The Physiological Importance of Glucosinolates on Plant Response to Abiotic Stress in Brassica
Abstract
:1. Introduction
2. Changes in the Glucosinolate Profile under Abiotic Stress: Implications for Plant Adaptation
2.1. Salinity
2.2. Drought
2.3. Extreme Temperatures and Light Cycling
2.4. Nutritional Deficiencies
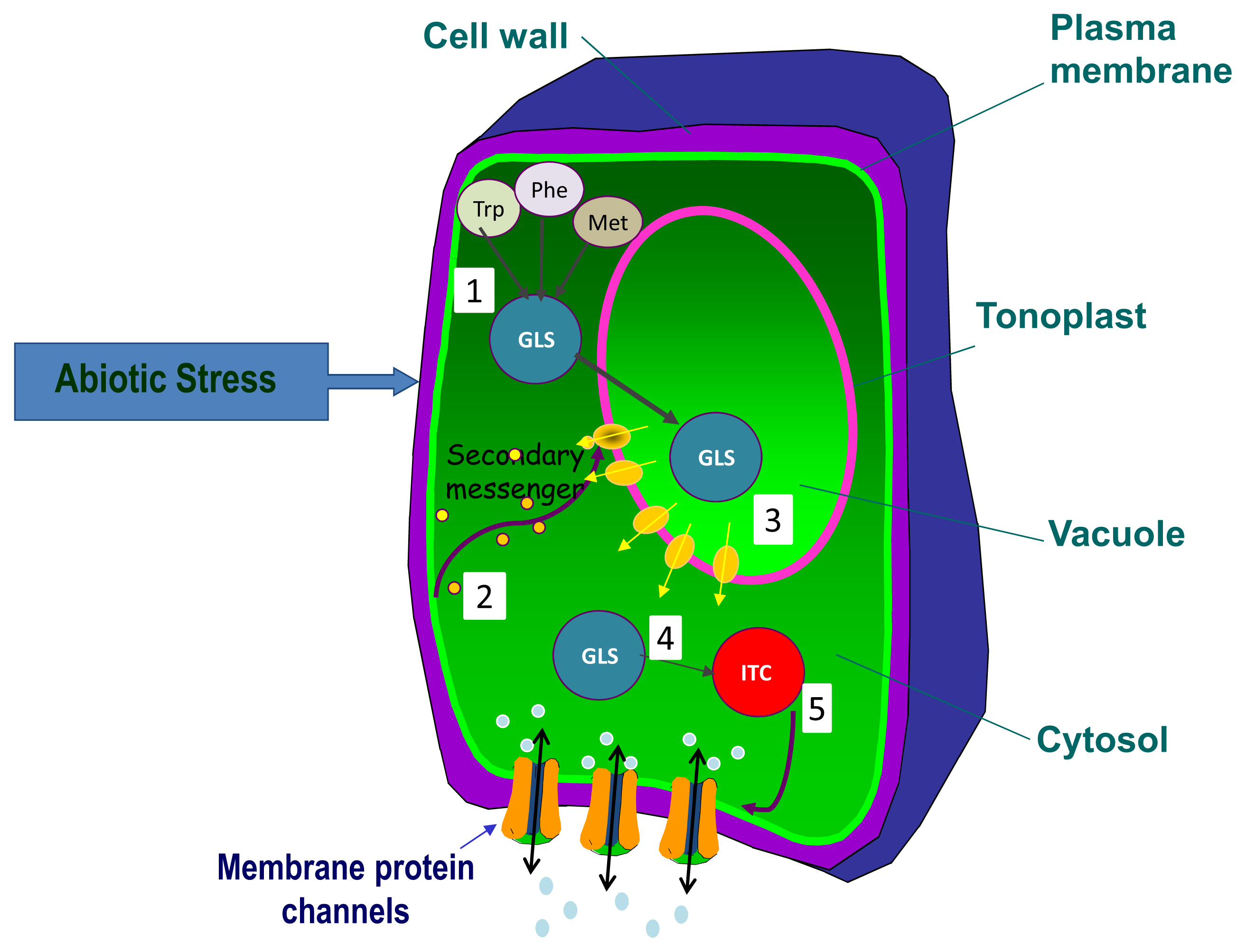
3. Glucosinolate Organ or Tissue Allocation and Transport under Abiotic Stress
4. Concluding Remarks
Acknowledgments
References
- Fahey, J.W.; Zalcmann, A.T.; Talalay, P. The chemical diversity and distribution of glucosinolates and isothiocyanates among plants. Phytochemistry 2001, 56, 5–51. [Google Scholar]
- Wittstock, U.; Halkier, B.A. Glucosinolate research in the Arabidopsis era. Trends Plant Sci 2002, 7, 263–270. [Google Scholar]
- Bones, A.M.; Rossiter, J.T. The enzymic and chemically induced decomposition of glucosinolates. Phytochemistry 2006, 67, 1053–1067. [Google Scholar]
- Mithen, R.F.; Dekker, M.; Verkerk, R.; Rabot, S.; Johnson, I.T. The nutritional significance, biosynthesis and bioavailability of glucosinolates in human foods. J. Sci. Food Agric 2000, 80, 967–984. [Google Scholar]
- Traka, M.; Mithen, R. Glucosinolates, isothiocyanates and human health. Phytochem. Rev 2009, 8, 269–282. [Google Scholar]
- Schnug, E. Sulfur nutrition and quality of vegetables. Sulphur Agric 1990, 16, 3–7. [Google Scholar]
- Schnug, E. Pysiological Functions and Environmental Relevance of Sulphur-Containing Secondary Metabolites. In Sulfur Nutrition and Assimilation in Higher Plants; de Kok, L., Stulen, I., Rennenberg, H., Eds.; Academic Publishing: The Hague, The Netherlands, 1993; pp. 179–190. [Google Scholar]
- Ernst, W. Ecological Aspects of Sulfur in Higher Plants: The Impact of SO2, the Evolution of the Biosynthesis of Organic Sulfur Compounds on Populations, Ecosystems. De Kok, L.J., Stulen, I., Rennenberg, H., Brunold, C., Rauser, W.E., Eds.; Eds.SPB Academic Publishing: The Hague, The Netherlands, 1993; pp. 125–138. [Google Scholar]
- Van der Kooij, T.A.W.; de Kok, L.J.; Haneklaus, S.; Schnug, E. Uptake, metabolism of sulphur dioxide by Arabidopsis thaliana. New Phytol 1997, 137, 101–107. [Google Scholar]
- Westerman, S.; Stulen, I.; Suter, M.; Brunold, C.; de Kok, L.J. Atmospheric H2S as sulfur source for Brassica oleracea: Consequences for the activity of the enzymes of the assimilatory sulfate reduction pathway. Plant Physiol. Biochem 2001, 39, 425–432. [Google Scholar]
- Khan, N.A.; Nazar, R.; Anjum, N.A. Growth, photosynthesis and antioxidant metabolism in mustard (Brassica juncea L.) cultivars differing in ATP-sulfurylase activity under salinity stress. Sci. Hort 2009, 122, 455–460. [Google Scholar]
- Chen, Y.Z.; Pang, Q.Y.; Hea, Y.; Zhua, N.; Branstroma, I.; Yanb, X.F.; Chen, S. Proteomics and metabolomics of arabidopsis esponses to perturbation of glucosinolate biosynthesis. Mol. Plant 2012, 5, 1138–1150. [Google Scholar]
- Endara, M.J.; Coley, P.D. The resource availability hypothesis revisited: A meta-analysis. Funct. Ecol 2011, 25, 389–398. [Google Scholar]
- Engelen-Eigles, G.; Holden, G.; Cohen, J.D.; Gardner, G. The effect of temperature, photoperiod, and light quality on gluconasturtiin concentration in watercress (Nasturtium officinale R. Br.). J. Agric. Food Chem 2006, 54, 328–334. [Google Scholar]
- Velasco, P.; Cartea, M.E.; Gonzalez, C.; Vilar, M.; Ordas, A. Factors affecting the glucosinolate content of kale (Brassica oleracea acephala group). J. Agric. Food Chem 2007, 55, 955–962. [Google Scholar]
- Qasim, M.; Ashraf, M.; Ashraf, M.Y.; Rehman, S.U.; Rha, E.S. Salt induced changes in two canola cultivars differing in salt tolerance. Biol. Plantarum 2003, 46, 629–632. [Google Scholar]
- López-Berenguer, C.; Martínez-Ballesta, M.C.; Moreno, D.A.; Carvajal, M.; García-Viguera, C. Growing hardier crops for better health: Salinity tolerance and the nutritional value of broccoli. J. Agric. Food Chem 2009, 57, 572–578. [Google Scholar]
- Gardner, G. The Center for Plants and Human Health: An Interdisciplinary Approach. NABC Report on Foods for Health: Integrating Agriculture, Medicine and Food for Future Health. In National Agricultural Biotechnology Council; Eaglesham, A., Carlson, C., Hardy, R.W.F., Eds.; USG Publishers: Ithaca, NY, USA, 2002; pp. 299–308. [Google Scholar]
- Radovich, T.J.K.; Kleinhenz, M.D.; Streeter, J.G. Irrigation timing relative to head development influences yield components, sugar levels, and glucosinolate concentrations in cabbage. J. Am. Soc. Hortic. Sci 2005, 130, 943–949. [Google Scholar]
- Zhu, J.K. Plant salt tolerance. Trends Plant Sci 2001, 6, 66–71. [Google Scholar]
- Munns, R.; Tester, M. Mechanisms of salinity tolerance. Annu. Rev. Plant Physiol 2008, 59, 651–681. [Google Scholar]
- López-Berenguer, C.; Martínez-Ballesta, M.C.; García-Viguera, C.; Carvajal, M. Leaf water balance mediated by aquaporins under salt stress and associated glucosinolate synthesis in broccoli. Plant Sci 2008, 174, 321–328. [Google Scholar]
- Yuan, G.F.; Wang, X.P.; Guo, R.F.; Wang, Q.M. Effect of salt stress on phenolic compounds, glucosinolates, myrosinase and antioxidant activity in radish sprouts. Food Chem 2010, 121, 1014–1019. [Google Scholar]
- Pang, Q.; Guo, J.; Chen, S.; Chen, Y.; Zhang, L.; Fei, M.; Jin, S.; Li, M.; Wang, Y.; Yan, X. Effect of salt treatment on the glucosinolate-myrosinase system in Thellungiella salsuginea. Plant Soil 2012, 355, 363–374. [Google Scholar]
- López-Pérez, L.; Martínez Ballesta, M.C.; Maurel, C.; Carvajal, M. Changes in plasma membrane composition of broccoli roots as an adaptation to increase water transport under salinity. Phytochemistry 2009, 70, 492–500. [Google Scholar]
- Brown, P.D.; Tokuhisa, J.G.; Reichelt, M.; Gershenzon, J. Variation of glucosinolate accumulation among different organs and developmental stages of Arabidopsis thaliana. Phytochemistry 2003, 62, 471–481. [Google Scholar]
- Pang, Q.; Chen, S.; Li, L.; Yan, X. Characterization of glucosinolate-myrosinase system in developing salt cress Thellungiella Halophila. Physiol. Plantarum 2009, 136, 1–9. [Google Scholar]
- Halkier, B.A.; Gershenzon, J. Biology and biochemistry of glucosinolates. Annu. Rev. Plant Biol 2006, 57, 303–333. [Google Scholar]
- Yan, X.; Chen, S. Regulation of plant glucosinolate metabolism. Planta 2007, 226, 1343–1352. [Google Scholar]
- Galletti, S.; Barillari, J.; Iori, R.; Venturi, G. Glucobrassicin enhancement in woad (Isatis tinctoria) leaves by chemical and physical treatments. J. Sci. Food Agric 2006, 86, 1833–1838. [Google Scholar]
- Andersson, D.; Chakrabarty, R.; Bejai, S.; Zhang, J.; Rask, L.; Meijer, J. Myrosinases from root and leaves of Arabidopsis thaliana have different catalytic properties. Phytochemistry 2009, 70, 1345–1354. [Google Scholar]
- Steinbrenner, A.D.; Agerbirk, N.; Orians, C.M.; Chew, F.S. Transient abiotic stresses lead to latent defense and reproductive responses over the Brassica rapa life cycle. Chemoecology 2012, 22, 239–250. [Google Scholar]
- Champolivier, L.; Merrien, A. Effects of water stress applied at different growth stages to Brassica napus L. var. oleifera on yield, yield components and seed quality. Eur. J. Agron 1996, 5, 153–160. [Google Scholar]
- Paschold, P.J.; Kleber, J.; Adam, S.T.; Bognar, A.; Tauscher, B. Einfluss von bewasserung und N-dungung auf ertrag und sulforaphangehalt von brokkoli (Brassica oleracea). In Proceedings of Deutsche Gesellschaft für Qualit atsforschung; Deutsche Gesellschaft für Qualit atsforschung: Karlsruhe, Germany, 2000; pp. 57–66. [Google Scholar]
- Jensen, C.R.; Mogensen, V.; Mortensen, G.; Fieldsen, J.; Milford, G.; Andersen, M.; Thage, J. Seed glucosinolate, oil and protein contents of field-grown rape (Brasica napus L.) affected by soil drying and evaporative demand. Field Crops Res 1996, 47, 93–105. [Google Scholar]
- Zhang, H.; Schonhof, I.; Krurnbein, A.; Gutezeit, B.; Li, L.; Stuzel, H.; Schreiner, M. Water supply and growing season influence glucosinolate concentration and composition in turnip root (Brassica rapa ssp rapifera L.). J. Plant Nutr. Soil Sci 2008, 171, 255–265. [Google Scholar]
- Schreiner, M.; Beyene, B.; Krumbein, A.; Stutzel, H. Ontogenetic changes of 2-propenyl and 3-indolylmethyl glucosinolates in Brassica carinata leaves as affected by water supply. J. Agric. Food Chem 2009, 57, 7259–7263. [Google Scholar]
- Jones, C.G.; Hartley, S.E. A protein competition model of phenolic allocation. Oikos 1999, 86, 27–44. [Google Scholar]
- Gutbrodt, B.; Dorn, S.; Unsicker, S.B.; Mody, K. Species-specific responses of herbivores to within-plant and environmentally mediated between-plant variability in plant chemistry. Chemoecology 2012, 22, 101–111. [Google Scholar]
- Robbins, R.J.; Keck, A.S.; Banuelos, G.; Finley, J.W. Cultivation conditions and selenium fertilization alter the phenolic profile, glucosinolate, and sulforaphane content of broccoli. J. Med. Food 2005, 8, 204–214. [Google Scholar]
- Gutbrodt, B.; Mody, K.; Dorn, S. Drought changes plant chemistry and causes contrasting responses in lepidopteran herbivores. Oikos 2011, 120, 1732–1740. [Google Scholar]
- Khan, M.A.M.; Ulrichs, C.h.; Mewis, I. Drought stress—Impact on glucosinolate profile and performance of phloem feeding cruciferous insects. Acta Hort 2011, 917, 111–117. [Google Scholar]
- Ren, H.; Zhong, H.X.; Dai, S.J.; Chen, S.X.; Yan, X.F. Water stress on glucosinolate contents in Arabidopsis rosette leaves. Acta Ecol. Sin 2009, 8, 4372–4379. [Google Scholar]
- Hartley, S.E.; Jones, T.H. Plant diversity and insect herbivores: Effects of environmental change in contrasting model systems. Oikos 2003, 101, 6–17. [Google Scholar]
- Hale, B.K.; Herms, D.A.; Hansen, R.C.; Clausen, T.P.; Arnold, D. Effects of drought stress and nutrient availability on dry matter. J. Chem. Ecol 2005, 11, 2601–2620. [Google Scholar]
- Khan, M.A.M.; Ulrichs, C.h.; Mewis, I. Water stress alters aphid-induced glucosinolate response in Brassica oleracea var. italica differently. Chemoecology 2011b, 21, 235–242. [Google Scholar]
- Mewis, I.; Mohammed, A.; Khan, M.; Glawischnig, E.; Schreiner, M.; Ulrichs, C. Water stress and aphid feeding differentially influence metabolite composition in Arabidopsis thaliana (L.). PLoS One 2012, 7, 1–15. [Google Scholar]
- Tariq, M.; Rossiter, J.T.; Wright, D.J.; Staley, J.T. Drought alters interactions between root and foliar herbivores. Oecologia 2013. [Google Scholar] [CrossRef]
- Ton, J.; Flors, V.; Mauch-Mani, B. The multifaceted role of ABA in disease resistance. Trends Plant Sci 2009, 14, 310–317. [Google Scholar]
- Beattie, G.A. Water relations in the interaction of foliar bacterial pathogens with plants. Annu. Rev. Phytopathol 2011, 49, 533–555. [Google Scholar]
- Atkinson, N.J.; Urwin, P.E. The interaction of plant biotic and abiotic stresses: From genes to the field. J. Exp. Bot 2012, 63, 3523–3544. [Google Scholar]
- Baerenfaller, K.; Massonnet, C.; Walsh, S.; Baginsky, S.; Buhlmann, P.; Hennig, L.; Hirsch-Hoffmann, M.; Howell, K.A.; Kahlau, S.; Radziejwoski, A.; et al. Systems-based analysis of Arabidopsis leaf growth reveals adaptation to water deficit. Mol. Syst. Biol 2012, 8, 1–18. [Google Scholar]
- Zhao, Z.; Zhang, W.; Stanley, B.A.; Assmann, S.M. Functional proteomics of Arabidopsis thaliana guard cells uncovers new stomatal signaling pathways WOA. Plant Cell 2008, 20, 3210–3226. [Google Scholar]
- Khokon, M.A.; Jahan, M.S.; Rahman, T.; Hossain, M.A.; Muroyama, D.; Minami, I.; Munemasa, S.; Mori, I.C.; Nakamura, Y.; Murata, Y. Allyl isothiocyanate (AITC) induces stomatal closure in Arabidopsis. Plant Cell Environ 2011, 34, 1900–1906. [Google Scholar]
- Vadassery, J.; Reichelt, M.; Hause, B.; Gershenzon, J.; Boland, W.; Mithöfer, A. CML42-mediated calcium signaling coordinates responses to Spodoptera herbivory and abiotic stresses in Arabidopsis. Plant Physiol 2012, 159, 1159–1175. [Google Scholar]
- Siemens, D.; Haugen, R.; Matzner, S.; Vanasma, N. 2009. Plant chemical defense allocation constrains evolution of local range. Mol. Ecol 2009, 18, 4974–4983. [Google Scholar]
- Siemens, D.H.; Duvall-Jisha, J.; Jacobs, J.; Manthey, J.; Haugen, R.; Matzner, S. Water deficiency induces evolutionary tradeoff between stress tolerance and chemical defense allocation that may help explain range limits in plants. Oikos 2012, 121, 790–800. [Google Scholar]
- Schreiner, M.; Huyskens-Keil, S.; Peters, P.; Schonhof, I.; Krumbein, A.; Widell, S. Seasonal climate effects on root colour and compounds of red radish. J. Sci. Food Agric 2002, 82, 1325–1333. [Google Scholar]
- Ito, H.; Kimura, M. Pre-harvest effects on naturally occurred isothiocyanates (ITCs) of cruciferous sprouts. Acta Hort 2006, 712, 497–503. [Google Scholar]
- Rosa, E.A.S.; Heaney, R.K.; Portas, C.A.M.; Fenwick, G.R. Changes in glucosinolate concentrations in Brassica crops (B. oleracea and B. napus) throughout growing seasons. J. Sci. Food Agric 1996, 71, 237–244. [Google Scholar]
- Sarwar, M.; Kirkegaard, J.A. Biofumigation potential of brassicas II: Effect of environment and ontogeny on glucosinolate production and implications for screening. Plant Soil 1998, 201, 91–101. [Google Scholar]
- Padilla, G.; Cartea, M.E.; Velasco, P.; Haro, A.d.; Ordás, A. Variation of glucosinolates in vegetable crops of Brassica rapa. Phytochemistry 2007, 68, 536–545. [Google Scholar]
- Charron, C.S.; Sams, C.E. Glucosinolate content and myrosinase activity in rapid cycling Brassica oleracea grown in a controlled environment. J. Am. Soc. Hort. Sci 2004, 129, 321–330. [Google Scholar]
- Farnham, M.W.; Wilson, P.E.; Stephenson, K.K.; Fahey, J.W. Genetic and environmental effects on glucosinolate content and chemoprotective potency of broccoli. Plant Breed 2004, 123, 60–65. [Google Scholar]
- Charron, C.S.; Saxton, A.M.; Sams, C.E. Relationship of climate and genotype to seasonal variation in the glucosinolate–myrosinase system I: Glucosinolate content in ten cultivars of Brassica oleracea grown in fall and spring seasons. J. Sci. Food Agric 2005, 85, 671–681. [Google Scholar]
- Nilsson, J.; Olsson, K.; Engqvist, G.; Ekvall, J.; Olsson, M.; Nyman, M.; Åkesson, B. Variation in the content of glucosinolates, hydroxycinnamic acids, carotenoids, total antioxidant capacity and low-molecular-weight carbohydrates in Brassica vegetables. J. Sci. Food Agric 2006, 86, 528–538. [Google Scholar]
- Cartea, M.E.; Velasco, P.; Obregon, S.; Padilla, G.; de Haro, A. Seasonal variation in glucosinolate content in Brassica oleracea crops grown in northwestern Spain. Phytochemistry 2008, 69, 403–410. [Google Scholar]
- Justen, V.L.; Fritz, V.A. Temperature-induced glucosinolate accumulation is associated with expression of BrMYB transcription factors. Hortscience 2013, 48, 47–52. [Google Scholar]
- Justen, V.L.; Cohen, J.D.; Gardner, G.; Fritz, V.A. Seasonal variation in glucosinolate accumulation in turnip cultivars grown with colored plastic mulches. Hortscience 2011, 46, 1608–1614. [Google Scholar]
- Bohinc, T.; Trdan, S. Environmental factors affecting the glucosinolate content in Brassicaceae. J. Food Agric. Environ. 2012, 10, 357–360. [Google Scholar]
- Rosa, A.S.; Rodrigues, P.M.F. The effect of light and temperature on glucosinolate concentration in the leaves and roots of cabbage seedlings. J. Sci. Food Agric 1998, 78, 208–212. [Google Scholar]
- Rosa, E.A.S.; Heaney, R.K.; Rego, F.C.; Fenwick, G.R. The variation of glucosinolate concentration during a single day in young plants of Brassica oleracea var. Acephala and Capitata. J. Sci. Food Agric 2006, 66, 457–463. [Google Scholar]
- Rosa, E.A.S. Daily variation in glucosinolate concentrations in the leaves and roots of cabbage seedlings in two constant temperature regimes. J. Sci. Food Agric 1997, 73, 364–368. [Google Scholar]
- Huseby, S.; Koprivova, A.; Lee, B.R.; Saha, S.; Mithen, R.; Wold, A.B.; Bengtsson, G.B.; Kopriva, S. Diurnal and light regulation of sulphur assimilation and glucosinolate biosynthesis in Arabidopsis. J. Exp. Bot. 2013. [Google Scholar] [CrossRef]
- Pérez-Balibrea, S.; Moreno, D.A.; García-Viguera, C. Influence of light on health promoting phytochemicals of broccoli sprouts. J. Sci. Food Agric 2008, 88, 904–910. [Google Scholar]
- Björkman, M.; Klingen, I.; Birch, A.N.E.; Bones, A.M.; Bruce, T.J.A.; Johansen, T.J.; Meadow, R.; Mølmann, J.; Seljåsen, R.; Smart, L.E.; et al. Phytochemicals of Brassicaceae in plant protection and human health-Influences of climate, environment and agronomic practice. Phytochemistry 2011, 72, 538–556. [Google Scholar]
- Sønderby, I.E.; Hansen, B.G.; Bjarnholt, N.; Ticconi, C.; Halkier, B.A.; Kliebenstein, D.J. A systems biology approach identifies a R2R3 MYB gene subfamily with distinct and overlapping functions in regulation of aliphatic glucosinolates. PLoS One 2007, 2, e1322. [Google Scholar]
- Gigolashvili, T.; Engqvist, M.; Yatusevich, R.; Muller, C.; Flugge, U.I. HAG2/MYB76 and HAG3/MYB29 exert a specific and coordinated control on the regulation of aliphatic glucosinolate biosynthesis in Arabidopsis thaliana. New Phytol 2008, 177, 627–642. [Google Scholar]
- Malitsky, S.; Blum, E.; Less, H.; Venger, I.; Elbaz, M.; Morin, S.; Eshed, Y.; Aharoni, A. The transcript and metabolite networks affected by the two clades of Arabidopsis glucosinolate biosynthesis regulators. Plant Physiol 2008, 148, 2021–2049. [Google Scholar]
- Kooke, R.; Keurentjes, J.JB. Multi-dimensional regulation of metabolic networks shaping plant development and performance. J. Exp. Bot. 2012, 63, 3353–3365. [Google Scholar]
- Kerwin, R.E.; Jimenez-Gomez, J.M.; Fulop, D.; Harmer, S.L.; Maloof, J.N.; Kliebenstein, D.J. Network quantitative trait loci mapping of circadian clock outputs identifies metabolic pathway-to-clock linkages in Arabidopsis. Plant Cell 2011, 23, 471–485. [Google Scholar]
- Klein, M.; Reichelt, M.; Gershenzo, J.; Papenbrock, J. The three desulfoglucosinolate sulfotransferase proteins in Arabidopsis have different substrate specificities and are differentially expressed. FEBS J 2006, 273, 122–136. [Google Scholar]
- Schuster, J.; Knill, T.; Reichelt, M.; Gershenzon, J.; Binder, S. Branched-chain aminotransferase 4 is part of the chain elongation pathway in the biosynthesis of methionine-derived glucosinolates in Arabidopsis. Plant Cell 2006, 18, 2664–2679. [Google Scholar]
- Müller, J.L.; Krishna, P.; Foreiter, C. A glucosinolate mutant of Arabidopsis is thermosensitive and defective in cytosolic Hsp90 expression after heat stress. Plant Physiol 2000, 123, 949–958. [Google Scholar]
- Hara, M.; Yatsuzuka, Y.; Tabata, K.; Kuboi, T. Exogenously applied isothiocyanates enhance glutathione S-transferase expression in Arabidopsis but act as herbicides at higher concentrations. J. Plant Physiol 2010, 167, 643–649. [Google Scholar]
- Hara, M.; Harazaki, A.; Tabata, K. Administration of isothiocyanates enhances heat tolerance in Arabidopsis thaliana. Plant Growth Regul 2013, 69, 71–77. [Google Scholar]
- Horváth, E.; Szalai, G.; Janda, T. Induction of abiotic stress tolerance by salicylic acid signaling. J. Plant Growth Regul 2007, 26, 290–300. [Google Scholar]
- Coulombe, J.; Villeneuve, S.; Lamy, P.; Yelle, S.; Bélec, C.; Tremblay, N. Evaluation of soil and petiole sap nitrate quick tests for broccoli in Québec. Acta Hortic 1998, 506, 147–152. [Google Scholar]
- Vågen, I.M. Nitrogen uptake in a broccoli crop. Nitrogen dynamics on a relative time scale. Acta Hortic 2003, 627, 195–202. [Google Scholar]
- Fabek, S.; Toth, N.; Radojcic-Redovnikovic, I.; Herak-Custic, M.; Benkoand, B.; Zutic, I. The effect of nitrogen fertilization on nitrate accumulation, and the content of minerals and glucosinolates in broccoli cultivars. Food Technol. Biotechnol. 2012, 50, 83–191. [Google Scholar]
- Schonhof, I.; Blankenburg, D.; Müller, S.; Krumbein, A. Sulfur and nitrogen supply influence growtn, product appearance, and glucosinolate concentration of broccoli. J. Plant Nutr. Soil Sci 2007, 170, 65–72. [Google Scholar]
- Koprivova, A.; Suter, M.; Op den Camp, R.; Brunold, C.; Kopriva, S. Regulation of sulfate assimilation by nitrogen in Arabidopsis. Plant Physiol 2000, 122, 737–746. [Google Scholar]
- Leustek, T.; Martin, M.N.; Bick, J.A.; Davies, J.P. Pathways and regulation of sulfur metabolism revealed through molecular and genetic studies. Annu. Rev. Plant Physiol. Plant Mol. Biol 2000, 51, 141–165. [Google Scholar]
- Omirou, M.D.; Papadopoulou, K.K.; Papastylianou, I.; Constantinou, M.; Karpouzas, D.G.; Asimakopoulos, I.; Ehaliotis, C. Impact of nitrogen and sulfur fertilization on the composition of glucosinolates in relation to sulfur assimilation in different plant organs of broccoli. J. Agric. Food Chem 2009, 57, 9408–9417. [Google Scholar]
- Li, S.; Schonhof, I.; Krumbein, A.; Li, L.; Stützel, H.; Schreiner, M. Glucosinolate concentration in turnip (Brassica rapa ssp. rapifera L.) roots as affected by nitrogen and sulfur supply. J. Agric. Food Chem 2007, 55, 8452–8457. [Google Scholar]
- Krumbein, A.; Schonhof, I.; Rühlmann, J.; Widell, S. Influence of Sulphur and Nitrogen Supply on Flavour and Health-Affecting Compounds in Brassicacea. In Plant Nutrition. Food Security and Sustainability of Agro-Ecosystems Through Basic and Applied Research; Horst, W.J., Schenk, M.K., Bürkert, A., Claassen, N., Flessa, H., Frommer, W.B., Goldbach, H., Olfs, H.W., Schubert, S., Wittenmayer, L., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2001; pp. 294–295. [Google Scholar]
- Kaur, S.; Gupta, S.K.; Sukhija, P.S.; Munshi, S.K. Accumulation of glucosinolates in developing mustard (Brassica juncea L.) seeds in response to sulphur application. Plant Sci 1990, 66, 181–184. [Google Scholar]
- Vallejo, F.; Tomás-Barberán, F.A.; Benavente-García, A.G.; García-Viguera, C. Total and individual glucosinolate contents in inflorescences of eight broccoli cultivars grown under various climatic and fertilisation conditions. J. Sci. Food Agric 2003, 83, 307–331. [Google Scholar]
- Troufflard, S.; Mullen, W.; Larson, T.R.; Graham, I.A.; Crozier, A.; Amtmann, A.; Armengaud, P. Potassium deficiency induces the biosynthesis of oxylipins and glucosinolates in Arabidopsis thaliana. BMC Plant Biol 2010, 10, 172–185. [Google Scholar]
- Kim, H.S.; Juvik, J.A. Effect of selenium fertilization and methyl jasmonate treatment on glucosinolate accumulation in broccoli florets. J. Am. Soc. Hort. Sci 2011, 136, 239–246. [Google Scholar]
- Shelp, B.J.; Shattuck, V.I.; McLellan, D.; Liu, L. Boron nutrition and the composition of glucosinolates and soluble nitrogen compounds in two broccoli (Brassica oleracea var. italica) cultivars. Can. J. Plant Sci 1992, 72, 889–899. [Google Scholar]
- Van der Meijden, E. Plant defense, an evolutionary dilemma: Contrasting effects of (specialist and generalist) herbivores and natural enemies. Entomol. Exp. Appl 1996, 80, 307–310. [Google Scholar]
- Stamp, N. Out of the quagmire of plant defense hypotheses. Q. Rev. Biol 2003, 78, 23–55. [Google Scholar]
- Frost, C.; Hunter, M. Herbivore induced shifts in carbon and nitrogen allocation in red oak seedlings. New Phytol 2008, 178, 835–845. [Google Scholar]
- Porter, A.J.R.; Morton, A.M.; Kiddle, G.; Doughty, K.J.; Wallsgrove, R.M. Variation in the glucosinolate content of oilseed rape (Brassica napus L.). I. Effect of leaf age and position. Ann. Appl. Biol 1991, 118, 461–467. [Google Scholar]
- Fieldsend, J.; Milford, G.F.J. Changes in glucosinolates during crop development in single- and double-low genotypes of winter oilseed rape (Brassica napus). I. Production and distribution in vegetative tissues and developing pods during development and potential role in the recycling of sulphur within the crop. Ann. Appl. Biol 1994, 124, 531–542. [Google Scholar]
- Van Dam, N.M.; Tytgat, T.O.G.; Kirkegaard, J.A. Root and shoot glucosinolates: A comparison of their diversity, function and interactions in natural and managed ecosystems. Phytochem. Rev 2009, 8, 171–186. [Google Scholar]
- Doughty, K.J.; Porter, A.J.R.; Morton, A.M.; Kiddle, G.; Bock, C.H.; Wallsgrove, R.M. Variation in the glucosinolate content of oilseed rape (Brassica napus L.) leaves. II. Response to infection by Alternaria brassicae (Berk.) Sacc. Ann. Appl. Biol 1991, 118, 469–477. [Google Scholar]
- Koritsas, V.M.; Lewis, J.A.; Fenwick, G.R. Glucosinolate responses of oilseed rape, mustard and kale to mechanical wounding and infestation by cabbage stem flea beetle (Psylliodes chrysocephala). Ann. Appl. Biol 1991, 118, 209–221. [Google Scholar]
- Bodnaryk, R.P. Effects of wounding on glucosinolates in the cotyledons of oilseed rape and mustard. Phytochemistry 1992, 31, 2671–2677. [Google Scholar]
- Falk, K.L.; Tokuhisa, J.G.; Gershenzon, J. The effect of sulfur nutrition on plant glucosinolate content: Physiology and molecular mechanisms. Plant Biol 2007, 9, 573–581. [Google Scholar]
- Pereira, F.; Rosa, E.; Fahey, J.W.; Stephenson, K.; Carvalho, R.; Aires, A. Influence of temperature and ontogeny on the levels of glucosinolates in broccoli (Brassica oleracea var. italica) sprouts and their effect on the induction of mammalian phase 2 enzymes. J Agric. Food Chem 2002, 50, 6239–6244. [Google Scholar]
- Gols, R.; Raaijmakers, C.E.; van Dam, N.M.; Dicke, M.; Bukovinszky, T.; Harvey, J.A. Temporal changes affect plant chemistry and tritrophic interactions. Basic Appl. Ecol 2007, 8, 421–433. [Google Scholar]
- Petersen, B.L.; Chen, S.X.; Hansen, C.H.; Olsen, C.E.; Halkier, B.A. Composition and content of glucosinolates in developing Arabidopsis thaliana. Planta 2002, 214, 562–571. [Google Scholar]
- Pongrac, P.; Vogel-Mikus, K.; Regvar, M.; Tolra, R.; Poschenrieder, C.; Barcelo, J. Glucosinolate profiles change during the life cycle and mycorrhizal colonization in a Cd/Zn hyperaccumulator Thlaspi praecox (Brassicaceae). J. Chem. Ecol 2008, 34, 1038–1044. [Google Scholar]
- Bekaert, M.; Edger, P.P.; Hudson, C.M.; Pires, J.C.; Conant, G.C. Metabolic and evolutionary costs of herbivory defense: Systems biology of glucosinolates synthesis. New Phytol 2012, 196, 596–605. [Google Scholar]
| Abiotic stress conditions | Plant cultivar | Glucosinolate content | References |
|---|---|---|---|
| Saline stress | |||
| NaCl (40, 80 mM), during two weeks | Brassica oleracea L. var. italica | Increase | López-Berenguer et al., 2008 [22] |
| NaCl (20,40, 60 mM), during 5 d | Brassica rapa L. | Increase | Steinbrenner et al., 2012 [32] |
| Drought | |||
| Severe stress two weeks | Brassica oleracea L. var. capitata | Increase | Radovich et al., 2005 [19] |
| Severe stress two weeks | Brassica oleracea L. var. italica | Increase | Champolivier and Merrien 1996 [33] |
| Severe stress more than one week | Brassica napus L. | Increase | Jensen et al., 1996 [35] |
| Mild stress-25% of available water | Brassica rapa ssp. rapifera L. | Increase | Zhang et al., 2008 [36] |
| Mild and severe stress (40, 23, 17 and 15% of available water) | Brassica carinata L. | Increase /No effect | Schreiner et al., 2009 [37] |
| Mild stress (30% of available water) | Brassica oleracea L. var. gemmifera | No effect | Gutbrodt et al., 2012 [39] |
| Mild stress | Brassica napus L. | No effect | Jensen et al., 1996 [35] |
| Mild and severe stress (40%–45% of available water) | Brassica oleracea L. | Decrease | Gutbrodt et al., 2011a Khan et al., 2011a [41,42] |
| Severe stress | Arabidopsis thaliana L. | Decrease | Ren et al., 2009 [43] |
| Temperature | |||
| Elevated temperature (21–34 °C) | Brassica rapa L. | Increase | Justen and Fritz 2013 [68] |
| Low-medium temperature (15–27 °C) | Brassica rapa L. | Decrease | Justen and Fritz 2013 [68] |
| Elevated temperature (32 °C) | Brassica oleracea L. | Increase | Charron et al., 2004, 2005 [63,65] |
| Light cycling | |||
| 14 h/10 h d/n * | Brassica oleracea L. | Decrease during day/increase during night | Rosa et al., 1997, 1998 [71,73] |
| 16 h/8 h d/n or continuous darkness | Arabidopsis thaliana L. | Increase upon light /decrease upon darkness | Huseby et al., 2013 [74] |
| 16 h/8 h d/n or continuous darkness | Brassica oleracea L. var. italica | Increase upon light | Pérez-Balibrea et al., 2008 [75] |
| Nutrient availability | |||
| N-limitation (1 gr N pot−1) | Brassica oleracea L. var. italica | Increase | Schonhof et al., 2007 [91] |
| S-supply (60 kg S ha−1) | Brassica rapa ssp. rapifera L | Increase | Li et al., 2007 [95] |
| S-supply (150 kg/ha) | Brassica oleracea L. var. italica | No effect | Vallejo et al., 2003 [98] |
| S-limitation (15 kg/ha) | Brassica oleracea L. var. italica | No effect | Vallejo et al., 2003 [98] |
| K-deficiency( lack KNO3 for two weeks) | Arabidopsis thaliana L. | Increase | Troufflard et al., 2010 [99] |
| K-deficiency ( lack of nutrient solution for five days) | Brassica rapa L. | Decrease | Steinbrenner et al., 2012 [32] |
| Se-supply (5.2 mM Na2SeO4) | Brassica oleracea L. var. italica | Increase | Kim et al., 2011 [100] |
| B-deficiency (9–12 μg gr DW−1) | Brassica oleracea L. var. italica | Shelp et al., 1992 [101] | |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Del Carmen Martínez-Ballesta, M.; Moreno, D.A.; Carvajal, M. The Physiological Importance of Glucosinolates on Plant Response to Abiotic Stress in Brassica. Int. J. Mol. Sci. 2013, 14, 11607-11625. https://doi.org/10.3390/ijms140611607
Del Carmen Martínez-Ballesta M, Moreno DA, Carvajal M. The Physiological Importance of Glucosinolates on Plant Response to Abiotic Stress in Brassica. International Journal of Molecular Sciences. 2013; 14(6):11607-11625. https://doi.org/10.3390/ijms140611607
Chicago/Turabian StyleDel Carmen Martínez-Ballesta, María, Diego A. Moreno, and Micaela Carvajal. 2013. "The Physiological Importance of Glucosinolates on Plant Response to Abiotic Stress in Brassica" International Journal of Molecular Sciences 14, no. 6: 11607-11625. https://doi.org/10.3390/ijms140611607
APA StyleDel Carmen Martínez-Ballesta, M., Moreno, D. A., & Carvajal, M. (2013). The Physiological Importance of Glucosinolates on Plant Response to Abiotic Stress in Brassica. International Journal of Molecular Sciences, 14(6), 11607-11625. https://doi.org/10.3390/ijms140611607








