Protein Contribution to Plant Salinity Response and Tolerance Acquisition
Abstract
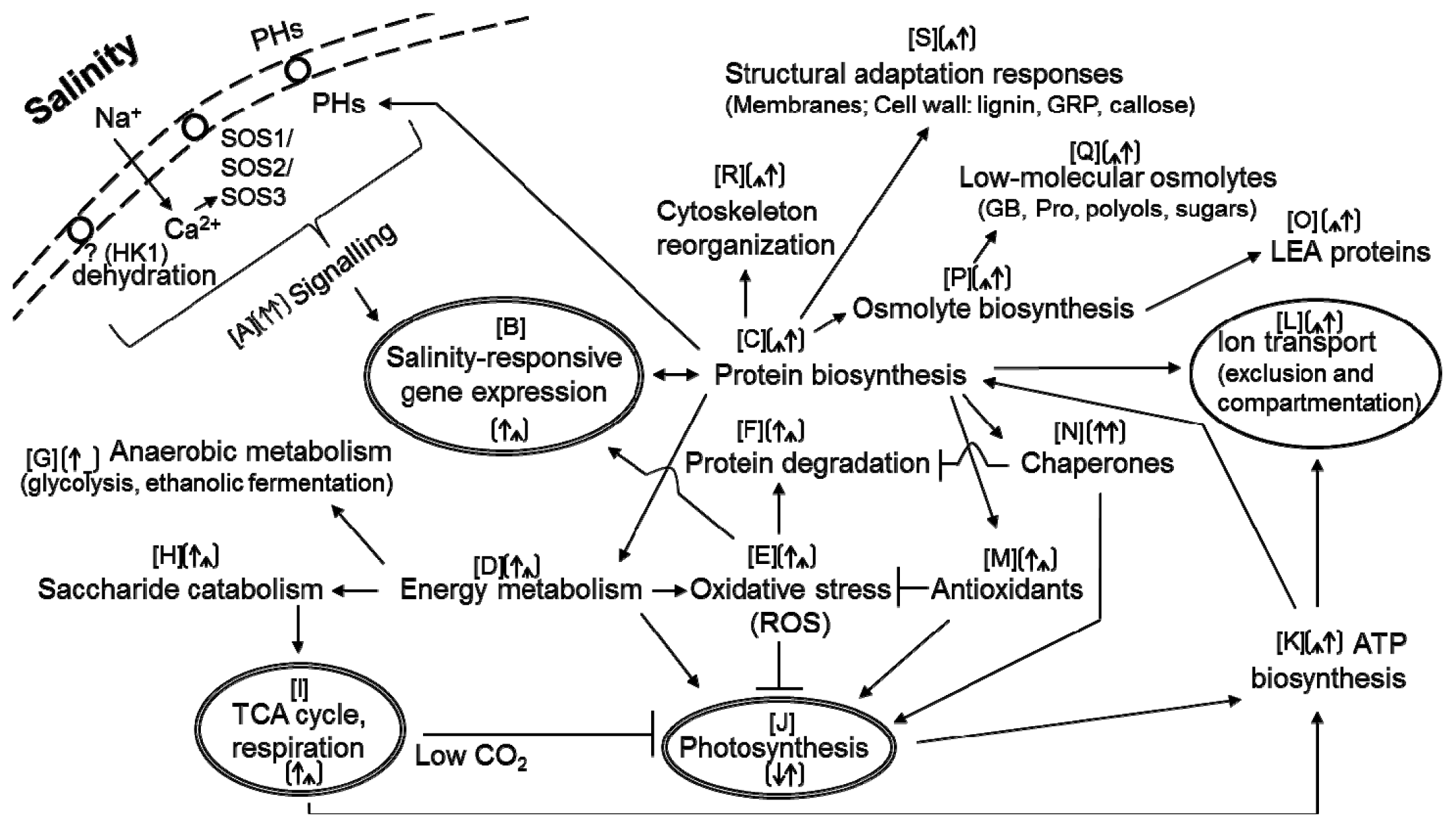
:1. Introduction
2. Plant Adaptation to Salinity—The Role of Proteomics Regarding the Other Aspects of Plant Salinity Response
- (1)
- Genomic level: Tolerant plants may possess some unique stress-responsive genes which are absent in susceptible plants (differences at genome structure level).
- (2)
- Transcriptomic level: Tolerant plants reveal altered regulation of gene expression of important stress-responsive genes than susceptible plants (qualitative and quantitative differences at gene expression level);
- (3)
- Proteomic level: Proteins involved in stress response reveal an altered activity in tolerant plants than in susceptible ones (differences in protein structure and activity level).
3. Effects of Salinity Stress on Plants (Physiological Level)
3.1. Osmotic Effect
3.1.1. Osmotic Stress-Related Signalling
3.2. Ionic Effect
3.2.1. Ion-Related Signalling
3.3. Further Aspects of Salinity Stress
4. Genomic Level of Adaptation to Salinity
5. Transcriptomic Level of Adaptation to Salinity—Comparative Studies
6. Proteomic Level of Adaptation to Salinity
6.1. Signalling
6.2. Gene Expression and Protein Metabolism
6.3. Energy Metabolism
6.4. Oxidative Stress and Stress-Related Proteins
6.5. Osmolyte Metabolism
6.6. Ion Transport
6.7. Mechanical Stress-Related Proteins
6.8. Phytohormone Metabolism
6.9. Lipid Metabolism
6.10. Secondary Metabolism
6.11. Postranslational Modifications
7. Conclusions and Future Perspectives
Supplementary Information
ijms-14-06757-s001.docAcknowledgements
Abbreviations
| 2D-DIGE | two-dimensional difference gel electrophoresis |
| ABA | abscisic acid |
| ACP | acyl carrier protein |
| ADH | alcohol dehydrogenase |
| ADK | adenylate kinase |
| ALDH | betaine aldehyde dehydrogenase |
| AOC | allene oxide cyclase |
| AOX | alternative oxidase |
| APX | ascorbate peroxidase |
| CAB | chlorophyll a/b binding protein |
| CaM | calmodulin |
| CAT | catalase |
| Chl | chlorophyll |
| CK | cytokinin |
| CCOMT | caffeic acid 3-O-methyltransferase |
| CMO | choline monooxygenase |
| COMT | caffeoyl-O-methyltransferase |
| DHAR | dehydroascorbate reductase |
| DHN | dehydrin |
| DNPH | dinitrophenylhydrazine |
| eEF | eukaryotic elongation translation factor |
| eIF | eukaryotic initiation translation factor |
| ENO | enolase |
| ENR | enoyl-ACP reductase |
| FBP | fructose bisphosphate |
| FFZE | free flow zonal electrophoresis |
| GA | gibberellin |
| GAPDH | glyceraldehyde-3-phosphate dehydrogenase |
| GB | glycine betaine |
| GRP | glycine-rich protein |
| GS | glutamine synthetase |
| GST | glutathione-S-transferase |
| HK | histidine kinase |
| HKT | high-affinity potassium transporter |
| HSP | heat shock protein |
| IDI | iron deficiency-induced |
| IDS | iron deficiency-specific |
| INPS | l-myo-inositol-1-phosphate synthase |
| JA | jasmonic acid |
| LEA | Late embryogenesis abundant |
| LHC | light-harvesting complex |
| LOX | lipoxygenase |
| LRR | leucine-rich repeat |
| LTP | lipid transfer protein |
| MAPK | mitogen-activated protein kinase |
| MDAR | monodehydroascorbate reductase |
| MDH | malate dehydrogenase |
| MRL | mannose-binding RICE lectin |
| NCED | 9-cis-epoxy-dioxygenase |
| NDPK | nucleotide diphosphate kinase |
| NHX | Na+/H+ exchanger |
| OEC | oxygen evolving complex |
| OEE | oxygen evolving enhancer |
| P5CS | pyrroline-5-carboxylate synthase |
| PA | phosphatidic acid |
| PC | plastocyanin |
| PDH | proline dehydrogenase |
| PEPCase | phosphoenolpyruvate carboxylase |
| PGK | phosphoribulokinase |
| PH | phytohormone |
| PK | protein kinase |
| PLC | phospholipase C |
| PLD | phospholipase D |
| PPase | pyrophosphatase |
| PPP | pentose phosphate pathway |
| PR | pathogenesis-related |
| PRK | phosphoribulokinase |
| PRX | peroxiredoxin |
| PS | photosystem |
| PS RC | photosystem reaction center |
| PTM | posttranslational modification |
| ROS | reactive oxygen species |
| RubisCO | ribuloso-1,5-bisphosphate carboxylase/oxygenase |
| RubisCO LSU | RubisCO large subunit |
| RubisCO SSU | RubisCO small subunit |
| SA | salicylic acid |
| SAM | S-adenosylmethionine |
| SAMS | S-adenosylmethionine synthetase |
| SBP | sedoheptulose-1,7-bisphosphate |
| SOD | superoxide dismutase |
| SOS | salt overly sensitive |
| SUS | sucrose synthase |
| TCA | tricarboxylic acid cycle |
| TPI | triose phosphate isomerase |
| TPR | tetratricopeptide repeat |
| Trx | thioredoxin |
| V-ATPase | vacuolar ATPase |
| VDAC | voltage dependent anion channel |
| XET | xyloglucan endotransglycosylase |
Conflict of Interest
References
- Munns, R. Genes and salt tolerance: Bringing them together. Tansley. Rev. New Phytol 2005, 167, 645–663. [Google Scholar]
- Munns, R. Comparative physiology of salt and water stress. Plant Cell Environ 2002, 25, 239–250. [Google Scholar]
- Munns, R.; Tester, M. Mechanisms of salinity tolerance. Annu. Rev. Plant Biol 2008, 59, 651–681. [Google Scholar]
- Kosová, K.; Vítámvás, P.; Prášil, I.T.; Renaut, J. Plant proteome changes under abiotic stress— contribution of proteomics studies to understanding plant stress response. J. Proteomics 2011, 74, 1301–1322. [Google Scholar]
- Flowers, T.J.; Hajibagheri, M.A.; Clipson, N.J.W. Halophytes. Q. Rev. Biol 1986, 61, 313–337. [Google Scholar]
- Flowers, T.J.; Colmer, T.D. Salinity tolerance in halophytes. Tansley Review. New Phytol 2008, 179, 945–963. [Google Scholar]
- Colmer, T.D.; Flowers, T.J.; Munns, R. Use of wild relatives to improve salt tolerance in wheat. J. Exp. Bot 2006, 57, 1059–1078. [Google Scholar]
- Hasegawa, P.M.; Bressan, R.A.; Zhu, J.K.; Bohnert, H.J. Plant cellular and molecular responses to high salinity. Annu. Rev. Plant Physiol. Plant Mol. Biol 2000, 51, 463–499. [Google Scholar]
- Flowers, T.J. Improving crop salt tolerance. J. Exp. Bot 2004, 55, 307–319. [Google Scholar]
- Munns, R.; James, R.A.; Läuchli, A. Approaches to increasing the salt tolerance of wheat and other cereals. J. Exp. Bot 2006, 57, 1025–1043. [Google Scholar]
- Zhu, J.K. Salt and drought stress signal transduction in plants. Annu. Rev. Plant. Biol 2002, 53, 247–273. [Google Scholar]
- Yamaguchi-Shinozaki, K.; Shinozaki, K. Transcriptional regulatory networks in cellular responses and tolerance to dehydration and cold stresses. Annu. Rev. Plant Biol 2006, 57, 781–803. [Google Scholar]
- Sobhanian, H.; Aghaei, K.; Komatsu, S. Changes in the plant proteome resulting from salt stress: Toward the creation of salt-tolerant crops? J. Proteomics 2011, 74, 1323–1337. [Google Scholar]
- Zhang, H.; Han, B.; Wang, T.; Chen, S.; Li, H.; Zhang, Y.; Dai, S. Mechanisms of plant salt response: Insights from proteomics. J. Proteome Res 2012, 11, 49–67. [Google Scholar]
- Inan, G.; Zhang, Q.; Li, P.; Wang, Z.; Cao, Z.; Zhang, H.; Zhang, C.; Quist, T.M.; Goodwin, M.S.; Zhu, J.; et al. Salt cress. A halophyte and cryophyte Arabidopsis relative model systém and its applicability to molecular genetic analyses of growth and development of extremophiles. Plant Physiol 2004, 135, 1718–1737. [Google Scholar]
- Sengupta, S.; Majumder, A.L. Insight into the salt tolerance factors of a wild halophytic rice, Porteresia. coarctata: A physiological and proteomic approach. Planta 2009, 229, 911–929. [Google Scholar]
- Sengupta, S.; Majumder, A.L. Porteresia. coarctata (Roxb.) Tateoka, a wild rice: A potential model for studying salt-stress biology in rice. Plant Cell Environ 2010, 33, 526–542. [Google Scholar]
- Dassanayake, M.; Oh, D.H.; Haas, J.S.; Hernandez, A.; Hong, H.; Ali, S.; Yun, D.J.; Bressan, R.A.; Zhu, J.K.; Bohnert, H.J.; et al. The genome of the extremophile crucifer Thellungiella. parvula. Nat. Genet 2011, 43, 913–918. [Google Scholar]
- Taji, T.; Seki, M.; Satou, M.; Sakurai, T.; Kobayashi, M.; Ishiyama, K.; Narusaka, Y.; Narusaka, M.; Zhu, J.K.; Shinozaki, K. Comparative genomics in salt tolerance between Arabidopsis and Arabidopsis-related halophyte salt cress using Arabidopsis microarray. Plant Physiol 2004, 135, 1697–1709. [Google Scholar]
- Gong, Q.; Li, P.; Ma, S.; Rupassara, S.I.; Bohnert, H.J. Salinity stress adaptation competence in the extremophile Thellungiella halophila in comparison with its relative Arabidopsis thaliana. Plant. J 2005, 44, 826–839. [Google Scholar]
- Kant, S.; Kant, P.; Raveh, E.; Barak, S. Evidence that differential gene expression between the halophyte, Thellungiella halophila, and Arabidopsis thaliana is responsible for higher levels of the compatible osmolyte proline and tight control of Na+ uptake in T. halophila. Plant. Cell Environ 2006, 29, 1220–1234. [Google Scholar]
- Pang, Q.; Chen, S.; Dai, S.; Chen, Y.; Wang, Y.; Yan, X. Comparative proteomics of salt tolerance in Arabidopsis thaliana and Thellungiella halophila. J. Proteome Res 2010, 9, 2584–2599. [Google Scholar]
- Wang, M.C.; Peng, Z.Y.; Li, C.L.; Li, F.; Liu, C.; Xia, G.M. Proteomic analysis on a high salt tolerance introgression strain of Triticum aestivum/Thinopyrum ponticum. Proteomics 2008, 8, 1470–1489. [Google Scholar]
- Wakeel, A.; Asif, A.R.; Pitann, B.; Schubert, S. Proteome analysis of sugar beet (Beta vulgaris L.) elucidates constitutive adaptation during the first phase of salt stress. J. Plant Physiol 2011, 168, 519–526. [Google Scholar]
- Jellouli, N.; Ben Jouira, H.; Skouri, H.; Ghorbel, A.; Gourgouri, A.; Mliki, A. Proteomic analysis of Tunisian grapevine cultivar Razegui under salt stress. J. Plant Physiol 2008, 165, 471–481. [Google Scholar]
- Askari, H.; Edqvist, J.; Hajheidari, M.; Kafi, M.; Salekdeh, G.H. Effects of salinity levels on proteome of Suaeda aegyptiaca leaves. Proteomics 2006, 6, 2542–2554. [Google Scholar]
- Wang, X.; Fan, P.; Song, H.; Chen, X.; Li, X.; Li, Y. Comparative proteomic analysis of differentially expressed proteins in shoots of Salicornia europaea under different salinity. J. Proteome Res 2009, 8, 3331–3345. [Google Scholar]
- Yu, J.; Chen, S.; Zhao, Q.; Wang, T.; Yang, C.; Diaz, C.; Sun, G.; Dai, S. Physiological and proteomic analysis of salinity tolerance in Puccinellia tenuiflora. J. Proteome Res 2011, 10, 3852–3870. [Google Scholar]
- Sobhanian, H.; Motamed, N.; Jazii, F.R.; Nakamura, T.; Komatsu, S. Salt stress induced differential proteome and metabolome response in the shoots of Aeluropus lagopoides (Poaceae), a halophyte C4 plant. J. Proteome Res 2010, 9, 2882–2897. [Google Scholar]
- Caruso, G.; Cavaliere, C.; Guarino, C.; Gubbiotti, R.; Foglia, P.; Lagana, A. Identification of changes in Triticum. durum L. leaf proteome in response to salt stress by two-dimensional electrophoresis and MALDI-TOF mass spektrometry. Anal. Bioanal. Chem 2008, 391, 381–390. [Google Scholar]
- Liska, A.J.; Shevchenko, A.; Pick, U.; Katz, A. Enhanced photosynthesis and redox energy production contribute to salinity tolerance in Dunaliella as revealed by homology-based proteomics. Plant Physiol 2004, 136, 2806–2817. [Google Scholar]
- Katz, A.; Waride, P.; Shevchenko, A.; Pick, U. Salt-induced changes in the plasma membrane proteome of the halotolerant alga Dunaliella. salina as revealed by blue native gel electrophoresis and nano-LC-MS/MS analysis. Mol. Cell Proteomics 2007, 6, 1459–1472. [Google Scholar]
- Mehta, P.A.; Rebala, K.C.; Venkataraman, G.; Parida, A. A diurnally regulated dehydrin from Avicennia marina that shows nucleo-cytoplasmic localization and is phosphorylated by Casein kinase II in vitro. Plant Physiol. Biochem 2009, 47, 701–709. [Google Scholar]
- Manaa, A.; Ahmed, H.B.; Valot, B.; Bouchet, J.P.; Aschi-Smiti, S.; Causse, M.; Faurobet, M. Salt and genotype impact on plant physiology and root proteome variations in tomato. J. Exp. Bot 2011, 62, 2797–2813. [Google Scholar]
- Tada, Y.; Kashimura, T. Proteomic analysis of salt-responsive proteins in the mangrove plant, Bruguiera gymnorhiza. Plant Cell Physiol 2009, 50, 439–446. [Google Scholar]
- Levitt, J. Responses of Plants to Environmental Stress. In Chilling, Freezing and High Temperature Stress, 2nd ed; Academic Press: New York NY, USA, 1980. [Google Scholar]
- Larcher, W. Physiological Plant. Ecology, 4th ed; Springer Verlag: Berlin/Heidelberg, Germany, 2003. [Google Scholar]
- Urao, T.; Yakubov, B.; Satoh, R.; Yamaguchi-Shinozaki, K.; Seki, M.; Hirayama, T.; Shinozaki, K. A transmembrane hybrid-type histidine kinase in Arabidopsis functions as an osmosensor. Plant Cell 1999, 11, 1743–1754. [Google Scholar]
- Finkelstein, R. Abscisic acid: A seed maturation and antistress signal. In Plant Physiology, 4th ed; Taiz, L., Zeigler, E., Eds.; Sinauer Associates, Inc: Sunderland, MA, USA, 2006; pp. 593–616. [Google Scholar]
- Ma, Y.; Szostkiewicz, I.; Korte, A.; Moes, D.; Yang, Y.; Christmann, A.; Grill, E. Regulators of PP2C phosphatase activity function as abscisic acid sensors. Science 2009, 324, 1064–1068. [Google Scholar]
- Park, S.Y.; Fung, P.; Nishimura, N.; Jensen, D.R.; Fujii, H.; Zhao, Y.; Lumba, S.; Santiago, J.; Rodrigues, A.; Chow, T.F.F.; et al. Abscisic acid inhibits type 2C protein phosphatases via the PYR/PYL family of START proteins. Science 2009, 324, 1068–1071. [Google Scholar]
- Klingler, J.P.; Batelli, G.; Zhu, J.K. ABA receptors: The START of a new paradigm in phytohormone signalling. J. Exp. Bot 2010, 61, 3199–3210. [Google Scholar]
- Pandey, S.; Nelson, D.C.; Assmann, S.A. Two novel GPCR-type G proteins are abscisic acid receptor in Arabidopsis. Cell 2009, 136, 136–148. [Google Scholar]
- Kosová, K.; Vítámvás, P.; Prášil, I.T. The role of dehydrins in plant stress response. In Handbook of Plant. and Crop. Stress, 3rd ed.; Pessarakli, M., Ed.; CRC Press, Taylor and Francis: Boca Raton, FL, USA, 2010; pp. 239–285. [Google Scholar]
- Liu, J.; Ishitani, M.; Halfter, U.; Kim, C.S.; Zhu, J.K. The Arabidopsis thaliana SOS2 gene encodes a protein kinase that is required for salt tolerance. Proc. Natl. Acad. Sci. USA 2000, 97, 3730–3734. [Google Scholar]
- Shi, H.; Ishitani, M.; Kim, C.; Zhu, J.K. The Arabidopsis thaliana salt tolerance gene SOS1 encodes a putative Na+/H+ antiporter. Proc. Natl. Acad. Sci. USA 2000, 97, 6896–6901. [Google Scholar]
- Shi, H.; Quintero, F.J.; Pardo, J.M.; Zhu, J.K. The putative plasma membrane Na+/H+ antiporter SOS1 controls long-distance Na+ transport in plants. Plant. Cell 2002, 14, 465–477. [Google Scholar]
- Shi, H.; Zhu, J.K. SOS4, a pyridoxal kinase gene, is required for root hair development in Arabidopsis. Plant Physiol 2002, 129, 585–593. [Google Scholar]
- Mohr, H.; Schopfer, P. Plant Physiology, 4th ed; Springer-Verlag: Berlin/Heidelberg, Germany, 1995. [Google Scholar]
- Kumari, S.; Panjabi, V.; Kushwaha, H.R.; Sopory, S.K.; Singla-Pareek, S.L.; Pareek, A. Transcriptome map for seedling stage specific salinity stress response indicates a specific set of genes as candidate for saline tolerance in Oryza sativa L. Funct. Integr. Genomics 2009, 9, 109–123. [Google Scholar]
- Jiang, Y.; Yang, B.; Harris, N.S.; Deyholos, M.K. Comparative proteomic analysis of NaCl stress-responsive proteins in Arabidopsis roots. J. Exp. Bot 2007, 58, 3591–3607. [Google Scholar]
- Ndimba, B.K.; Chivasa, S.; Simon, W.J.; Slabas, A.R. Identification of Arabidopsis salt and osmotic stress responsive proteins using two-dimensional difference gel electrophoresis and mass spectrometry. Proteomics 2005, 5, 4185–4196. [Google Scholar]
- Dani, V.; Simon, W.J.; Duranti, M.; Croy, R.R.D. Changes in the tobacco leaf apoplast proteome in response to salt stress. Proteomics 2005, 5, 737–745. [Google Scholar]
- Razavizadeh, R.; Ehsanpour, A.A.; Ahsan, N.; Komatsu, S. Proteome analysis of tobacco leaves under salt stress. Peptides 2009, 30, 1651–1659. [Google Scholar]
- Chen, F.; Zhang, S.; Jiang, H.; Ma, W.; Korpelainen, H.; Li, C. Comparative proteomics analysis of salt response reveals sex-related photosynthetic inhibition by salinity in Populus cathayana cuttings. J. Proteome Res 2011, 10, 3944–3958. [Google Scholar]
- Chattopadhyay, A.; Subba, P.; Pandey, A.; Bhushan, D.; Kumar, R.; Datta, A.; Chakraborty, S.; Chakraborty, N. Analysis of the grasspea proteome and identification of stress-responsive proteins upon exposure to high salinity, low temperature, and abscisic acid treatment. Phytochemistry 2011, 72, 1293–1307. [Google Scholar]
- Xu, C.; Sibicky, T.; Huang, B. Protein profile analysis of salt-responsive proteins in leaves and roots in two cultivars of creeping bentgrass differing in salinity tolerance. Plant Cell Rep 2010, 29, 595–615. [Google Scholar]
- Abbasi, F.M.; Komatsu, S. A proteomic approach to analyze salt-responsive proteins in rice leaf sheath. Proteomics 2004, 4, 2072–2081. [Google Scholar]
- Chitteti, B.; Peng, Z. Proteome and phosphoproteome differential expression under salinity stress in rice (Oryza sativa) roots. J. Proteome Res 2007, 6, 1718–1727. [Google Scholar]
- Dooki, A.D.; Mayer-Posner, F.J.; Askari, H.; Zaiee, A.A.; Salekdeh, G.H. Proteomic responses of rice young panicles to salinity. Proteomics 2006, 6, 6498–6507. [Google Scholar]
- Kim, D.W.; Rakwal, R.; Agrawal, G.K.; Jung, Y.H.; Shibato, J.; Jwa, N.S.; Iwahashi, Y.; Iwahashi, H.; Kim, D.H.; Shim, I.S.; et al. A hydroponic rice seedling culture model system for investigating proteome of salt stress in rice leaf. Electrophoresis 2005, 26, 4521–4539. [Google Scholar]
- Yan, S.; Tang, Z.; Su, W.; Sun, W. Proteomic analysis of salt stress-responsive proteins in rice root. Proteomics 2005, 5, 235–244. [Google Scholar]
- Jacoby, R.P.; Millar, A.H.; Taylor, N.L. Wheat mitochondrial proteomes provide new links between antioxidant defense and plant salinity tolerance. J. Proteome Res 2010, 9, 6595–6604. [Google Scholar]
- Peng, Z.; Wang, M.; Li, F.; Lv, H.; Li, C.; Xia, G. A proteomic study of the response to salinity and drought stress in an introgression strain of bread wheat. Mol. Cell Proteomics 2009, 8, 2676–2686. [Google Scholar]
- Fatehi, F.; Hosseinzadeh, A.; Alizadeh, H.; Brimavandi, T.; Struik, P.C. The proteome response of salt-resistant and salt-sensitive barley genotypes to long-term salinity stress. Mol. Biol. Rep 2012, 39, 6387–6397. [Google Scholar]
- Rasoulnia, A.; Bihamta, M.R.; Peyghambari, S.A.; Alizadeh, H.; Rahnama, A. Proteomic response of barley leaves to salinity. Mol. Biol. Rep 2011, 38, 5055–5063. [Google Scholar]
- Sugimoto, M.; Takeda, K. Proteomic analysis of specific proteins in the root of salt-tolerant barley. Biosci. Biotech. Biochem 2009, 73, 2762–2765. [Google Scholar]
- Witzel, K.; Weidner, A.; Surabhi, G.K.; Börner, A.; Mock, H.P. Salt stress-induced alterations in the root proteome of barley genotypes with contrasting response towards salinity. J. Exp. Bot 2009, 60, 3546–3557. [Google Scholar]
- Witzel, K.; Weidner, A.; Surabhi, G.K.; Varshney, R.K.; Kunze, G.; Buck-Sorlin, G.H.; Börner, A.; Mock, H.P. Comparative analysis of the grain proteome fraction in barley genotypes with contrasting salinity tolerance during germination. Plant Cell Environ 2010, 33, 211–222. [Google Scholar]
- Veeranagamallaiah, G.; Jyothsnakumari, G.; Thippeswamy, M.; Reddy, P.C.O.; Surabhi, G.K.; Sriranganayakulu, G.; Mahesh, Y.; Rajasekhar, B.; Madhurarekha, C.; Sudhakar, C. Proteomic analysis of salt stress responses in foxtail millet (Setaria italica L. cv. Prasad) seedlings. Plant Sci 2008, 175, 631–641. [Google Scholar]
- Bandehagh, A.; Salekdeh, G.H.; Toorchi, M.; Mohammadi, A.; Komatsu, S. Comparative proteomic analysis of canola leaves under salinity stress. Proteomics 2011, 11, 1965–1975. [Google Scholar]
- Aghaei, K.; Ehsanpour, A.A.; Shah, A.H.; Komatsu, S. Proteome analysis of soybean hypokotyl and root under salt stress. Amino Acids 2009, 36, 91–98. [Google Scholar]
- Sobhanian, H.; Razavizadeh, R.; Nanjo, Y.; Ehsanpour, A.A.; Jazii, F.R.; Motamed, N.; Komatsu, S. Proteome analysis of soybean leaves, hypocotyls and roots under salt stress. Proteome Sci 2010, 8, 19. [Google Scholar]
- Kav, N.N.V.; Srivastava, S.; Goonewardende, L.; Blade, S.F. Proteome-level changes in the roots of Pisum sativum in response to salinity. Ann. Appl. Biol 2004, 145, 217–230. [Google Scholar]
- Jain, S.; Srivastava, S.; Sarin, N.B.; Kav, N.N.V. Proteomics reveals elevated levels of PR10 proteins in saline-tolerant peanut (Arachis hypogaea) calli. Plant Physiol. Biochem 2006, 44, 253–259. [Google Scholar]
- Kumar Swami, A.; Alam, S.I.; Sengupta, N.; Sarin, R. Differential proteomic analysis of salt response in Sorghum bicolor leaves. Environ. Exp. Bot 2011, 71, 321–328. [Google Scholar]
- Ngara, R.; Ndimba, R.; Borch-Jensen, J.; Jensen, O.N.; Ndimba, B. Identification and profiling of salinity stress-responsive proteins in Sorghum bicolor seedlings. J. Proteomics 2012, 75, 4139–4150. [Google Scholar]
- Zörb, C.; Herbst, R.; Forreite, C.; Schubert, S. Short-term effects of salt exposure on the maize chloroplast protein pattern. Proteomics 2009, 9, 4209–4220. [Google Scholar]
- Zörb, C.; Schmitt, S.; Mühling, K.H. Proteomic changes in maize roots after short-term adjustment to saline growth conditions. Proteomics 2010, 10, 4441–4449. [Google Scholar]
- Chen, S.; Gollop, N.; Heuer, B. Proteomic analysis of salt-stressed tomato (Solanum lycopersicum) seedlings: effect of genotype and exogenous application of glycinebetaine. J. Exp. Bot 2009, 60, 2005–2019. [Google Scholar]
- Aghaei, K.; Ehsanpour, A.A.; Komatsu, S. Proteome analysis of potato under salt stress. J. Proteome Res 2008, 7, 4858–4868. [Google Scholar]
- Du, C.X.; Fan, H.F.; Guo, S.R.; Tezuka, T.; Li, J. Proteomic analysis of cucmber seedling roots subjected to salt stress. Phytochemistry 2010, 71, 1450–1459. [Google Scholar]
- Tanou, G.; Job, C.; Rajjou, L.; Arc, E.; Belghazi, M.; Diamantidis, G.; Molassiotis, A.; Job, D. Proteomics reveals the overlapping roles of hydrogen peroxide and nitric oxide in the acclimation of citrus plants to salinity. Plant J 2009, 60, 795–804. [Google Scholar]
- Vincent, D.; Ergül, A.; Bohlman, M.C.; Tattersall, E.A.R.; Tillett, R.L.; Wheatley, M.D.; Woolsey, R.; Quilici, D.R.; Joets, J.; Schlauch, K.; et al. Proteomic analysis reveals differences between Vitis. vinifera L. cv. Chardonnay and cv. Cabernet Sauvignon and their responses to water deficit and salinity. J. Exp. Bot 2007, 58, 1873–1892. [Google Scholar]
- Salekdeh, G.H.; Komatsu, S. Crop proteomics: Aim at sustainable agriculture of tomorrow. Proteomics 2007, 7, 2976–2996. [Google Scholar]
- Li, W.; Zhang, C.; Lu, Q.; Wen, X.; Lu, C. The combined effect of salt stress and heat shock on proteome profiling in Suaeda salsa. J. Plant. Physiol 2011, 168, 1743–1752. [Google Scholar]
- Barkla, B.J.; Vera-Estrella, R.; Hernandez-Coronado, M.; Pantoja, O. Quantitative proteomics of the tonoplast reveals a role for glycolytic enzymes in salt tolerance. Plant Cell 2009, 21, 4044–4058. [Google Scholar]
- Geissler, N.; Hussin, S.; Koyro, H.W. Elevated atmospheric CO2 concentration enhances salinity tolerance in Aster tripolium L. Planta 2010, 231, 583–594. [Google Scholar]
- Wang, X.; Yang, P.; Gao, Q.; Liu, X.; Kuang, T.; Shen, S.; He, Y. Proteomic analysis of the response to high-salinity stress in Physcomitrella patens. Planta 2008, 228, 167–177. [Google Scholar]
- Huang, F.; Fulda, S.; Hagermann, M.; Norling, B. Proteomic screening of salt-stress-induced changes in plasma membranes of Synechocystis sp. strain PCC6803. Proteomics 2006, 6, 910–920. [Google Scholar]
- Cheng, Y.; Qi, Y.; Zhu, Q.; Chen, X.; Wang, N.; Zhao, X.; Chen, H.; Cui, X.; Xu, L.; Zhang, W. New changes in the plasma-membrane-associated proteome of rice roots under salt stress. Proteomics 2009, 9, 3100–3114. [Google Scholar]
- Caruso, A.; Morabito, D.; Delmotte, F.; Kahlem, G.; Carpin, S. Dehydrin induction during drought and osmotic stress in Populus. Plant Physiol. Biochem 2002, 40, 1033–1042. [Google Scholar]
- Godoy, J.A.; Lunar, S.; Torres-Schumann, J.; Moreono, J.; Rodrigo, R.M.; Pintor-Toro, J.A. Expression, tissue distribution and subcellular localization of dehydrin TAS14 in salt-stressed tomato plants. Plant Mol. Biol 1994, 26, 1921–1934. [Google Scholar]
- Moons, A.; Bauw, G.; Prinsen, E.; van Montagu, M.; van Der Straeten, D. Molecular and physiological responses to abscisic acid and salts in roots of salt-sensitive and salt-tolerant Indica rice varietites. Plant Physiol 1995, 107, 177–186. [Google Scholar]
- Brini, F.; Hanin, M.; Lumbreras, V.; Irar, S.; Pages, M.; Masmoudi, K. Functional characterization of DHN-5, a dehydrin showing a differential phosphorylation pattern in two Tunisian durum wheat (Triticum durum Desf.) varieties with marked differences in salt and drought tolerance. Plant Sci 2007, 172, 20–28. [Google Scholar]
- Agarwal, P.; Reddy, M.K.; Sopory, S.K.; Agarwal, P.K. Plant Rabs: Characterization, functional diversity, and role in stress tolerance. Plant Mol. Biol. Rep 2009, 27, 417–430. [Google Scholar]
- Pandey, A.; Chakraborty, S.; Datta, A.; Chakraborty, N. Proteomics approach to identify dehydration responsive nuclear proteins from chickpea (Cicer arietinum L.). Mol. Cell. Proteomics 2008, 7, 88–107. [Google Scholar]
- Singh, R.; Green, M.R. Sequence-specific binding of transfer RNA by glyceraldehyde-3-phosphate dehydrogenase. Science 1993, 259, 365–368. [Google Scholar]
- Ronai, Z.; Robinson, R.; Rutberg, S.; Lazarus, P.; Sardana, M. Aldolase DNA interactions in a SEWA cell system. Biochim. Biophys. Acta 1992, 1130, 20–28. [Google Scholar]
- Lee, H.; Guo, Y.; Ohta, M.; Xiong, L.; Stevenson, B.; Zhu, J.K. LOS2, a genetic locus required for cold-responsive gene transcription encodes a bi-functional enolase. EMBO J 2002, 21, 2692–2702. [Google Scholar]
- Moon, H.; Lee, B.; Choi, G.; Shin, S.; Prasad, D.T.; Lee, O.; Kwak, S.S.; Kim, D.H.; Nam, J.; Bahk, J.; et al. NDP kinase 2 interacts with two oxidative stress-acivated MAPKs to regulate cellular redox state and enhances multiple stress tolerance in transgenic plants. Proc. Natl. Acad. Sci. USA 2003, 100, 358–363. [Google Scholar]
- Hashimoto, M.; Kisseleva, L.; Sawa, S.; Furukawa, T.; Komatsu, S.; Koshiba, T. A novel rice PR10 protein, RSOsPR10, specifically induced in roots by biotic and abiotic stress, possibly via the jasmonic acid signalling pathway. Plant Cell Physiol 2004, 45, 550–559. [Google Scholar]
- Park, C.J.; Kim, K.J.; Shin, R.; Park, J.M.; Shin, Y.C.; Paek, K.H. Pathogenesis-related protein 10 isolated from hot pepper functions as a ribonuclease in an antiviral pathway. Plant J 2004, 37, 186–198. [Google Scholar]
- Mogensen, J.E.; Wimmer, R.; Larsen, J.N.; Spangfort, M.D. The major birch allergen, Bet v 1, shows affinity for a broad spectrum of physiological ligands. J. Biol. Chem 2002, 277, 684–692. [Google Scholar]
- Zhang, W.; Peumans, W.J.; Barre, A.; Astoul, C.H.; Rovira, P.; Rougé, P.; Proost, P.; Truffa-Bachi, P.; Jalali, A.A.H.; van Damme, E.J.M. Isolation and characterization of a jacalin-related mannose-binding lectin from salt-stressed rice (Oryza sativa) plants. Planta 2000, 210, 970–978. [Google Scholar]
- Carpita, N.; Sabularse, D.; Montezinos, D.; Delmer, D.P. Determination of the pore size of cell walls of living plants. Science 1979, 205, 1144–1147. [Google Scholar]
- Battaglia, M.; Olvera-Carillo, Y.; Garciarrubio, A.; Covarrubias, A.A. The enigmatic LEA proteins and other hydrophilins. Plant Physiol 2008, 48, 6–24. [Google Scholar]
- Hundertmark, M.; Hincha, D.K. LEA (late embryogenesis abundant) proteins and their encoding egenes in Arabidopsis thaliana. BMC Genomics 2008, 9, 118–139. [Google Scholar]
- Batelli, G.; Verslues, P.E.; Agius, F.; Qiu, Q.; Fujii, H.; Pan, S.Q.; Schumaker, K.; Grillo, S.; Zhu, J.K. SOS2 promotes salt tolerance in part by interacting with the vacuolar H+-ATPase and upregulating its transport activity. Mol. Cell. Biol 2007, 27, 7781–7790. [Google Scholar]
- Garcia-Olmedo, F.; Molina, A.; Segura, A.; Moreno, M. The defensive role of nonspecific lipid-transfer proteins in plants. Trends Microbiol 1995, 3, 72–74. [Google Scholar]
- Bugos, R.C.; Hieber, A.D.; Yamamoto, H.Y. Xanthophyll cycle enzymes are members of the lipocalin family, the first identified from plants. J. Biol. Chem 1998, 273, 15321–15324. [Google Scholar]
- Vítámvás, P.; Prášil, I.T.; Kosová, K.; Planchon, S.; Renaut, J. Analysis of proteome and frost tolerance in chromosome 5A and 5B reciprocal substitution lines between two winter wheats during long-term cold acclimation. Proteomics 2012, 12, 68–85. [Google Scholar]
- Vítámvás, P.; Kosová, K.; Prášilová, P.; Prášil, I.T. Accumulation of WCS120 protein in wheat cultivars grown at 9 °C or 17 °C in relation to their winter survival. Plant Breeding 2010, 129, 611–616. [Google Scholar]

| Level of study | Salinity adaptation | Ref. |
|---|---|---|
| Genomic | Gene duplication (increased gene copy number) and promoter adaptation of several salinity-responsive genes (transcription factors: Myb24, ATPase AVP1, ion transporters: SOS1, NHX; ABC) | [18] |
| Transcriptomic | Enhanced constitutive expression of several salinity-responsive transcripts (SOS1, SOD, P5CS, GS, INPS, cytochrome P450, heat shock protein ) Hsc70-3, antifugal protein PDF1.2) | [19–21] |
| Proteomic | Enhanced abundance of several stress- and defence-related proteins (LEA, redox, PR), ion transporters, protective proteins involved in activation of photosynthesis (D2 protein) and protein biosynthesis, activation of biosynthesis of protective compounds (lignin) | [22–27] |
| Metabolomic | Alterations in carbohydrate metabolism—activation of catabolism (glycolysis, Krebs cycle, starch degradation), enhanced biosynthesis of organic osmolytes, phenolic compounds, lignin) | [20,22, 28,29] |
| Osmotic effect of salinity | ||
| Functional (physiological) level | - osmotic adjustment (accumulation of low-molecular organic osmolytes and proteins—LEA proteins) | [21,30–33] |
| - adjustment to mechanical stress (increased cell wall lignification, accumulation and oligomerization of several coat proteins in plasmamembrane of Dunaliella salina) | [27,28, 32,34] | |
| Ion-related effects of salinity | ||
| - salt ion exclusion (increased abundance and activity of plasma membrane ion transporters (SOS1), increased lignification of xylem vessels (long-distance transport of excluded salt ions via transpiration stream— Salicornia europaea) | [27] | |
| - salt ion intracellular compartmentation (salt import into vacuoles— an enhanced abundance of tonoplast ion transporters NHX, support of ion transport - H+-ATPase and FBP aldolase activity) | [24,35] | |
| Protein functional group | Glycophytes | Halophytes | ||
|---|---|---|---|---|
| Increased protein | Decreased protein | Increased protein | Decreased protein | |
| Signalling | Annexin [22], calmodulin, OsRPK1 [91], calreticulin [51,84] MAPK [56,76], large GTP binding protein; β subunit of heterotrimeric G protein, OsRac2 [22,23,51,52,59], 14-3-3 [23,56,79,89] | Ras GTPase, 14-3-3 [52] | Annexin [22,27], β subunit of heterotrimeric G protein [23], phototropin [89], alpha-SNAP [28] | CBL-interacting protein kinase [27], 14-3-3 [28] |
| Gene expression regulation, cell growth and division | NAC-α, HB1B, OSAP1 RNA helicase [30,62,65,80] | NAC-α, polyA-binding protein [51] | CRT/DREB [16], Cell division protein fts homolog [89] | SKP1-like protein [89], MorR homolog [90], TF APFI [27] |
| Protein biosynthesis and degradation | eIF, eEF, eIF5A3, ribosomal proteins L12, L31, S29 [22], Proteasome 20S [60], 26S [70], GS [62] | eIF-4E2, eEF [52], L10, L12, S3a, S12 [84], L29, S5 [22], GS [81] | eIF3A, S7, S24, S15a [22] Alpha type 6, Beta type 1, GS [22,27] | 20S, Proteasome subunit alpha type 2, 4, 6 [28] |
| Protein folding | DnaK chaperone, HSP70, small HSP [80], RubisCO binding protein subunit β (CPN60-β) [30,65,66] | HSP90 [57] | Small HSP [88], HSP70 [89], CPN60-α [28], HSP60 [27] | HSP90 [28] |
| Photosynthesis | 23 kDa (PsbP), ferredoxin-NADPH reductase [78], OEE2 [65,66], RubisCO activase [61,65,92], TPI, GAPDH, Glucose-6-P dehydrogenase [56,58,73] | LHC, PC, OEE1, OEE2, RubisCO LSU and SSU, RubisCO activase [22,56,71,73,81], carbonic anhydrase; GAPDH, SBP, PGK, PRK, TK [61,71,92] | LHC, OEE2, RubisCO LSU and SSU, RubisCO activase [16,89], D2 protein PSII [26], CP24, CP47, PSI subunit IV [16], carbonic anhydrase; SBP, PGK [29,87] | LHC, OEE1, RubisCO LSU and SSU, RubisCO activase [28], PsbO [90], SBP [26,86] |
| Respiratory pathway and sucrose matabolism | FBP aldolase, GAPDH, TPI, ENO [61], Succinyl-CoA ligase β subunit, MDH (NAD), cytochrome c oxidase subunit 6b-1 [22], ATP synthase CF1β,δ,ɛ ; ADK, NDPK1 [22,63,74,78], ADH [73] | ATP synthase CF1α,β-3, mtATP β-3 [77] | FBP aldolase [35], ATP synthase CF1β,ɛ, ADK, NDPK1 [86], Fructokinase-like protein [89], GAPDH, MDH [27] | ATP synthase CF1α,β [89], MDH [27], sucrose synthase [28] |
| Redox metabolism | GST, APX, Cu/Zn-SOD, Mn-SOD, PRX, Trx h, protein disulfide isomerase (practically all papers) | APX, MDAR [51] | GST, APX, Cu/Zn–SOD, 2-Cys PRX, MDAR (practically all papers) | APX chain A [86], MDAR [27], GST-I, Thioredoxin [28] |
| Metal binding proteins | Ferritin, IDI2, IDS2, IDS3 [68], magnesium chelatase [66], metallothionein [70] | Ferritin, Voltage-dependent anion channel protein [27], transferrin-like [31], FutA1, FutA2 [90] | Ferritin [27] | |
| Defence-related proteins | PR5, PR10 [22,25,67,72,74,75,82,84,92], β-1,3- glucanase, cyanogenic β-glucosidase dhurrinase [77], (1–3)-b-Glucanase GV [68], Jacalin lectin, mannose-binding RICE lectin [59], lectin-like [56,76], Germin-like [51,65], SAMS [68] | Lectin-like [72], 23 kDa jasmonate-induced protein, F23N19.10 stressinducible protein [68], SAMS [73], TSI-1 [34] | NBS-LRR disease resistance [27], SAMS [27,28], Lipocalin-like protein [89] | Disease resistance protein [27] |
| Transport | PM H+- ATPase, V-ATPase subunit β (VHA-B), PPase, NHX [22–24,51,57,82], ABC [23] | V-ATPase subunit β (VHA-B) [34] | HKT, V-ATPase subunit β (VHA-B), PPase, NHX [22,23], ABC [86,90] | V-ATPase subunit β [28], Putative porin [90] |
| Cytoskeleton-related | Profilin [65], β-tubulin [22,51] | Tubulin, actin [83] | Profilin [26] | |
| Structural proteins | Myosin VIII, remorin [91], OSR40c1, β-d-glucan exohydrolase, GRP [57,60,82] | XET [84] | PM coat proteins [32], cellulose synthase [16] | |
| Lipid metabolism | 3-ketoacyl-acyl carrier protein synthase I, phospholipase/carboxyesterase family protein, dihydrolipoamide dehydrogenase, ENR [60], UDP-sulfoquinovose synthase [57] | Monogalactosyl diacylglycerol synthase [78] | Long-chain-fatty-acid-CoA ligase [22] | Glycerophosphodiesterase [22] |
| Phytohormone metabolism | JA biosynthesis (AOC, LOX) [22,51] | ABA biosynthesis (NCED) [19], GA biosynthesis (DWARF3) [23,64] | ||
| Lignin biosynthesis | COMT, CCOMT [34,67,70] | COMT, CCOMT [73] | COMT, CCOMT [27,28] | |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kosová, K.; Prášil, I.T.; Vítámvás, P. Protein Contribution to Plant Salinity Response and Tolerance Acquisition. Int. J. Mol. Sci. 2013, 14, 6757-6789. https://doi.org/10.3390/ijms14046757
Kosová K, Prášil IT, Vítámvás P. Protein Contribution to Plant Salinity Response and Tolerance Acquisition. International Journal of Molecular Sciences. 2013; 14(4):6757-6789. https://doi.org/10.3390/ijms14046757
Chicago/Turabian StyleKosová, Klára, Ilja T. Prášil, and Pavel Vítámvás. 2013. "Protein Contribution to Plant Salinity Response and Tolerance Acquisition" International Journal of Molecular Sciences 14, no. 4: 6757-6789. https://doi.org/10.3390/ijms14046757
APA StyleKosová, K., Prášil, I. T., & Vítámvás, P. (2013). Protein Contribution to Plant Salinity Response and Tolerance Acquisition. International Journal of Molecular Sciences, 14(4), 6757-6789. https://doi.org/10.3390/ijms14046757




