 Hz, H-4 and 6) | 4.94 |
| Proline | 2.34 (m, H-3) | 4.61 |
| Malic acid | 2.80 (dd, J = 15.9 Hz, 4.5 Hz, H-b) | 2.33 |
| Cell lines | Infected plants (IC50 μg/mL) | Non infected plants (IC50 μg/mL) | ||||||
|---|---|---|---|---|---|---|---|---|
| Water | Methanol | Chloroform | Hexan | Water | Methanol | Chloroform | Hexan | |
| A549 | NA | >100 | 69.8 ± 0.9 | >100 | NA | NA | >100 | >100 |
| MCF-7 | NA | NA | 85.6 ± 1.0 | >100 | NA | NA | >100 | NA |
| PC3 | NA | NA | 84.5 ± 4.3 | >100 | NA | NA | >100 | NA |
| U373 | NA | >100 | 58.9 ± 2.0 | >100 | NA | NA | >100 | >100 |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Nacoulma, A.P.; Vandeputte, O.M.; De Lorenzi, M.; El Jaziri, M.; Duez, P. Metabolomic-Based Study of the Leafy Gall, the Ecological Niche of the Phytopathogen Rhodococcus fascians, as a Potential Source of Bioactive Compounds. Int. J. Mol. Sci. 2013, 14, 12533-12549. https://doi.org/10.3390/ijms140612533
Nacoulma AP, Vandeputte OM, De Lorenzi M, El Jaziri M, Duez P. Metabolomic-Based Study of the Leafy Gall, the Ecological Niche of the Phytopathogen Rhodococcus fascians, as a Potential Source of Bioactive Compounds. International Journal of Molecular Sciences. 2013; 14(6):12533-12549. https://doi.org/10.3390/ijms140612533
Chicago/Turabian StyleNacoulma, Aminata P., Olivier M. Vandeputte, Manuella De Lorenzi, Mondher El Jaziri, and Pierre Duez. 2013. "Metabolomic-Based Study of the Leafy Gall, the Ecological Niche of the Phytopathogen Rhodococcus fascians, as a Potential Source of Bioactive Compounds" International Journal of Molecular Sciences 14, no. 6: 12533-12549. https://doi.org/10.3390/ijms140612533




