A Dual Role of Strigolactones in Phosphate Acquisition and Utilization in Plants
Abstract
:1. Introduction
2. Phosphorus Availability Is a Major Factor Limiting Plant Growth, Development and Reproduction
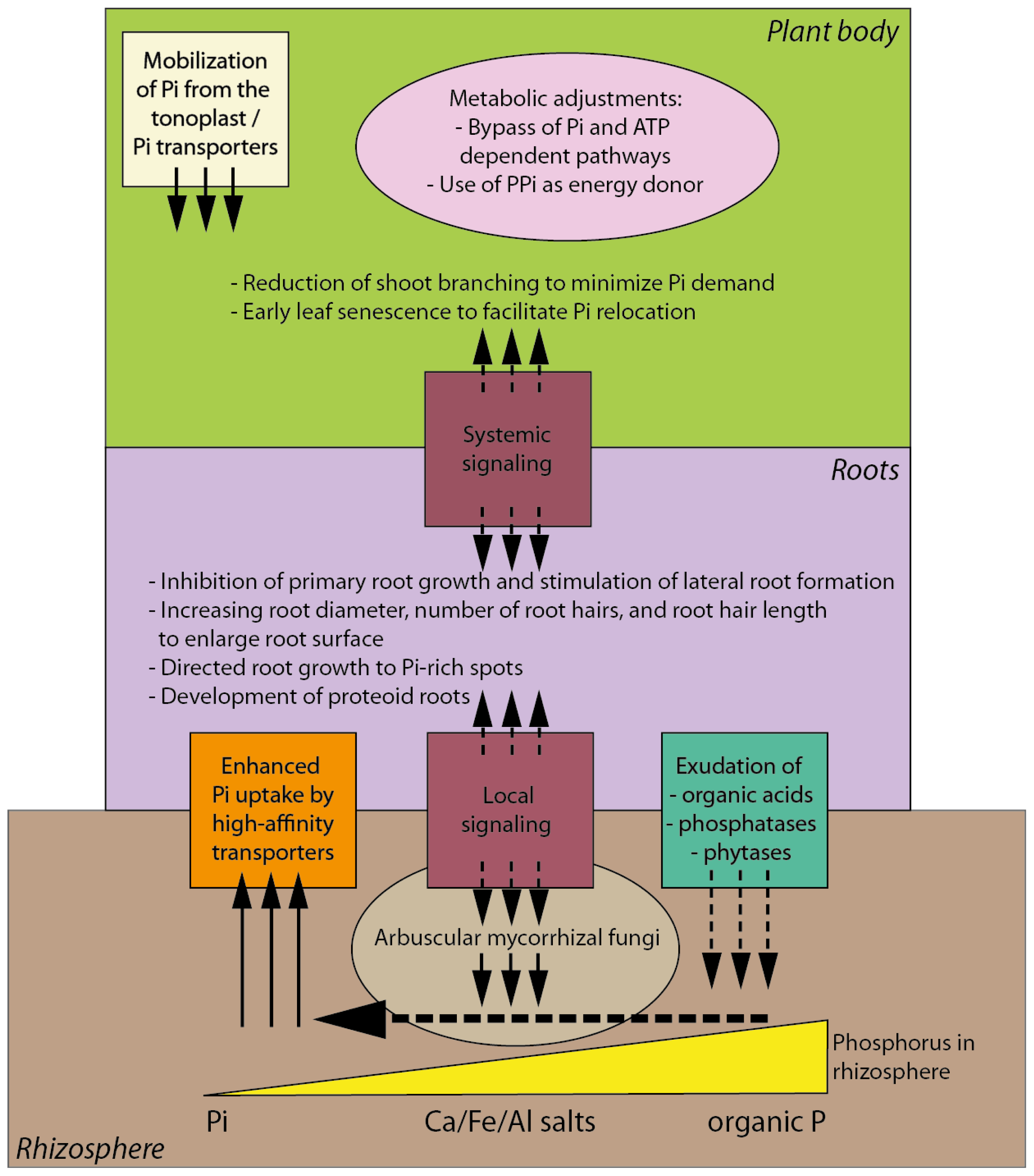
3. Morphological, Physiological and Biochemical Responses of Plants to Pi Deficiency
4. Complex Signaling Network Underlying Plant Response to Pi Deficiency
5. Strigolactones: Physiological Roles, Biosynthesis and Signaling
6. Pi Deficiency Stimulates SL Biosynthesis and Exudation
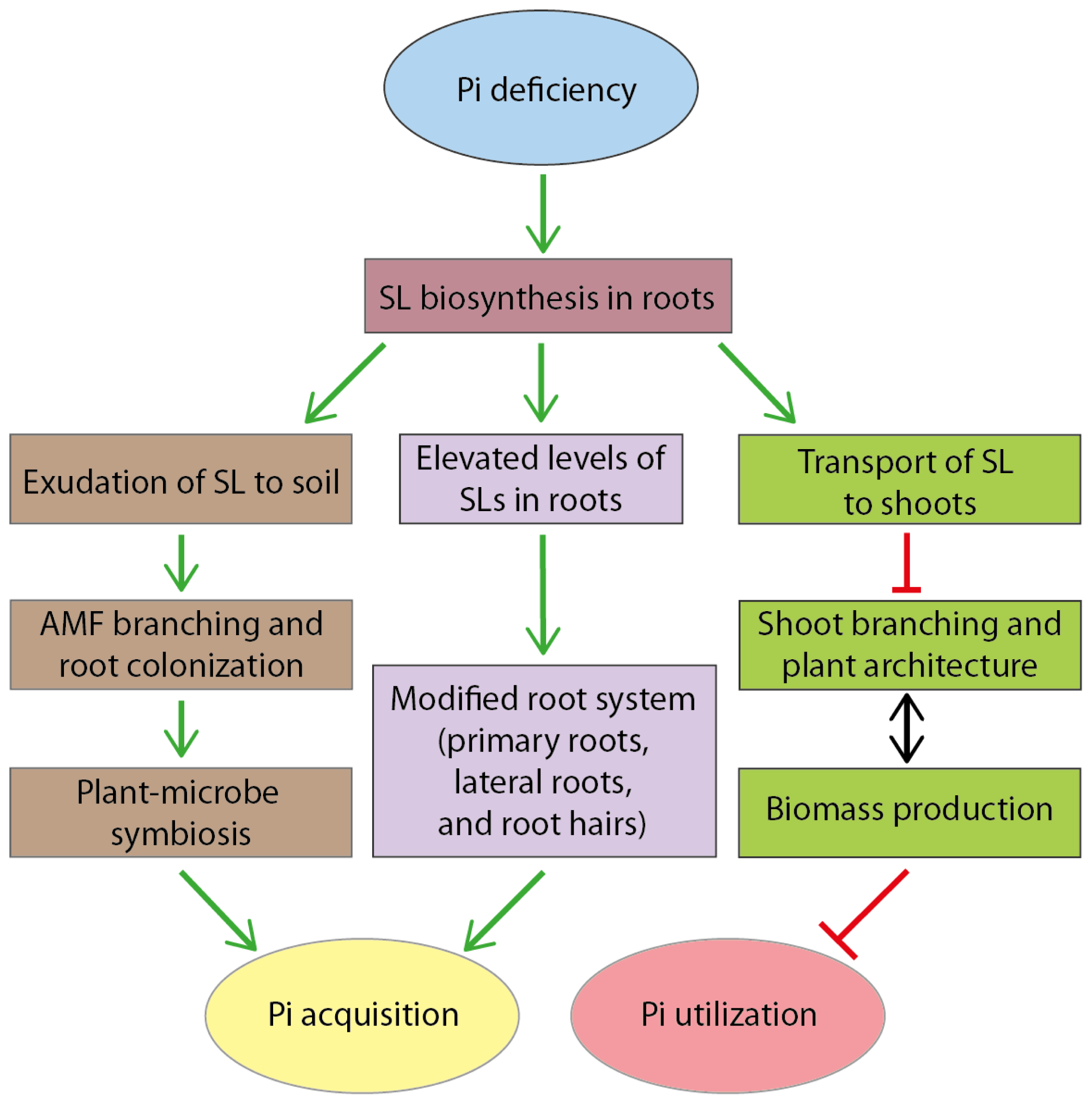
7. Model: A Dual Role of Strigolactones in Pi Acquisition and Utilization under Pi Deficiency Conditions
Acknowledgments
Conflict of Interest
References
- Gomez-Roldan, V.; Fermas, S.; Brewer, P.B.; Puech-Pages, V.; Dun, E.A.; Pillot, J.P.; Letisse, F.; Matusova, R.; Danoun, S.; Portais, J.C.; et al. Strigolactone inhibition of shoot branching. Nature 2008, 455, 189–194. [Google Scholar]
- Umehara, M.; Hanada, A.; Yoshida, S.; Akiyama, K.; Arite, T.; Takeda-Kamiya, N.; Magome, H.; Kamiya, Y.; Shirasu, K.; Yoneyama, K.; et al. Inhibition of shoot branching by new terpenoid plant hormones. Nature 2008, 455, 195–200. [Google Scholar]
- Yoneyama, K.; Xie, X.N.; Kusumoto, D.; Sekimoto, H.; Sugimoto, Y.; Takeuchi, Y.; Yoneyama, K. Nitrogen deficiency as well as phosphorus deficiency in sorghum promotes the production and exudation of 5-deoxystrigol, the host recognition signal for arbuscular mycorrhizal fungi and root parasites. Planta 2007, 227, 125–132. [Google Scholar]
- Yoneyama, K.; Yoneyama, K.; Takeuchi, Y.; Sekimoto, H. Phosphorus deficiency in red clover promotes exudation of orobanchol, the signal for mycorrhizal symbionts and germination stimulant for root parasites. Planta 2007, 225, 1031–1038. [Google Scholar]
- Umehara, M.; Hanada, A.; Magome, H.; Takeda-Kamiya, N.; Yamaguchi, S. Contribution of strigolactones to the inhibition of tiller bud outgrowth under phosphate deficiency in rice. Plant Cell Physiol 2010, 51, 1118–1126. [Google Scholar]
- Akiyama, K.; Matsuzaki, K.; Hayashi, H. Plant sesquiterpenes induce hyphal branching in arbuscular mycorrhizal fungi. Nature 2005, 435, 824–827. [Google Scholar]
- Schlesinger, W.H.; Bernhard, E.S. Biogeochemistry: An analysis of Global Change, 3rd ed; Academic Press: Waltham, MA, USA, 2013. [Google Scholar]
- Epstein, E. The anomaly of silicon in plant biology. Proc. Natl. Acad. Sci. USA 1994, 91, 11–17. [Google Scholar]
- Bieleski, R.L. Phosphate pools, phosphate pransport, and phosphate availability. Annu. Rev. Plant Physiol 1973, 24, 225–252. [Google Scholar]
- Paytan, A.; McLaughlin, K. The oceanic phosphorus cycle. Chem. Rev 2007, 107, 563–576. [Google Scholar]
- Holford, I.C.R. Soil phosphorus: Its measurement, and its uptake by plants. Aust. J. Soil Res 1997, 35, 227–239. [Google Scholar]
- Shen, J.B.; Yuan, L.X.; Zhang, J.L.; Li, H.G.; Bai, Z.H.; Chen, X.P.; Zhang, W.F.; Zhang, F.S. Phosphorus dynamics: from soil to plant. Plant Physiol 2011, 156, 997–1005. [Google Scholar]
- Schachtman, D.P.; Reid, R.J.; Ayling, S.M. Phosphorus uptake by plants: From soil to cell. Plant Physiol 1998, 116, 447–453. [Google Scholar]
- Marschner, P. Marschner’s Mineral Nutrition of Higher Plants, 3rd ed; Academic Press: San Diego, CA USA, 2012. [Google Scholar]
- Gilbert, N. Environment: The disappearing nutrient. Nature 2009, 461, 716–718. [Google Scholar]
- Vance, C.P.; Uhde-Stone, C.; Allan, D.L. Phosphorus acquisition and use: critical adaptations by plants for securing a nonrenewable resource. New Phytol 2003, 157, 423–447. [Google Scholar]
- Ae, N.; Arihara, J.; Okada, K.; Yoshihara, T.; Johansen, C. Phosphorus uptake by pigeon pea and its role in cropping systems of the Indian subcontinent. Science 1990, 248, 477–480. [Google Scholar]
- Lambers, H.; Raven, J.A.; Shaver, G.R.; Smith, S.E. Plant nutrient-acquisition strategies change with soil age. Trends Ecol. Evol 2008, 23, 95–103. [Google Scholar]
- Bennett, W.F. Nutrient Deficiencies & Toxocotoes in Crop Plants; APS Press: St. Paul, MN, USA, 1993. [Google Scholar]
- Dietz, K.J.; Heilos, L. Carbon metabolism in spinach leaves as affected by leaf age and phosphorus and sulfur nutrition. Plant Physiol 1990, 93, 1219–1225. [Google Scholar]
- Miginiacmaslow, M.; Hoarau, A. Variations in the adenylate levels during phosphate-depletion in isolated soybean cells and wheat leaf fragments. Z. Pflanzenphysiol 1982, 107, 427–436. [Google Scholar]
- Mikulska, M.; Bomsel, J.L.; Rychter, A.M. The influence of phosphate deficiency on photosynthesis, respiration and adenine nucleotide pool in bean leaves. Photosynthetica 1998, 35, 79–88. [Google Scholar]
- Plaxton, W.C. The organization and regulation of plant glycolysis. Annu. Rev. Plant Physiol 1996, 47, 185–214. [Google Scholar]
- Natr, L. Mineral nutrients—A ubiquitous stress factor for photosynthesis. Photosynthetica 1992, 27, 271–294. [Google Scholar]
- Raghothama, K.G. Phosphate acquisition. Annu. Rev. Plant Physiol 1999, 50, 665–693. [Google Scholar]
- Lynch, J. Root architecture and plant productivity. Plant Physiol 1995, 109, 7–13. [Google Scholar]
- Williamson, L.C.; Ribrioux, S.P.C.P.; Fitter, A.H.; Leyser, H.M.O. Phosphate availability regulates root system architecture in Arabidopsis. Plant Physiol 2001, 126, 875–882. [Google Scholar]
- Fohse, D.; Claassen, N.; Jungk, A. Phosphorus efficiency of plants. II. Significance of root radius, root hairs and cation-anion balance for phosphorus influx in 7 plant-species. Plant Soil 1991, 132, 261–272. [Google Scholar]
- Gahoonia, T.S.; Nielsen, N.E. Direct evidence on participation of root hairs in phosphorus (P-32) uptake from soil. Plant Soil 1998, 198, 147–152. [Google Scholar]
- Bates, T.R.; Lynch, J.P. Stimulation of root hair elongation in Arabidopsis thaliana by low phosphorus availability. Plant Cell Environ 1996, 19, 529–538. [Google Scholar]
- Brown, L.K.; George, T.S.; Dupuy, L.X.; White, P.J. A conceptual model of root hair ideotypes for future agricultural environments: What combination of traits should be targeted to cope with limited P availability? Ann. Bot. 2012. [Google Scholar] [CrossRef]
- Ma, Z.; Bielenberg, D.G.; Brown, K.M.; Lynch, J.P. Regulation of root hair density by phosphorus availability in Arabidopsis thaliana. Plant Cell Environ 2001, 24, 459–467. [Google Scholar]
- Lopez-Bucio, J.; Cruz-Ramirez, A.; Herrera-Estrella, L. The role of nutrient availability in regulating root architecture. Curr. Opin. Plant Biol 2003, 6, 280–287. [Google Scholar]
- Peret, B.; Clement, M.; Nussaume, L.; Desnos, T. Root developmental adaptation to phosphate starvation: Better safe than sorry. Trends Plant Sci 2011, 16, 442–450. [Google Scholar]
- Drew, M.C.; Saker, L.R. Nutrient supply and growth of seminal root-system in barley. III. Compensatory increases in growth of lateralroots, and in rates of phosphate uptake, in response to a localized supply of phosphate. J. Exp. Bot 1978, 29, 435–451. [Google Scholar]
- Jackson, R.B.; Manwaring, J.H.; Caldwell, M.M. Rapid physiological adjustment of roots to localized soil enrichment. Nature 1990, 344, 58–60. [Google Scholar]
- Smith, S.E.; Smith, F.A.; Jakobsen, I. Mycorrhizal fungi can dominate phosphate supply to plants irrespective of growth responses. Plant Physiol 2003, 133, 16–20. [Google Scholar]
- Bonfante, P.; Anca, I.A. Plants, mycorrhizal fungi, and bacteria: A network of interactions. Annu. Rev. Microbiol 2009, 63, 363–383. [Google Scholar]
- Purnell, H. Studies of the family proteaceae. I. Anatomy and morphology of the roots of some victorian species. Aust. J. Bot 1960, 8, 38–50. [Google Scholar]
- Gardner, W.K.; Barber, D.A.; Parbery, D.G. Effect of microorganisms on the formation and activity of proteoid roots of Lupinus albus L. Aust. J. Bot 1982, 30, 303–309. [Google Scholar]
- Johnson, J.F.; Allan, D.L.; Vance, C.P. Phosphorus stress-induced proteoid roots show altered metabolism in Lupinus albus. Plant Physiol 1994, 104, 657–665. [Google Scholar]
- Malajczu, N.; Bowen, G.D. Proteoid roots are microbially induced. Nature 1974, 251, 316–317. [Google Scholar]
- Watt, M.; Evans, J.R. Proteoid roots. Physiology and development. Plant Physiol 1999, 121, 317–323. [Google Scholar]
- Jones, D.L. Organic acids in the rhizosphere—A critical review. Plant Soil 1998, 205, 25–44. [Google Scholar]
- Li, M.G.; Osaki, M.; Rao, I.M.; Tadano, T. Secretion of phytase from the roots of several plant species under phosphorus-deficient conditions. Plant Soil 1997, 195, 161–169. [Google Scholar]
- Wang, X.R.; Wang, Y.X.; Tian, J.; Lim, B.L.; Yan, X.L.; Liao, H. Overexpressing AtPAP15 enhances phosphorus efficiency in soybean. Plant Physiol 2009, 151, 233–240. [Google Scholar]
- Neumann, G.; George, T.S.; Plassard, C. Strategies and methods for studying the rhizosphere-the plant science toolbox. Plant Soil 2009, 321, 431–456. [Google Scholar]
- Duff, S.M.G.; Sarath, G.; Plaxton, W.C. The role of acid-phosphatases in plant phosphorus-metabolism. Physiol. Plant 1994, 90, 791–800. [Google Scholar]
- Tadano, T.; Sakai, H. Secretion of acid-phosphatase by the roots of several crop species under phosphorus-deficient conditions. Soil Sci. Plant Nutr 1991, 37, 129–140. [Google Scholar]
- Starnes, D.L.; Padmanabhan, P.; Sahi, S.V. Effect of P sources on growth, P accumulation and activities of phytase and acid phosphatases in two cultivars of annual ryegrass (Lolium multiflorum L.). Plant Physiol. Biochem 2008, 46, 580–589. [Google Scholar]
- Plaxton, W.C.; Tran, H.T. Metabolic adaptations of phosphate-starved plants. Plant Physiol 2011, 156, 1006–1015. [Google Scholar]
- Ai, P.H.; Sun, S.B.; Zhao, J.N.; Fan, X.R.; Xin, W.J.; Guo, Q.; Yu, L.; Shen, Q.R.; Wu, P.; Miller, A.J.; et al. Two rice phosphate transporters, OsPht1;2 and OsPht1;6, have different functions and kinetic properties in uptake and translocation. Plant J 2009, 57, 798–809. [Google Scholar]
- Shin, H.; Shin, H.S.; Dewbre, G.R.; Harrison, M.J. Phosphate transport in Arabidopsis: Pht1;1 and Pht1; 4 play a major role in phosphate acquisition from both low- and high-phosphate environments. Plant J 2004, 39, 629–642. [Google Scholar]
- Daram, P.; Brunner, S.; Rausch, C.; Steiner, C.; Amrhein, N.; Bucher, M. Pht2;1 encodes a low-affinity phosphate transporter from Arabidopsis. Plant Cell 1999, 11, 2153–2166. [Google Scholar]
- Poirier, Y.; Bucher, M. Phosphate transport and homeostasis in Arabidopsis. The Arabidopsis B 2002, 1, e0024. [Google Scholar]
- Tu, S.I.; Cavanaugh, J.R.; Boswell, R.T. Phosphate-uptake by excised maize root-tips studied by in vivo P-31 nuclear-magnetic-resonance spectroscopy. Plant Physiol 1990, 93, 778–784. [Google Scholar]
- Pratt, J.; Boisson, A.M.; Gout, E.; Bligny, R.; Douce, R.; Aubert, S. Phosphate (Pi) starvation effect on the cytosolic Pi concentration and Pi exchanges across the tonoplast in plant cells: An in vivo P-31-nuclear magnetic resonance study using methylphosphonate as a Pi analog. Plant Physiol 2009, 151, 1646–1657. [Google Scholar]
- Duff, S.M.G.; Moorhead, G.B.G.; Lefebvre, D.D.; Plaxton, W.C. Phosphate starvation inducible bypasses of adenylate and phosphate dependent glycolytic-enzymes in Brassica nigra suspension cells. Plant Physiol 1989, 90, 1275–1278. [Google Scholar]
- Rychter, A.M.; Mikulska, M. The relationship between phosphate status and cyanide-resistant respiration in bean roots. Physiol. Plant 1990, 79, 663–667. [Google Scholar]
- Hartel, H.; Dormann, P.; Benning, C. DGD1-independent biosynthesis of extraplastidic galactolipids after phosphate deprivation in Arabidopsis. Proc. Natl. Acad. Sci. USA 2000, 97, 10649–10654. [Google Scholar]
- Stitt, M. Pyrophosphate as an energy donor in the cytosol of plant cells: An enigmatic alternative to ATP. Bot. Acta 1998, 111, 167–175. [Google Scholar]
- Weiner, H.; Stitt, M.; Heldt, H.W. Subcellular compartmentation of pyrophosphate and alkaline pyrophosphatase in leaves. Biochim. Biophys. Acta 1987, 893, 13–21. [Google Scholar]
- Chiou, T.J.; Lin, S.I. Signaling network in sensing phosphate availability in plants. Annu. Rev. Plant Biol 2011, 62, 185–206. [Google Scholar]
- Hu, B.; Chu, C. Phosphate starvation signaling in rice. Plant Signal. Behav 2011, 6, 927–929. [Google Scholar]
- Yang, X.J.; Finnegan, P.M. Regulation of phosphate starvation responses in higher plants. Ann. Bot 2010, 105, 513–526. [Google Scholar]
- Rouached, H.; Arpat, A.B.; Poirier, Y. Regulation of phosphate starvation responses in plants: Signaling players and cross-talks. Mol. Plant 2010, 3, 288–299. [Google Scholar]
- Yuan, H.; Liu, D. Signaling components involved in plant responses to phosphate starvation. J. Integr. Plant Biol 2008, 50, 849–859. [Google Scholar]
- Ticconi, C.A.; Delatorre, C.A.; Abel, S. Attenuation of phosphate starvation responses by phosphite in arabidopsis. Plant Physiol 2001, 127, 963–972. [Google Scholar]
- Varadarajan, D.K.; Karthikeyan, A.S.; Matilda, P.D.; Raghothama, K.G. Phosphite, an analog of phosphate, suppresses the coordinated expression of genes under phosphate starvation. Plant Physiol 2002, 129, 1232–1240. [Google Scholar]
- Linkohr, B.I.; Williamson, L.C.; Fitter, A.H.; Leyser, H.M.O. Nitrate and phosphate availability and distribution have different effects on root system architecture of Arabidopsis. Plant J 2002, 29, 751–760. [Google Scholar] [Green Version]
- Svistoonoff, S.; Creff, A.; Reymond, M.; Sigoillot-Claude, C.; Ricaud, L.; Blanchet, A.; Nussaume, L.; Desnos, T. Root tip contact with low-phosphate media reprograms plant root architecture. Nat. Genet 2007, 39, 792–796. [Google Scholar]
- Ticconi, C.A.; Delatorre, C.A.; Lahner, B.; Salt, D.E.; Abel, S. Arabidopsis pdr2 reveals a phosphate-sensitive checkpoint in root development. Plant J 2004, 37, 801–814. [Google Scholar]
- Ticconi, C.A.; Lucero, R.D.; Sakhonwasee, S.; Adamson, A.W.; Creff, A.; Nussaume, L.; Desnos, T.; Abel, S. ER-resident proteins PDR2 and LPR1 mediate the developmental response of root meristems to phosphate availability. Proc. Natl. Acad. Sci. USA 2009, 106, 14174–14179. [Google Scholar]
- Sanchez-Calderon, L.; Lopez-Bucio, J.; Chacon-Lopez, A.; Gutierrez-Ortega, A.; Hernandez-Abreu, E.; Herrera-Estrella, L. Characterization of low phosphorus insensitive mutants reveals a crosstalk between low phosphorus-induced determinate root development and the activation of genes involved in the adaptation of Arabidopsis to phosphorus deficiency. Plant Physiol 2006, 140, 879–889. [Google Scholar]
- Mayzlish-Gati, E.; De-Cuyper, C.; Goormachtig, S.; Beeckman, T.; Vuylsteke, M.; Brewer, P.B.; Beveridge, C.A.; Yermiyahu, U.; Kaplan, Y.; Enzer, Y.; et al. Strigolactones are involved in root response to low phosphate conditions in Arabidopsis. Plant Physiol 2012, 160, 1329–1341. [Google Scholar]
- Perez-Torres, C.A.; Lopez-Bucio, J.; Cruz-Ramirez, A.; Ibarra-Laclette, E.; Dharmasiri, S.; Estelle, M.; Herrera-Estrella, L. Phosphate availability alters lateral root development in Arabidopsis by modulating auxin sensitivity via a mechanism involving the TIR1 auxin receptor. Plant Cell 2008, 20, 3258–3272. [Google Scholar]
- Gilbert, G.A.; Knight, J.D.; Vance, C.P.; Allan, D.L. Proteoid root development of phosphorus deficient lupin is mimicked by auxin and phosphonate. Ann. Bot 2000, 85, 921–928. [Google Scholar]
- Franco-Zorrilla, J.M.; Martin, A.C.; Leyva, A.; Par-Ares, J.P. Interaction between phosphate-starvation, sugar, and cytokinin signaling in Arabidopsis and the roles of cytokinin receptors CRE1/AHK4 and AHK3. Plant Physiol 2005, 138, 847–857. [Google Scholar]
- Jiang, C.F.; Gao, X.H.; Liao, L.; Harberd, N.P.; Fu, X.D. Phosphate starvation root architecture and anthocyanin accumulation responses are modulated by the gibberellin-DELLA signaling pathway in Arabidopsis. Plant Physiol 2007, 145, 1460–1470. [Google Scholar]
- Gibson, S.I. Sugar and phytohormone response pathways: navigating a signalling network. J. Exp. Bot 2004, 55, 253–264. [Google Scholar]
- Kircher, S.; Schopfer, P. Photosynthetic sucrose acts as cotyledon-derived long-distance signal to control root growth during early seedling development in Arabidopsis. Proc. Natl. Acad. Sci. USA 2012, 109, 11217–11221. [Google Scholar]
- Aung, K.; Lin, S.I.; Wu, C.C.; Huang, Y.T.; Su, C.L.; Chiou, T.J. pho2, a phosphate overaccumulator, is caused by a nonsense mutation in a microRNA399 target gene. Plant Physiol 2006, 141, 1000–1011. [Google Scholar]
- Chiou, T.J.; Aung, K.; Lin, S.I.; Wu, C.C.; Chiang, S.F.; Su, C.L. Regulation of phosphate homeostasis by microRNA in Arabidopsis. Plant Cell 2006, 18, 412–421. [Google Scholar]
- Bari, R.; Datt Pant, B.; Stitt, M.; Scheible, W.R. PHO2, microRNA399, and PHR1 define a phosphate-signaling pathway in plants. Plant Physiol 2006, 141, 988–999. [Google Scholar]
- Pant, B.D.; Buhtz, A.; Kehr, J.; Scheible, W.R. MicroRNA399 is a long-distance signal for the regulation of plant phosphate homeostasis. Plant J 2008, 53, 731–738. [Google Scholar]
- Lin, S.I.; Chiang, S.F.; Lin, W.Y.; Chen, J.W.; Tseng, C.Y.; Wu, P.C.; Chiou, T.J. Regulatory network of microRNA399 and PHO2 by systemic signaling. Plant Physiol 2008, 147, 732–746. [Google Scholar]
- Matusova, R.; Rani, K.; Verstappen, F.W.A.; Franssen, M.C.R.; Beale, M.H.; Bouwmeester, H.J. The strigolactone germination stimulants of the plant-parasitic Striga and Orobanche spp. are derived from the carotenoid pathway. Plant Physiol 2005, 139, 920–934. [Google Scholar]
- Alder, A.; Jamil, M.; Marzorati, M.; Bruno, M.; Vermathen, M.; Bigler, P.; Ghisla, S.; Bouwmeester, H.; Beyer, P.; Al-Babili, S. The path from beta-carotene to carlactone, a strigolactone-like plant hormone. Science 2012, 335, 1348–1351. [Google Scholar]
- Cook, C.E.; Whichard, L.P.; Turner, B.; Wall, M.E. Germination of witchweed (Striga lutea Lour.)—Isolation and properties of a potent stimulant. Science 1966, 154, 1189–1190. [Google Scholar]
- Besserer, A.; Puech-Pages, V.; Kiefer, P.; Gomez-Roldan, V.; Jauneau, A.; Roy, S.; Portais, J.C.; Roux, C.; Becard, G.; Sejalon-Delmas, N. Strigolactones stimulate arbuscular mycorrhizal fungi by activating mitochondria. PLoS Biol 2006, 4, 1239–1247. [Google Scholar]
- Kohlen, W.; Charnikhova, T.; Liu, Q.; Bours, R.; Domagalska, M.A.; Beguerie, S.; Verstappen, F.; Leyser, O.; Bouwmeester, H.; Ruyter-Spira, C. Strigolactones are transported through the xylem and play a key role in shoot architectural response to phosphate deficiency in nonarbuscular mycorrhizal host Arabidopsis. Plant Physiol 2011, 155, 974–987. [Google Scholar]
- Kretzschmar, T.; Kohlen, W.; Sasse, J.; Borghi, L.; Schlegel, M.; Bachelier, J.B.; Reinhardt, D.; Bours, R.; Bouwmeester, H.J.; Martinoia, E. A petunia ABC protein controls strigolactone-dependent symbiotic signalling and branching. Nature 2012, 483, 341–344. [Google Scholar] [Green Version]
- Beveridge, C.A.; Kyozuka, J. New genes in the strigolactone-related shoot branching pathway. Curr. Opin. Plant Biol 2010, 13, 34–39. [Google Scholar]
- Domagalska, M.A.; Leyser, O. Signal integration in the control of shoot branching. Nat. Rev. Mol. Cell Biol 2011, 12, 211–221. [Google Scholar]
- Tsuchiya, Y.; McCourt, P. Strigolactones as small molecule communicators. Mol. Biosystems 2012, 8, 464–469. [Google Scholar]
- Xie, X.N.; Yoneyama, K.; Yoneyama, K. The strigolactone story. Annu. Rev. Phytopathol 2010, 48, 93–117. [Google Scholar]
- Koltai, H. Strigolactones are regulators of root development. New Phytol 2011, 190, 545–549. [Google Scholar]
- Wang, Y.H.; Li, J.Y. Branching in rice. Curr. Opin. Plant Biol 2011, 14, 94–99. [Google Scholar]
- Seto, Y.; Kameoka, H.; Yamaguchi, S.; Kyozuka, J. Recent advances in strigolactone research: Chemical and biological aspects. Plant Cell Physiol 2012, 53, 1843–1853. [Google Scholar]
- Ruyter-Spira, C.; Al-Babili, S.; van der Krol, S.; Bouwmeester, H. The biology of strigolactones. Trends Plant Sci 2012, 18, 72–83. [Google Scholar]
- Brewer, P.B.; Koltai, H.; Beveridge, C.A. Diverse roles of strigolactones in plant development. Mol. Plant 2013, 6, 18–28. [Google Scholar]
- Booker, J.; Auldridge, M.; Wills, S.; McCarty, D.; Klee, H.; Leyser, O. MAX3/CCD7 is a carotenoid cleavage dioxygenase required for the synthesis of a novel plant signaling molecule. Curr. Biol 2004, 14, 1232–1238. [Google Scholar]
- Sorefan, K.; Booker, J.; Haurogne, K.; Goussot, M.; Bainbridge, K.; Foo, E.; Chatfield, S.; Ward, S.; Beveridge, C.; Rameau, C.; et al. MAX4 and RMS1 are orthologous dioxygenase-like genes that regulate shoot branching in Arabidopsis and pea. Genes Dev 2003, 17, 1469–1474. [Google Scholar]
- Booker, J.; Sieberer, T.; Wright, W.; Williamson, L.; Willett, B.; Stirnberg, P.; Turnbull, C.; Srinivasan, M.; Goddard, P.; Leyser, O. MAX1 encodes a cytochrome P450 family member that acts downstream of MAX3/4 to produce a carotenoid-derived branch-inhibiting hormone. Dev. Cell 2005, 8, 443–449. [Google Scholar]
- Snowden, K.C.; Simkin, A.J.; Janssen, B.J.; Templeton, K.R.; Loucas, H.M.; Simons, J.L.; Karunairetnam, S.; Gleave, A.P.; Clark, D.G.; Klee, H.J. The Decreased apical dominance 1/petunia hybrida carotenoid cleavage dioxygenase 8 gene affects branch production and plays a role in leaf senescence, root growth, and flower development. Plant Cell 2005, 17, 746–759. [Google Scholar]
- Lin, H.; Wang, R.X.; Qian, Q.; Yan, M.X.; Meng, X.B.; Fu, Z.M.; Yan, C.Y.; Jiang, B.; Su, Z.; Li, J.Y.; et al. DWARF27, an iron-containing protein required for the biosynthesis of strigolactones, regulates rice tiller bud outgrowth. Plant Cell 2009, 21, 1512–1525. [Google Scholar]
- Waters, M.T.; Brewer, P.B.; Bussell, J.D.; Smith, S.M.; Beveridge, C.A. The Arabidopsis ortholog of rice DWARF27 acts upstream of MAX1 in the control of plant development by strigolactones. Plant Physiol 2012, 159, 1073–1085. [Google Scholar]
- Liu, W.; Kohlen, W.; Lillo, A.; Op den Camp, R.; Ivanov, S.; Hartog, M.; Limpens, E.; Jamil, M.; Smaczniak, C.; Kaufmann, K.; et al. Strigolactone biosynthesis in Medicago truncatula and rice requires the symbiotic GRAS-type transcription factors NSP1 and NSP2. Plant Cell 2011, 23, 3853–3865. [Google Scholar]
- Stirnberg, P.; van de Sande, K.; Leyser, H.M.O. MAX1 and MAX2 control shoot lateral branching in Arabidopsis. Development 2002, 129, 1131–1141. [Google Scholar]
- Arite, T.; Umehara, M.; Ishikawa, S.; Hanada, A.; Maekawa, M.; Yamaguchi, S.; Kyozuka, J. d14, a strigolactone-insensitive mutant of rice, shows an accelerated outgrowth of tillers. Plant Cell Physiol 2009, 50, 1416–1424. [Google Scholar]
- Waters, M.T.; Nelson, D.C.; Scaffidi, A.; Flematti, G.R.; Sun, Y.K.M.; Dixon, K.W.; Smith, S.M. Specialisation within the DWARF14 protein family confers distinct responses to karrikins and strigolactones in Arabidopsis. Development 2012, 139, 1285–1295. [Google Scholar]
- Hamiaux, C.; Drummond, R.S.M.; Janssen, B.J.; Ledger, S.E.; Cooney, J.M.; Newcomb, R.D.; Snowden, K.C. DAD2 is an alpha/beta hydrolase likely to be involved in the perception of the plant branching hormone, strigolactone. Curr. Biol 2012, 22, 2032–2036. [Google Scholar]
- Santner, A.; Estelle, M. Recent advances and emerging trends in plant hormone signalling. Nature 2009, 459, 1071–1078. [Google Scholar]
- Ueguchi-Tanaka, M.; Ashikari, M.; Nakajima, M.; Itoh, H.; Katoh, E.; Kobayashi, M.; Chow, T.Y.; Hsing, Y.I.C.; Kitano, H.; Yamaguchi, I.; et al. GIBBERELLIN INSENSITIVE DWARF1 encodes a soluble receptor for gibberellin. Nature 2005, 437, 693–698. [Google Scholar]
- Gaiji, N.; Cardinale, F.; Prandi, C.; Bonfante, P.; Ranghino, G. The computational-based structure of Dwarf14 provides evidence for its role as potential strigolactone receptor in plants. BMC Res. Notes 2012, 5, 307. [Google Scholar]
- Zhao, L.H.; Zhou, X.E.; Wu, Z.S.; Yi, W.; Xu, Y.; Li, S.; Xu, T.H.; Liu, Y.; Chen, R.Z.; Kovach, A.; et al. Crystal structures of two phytohormone signal-transducing alpha/beta hydrolases: Karrikin-signaling KAI2 and strigolactone-signaling DWARF14. Cell Res 2013, 23, 436–439. [Google Scholar]
- Kagiyama, M.; Hirano, Y.; Mori, T.; Kim, S.Y.; Kyozuka, J.; Seto, Y.; Yamaguchi, S.; Hakoshima, T. Structures of D14 and D14L in the strigolactone and karrikin signaling pathways. Genes Cells 2013, 18, 147–160. [Google Scholar]
- Minakuchi, K.; Kameoka, H.; Yasuno, N.; Umehara, M.; Luo, L.; Kobayashi, K.; Hanada, A.; Ueno, K.; Asami, T.; Yamaguchi, S.; et al. FINE CULM1 (FC1) works downstream of strigolactones to inhibit the outgrowth of axillary buds in rice. Plant Cell Physiol 2010, 51, 1127–1135. [Google Scholar]
- Takeda, T.; Suwa, Y.; Suzuki, M.; Kitano, H.; Ueguchi-Tanaka, M.; Ashikari, M.; Matsuoka, M.; Ueguchi, C. The OsTB1 gene negatively regulates lateral branching in rice. Plant J 2003, 33, 513–520. [Google Scholar]
- Doebley, J.; Stec, A.; Hubbard, L. The evolution of apical dominance in maize. Nature 1997, 386, 485–488. [Google Scholar]
- Aguilar-Martinez, J.A.; Poza-Carrion, C.; Cubas, P. Arabidopsis BRANCHED1 acts as an integrator of branching signals within axillary buds. Plant Cell 2007, 19, 458–472. [Google Scholar]
- Braun, N.; de Saint Germain, A.; Pillot, J.P.; Boutet-Mercey, S.; Dalmais, M.; Antoniadi, I.; Li, X.; Maia-Grondard, A.; Le Signor, C.; Bouteiller, N.; et al. The pea TCP transcription factor PsBRC1 acts downstream of strigolactones to control shoot branching. Plant Physiol 2012, 158, 225–238. [Google Scholar]
- Breuillin, F.; Schramm, J.; Hajirezaei, M.; Ahkami, A.; Favre, P.; Druege, U.; Hause, B.; Bucher, M.; Kretzschmar, T.; Bossolini, E.; et al. Phosphate systemically inhibits development of arbuscular mycorrhiza in Petunia hybrida and represses genes involved in mycorrhizal functioning. Plant J 2010, 64, 1002–1017. [Google Scholar] [Green Version]
- Balzergue, C.; Puech-Pages, V.; Becard, G.; Rochange, S.F. The regulation of arbuscular mycorrhizal symbiosis by phosphate in pea involves early and systemic signalling events. J. Exp. Bot 2011, 62, 1049–1060. [Google Scholar]
- Foo, E.; Yoneyama, K.; Hugill, C.J.; Quittenden, L.J.; Reid, J.B. Strigolactones and the regulation of pea symbioses in response to nitrate and phosphate deficiency. Mol. Plant 2013, 6, 76–87. [Google Scholar]
- Ruyter-Spira, C.; Kohlen, W.; Charnikhova, T.; van Zeijl, A.; van Bezouwen, L.; de Ruijter, N.; Cardoso, C.; Lopez-Raez, J.A.; Matusova, R.; Bours, R.; et al. Physiological effects of the synthetic strigolactone analog GR24 on root system architecture in Arabidopsis: another belowground role for strigolactones? Plant Physiol 2011, 155, 721–734. [Google Scholar]
- Kapulnik, Y.; Delaux, P.M.; Resnick, N.; Mayzlish-Gati, E.; Wininger, S.; Bhattacharya, C.; Sejalon-Delmas, N.; Combier, J.P.; Becard, G.; Belausov, E.; et al. Strigolactones affect lateral root formation and root-hair elongation in Arabidopsis. Planta 2011, 233, 209–216. [Google Scholar]
- Rasmussen, A.; Mason, M.G.; De Cuyper, C.; Brewer, P.B.; Herold, S.; Agusti, J.; Geelen, D.; Greb, T.; Goormachtig, S.; Beeckman, T.; et al. Strigolactones suppress adventitious rooting in Arabidopsis and Pea. Plant Physiol 2012, 158, 1976–1987. [Google Scholar]
- Lopez-Raez, J.A.; Bouwmeester, H. Fine-tuning regulation of strigolactone biosynthesis under phosphate starvation. Plant Signal. Behav 2008, 3, 963–965. [Google Scholar]
- Yoneyama, K.; Xie, X.N.; Sekimoto, H.; Takeuchi, Y.; Ogasawara, S.; Akiyama, K.; Hayashi, H.; Yoneyama, K. Strigolactones, host recognition signals for root parasitic plants and arbuscular mycorrhizal fungi, from Fabaceae plants. New Phytol 2008, 179, 484–494. [Google Scholar]
- Lopez-Raez, J.A.; Charnikhova, T.; Gomez-Roldan, V.; Matusova, R.; Kohlen, W.; De Vos, R.; Verstappen, F.; Puech-Pages, V.; Becard, G.; Mulder, P.; et al. Tomato strigolactones are derived from carotenoids and their biosynthesis is promoted by phosphate starvation. New Phytol 2008, 178, 863–874. [Google Scholar]
- Jamil, M.; Charnikhova, T.; Cardoso, C.; Jamil, T.; Ueno, K.; Verstappen, F.; Asami, T.; Bouwmeester, H.J. Quantification of the relationship between strigolactones and Striga hermonthica infection in rice under varying levels of nitrogen and phosphorus. Weed Res 2011, 51, 373–385. [Google Scholar]
- Yoneyama, K.; Xie, X.N.; Kim, H.I.; Kisugi, T.; Nomura, T.; Sekimoto, H.; Yokota, T.; Yoneyama, K. How do nitrogen and phosphorus deficiencies affect strigolactone production and exudation? Planta 2012, 235, 1197–1207. [Google Scholar]
- Yoneyama, K.; Xie, X.N.; Kisugi, T.; Nomura, T.; Sekimoto, H.; Yokota, T.; Yoneyama, K. Characterization of strigolactones exuded by Asteraceae plantss. Plant Growth Regul 2011, 65, 495–504. [Google Scholar]
- Smith, S.E.; Read, D.J. Mycorrhizal Symbiosis, 3rd ed; Academic Press: San Diego, CA, USA, 2008. [Google Scholar]
- Harrison, M.J. Molecular and cellular aspects of the arbuscular mycorrhizal symbiosis. Annu. Rev. Plant Physiol 1999, 50, 361–389. [Google Scholar]
- Nagahashi, G.; Douds, D.D. Partial separation of root exudate components and their effects upon the growth of germinated spores of AM fungi. Mycol. Res 2000, 104, 1453–1464. [Google Scholar]
- Besserer, A.; Becard, G.; Jauneau, A.; Roux, C.; Sejalon-Delmas, N. GR24, a synthetic analog of strigolactones, stimulates the mitosis and growth of the arbuscular mycorrhizal fungus Gigaspora rosea by boosting its energy metabolism. Plant Physiol 2008, 148, 402–413. [Google Scholar]
- Yoshida, S.; Kameoka, H.; Tempo, M.; Akiyama, K.; Umehara, M.; Yamaguchi, S.; Hayashi, H.; Kyozuka, J.; Shirasu, K. The D3 F-box protein is a key component in host strigolactone responses essential for arbuscular mycorrhizal symbiosis. New Phytol 2012, 196, 1208–1216. [Google Scholar]
- Kang, J.; Hwang, J.U.; Lee, M.; Kim, Y.Y.; Assmann, S.M.; Martinoia, E.; Lee, Y. PDR-type ABC transporter mediates cellular uptake of the phytohormone abscisic acid. Proc. Natl. Acad. Sci. USA 2010, 107, 2355–2360. [Google Scholar] [Green Version]
- Woo, H.R.; Chung, K.M.; Park, J.H.; Oh, S.A.; Ahn, T.; Hong, S.H.; Jang, S.K.; Nam, H.G. ORE9, an F-box protein that regulates leaf senescence in Arabidopsis. Plant Cell 2001, 13, 1779–1790. [Google Scholar]
- Yan, H.; Saika, H.; Maekawa, M.; Takamure, I.; Tsutsumi, N.; Kyozuka, J.; Nakazono, M. Rice tillering dwarf mutant dwarf3 has increased leaf longevity during darkness-induced senescence or hydrogen peroxide-induced cell death. Genes Genet. Syst 2007, 82, 361–366. [Google Scholar]
- Crafts-Brandner, S.J. Phosphorus nutrition influence on leaf senescence in soybean. Plant Physiol 1992, 98, 1128–1132. [Google Scholar]

© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Czarnecki, O.; Yang, J.; Weston, D.J.; Tuskan, G.A.; Chen, J.-G. A Dual Role of Strigolactones in Phosphate Acquisition and Utilization in Plants. Int. J. Mol. Sci. 2013, 14, 7681-7701. https://doi.org/10.3390/ijms14047681
Czarnecki O, Yang J, Weston DJ, Tuskan GA, Chen J-G. A Dual Role of Strigolactones in Phosphate Acquisition and Utilization in Plants. International Journal of Molecular Sciences. 2013; 14(4):7681-7701. https://doi.org/10.3390/ijms14047681
Chicago/Turabian StyleCzarnecki, Olaf, Jun Yang, David J. Weston, Gerald A. Tuskan, and Jin-Gui Chen. 2013. "A Dual Role of Strigolactones in Phosphate Acquisition and Utilization in Plants" International Journal of Molecular Sciences 14, no. 4: 7681-7701. https://doi.org/10.3390/ijms14047681



