Disruption of AtWNK8 Enhances Tolerance of Arabidopsis to Salt and Osmotic Stresses via Modulating Proline Content and Activities of Catalase and Peroxidase
Abstract
:1. Introduction
2. Results and Discussion
2.1. Activities of the AtWNK8 Promoter at Tissue Level
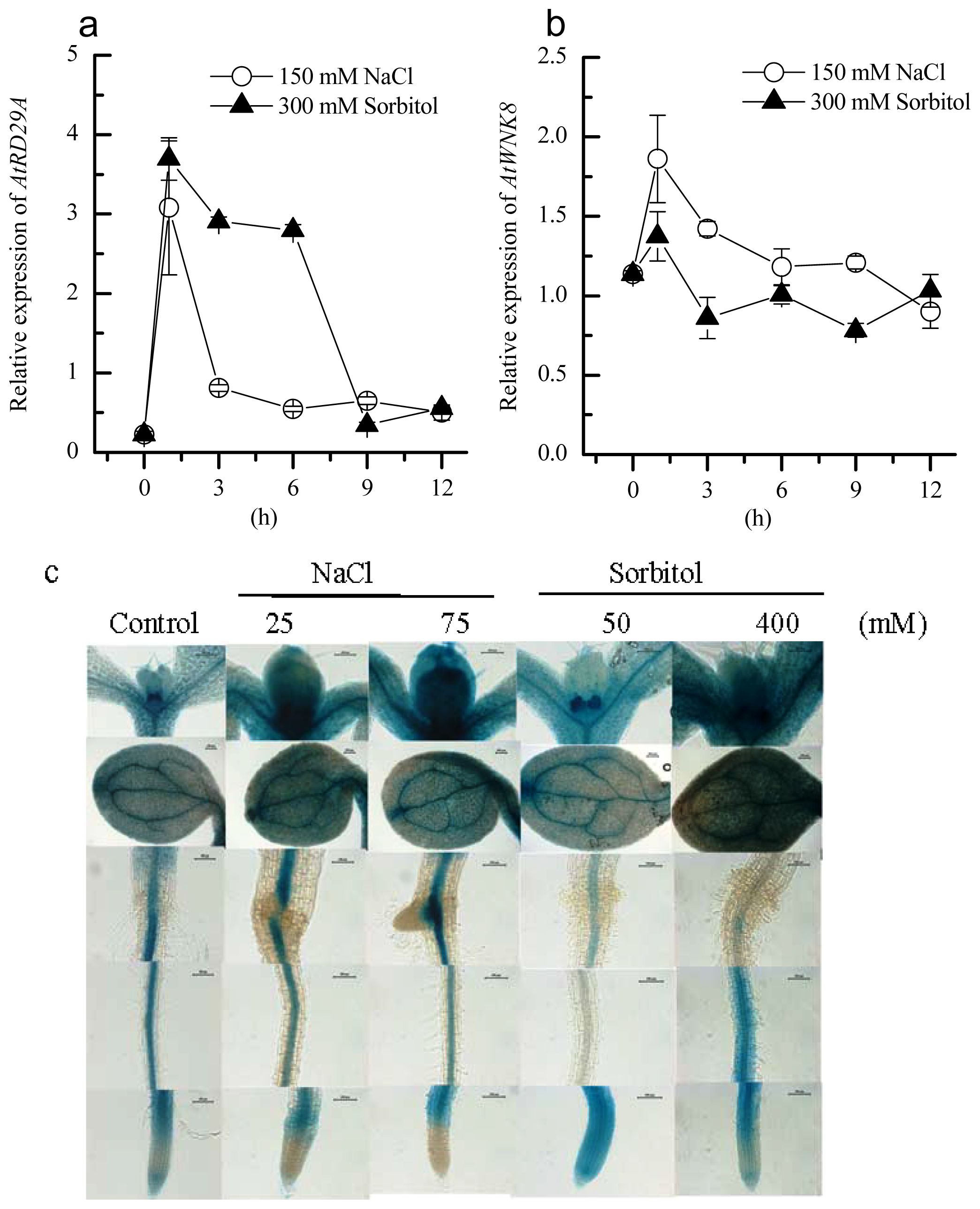
2.2. Induction of AtWNK8 by NaCl and Sorbitol Stresses in Arabidopsis
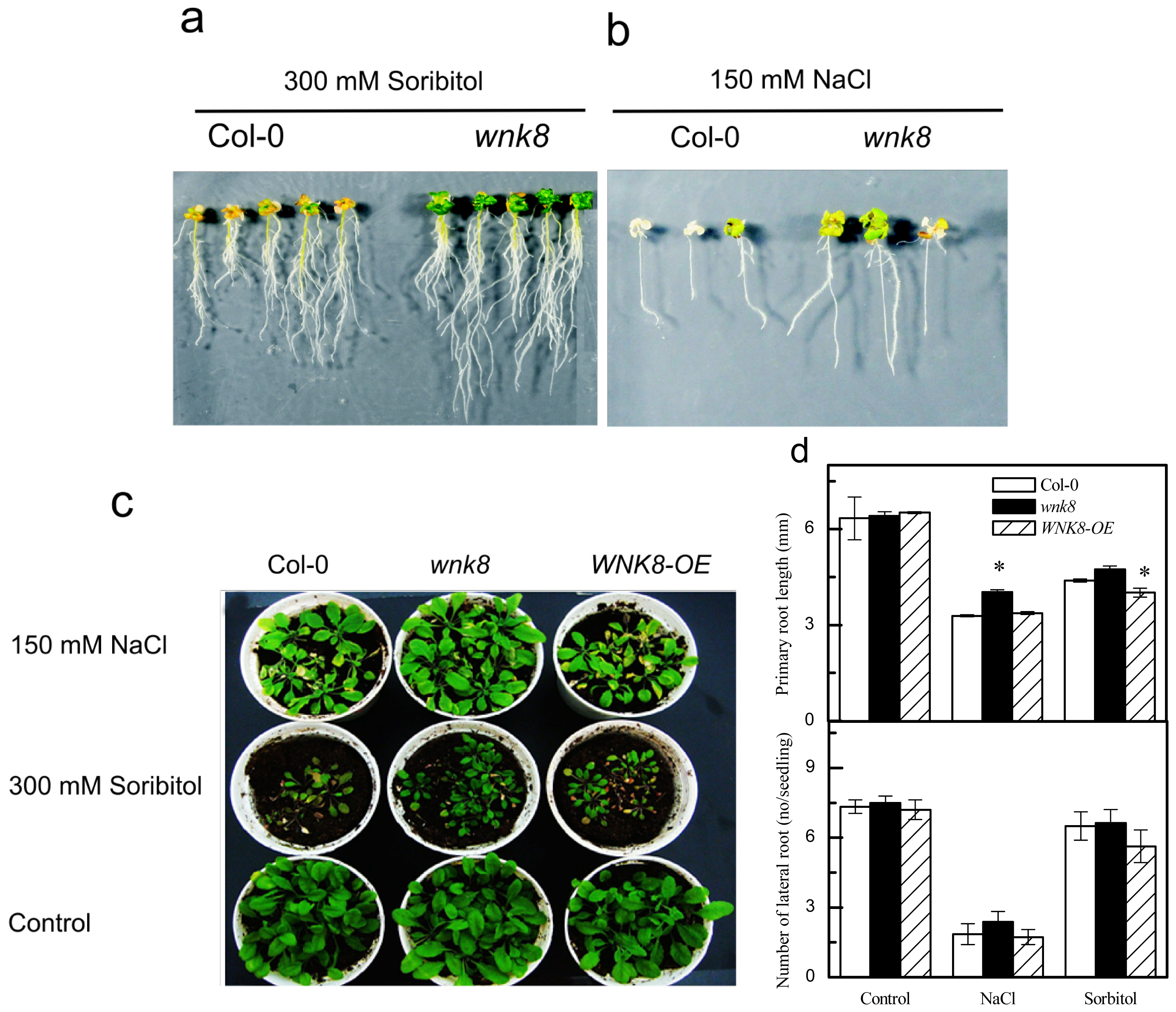
2.3. Responses of Knock-Out Mutant wnk8 and Overexpression Line of WNK8 (WNK8-OE) to Salt and Osmotic Stresses
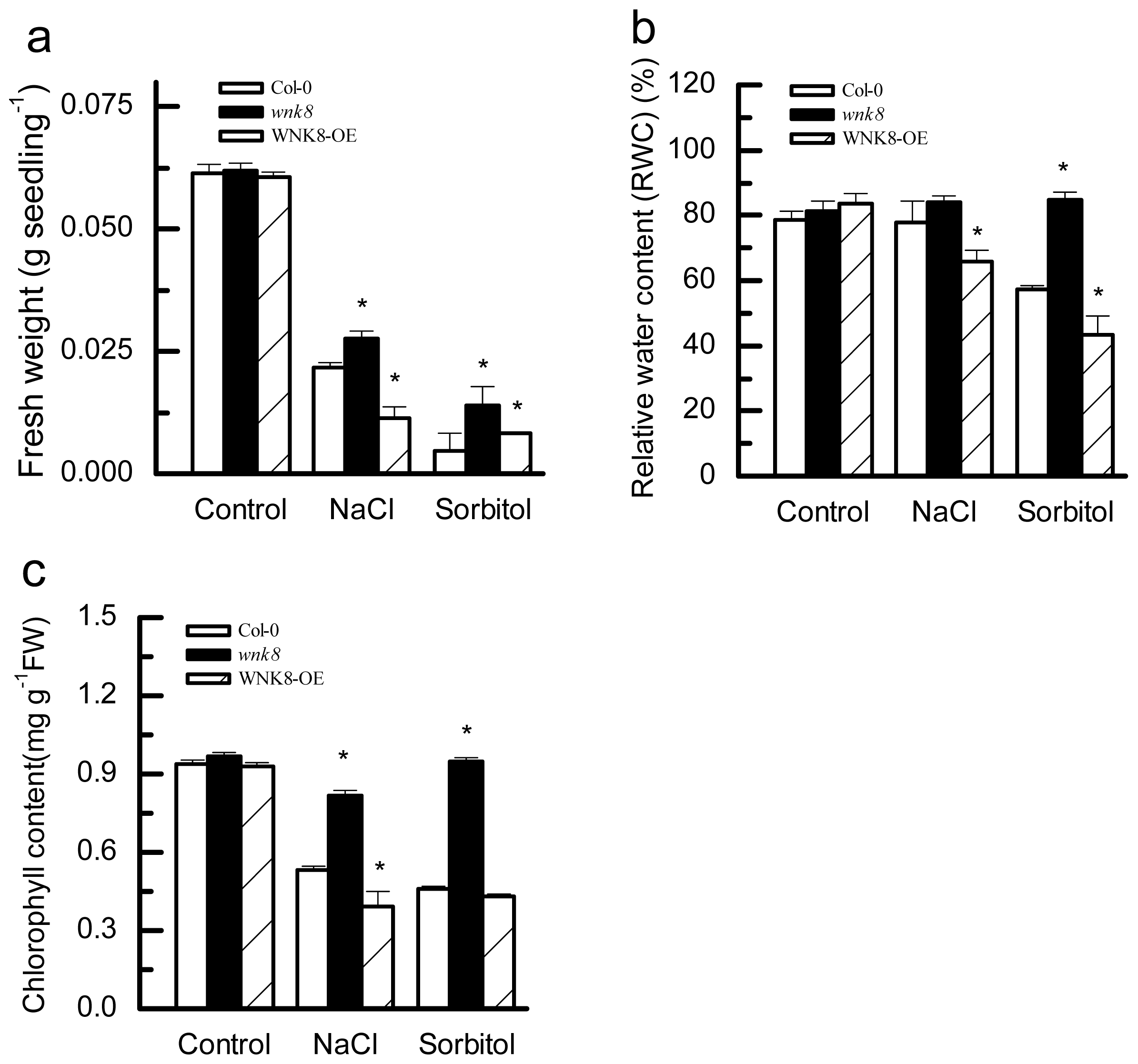
2.4. Effects of Salt and Osmotic Stresses on Fresh Weight (FW), Relative Water Content (RWC) and Chlorophyll Content of Col-0, wnk8 Mutant and WNK8-OE Plants
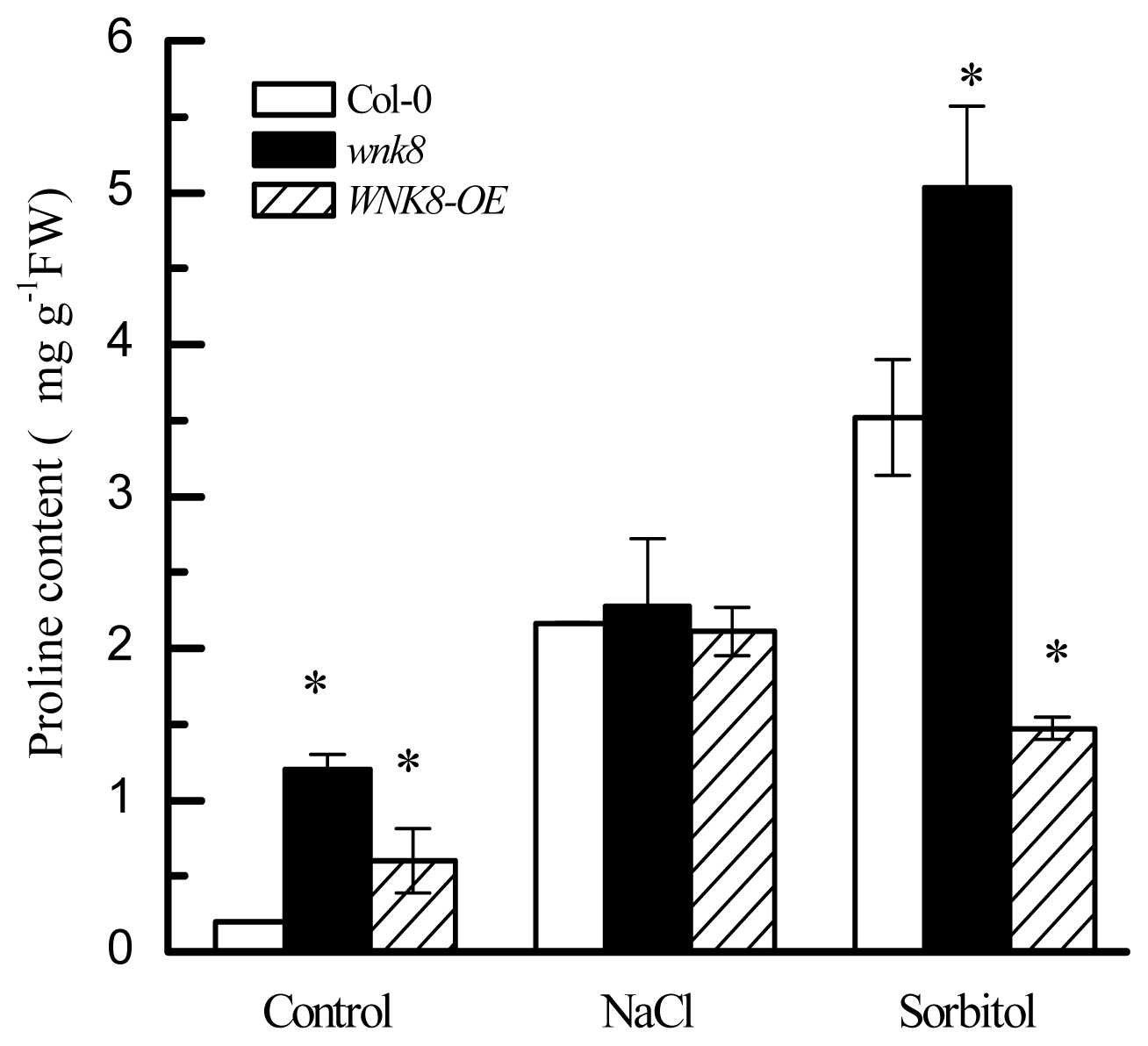
2.5. Higher Tolerance of wnk8 to Salt and Osmotic Stresses Was Partially Achieved by Accumulating More Proline
2.6. The wnk8 Mutant Maintained Higher Activities of CAT and POD in Salt and Osmotic Stresses
3. Experimental Section
3.1. Plant Materials and Growth Conditions
3.2. NaCl and Sorbitol Treatments
3.3. Determination of WNK8 Promoter Activity
3.4. Determination of WNK8 Transcript Level under Salt and Osmotic Stress
3.5. Determination of FW, RWC and Chlorophyll Content
3.6. Measurement of Proline Content and Activities of CAT and POD
3.7. Data Analysis
4. Conclusions
Supplementary Information
ijms-14-07032-s001.docAcknowledgements
Conflict of Interest
References
- Munns, R; Tester, M. Mechanisms of salinity tolerance. Annu. Rev. Plant Biol. 2008, 59, 651–681. [Google Scholar]
- Greenway, H; Munnsr, R. Mechanisms of salt tolerance in non-halophytes. Annu. Rev. Plant Physiol. 1980, 31, 149–190. [Google Scholar]
- Xiong, L; Schumaker, K.S; Zhu, J.K. Cell signaling during cold, drought, and salt stress. Plant Cell 2002, 14, S165–S183. [Google Scholar]
- Fridovich, I. Biological effects of superoxide radical. Arch. Biochem.Biophy 1986, 247, 1–11. [Google Scholar]
- Bowler, C; Montagu, M.V; Inze, D. Superoxide dismutase and stress tolerance. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1992, 43, 83–116. [Google Scholar]
- McCue, K.F; Hanson, D. Drought and salt tolerance: Towards understanding and application. Trends Biotechnol. 1990, 8, 358–362. [Google Scholar]
- Chaitanya, K.V.D; Sundar, P.P; Jutur, A; Reddy, R. Water stress effects on photosynthesis in different mulberry cultivars. Plant Growth Regul. 2003, 40, 75–80. [Google Scholar]
- Qiu, Q.S; Guo, Y; Dietrich, M.A; Schumaker, K.S; Zhu, J.K. Regulation of SOS1, a plasma membrane Na+/H+ exchanger in Arabidopsis thaliana, by SOS2 and SOS3. Proc. Natl. Acad. Sci. USA 2002, 99, 8436–8441. [Google Scholar]
- Wilson, F.H; Disse-Nicodeme, S; Choate, K.A; Ishikawa, K; Nelson-Williams, C; Desitter, I; Gunel, M; Milford, D.V; Lipkin, G.W; Achard, J.M.; et al. Science 2001, 293, 1107–1112.
- Xu, B.E; Min, X; Stippec, S; Lee, B.H; Goldsmith, E.J; Cobb, M.H. Regulation of WNK1 by an autoinhibitory domain and autophosphorylation. J. Biol. Chem. 2002, 277, 48456–48462. [Google Scholar]
- Georgina, C; Amir, G; O’Shaughnessy, K.M. WNK kinases and the control of blood pressure. Pharmacol. Ther. 2005, 106, 221–231. [Google Scholar]
- Veríssimo, F; Jordan, P. WNK kinases and a novel protein kinase subfamily in multi-cellular organisms. Oncogene 2001, 20, 5562–5569. [Google Scholar]
- Wang, Y; Liu, K; Liao, H; Zhuang, C; Ma, H; Yan, X. The plant WNK gene family and regulation of flowering time in Arabidopsis. Plant Biol. 2008, 10, 548–562. [Google Scholar]
- Murakami-Kojima, M; Nakamichi, N; Yamashino, T; Mizuno, T. The APRR3 component of the clock-associated APRR1/TOC1 quintet is phosphorylated by a novel protein kinase belonging to the WNK family and the gene for which is also transcribed rhythmically in Arabidopsis thaliana. Plant Cell Physiol. 2002, 43, 675–683. [Google Scholar]
- Hong-Hermesdorf, A; Brüx, A; Grüber, A; Grüber, G; Schumacher, K. A WNK kinase binds and phosphorylates V-ATPase subunit C. FEBS Lett. 2006, 580, 932–939. [Google Scholar]
- Tsuchiya, T; Eulgem, T. The Arabidopsis defense component EDM2 affects the floral transition in an FLC-dependent manner. Plant J. 2010, 62, 518–528. [Google Scholar]
- Wang, Y; Suo, H; Zheng, Y; Liu, K; Zhuang, C; Kahle, K.T; Ma, H; Yan, X. The soybean root-specific protein kinase GmWNK1 regulates stress-responsive ABA signaling on the root system architecture. Plant J. 2010, 64, 230–242. [Google Scholar]
- Yilmaz, A; Mejia-Guerra, M.K; Kurz, K; Liang, X; Welch, L; Grotewold, E. AGRIS: The Arabidopsis gene regulatory information server, an update. Nucleic Acids Res. 2011, 39, D1118–D1122. [Google Scholar]
- Hao, D; Yamasaki, K; Sarai, A; Ohme-Takagi, M. Determinants in the sequence specific binding of two plant transcription factors, CBF1 and NtERF2, to the DRE and GCC motifs. Biochemistry 2002, 41, 4202–4208. [Google Scholar]
- Zarka, D.G; Vogel, J.T; Cook, D; Thomashow, M.F. Cold induction of Arabidopsis CBF genes involves multiple ICE (inducer of CBF expression) promoter elements and a cold-regulatory circuit that is desensitized by low temperature. Plant Physiol. 2003, 133, 910–918. [Google Scholar]
- Schindler, U; Cashmore, A.R. Photoregulated gene expression may involve ubiquitous DNA binding proteins. EMBO J. 1990, 9, 3415–3427. [Google Scholar]
- Kahle, K.T; Ring, A.M; Lifton, R.P. Molecular physiology of the WNK kinases. Annu. Rev. Physiol. 2008, 70, 329–355. [Google Scholar]
- Narusaka, Y; Nakashima, K; Shinwari, Z.K; Sakuma, Y; Furihata, T; Abe, H; Narusaka, M; Shinozaki, K; Yamaguchi-Shinozaki, K. Interaction between two cis-acting elements, ABRE and DRE, in ABA-dependent expression of Arabidopsis rd29A gene in response to dehydration and high-salinity stresses. Plant J. 2003, 34, 137–148. [Google Scholar]
- Fitter, A; Strickland, T. Architectural analysis of plant root systems. II. Influence of nutrient supply on architecture in contrasting plant species. New Phytol. 1991, 118, 383–389. [Google Scholar]
- Deak, K.I; Malamy, J. Osmotic regulation of root system architecture. Plant J. 2005, 43, 17–28. [Google Scholar]
- Kishor, P.B.K; Hong, Z.L; Miao, G.H; Hu, C.A.A; Verma, D.P.S. Overexpression of delta-pyrroline-5-carboxylate synthetase increases proline production and confers osmotolerance in transgenic plants. Plant Physiol. 1995, 108, 1387–1394. [Google Scholar]
- Hare, P; Cress, W. Metabolic implications of stress-induced proline accumulation in plants. Plant Growth Regul. 1997, 21, 79–102. [Google Scholar]
- Deuschle, K; Funck, D; Forlani, G; Stransky, H; Biehl, A; Leister, D.; van der Graaff, E; Kunze, R; Frommer, W. The role of d1-pyrroline-5-carboxylate dehydrogenase in proline degradation. Plant Cell 2004, 16, 3413–3425. [Google Scholar]
- Scandalios, J.G. The rise of ROS. Trends Biochem. Sci 2002, 27, 483–486. [Google Scholar]
- Urano, D; Phan, N; Jones, J.C; Yang, J; Huang, J; Grigston, J; Philip Taylor, J; Jones, A.M. Endocytosis of the seven-transmembrane RGS1 protein activates G-protein-coupled signalling in Arabidopsis. Nat. Cell Biol. 2012, 14, 1079–1088. [Google Scholar]
- Wang, Y; Suo, H; Zhuang, C; Ma, H; Yan, X. Overexpression of the soybean GmWNK1 altered the sensitivity to salt and osmotic stress in Arabidopsis. J. Plant Physiol. 2011, 168, 2260–2270. [Google Scholar]
- Weigel, D; Glazebrook, J. Arabidopsis: A Laboratory Manual; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, New York, NY, USA, 2002. [Google Scholar]
- Jefferson, R.A; Kavanagh, T.A; Bevan, M.W. GUS fusions: beta-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. EMBO J. 1987, 6, 3901–3907. [Google Scholar]
- Clough, S.J; Bent, A.F. Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J. 1998, 16, 735–743. [Google Scholar]
- Alonso, J.M; Stepanova, A.N; Leisse, T.J; Kim, C.J; Chen, H; Shinn, P; Stevenson, D.K; Zimmerman, J; Barajas, P; Cheuk, R.; et al. Genome-wide insertional mutagenesis of Arabidopsis thaliana. Science 2003, 301, 653–657. [Google Scholar]
- Abramoff, M; Magelhaes, P; Ram, S. Image processing with ImageJ. Biophotonics Int. 2004, 11, 36–42. [Google Scholar]
- Lichtenthaler, H. Pigments of photosynthetic biomembranes. Methods Enzymol 1987, 148, 350–382. [Google Scholar]
- Smart, R.E. Rapid estimates of relative water content. Plant Physiol 1974, 53, 258–260. [Google Scholar]
- Bates, L.P; Waldren, R.P; Teare, I.D. Rapid determination of free proline for water-stress studies. Plant Soil 1973, 39, 205–208. [Google Scholar]
- Bradford, M. A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem 1976, 72, 248–254. [Google Scholar]
- Maehly, A.C; Chance, B. The assay of catalases and peroxidases. Methods Biochem. Anal. 1954, 1, 357–424. [Google Scholar]
- Shannon, L; Kay, E; Lew, J. Peroxidase isoenzymes from horseradish roots: isolation and physical properties. J. Biol. Chem. 1966, 241, 2166–2172. [Google Scholar]


| cis-Element | Sequence | Position |
|---|---|---|
| Phosphorus-related NIT-2 binding site | TATCTA(/G/T) | −1317, −1037, −917 |
| Phosphorus-related TATA box-like binding site | TATAAATA | −797 |
| Light responsive GT-1 binding site | GA(/G)A(/T)AAA(/T) | −1366, −979, −274 |
| Dehydration response DRE binding site | CCGAC | −1546, −1541 |
| Cold response element binding site | ACTCCG | −236 |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhang, B.; Liu, K.; Zheng, Y.; Wang, Y.; Wang, J.; Liao, H. Disruption of AtWNK8 Enhances Tolerance of Arabidopsis to Salt and Osmotic Stresses via Modulating Proline Content and Activities of Catalase and Peroxidase. Int. J. Mol. Sci. 2013, 14, 7032-7047. https://doi.org/10.3390/ijms14047032
Zhang B, Liu K, Zheng Y, Wang Y, Wang J, Liao H. Disruption of AtWNK8 Enhances Tolerance of Arabidopsis to Salt and Osmotic Stresses via Modulating Proline Content and Activities of Catalase and Peroxidase. International Journal of Molecular Sciences. 2013; 14(4):7032-7047. https://doi.org/10.3390/ijms14047032
Chicago/Turabian StyleZhang, Baige, Kaidong Liu, Yan Zheng, Yingxiang Wang, Jinxiang Wang, and Hong Liao. 2013. "Disruption of AtWNK8 Enhances Tolerance of Arabidopsis to Salt and Osmotic Stresses via Modulating Proline Content and Activities of Catalase and Peroxidase" International Journal of Molecular Sciences 14, no. 4: 7032-7047. https://doi.org/10.3390/ijms14047032





