Over-Expression of Deubiquitinating Enzyme USP14 in Lung Adenocarcinoma Promotes Proliferation through the Accumulation of β-Catenin
Abstract
:1. Introduction
2. Results and Discussion
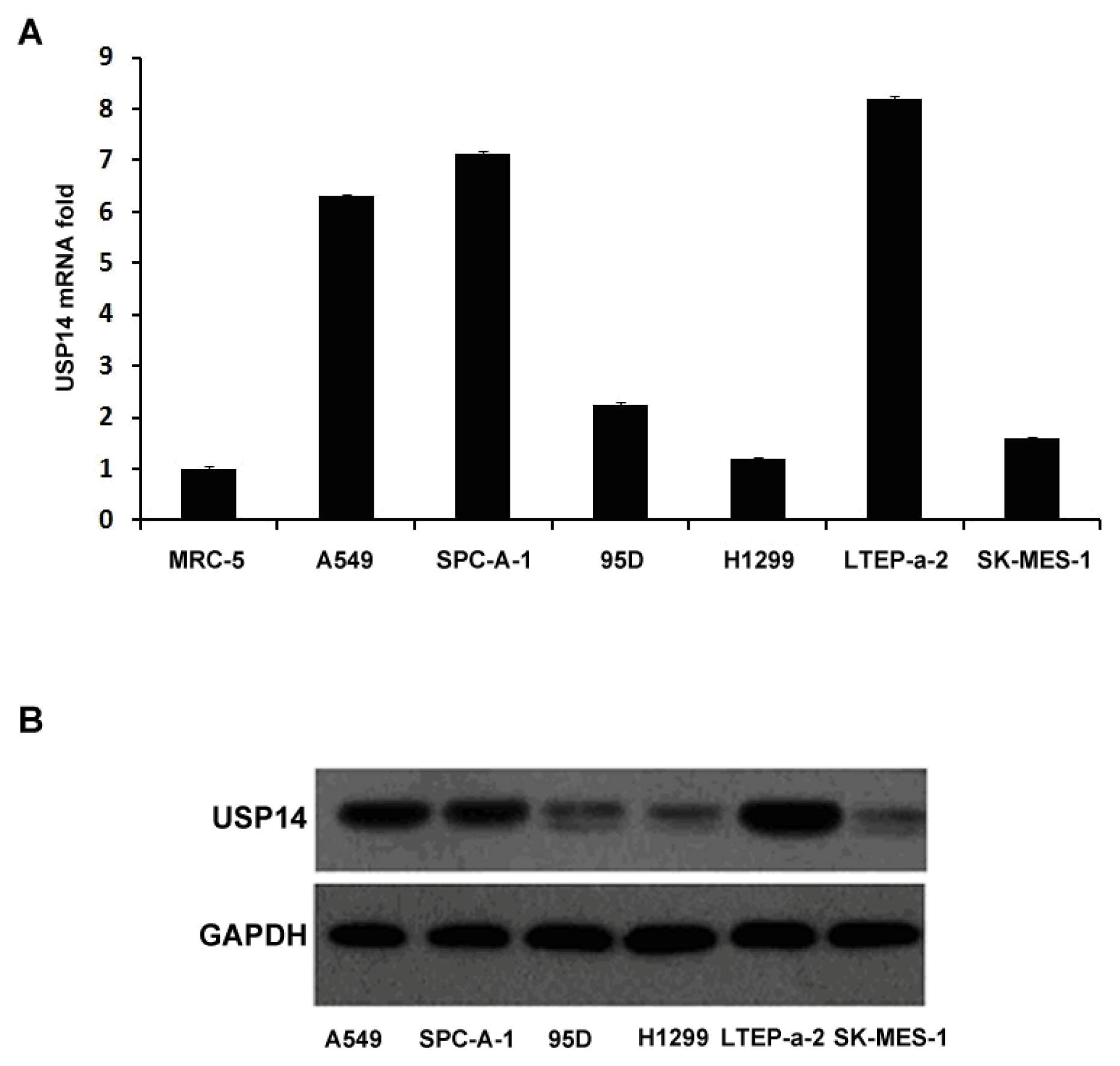
2.1. Expression of USP14 in Lung Adenocarcinoma Cell Lines
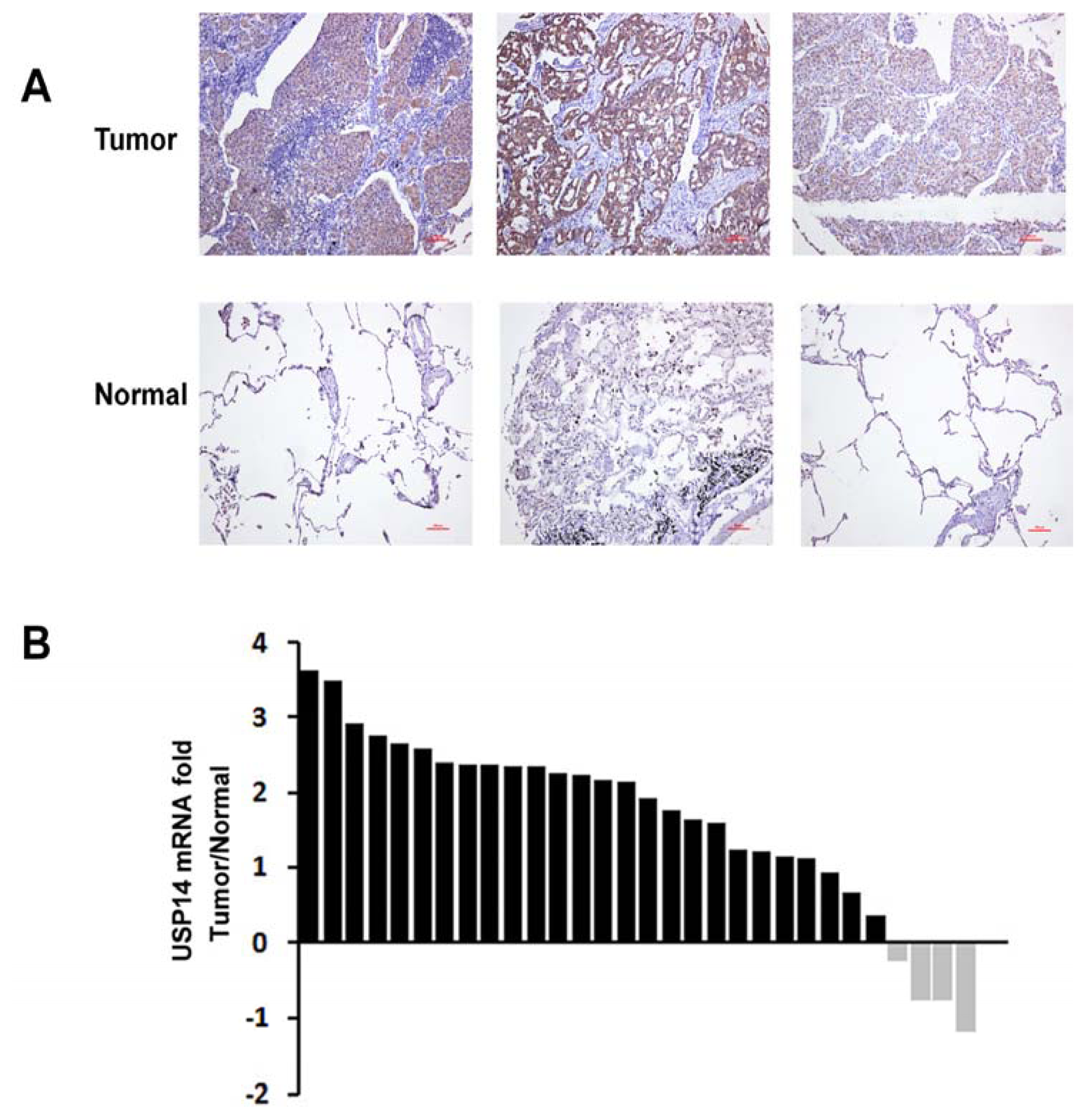
2.2. Expression of USP14 in NSCLC Tumor Tissues
2.3. Correlation between USP14 Expression and Overall Survival in Lung Adenocarcinoma Patients
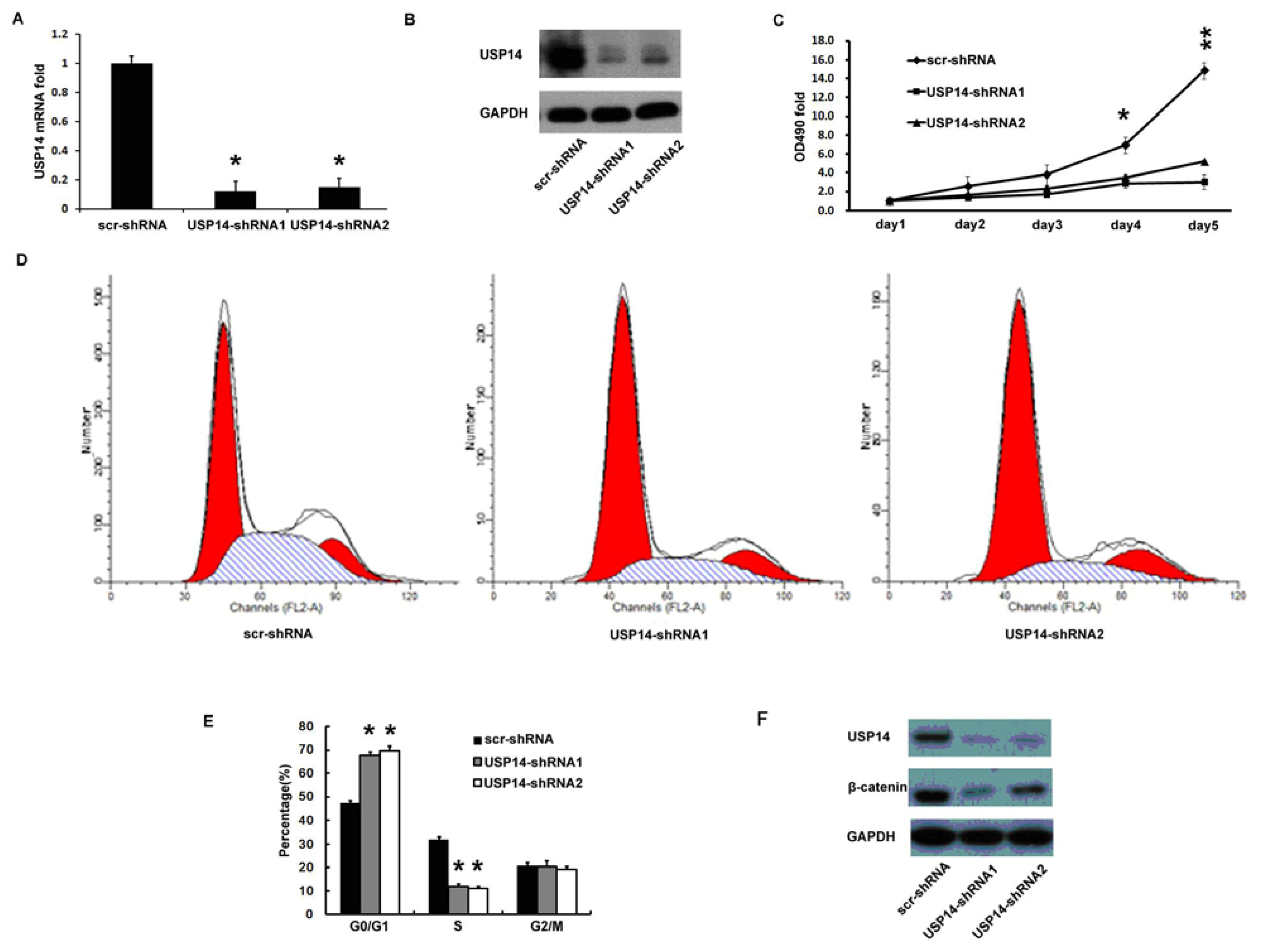
2.4. Silencing USP14 Impaired Lung Adenocarcinoma Cell Proliferation Coupling with β-Catenin Reduction
2.5. Discussion
3. Experimental Section
3.1. Patients
3.2. Cell Culture
3.3. Real-Time Quantitative Reverse Transcription Polymerase Chain Reaction
3.4. Western Blot
3.5. Immunohistochemistry
3.6. Cell Growth Assay
3.7. Cell Cycle Assay
3.8. Lentivirus Vectors
3.9. Statistical Analysis
4. Conclusions
Supplementary Information
ijms-14-10749-s001.pdfAcknowledgments
Conflict of Interest
References
- Ramalingam, S.S.; Owonikoko, T.K.; Khuri, F.R. Lung cancer: New biological insights and recent therapeutic advances. CA Cancer J. Clin 2011, 61, 91–112. [Google Scholar]
- Katlic, M.R.; Facktor, M.A.; Berry, S.A.; McKinley, K.E.; Bothe, A., Jr; Steele, G.D., Jr. Proven Care lung cancer: A multi-institutional improvement collaborative. CA Cancer J. Clin. 2011, 61, 382–396. [Google Scholar]
- Li, Xinxin; Wang, J.; Xu, Z.; Ahmad, A.; Li, E.; Wang, Y.; Qin, S.; Wang, Q. Expression of Sox2 and Oct4 and their clinical significance in human non-small-cell lung cancer. Int. J. Mol. Sci. 2012, 13, 7663–7675. [Google Scholar]
- Wen, C.; Dehnel, T. China wrestles with lung cancer. Lancet Oncol 2011, 12, 15. [Google Scholar]
- Wakelee, H.A.; Chang, E.T.; Gomez, S.L.; Keegan, T.H.; Feskanich, D.; Clarke, C.A.; Holmberg, L.; Yong, L.C.; Kolonel, L.N.; Gould, M.K.; West, D.W. Lung cancer incidence in never smokers. J. Clin. Oncol 2007, 25, 472–478. [Google Scholar]
- Fujii, T.; Dracheva, T.; Player, A.; Chacko, S.; Clifford, R.; Strausberg, R.L.; Buetow, K.; Azumi, N.; Travis, W.D.; Jen1, J. A preliminary transcriptome map of non-small cell lung cancer. Cancer Res 2002, 62, 3340–3346. [Google Scholar]
- Sowa, M.E.; Bennett, E.J.; Gygi, S.P.; Harper, J.W. Defining the human deubiquitinating enzyme interaction landscape. Cell 2009, 138, 389–403. [Google Scholar]
- Lee, B.H.; Lee, M.J.; Park, S.; Oh, D.-C.; Elsasser, S.; Chen, P.-C.; Gartner, C.; Dimova, N.; Hanna, J.; Gygi, S.P. Enhancement of proteasome activity by a small-molecule inhibitor of USP14. Nature 2010, 467, 179–184. [Google Scholar]
- Anderson, C.; Crimmins, S.; Wilson, J.A.; Korbel, G.A.; Ploegh, H.L.; Wilson, S.M. Loss of Usp14 results in reduced levels of ubiquitin in ataxia mice. J. Neurochem 2005, 95, 724–731. [Google Scholar]
- Nag, D.K.; Finley, D. A small-molecule inhibitor of deubiquitinating enzyme USP14 inhibits Dengue virus replication. Virus Res 2012, 165, 103–106. [Google Scholar]
- Peth, A.; Besche, H.C.; Goldberg, A.L. Ubiquitinated proteins activate the proteasome by binding to Usp14/Ubp6, which causes 20S gate opening. Mol. Cell 2009, 36, 794–804. [Google Scholar]
- Mines, M.A.; Goodwin, J.S.; Limbird, L.E.; Cui, F.F.; Fan, G.H. Deubiquitination of CXCR4 by USP14 is critical for both CXCL12-induced CXCR4 degradation and chemotaxis but not ERK ativation. J. Biol. Chem 2009, 284, 5742–5752. [Google Scholar]
- Wilson, S.M.; Bhattacharyya, B.; Rachel, R.A.; Coppola, V.; Tessarollo, L.; Householder, D.B.; Fletcher, C.F.; Miller, R.J.; Copeland, N.J.; Jenkins, N.A. Synaptic defects in ataxia mice result from a mutation in Usp14, encoding a ubiquitin-specific protease. Nat. Genet 2002, 32, 420–425. [Google Scholar]
- Borodovsky, A.; Kessler, B.M.; Casagrande, R.; Overkleeft, H.S.; Wilkinson, K.D.; Ploegh, H.L. A novel active site-directed probe specific for deubiquitylating enzymes reveals proteasome association of USP14. EMBO J 2001, 20, 5187–5196. [Google Scholar]
- Ishiwata, S.; Katayama, J.; Shindo, H.; Ozawa, Y.; Itoh, K.; Mizugaki, M. Increased expression of queuosine synthesizing enzyme, tRNA-guanine transglycosylase, and queuosine levels in tRNA of leukemic cells. J. Biochem 2001, 129, 13–17. [Google Scholar]
- Shinji, S.; Naito, Z.; Ishiwata, S.; Ishiwata, T.; Tanaka, N.; Furukawa, K.; Suzuki, H.; Seya, T.; Matsuda, A.; Katsuta, M.; Tajiri, T. Ubiquitin-specific protease 14 expression in colorectal cancer is associated with liver and lymph node metastases. Oncol. Rep 2006, 15, 539–543. [Google Scholar]
- Chuensumran, U.; Saelee, P.; Punyarit, P.; Wongkham, S.; Pairojkul, C.; Chauin, S.; Petmitr, S. Ubiquitin-specific protease 14 expression associated with intrahepatic cholangiocarcinoma cell differentiation. Asian Pac. J. Cancer Prev 2011, 12, 775–779. [Google Scholar]
- Yang, L.; Chen, Y.; Cui, T.; Knösel1, T.; Zhang, Q.; Albring, K.F.; Huber, O.; Petersen, I. Desmoplakin acts as a tumor suppressor by inhibition of the Wnt/beta-catenin signaling pathway in human lung cancer. Carcinogenesis 2012, 33, 1863–1870. [Google Scholar]
- Tsao, C.M.; Yan, M.D.; Shih, Y.L.; Yu, P.N.; Kuo, C.C.; Lin, W.C.; Li, H.J.; Lin, Y.W. SOX1 functions as a tumor suppressor through antagonizing the WNT/beta-catenin signaling pathway in hepatocellular carcinoma. Hepatology 2012, 56, 2277–2287. [Google Scholar]
- Subbaiah, V.K.; Narayan, N.; Massimi, P.; Banks, L. Regulation of the DLG tumor suppressor by beta-catenin. Int. J. Cancer 2012, 131, 2223–2233. [Google Scholar]
- Yoshioka, S.; King, M.L.; Ran, S.; Okuda, H.; MacLean, J.A.; McAsey, M.E.; Sugino, N.; Brard, L.; Watabe, K.; Hayashi, K. WNT7A regulates tumor growth and progression in ovarian cancer through the WNT/beta-catenin pathway. Mol. Cancer Res 2012, 10, 469–482. [Google Scholar]
- Shu, X.S.; Geng, H.; Li, L.; Ying, J.; Ma, C.; Wang, Y.; Poon, F.F.; Wang, X.; Ying, Y.; Yeo, W. The epigenetic modifier PRDM5 functions as a tumor suppressor through modulating WNT/beta-catenin signaling and is frequently silenced in multiple tumors. PLoS One 2011, 6, e27346. [Google Scholar]
- Wang, E.Y.; Yeh, S.H.; Tsai, T.F.; Huang, H.P.; Jeng, Y.M.; Lin, W.H.; Chen, W.C.; Yeh, K.H.; Chen, P.J.; Chen, D.S. Depletion of beta-catenin from mature hepatocytes of mice promotes expansion of hepatic progenitor cells and tumor development. Proc. Natl. Acad. Sci. USA 2011, 108, 18384–18389. [Google Scholar]
- Sadot, E.; Simcha, I.; Iwai, K.; Ciechanover, A.; Geiger, B.; Ben-Ze’ev, A. Differential interaction of plakoglobin and beta-catenin with the ubiquitin-proteasome system. Oncogene 2000, 19, 1992–2001. [Google Scholar]
- Tran, H.; Polakis, P. Reversible modification of adenomatous polyposis coli (APC) with K63-linked polyubiquitin regulates the assembly and activity of the beta-catenin destruction complex. J. Biol. Chem 2012, 287, 28552–28563. [Google Scholar]
- Jeong, W.J.; Yoon, J.; Park, J.C.; Lee, S.H.; Lee, S.H.; Kaduwal, S.; Kim, H.; Yoon, J.-B.; Choi, K.-Y. Ras stabilization through aberrant activation of Wnt/beta-catenin signaling promotes intestinal tumorigenesis. Sci. Signal. 2012, 5. [Google Scholar] [CrossRef]
- Chen, P.C.; Qin, L.N.; Li, X.M.; Walters, B.J.; Wilson, J.A.; Mei, L.; Wilson, S.M. The proteasome-associated deubiquitinating enzyme Usp14 is essential for the maintenance of synaptic ubiquitin levels and the development of neuromuscular junctions. J. Neurosci 2009, 29, 10909–10919. [Google Scholar]
- Hu, M.; Li, P.; Song, L.; Jeffrey, P.D.; Chernova, T.A.; Wilkinson, K.D.; Cohen, R.E.; Shi, Y. Structure and mechanisms of the proteasome-associated deubiquitinating enzyme USP14. EMBO J 2005, 24, 3747–3756. [Google Scholar]
- Inui, M.; Manfrin, A.; Mamidi, A.; Martello, G.; Morsut, L.; Soligo, S.; Enzo, E.; Moro, S.; Polo, S.; Dupont, S.; et al. USP15 is a deubiquitylating enzyme for receptor-activated SMADs. Nat. Cell Biol 2011, 13, 1368–1375. [Google Scholar]
- Eichhorn, P.J.; Rodon, L.; Gonzalez-Junca, A.; Dirac, A.; Gili, M.; Martínez-Sáez, E.; Aura, C.; Barba, I.; Peg, V.; Prat, A.; et al. USP15 stabilizes TGF-beta receptor I and promotes oncogenesis through the activation of TGF-beta signaling in glioblastoma. Nat. Med 2012, 18, 429–435. [Google Scholar]
- Wu, N.; Gu, H.J.; Li, Q. Effects of antidiabetic drug metformin on the migration and invasion abilities of human pulmonary adenocarcinoma A549 cell line in vitro. J. Thorac. Dis 2010, 2, 76–80. [Google Scholar]
- Hao, J.; Zhang, S.; Zhou, Y.; Hu, X.; Shao, C. MicroRNA 483-3p suppresses the expression of DPC4/Smad4 in pancreatic cancer. FEBS Lett 2011, 585, 207–213. [Google Scholar]
- Hao, J.; Zhang, S.; Zhou, Y.; Liu, C.; Hu, X.; Shao, C. MicroRNA 421 suppresses DPC4/Smad4 in pancreatic cancer. Biochem. Biophys. Res. Commun 2011, 406, 552–557. [Google Scholar]
- Liu, C.; Li, B.; Cheng, Y.; Lin, J.; Hao, J.; Zhang, S.; Mitchel, R.E.J.; Sun, D.; Ni, J.; Zhao, L.; et al. MiR-21 plays an important role in radiation induced carcinogenesis in BALB/c mice by directly targeting the tumor suppressor gene Big-h3. Int. J. Biol. Sci 2011, 7, 347–363. [Google Scholar]
- Chen, T.; Guo, J.; Yang, M.; Han, C.; Zhang, M.; Chen, W.; Liu, Q.; Wang, J.; Cao, X. Cyclosporin A impairs dendritic cell migration by regulating chemokine receptor expression and inhibiting cyclooxygenase-2 expression. Blood 2004, 103, 413–421. [Google Scholar]
- Xu, X.S.; Wang, L.; Abrams, J.; Wang, G. Histone deacetylases (HDACs) in XPC gene silencing and bladder cancer. J. Hematol. Oncol 2011, 4, 1–11. [Google Scholar]
- Lv, T.; Yuan, D.; Miao, X.; Lv, Y.; Zhan, P.; Shen, X.; Song, Y. Over-expression of LSD1 promotes proliferation, migration and invasion in non-small cell lung cancer. PLoS One 2012, 7, e35065. [Google Scholar]
- Liu, C.; Gao, F.; Li, B.; Mitchel, R.E.J.; Liu, X.; Lin, J.; Zhao, L.; Cai, J. TLR4 knockout protects mice from radiation-induced thymic lymphoma by downregulation of IL6 and miR-21. Leukemia 2011, 25, 1516–1519. [Google Scholar]
- Liu, C.; Zhou, C.; Gao, F.; Cai, S.; Zhang, C.; Zhao, L.; Zhao, F.; Cao, F.; Lin, J.; Yang, Y.; et al. MiR-34a in age and tissue related radio-sensitivity and serum miR-34a as a novel indicator of radiation injury. Int. J. Biol. Sci 2011, 7, 221–233. [Google Scholar]
- Liu, C.; Lin, J.; Zhao, L.; Yang, Y.; Gao, F.; Li, B.; Cui, J.; Cai, J. Gamma-ray irradiation impairs dendritic cell migration to CCL19 by down-regulation of CCR7 and induction of cell apoptosis. Int. J. Biol. Sci 2011, 7, 168–179. [Google Scholar]
- Hofig, I.; Atkinson, M.J.; Mall, S.; Krackhardt, A.M.; Thirion, C.; Anastasov, N. Poloxamer synperonic F108 improves cellular transduction with lentiviral vectors. J. Gene Med 2012, 14, 549–560. [Google Scholar]
- Luche, R.M.; Enssle, J.; Kiem, H.P. Abrogated cryptic activation of lentiviral transfer vectors. Sci. Rep 2012, 2. [Google Scholar] [CrossRef]
- Wu, D.T.; Seita, Y.; Zhang, X.; Lu, C.W.; Roth, M.J. Antibody-directed lentiviral gene transduction for live-cell monitoring and selection of human iPS and hES cells. PLoS One 2012, 7, e34778. [Google Scholar]

© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wu, N.; Liu, C.; Bai, C.; Han, Y.-P.; Cho, W.C.S.; Li, Q. Over-Expression of Deubiquitinating Enzyme USP14 in Lung Adenocarcinoma Promotes Proliferation through the Accumulation of β-Catenin. Int. J. Mol. Sci. 2013, 14, 10749-10760. https://doi.org/10.3390/ijms140610749
Wu N, Liu C, Bai C, Han Y-P, Cho WCS, Li Q. Over-Expression of Deubiquitinating Enzyme USP14 in Lung Adenocarcinoma Promotes Proliferation through the Accumulation of β-Catenin. International Journal of Molecular Sciences. 2013; 14(6):10749-10760. https://doi.org/10.3390/ijms140610749
Chicago/Turabian StyleWu, Ning, Cong Liu, Chong Bai, Yi-Ping Han, William C. S. Cho, and Qiang Li. 2013. "Over-Expression of Deubiquitinating Enzyme USP14 in Lung Adenocarcinoma Promotes Proliferation through the Accumulation of β-Catenin" International Journal of Molecular Sciences 14, no. 6: 10749-10760. https://doi.org/10.3390/ijms140610749
APA StyleWu, N., Liu, C., Bai, C., Han, Y.-P., Cho, W. C. S., & Li, Q. (2013). Over-Expression of Deubiquitinating Enzyme USP14 in Lung Adenocarcinoma Promotes Proliferation through the Accumulation of β-Catenin. International Journal of Molecular Sciences, 14(6), 10749-10760. https://doi.org/10.3390/ijms140610749





