Fabrication of Magnetic-Antimicrobial-Fluorescent Multifunctional Hybrid Microspheres and Their Properties
Abstract
:1. Introduction
2. Results and Discussion
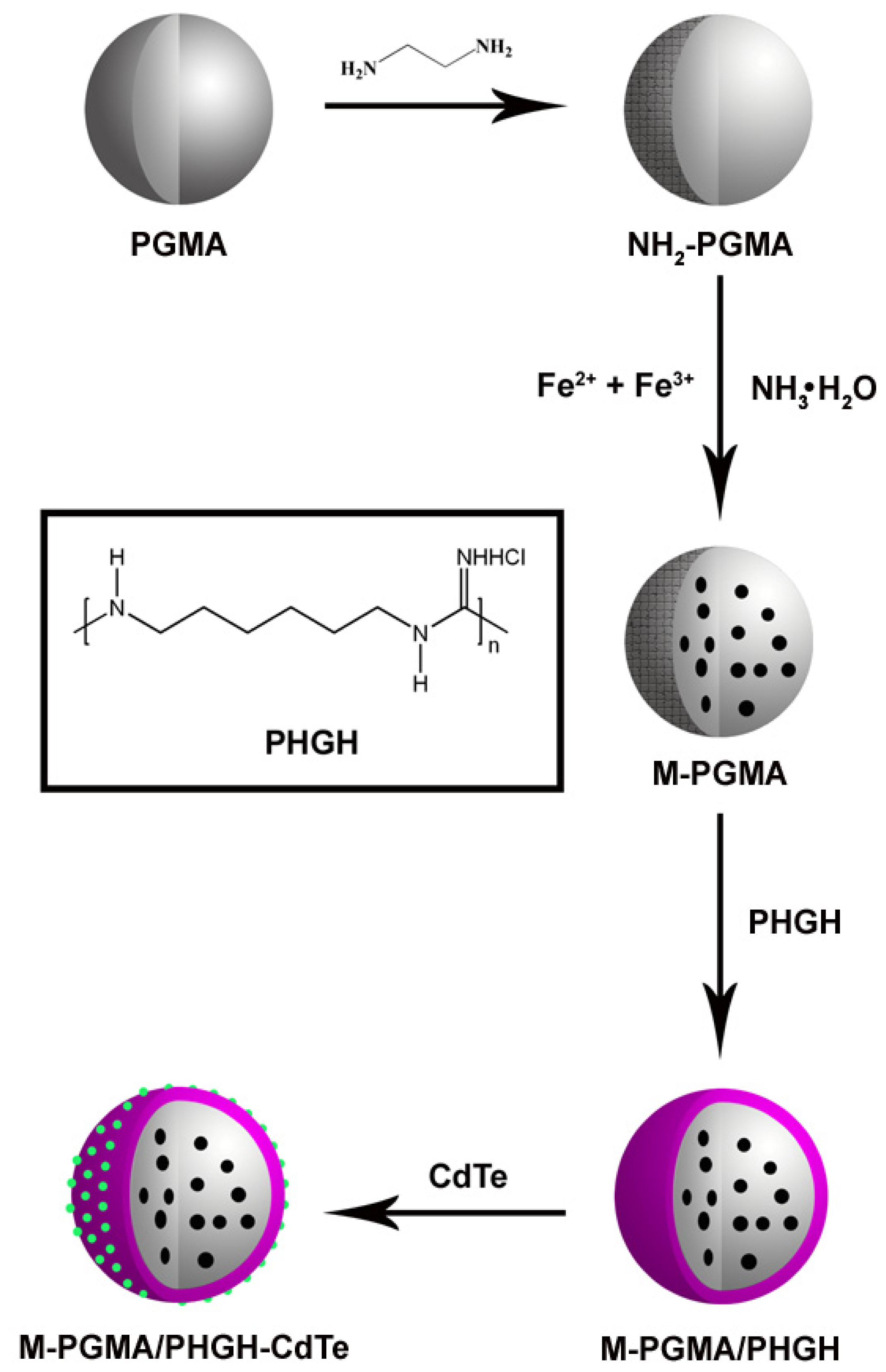
2.1. Description
2.2. SEM Test of the PGMA, NH2-PGMA and M-PGMA Microspheres
2.3. Characterization of the M-PGMA/PHGH-CdTe Microspheres
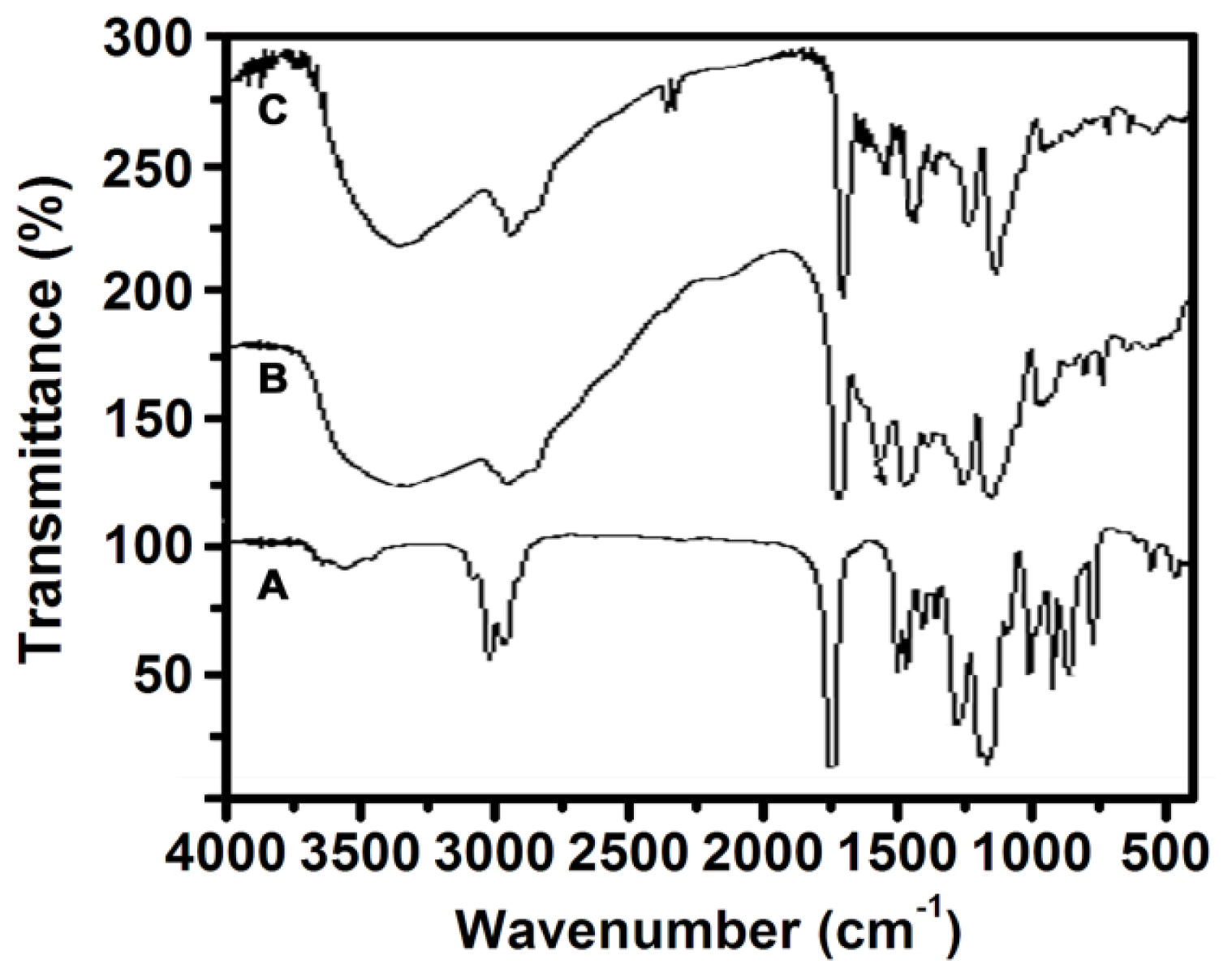
2.4. FT-IR Analysis of PGMA, NH2-PGMA and M-PGMA Microspheres
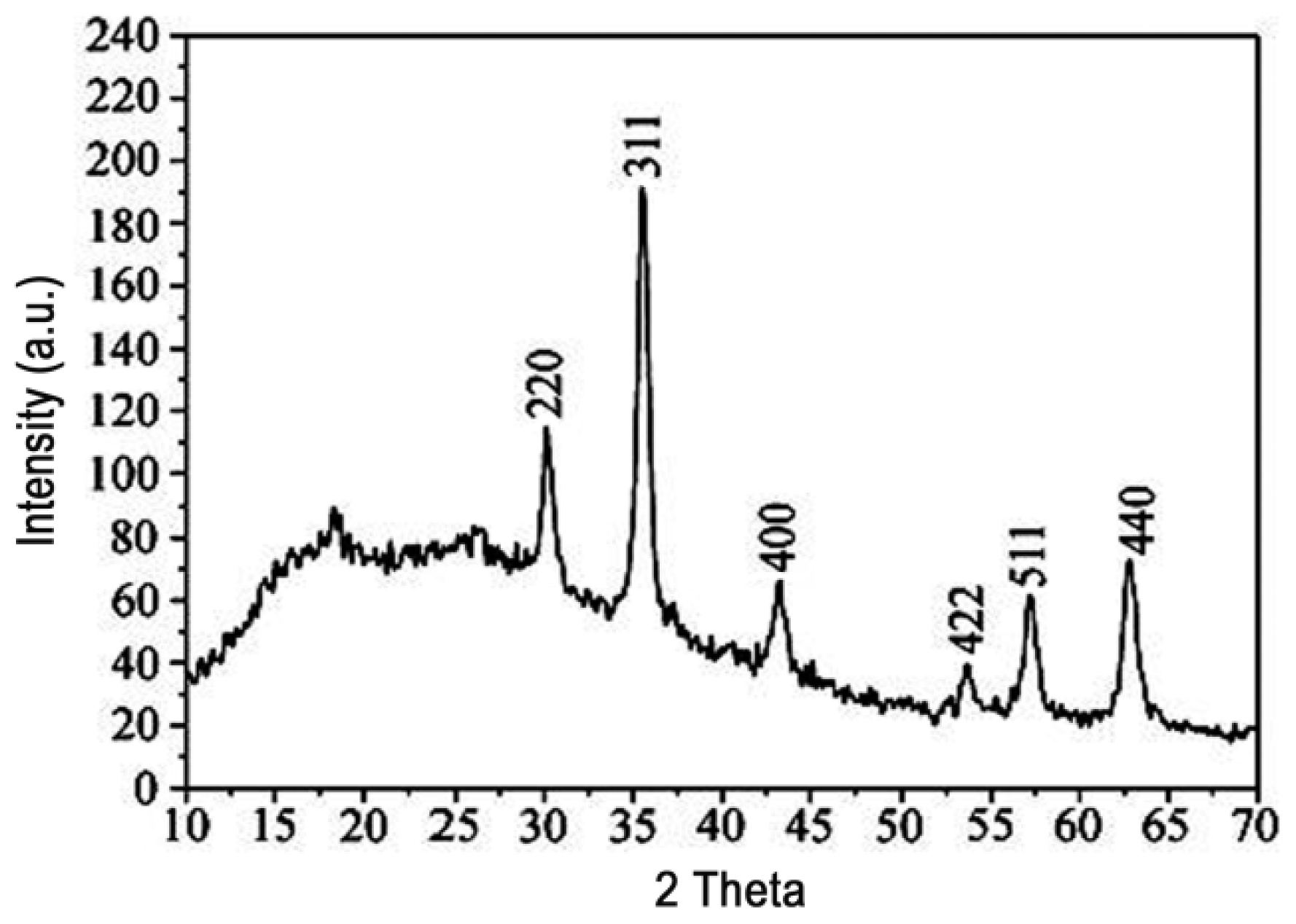
2.5. XRD Analysis of M-PGMA/PHGH Microspheres
2.6. Magnetism Assessment of M-PGMA/PHGH-CdTe Microspheres
2.7. The Fluorescence Assessment of M-PGMA/PHGH-CdTe Microspheres
2.8. Antimicrobial Functions
3. Experimental Section
3.1. Materials
3.2. Characterization
3.3. Preparation of PGMA Microspheres
3.4. Preparation of NH2-PGMA Microspheres
3.5. Preparation of M-PGMA Microspheres
3.6. Preparation of M-PGMA/PHGH Microspheres
3.7. Electrostatic Adsorption of Dot CdTe
3.8. Antibacterial Assessment
4. Conclusions
Acknowledgments
Conflict of interest
References
- Aleshina, E.Y; Yudanova, T.N; Skokova, I.F. Production and properties of polyvinyl alcohol spinning solutions containing protease C and polyhexamethylene guanidine. Fiber Chem. 2001, 33, 421–423. [Google Scholar]
- Wei, D.F; Ma, Q.X; Guan, Y; Hu, F.Z; Zheng, A; Zhang, X; Teng, Z; Jiang, H. Structural characterization and antibacterial activity of oligoguanidine (polyhexamethylene guanidine hydrochloride). Mater. Sci. Eng. C 2009, 29, 1776–1780. [Google Scholar]
- Hu, Y; Du, Y.M; Yang, J.H; Kennedy, J.F; Wang, X.H; Wang, L.S. Synthesis, characterization, and antibacterial activity of guanidinylated chitosan. Carbohydr. Polym. 2007, 67, 66–72. [Google Scholar]
- Broxton, P; Woodcock, P.M; Heatley, F; Gilbert, P. Interaction of some polyhexamethylene biguanides and membrane phospholipids in Escherichia coli. J. Appl. Bacteriol. 1984, 57, 115–124. [Google Scholar]
- Broxton, P; Woodcock, P.M; Gilbert, P. Binding of some polyhexamethylene biguanides to the cell envelope of Escherichia coli. Microbiology 1984, 41, 15–22. [Google Scholar]
- Zhang, Y.M; Jiang, J.M; Chen, Y.M. Synthesis and antimicrobial activity of polymeric guanidine and biguanidine salts. Polymer 1999, 40, 6189–6198. [Google Scholar]
- Guan, Y; Xian, H.N; Sullivan, H; Zheng, A. Antimicrobial-modified sulfite pulps prepared by in situ copolymerization. Carbohydr. Polym. 2007, 69, 688–696. [Google Scholar]
- Kenawy, E; Worley, S.D; Broughton, R. The chemistry and applications of antimicrobial polymers: A state-of-the-art review. Biomacromolecules 2007, 8, 1359–1384. [Google Scholar]
- Zou, H; Wu, S; Shen, J. Polymer/silica nanocomposites: Preparation, characterization, properties, and applications. Chem. Rev. 2008, 108, 3893–3957. [Google Scholar]
- Qian, L.Y; Guan, Y; He, B.H; Xiao, H.N. Modified guanidine polymers: Synthesis and antimicrobial mechanism revealed by AFM. Polymer 2008, 49, 2471–2475. [Google Scholar]
- Wang, Y; Maksimuk, S; Shen, R; Yang, H. Synthesis of γ-iron oxide nanoparticles using a freshly-made and recycled ionic liquid. Green Chem. 2007, 9, 1051–1056. [Google Scholar]
- Shukoor, M.I; Natalio, F; Tahir, M.N; Ksenofontov, V; Therese, H.A; Theato, P; Schröder, H.C; Müller, W.E.G; Tremel, W. Superparamagnetic γ-Fe2O3nanoparticles with tailored functionality for protein separation. Chem. Commun 2007, 4677–4679. [Google Scholar]
- Yu, C.H; Lo, C.C.H; Tam, K; Tsang, S.C. Monodisperse binary nanocomposite in silica with enhanced magnetization for magnetic separation. J. Phys. Chem. C 2007, 111, 7879–7882. [Google Scholar]
- Zhou, W; Yao, N; Yao, G; Deng, C; Zhang, X; Yang, P. Facile synthesis of aminophenylboronic acid-functionalized magnetic nanoparticles for selective separation of glycopeptides and glycoproteins. Chem. Commun. 2008, 5577–5579. [Google Scholar]
- Wang, C.X; Yin, L.W; Zhang, L.Y; Kang, L; Wang, X.F; Gao, R. Magnetic (γ-Fe2O3@SiO2)n@TiO2 functional hybrid nanoparticles with actived photocatalytic ability. J. Phys. Chem. C 2009, 113, 4008–4011. [Google Scholar]
- Kattnig, D.R; Rosspeintner, A; Grampp, G. Fully reversible interconversion between locally excited fluorophore, exciplex, and radical ion pair demonstrated by a new magnetic field effect. Angew. Chem. Int. Ed. 2008, 47, 960–962. [Google Scholar]
- Qian, H.S; Hu, Y; Li, Z.Q; Yang, X.Y; Li, L.C; Zhang, X.T; Xu, R. ZnO/ZnFe2O4 magnetic fluorescent bifunctional hollow nanospheres: Synthesis, characterization, and their optical/magnetic properties. J. Phys. Chem. C 2010, 114, 17455–17459. [Google Scholar]
- Wang, P; Shi, Q; Shi, Y; Clark, K.K; Stucky, G.D; Keller, A.A. Magnetic permanently confined micelle arrays for treating hydrophobic organic compound contamination. J. Am. Chem. Soc. 2009, 131, 182–188. [Google Scholar]
- Uematsu, T; Kitajima, H; Kohma, T; Torimoto, T; Tachibana, Y; Kuwabata, S. Tuning of the fluorescence wavelength of CdTe quantum dots with 2 nm resolution by size-selective photoetching. Nanotechnology 2009, 20, 215302, :1–215302:9.. [Google Scholar]
- Horák, D. Magnetic polyglycidylmethacrylate microspheres by dispersion polymerization. J. Polym. Sci. A 2001, 39, 3707–3715. [Google Scholar]
- Wang, W.C; Zhang, Q; Zhang, B.B; Li, D; Dong, X.Q; Zhang, L; Chang, J. Preparation of monodisperse, superparamagnetic, luminescent, and multifunctional PGMA microspheres with amino-groups. Chin. Sci. Bull. 2008, 53, 1165–1170. [Google Scholar]
- Dong, X; Zheng, Y; Huang, Y; Chen, X; Jing, X. Synthesis and characterization of multifunctional poly(glycidyl methacrylate) microspheres and their use in cell separation. Anal. Biochem. 2010, 405, 207–212. [Google Scholar]
- Sun, P; Zhang, H.Y; Liu, C; Fang, J; Wang, M; Chen, J; Zhang, J.P; Mao, C; Xu, S.K. Preparation and characterization of Fe3O4/CdTe magnetic/fluorescent nanocomposites and their applications in immuno-labeling and fluorescent imaging of cancer cells. Langmuir 2010, 26, 1278–1284. [Google Scholar]
- Chen, Z.B; Sun, Y.L. N-halamine-based antimicrobial additives for polymers: Preparation, characterization, and antimicrobial activity. Ind. Eng. Chem. Res. 2006, 45, 2634–2640. [Google Scholar]
- Lai, C; Wang, Y; Uttam, B.P; Chen, Y; Hsiao, J; Liu, C; Liu, H; Chen, C; Chou, P. One-pot solvothermal synthesis of FePt/Fe3O4core–shell nanoparticles. Chem. Commun. 2008, 5342–5344. [Google Scholar]
- Liu, J; Zhang, L; Shi, S; Chen, S; Zhou, N; Zhang, Z; Cheng, Z; Zhu, X. A novel and universal route to SiO2-supported organic/inorganic hybrid noble metal nanomaterials via surface RAFT polymerization. Langmuir 2010, 26, 14806–14813. [Google Scholar]
- Guo, J; Yang, W.L; Wang, C.C; He, J. Poly(N-isopropylacrylamide)-coated luminescent/magnetic silica microspheres: Preparation, characterization, and biomedical applications. Chem. Mater. 2006, 18, 5554–5562. [Google Scholar]
- Krishanu, R; Ramachandram, B; Joseph, R.L. Metal-enhanced fluorescence from CdTe nanocrystals: A single-molecule fluorescence study. J. Am. Chem. Soc. 2006, 128, 8998–8999. [Google Scholar]
- Cai, Z.S; Song, Z.Q; Yang, C.S; Shang, S.B; Yin, Y.B. Synthesis of 2-hydroxypropyl dimethylbenzylammonium N,O-(2-carboxyethyl) chitosan chloride and its antibacterial activity. J. Appl. Polym. Sci. 2009, 111, 3010–3015. [Google Scholar]
- Qian, L.Y; Guan, Y; He, B.H; Xiao, H.N. Synergy of wet strength and antimicrobial activity of cellulose paper induced by a novel polymer complex. Mater. Lett. 2008, 62, 3610–3612. [Google Scholar]
- Bao, H; Wang, E; Dong, S. One-pot synthesis of CdTe nanocrystals and shape control of luminescent CdTe-cystine nanocomposites. Small 2006, 2, 476–479. [Google Scholar]






| Antibacterial Result | Gram-negative | Gram-positive | ||
|---|---|---|---|---|
| E. coli ATCC 25922 | P. aeruginosa ATCC 27853 | S. aureus ATCC 25923 | B. subtilis ATCC 6633 | |
| MIC (μg/mL) | 64 | 500 | 32 | 16 |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Xiao, L.-H.; Wang, T.; Zhao, T.-Y.; Zheng, X.; Sun, L.-Y.; Li, P.; Liu, F.-Q.; Gao, G.; Dong, A. Fabrication of Magnetic-Antimicrobial-Fluorescent Multifunctional Hybrid Microspheres and Their Properties. Int. J. Mol. Sci. 2013, 14, 7391-7404. https://doi.org/10.3390/ijms14047391
Xiao L-H, Wang T, Zhao T-Y, Zheng X, Sun L-Y, Li P, Liu F-Q, Gao G, Dong A. Fabrication of Magnetic-Antimicrobial-Fluorescent Multifunctional Hybrid Microspheres and Their Properties. International Journal of Molecular Sciences. 2013; 14(4):7391-7404. https://doi.org/10.3390/ijms14047391
Chicago/Turabian StyleXiao, Ling-Han, Tao Wang, Tian-Yi Zhao, Xin Zheng, Li-Ying Sun, Ping Li, Feng-Qi Liu, Ge Gao, and Alideertu Dong. 2013. "Fabrication of Magnetic-Antimicrobial-Fluorescent Multifunctional Hybrid Microspheres and Their Properties" International Journal of Molecular Sciences 14, no. 4: 7391-7404. https://doi.org/10.3390/ijms14047391




