Aggregation of p-Sulfonatocalixarene-Based Amphiphiles and Supra-Amphiphiles
Abstract
:1. Introduction
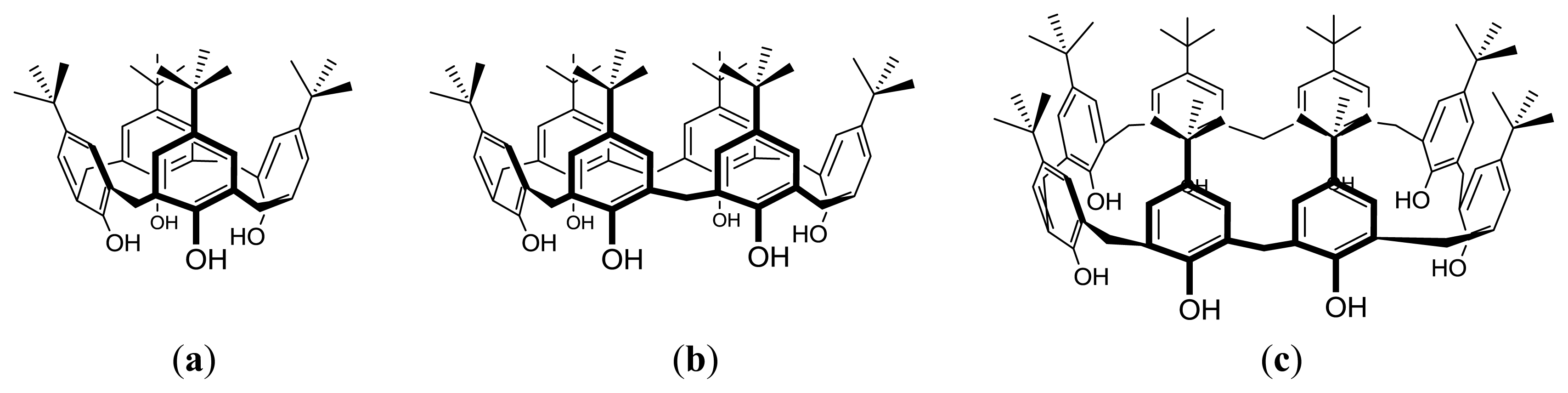
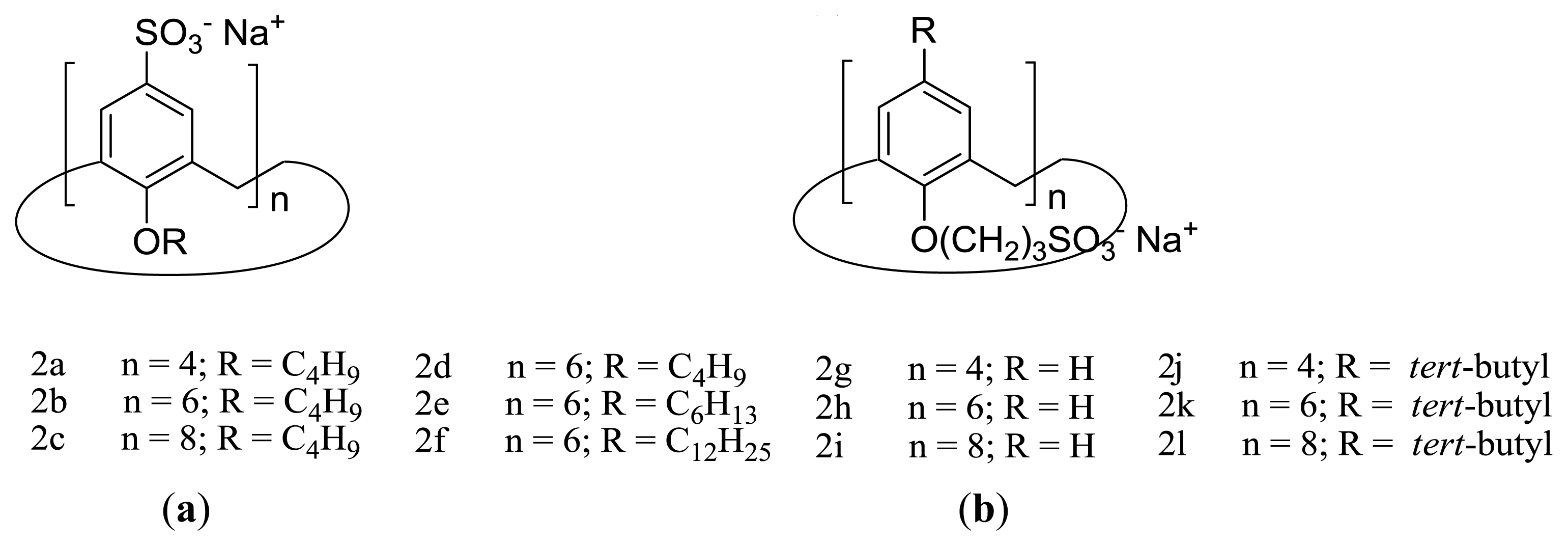
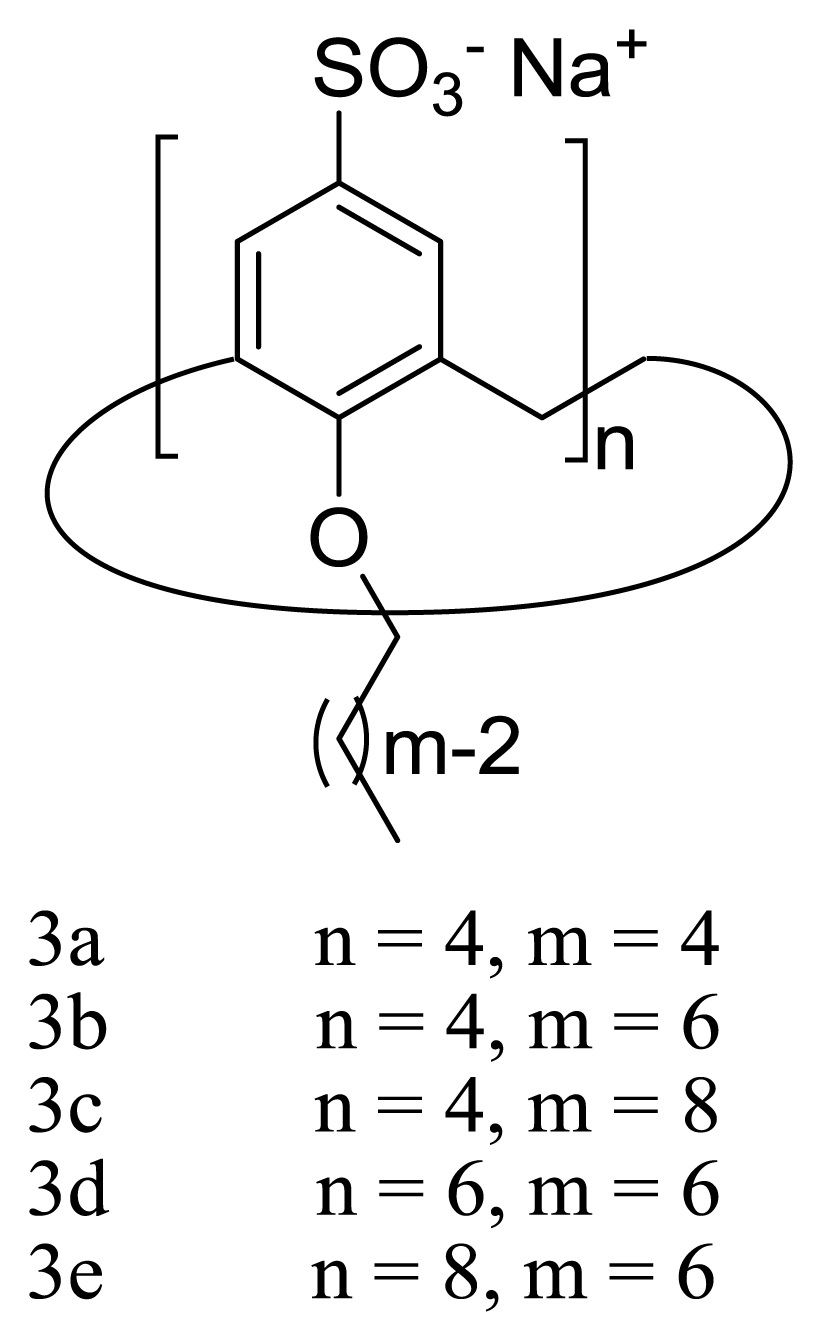
2. Amphiphilic Sulfonatocalixarenes: Structure-Aggregation Relationships
2.1. Earlier Work
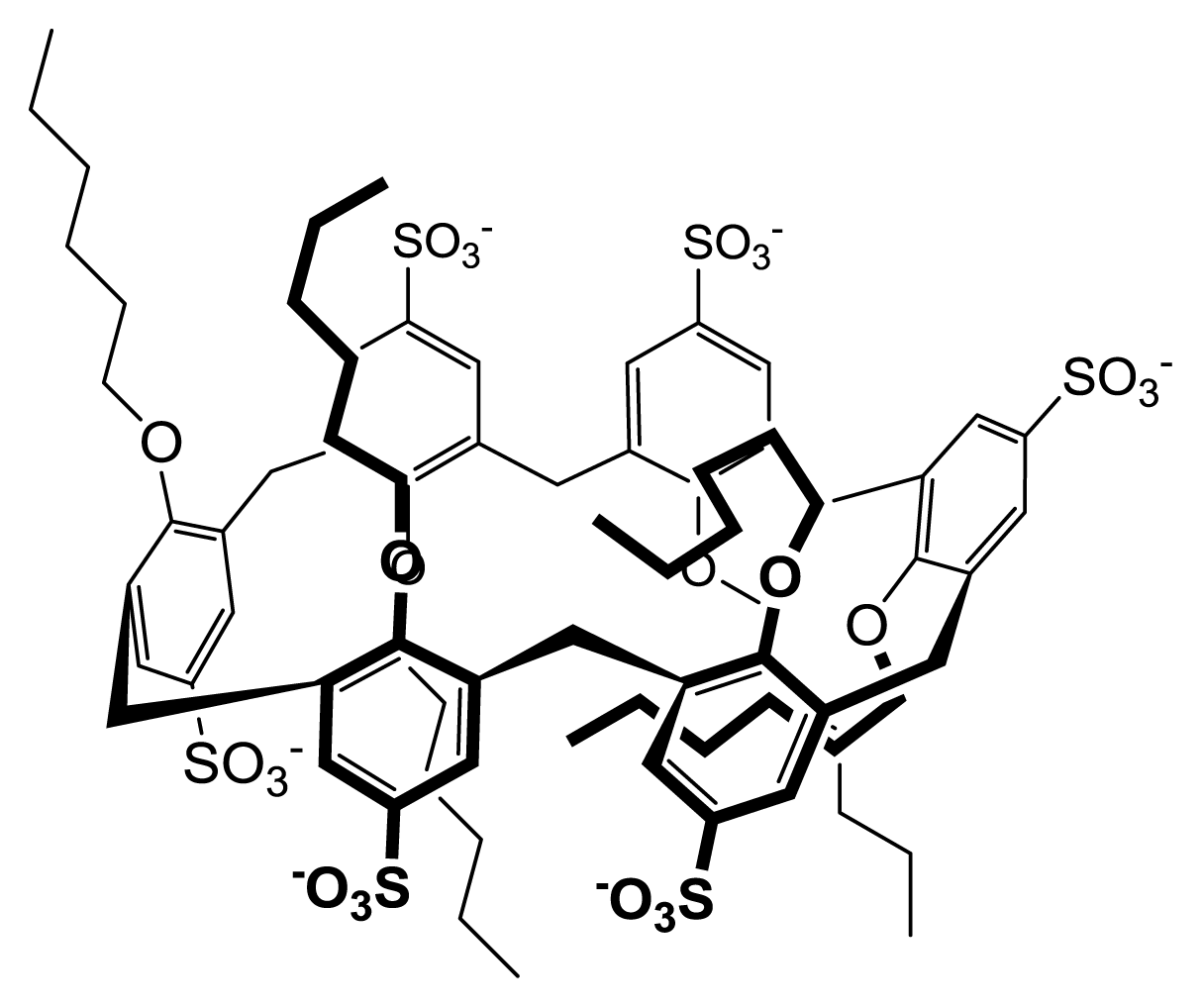
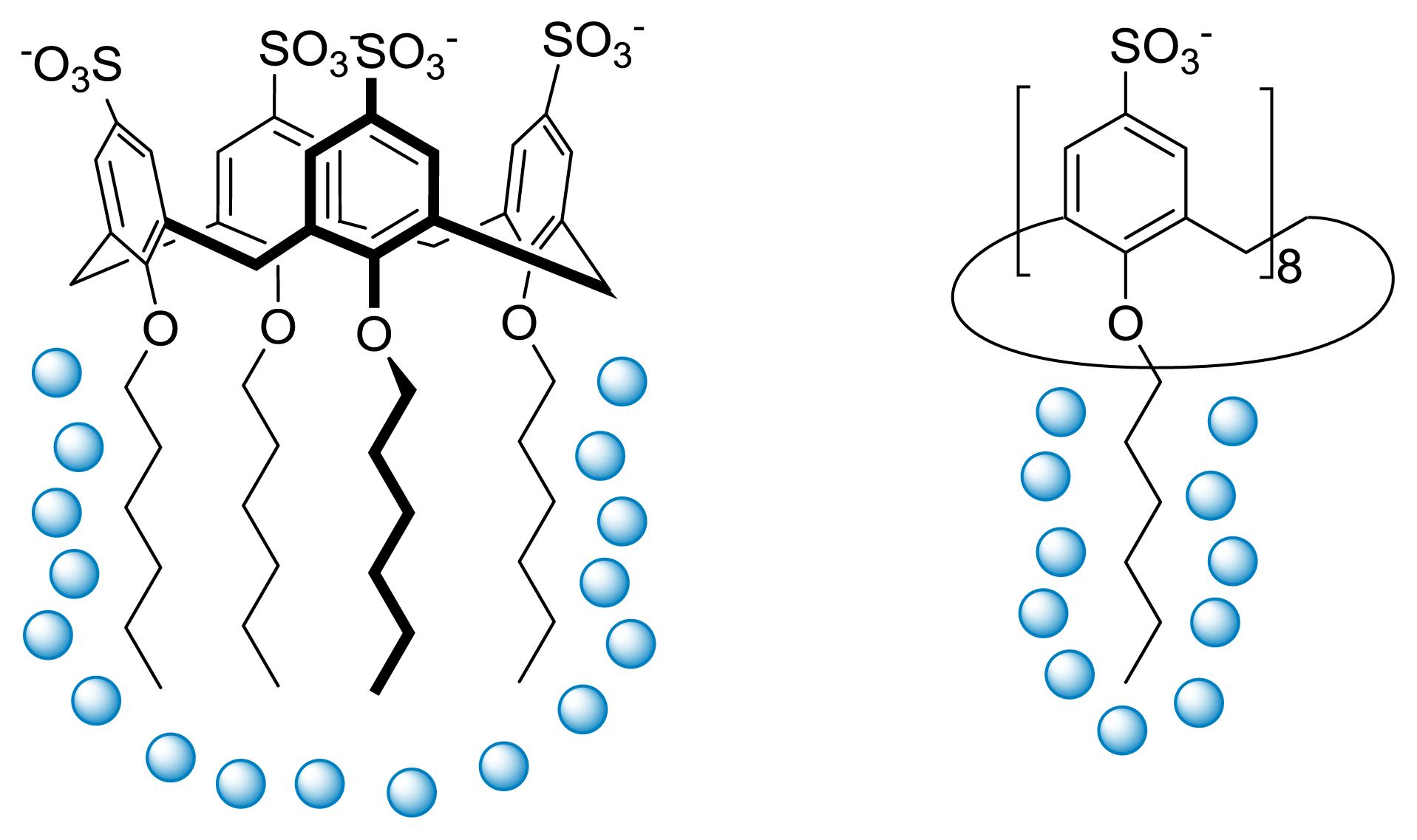
2.2. Conformational Reorganization upon Micellization
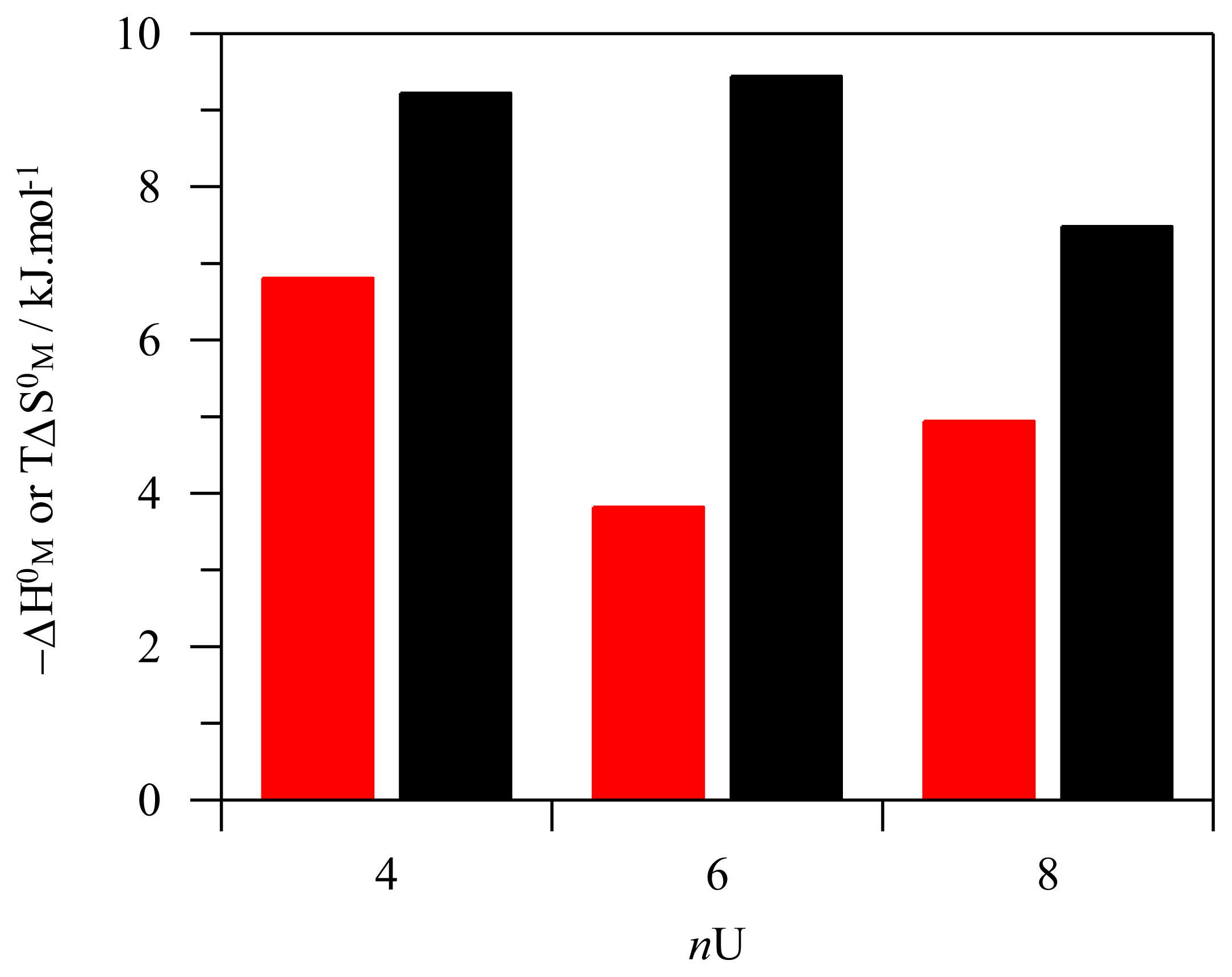
2.3. Thermodynamics of Micellization
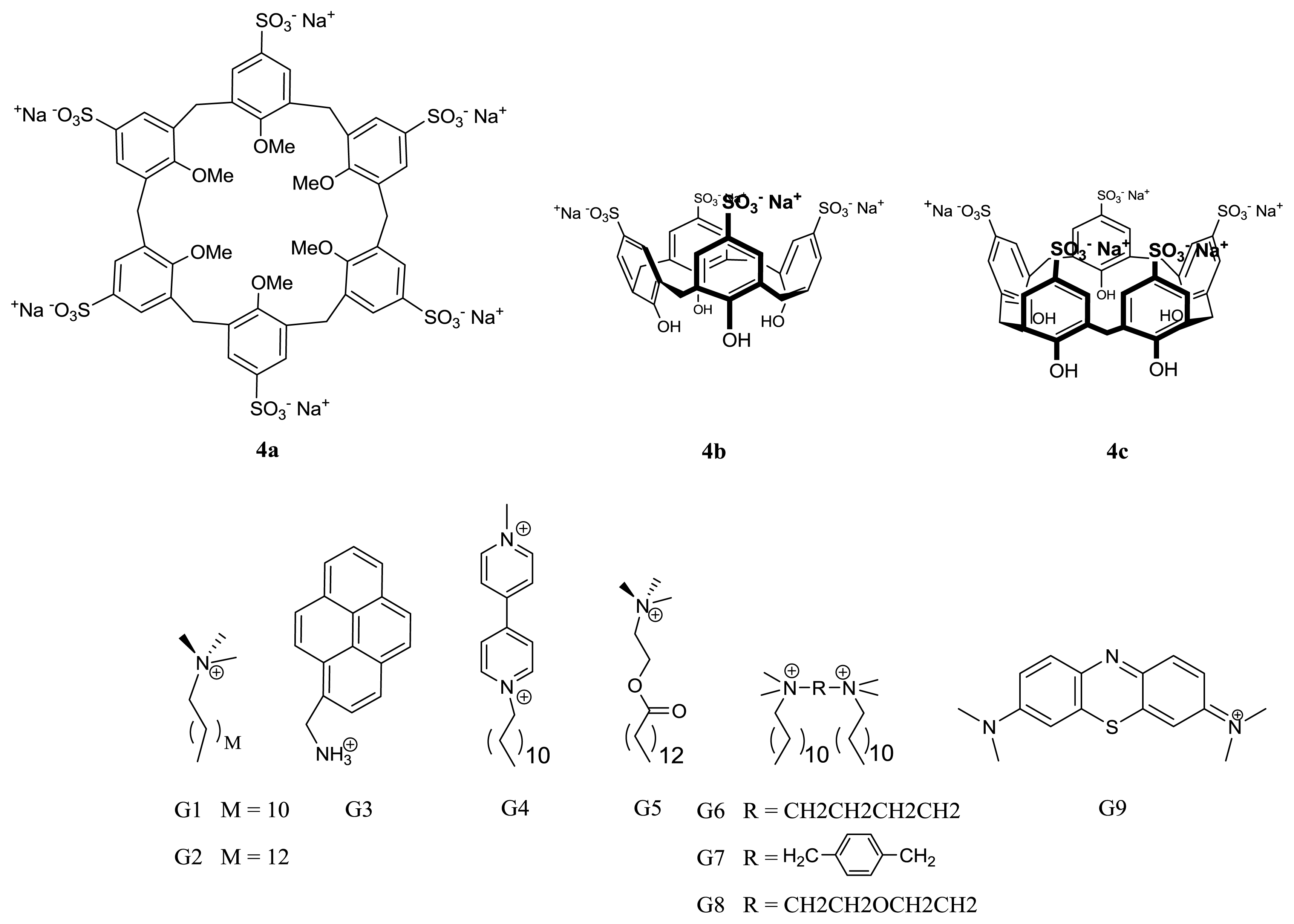
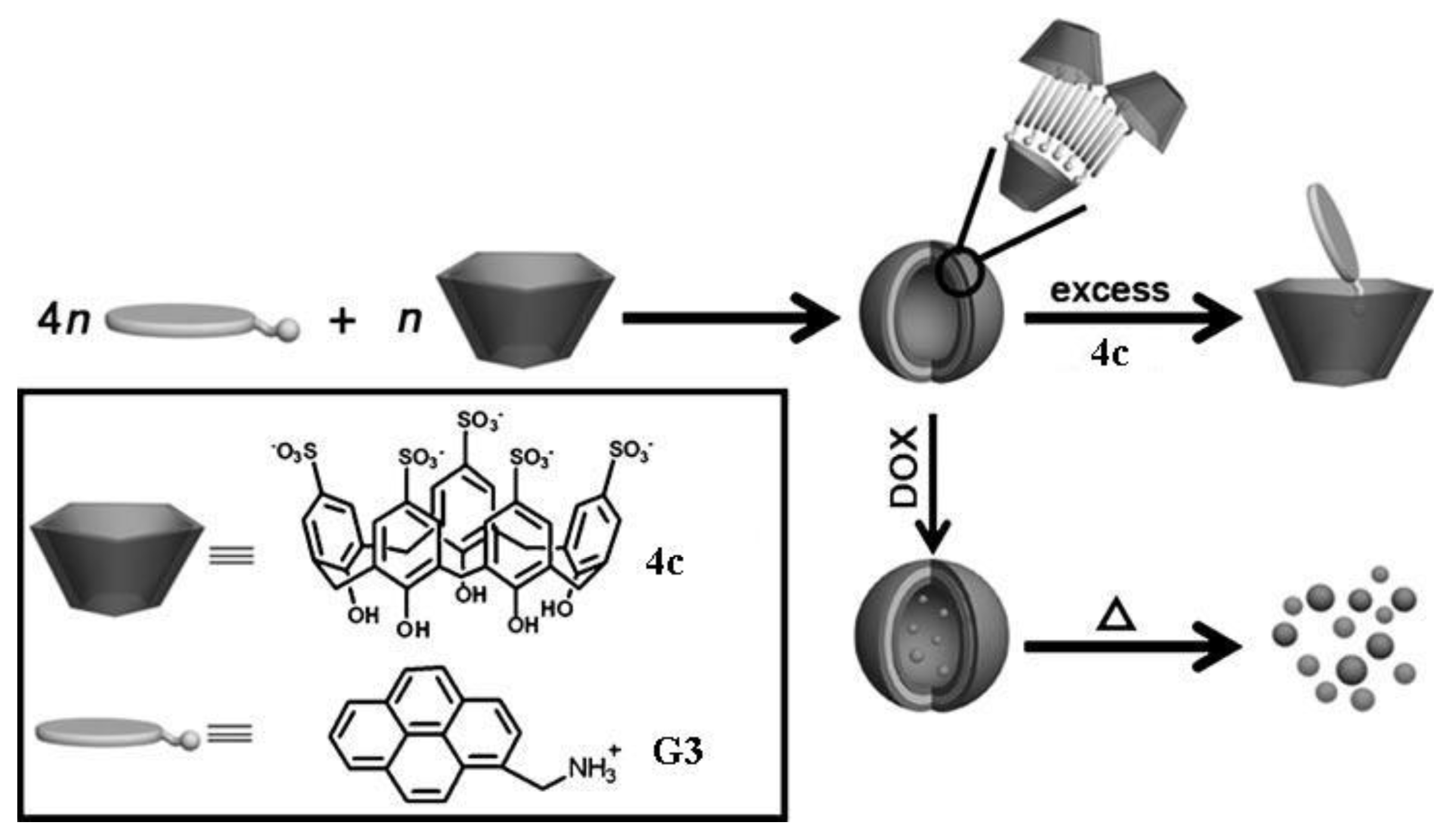
3. p-Sulfonatocalixarene-Based Supramolecular Amphiphiles
4. Conclusions and Outlook
Acknowledgments
Conflict of Interest
References
- Ikeda, A.; Shinkai, S. Novel cavity design using calix[n]arene skeletons: Toward molecular recognition and metal binding. Chem. Rev 1997, 97, 1713–1734. [Google Scholar]
- Böhmer, V. Calixarenes, macrocycles with (almost) unlimited possibilities. Angew. Chem. Int. Ed 1995, 34, 713–745. [Google Scholar]
- Stewart, D.R.; Gutsche, C.D. Isolation, characterization, and conformational characteristics of p-tert-butylcalix[9–20]arenes. J. Am. Chem. Soc 1999, 121, 4136–4146. [Google Scholar]
- Arduini, A.; Ciesa, F.; Fragassi, M.; Pochini, A.; Secchi, A. Selective synthesis of two constitutionally isomeric oriented calix[6]arene-based rotaxanes. Angew. Chem. Int. Ed 2005, 44, 278–281. [Google Scholar]
- Leontiev, A.V.; Jemmett, C.A.; Beer, P.D. Anion recognition and cation-induced molecular motion in a heteroditopic[2]rotaxane. Chem. Eur. J 2011, 17, 816–825. [Google Scholar]
- Pierro, T.; Gaeta, C.; Talotta, C.; Casapullo, A.; Neri, P. Fixed or invertible calixarene-based directional shuttles. Org. Lett 2011, 13, 2650–2653. [Google Scholar]
- McConnell, A.J.; Serpell, C.J.; Thompson, A.L.; Allan, D.R.; Beer, P.D. Calix[4]arene-based rotaxane host systems for anion recognition. Chem. Eur. J 2010, 16, 1256–1264. [Google Scholar]
- Bogdan, A.; Rudzevich, Y.; Vysotsky, M.O.; Böhmer, V. Topologically novel multiple rotaxanes and catenanes based on tetraurea calix[4]arenes. Chem. Commun. 2006, 2941–2952. [Google Scholar]
- Talotta, C.; Gaeta, C.; Neri, P. Stereoprogrammed direct synthesis of calixarene-based[3]rotaxanes. Org. Lett 2012, 14, 3104–3117. [Google Scholar]
- Wei, A. Calixarene-encapsulated nanoparticles: Self-assembly into functional nanomaterials. Chem. Commun. 2006, 1581–1591. [Google Scholar]
- Kim, H.J.; Lee, M.H.; Mutihac, L.; Vicens, J.; Kim, J.S. Host-guest sensing by calixarenes on the surfaces. Chem. Soc. Rev 2012, 41, 1173–1190. [Google Scholar]
- Dsouza, R.N.; Hennig, A.; Nau, W.M. Supramolecular tandem enzyme assays. Chem. Eur. J 2012, 18, 3444–3459. [Google Scholar]
- Ghosh, I.; Nau, W.M. The strategic use of supramolecular pK(a) shifts to enhance the bioavailability of drugs. Adv. Drug Deliv. Rev 2012, 64, 764–783. [Google Scholar]
- Guo, D.; Liu, Y. Calixarene-based supramolecular polymerization in solution. Chem. Soc. Rev 2012, 41, 5907–5921. [Google Scholar]
- Helttunen, K.; Shahgaldian, P. Self-assembly of amphiphilic calixarenes and resorcinarenes in water. New J. Chem 2010, 34, 2704–2714. [Google Scholar]
- Gutsche, C.D.; Bauer, L.J. Calixarenes. 13. The conformational properties of calix[4]arenes, calix[6]arenes, calix[8]arenes, and oxacalixarenes. J. Am. Chem. Soc 1985, 107, 6052–6059. [Google Scholar]
- Guo, D.-S.; Wang, K.; Liu, Y. Selective binding behaviors of p-sulfonatocalixarenes in aqueous solution. J. Incl. Phenom. Macrocycl. Chem 2008, 62, 1–21. [Google Scholar]
- Perret, F.; Coleman, A.W. Biochemistry of anionic calix[n]arenes. Chem. Commun 2011, 47, 7303–7319. [Google Scholar]
- Perret, F.; Lazar, A.N.; Coleman, A.W. Biochemistry of the para-sulfonato-calix[n]arenes. Chem. Commun. 2006, 2425–2438. [Google Scholar]
- Komiyama, M.; Isaka, K.; Shinkai, S. Water-soluble calixarene as the first man-made catalyst for regioselective cleavage of ribonucleoside 2′,3′-cyclic phosphate. Chem. Lett. 1991, 937–940. [Google Scholar]
- Shirakawa, S.; Shimizu, S. Dehydrative amination of alcohols in water using a water-soluble calix[4]resorcinarene sulfonic acid. Synlett 2008, 10, 1539–1542. [Google Scholar]
- Karakhanov, E. Substrate selectivity in byphasic wacker-oxidation of alkenes in the presence of water-soluble calixarenes. J. Mol. Catal. A Chem 2002, 184, 11–17. [Google Scholar]
- Liu, Y.-L.; Liu, L.; Wang, Y.-L.; Han, Y.-C.; Wang, D.; Chen, Y.-J. Calix[n]arene sulfonic acids bearing pendant aliphatic chains as recyclable surfactant-type Brønsted acid catalysts for allylic alkylation with allyl alcohols in water. Green Chem 2008, 10, 635–640. [Google Scholar]
- Shimizu, S.; Shimada, N.; Sasaki, Y. Mannich-type reactions in water using anionic water-soluble calixarenes as recoverable and reusable catalysts. Green Chem 2006, 8, 608–614. [Google Scholar]
- Danylyuk, O.; Suwinska, K. Solid-state interactions of calixarenes with biorelevant molecules. Chem. Commun. 2009, 5799–5813. [Google Scholar]
- Menger, F.M.; Littau, C.A. Gemini surfactants: A new class of self-assembling molecules. J. Am. Chem. Soc 1993, 115, 10083–10090. [Google Scholar]
- Menger, F.; Keiper, J. Gemini Surfactants. Angew. Chem. Int. Ed 2000, 39, 1906–1920. [Google Scholar]
- Zana, R. Dimeric and oligomeric surfactants. Behavior at interfaces and in aqueous solution: A review. Adv. Colloid Interface Sci 2002, 97, 205–253. [Google Scholar]
- Zana, R.; Levy, H.; Papoutsi, D.; Beinert, G. Micellization of two triquaternary ammonium surfactants in aqueous solution. Langmuir 1995, 11, 3694–3698. [Google Scholar]
- Esumi, K.; Taguma, K.; Koide, Y. Aqueous properties of multichain quaternary cationic surfactants. Langmuir 1996, 12, 4039–4041. [Google Scholar]
- Menger, F.M.; Migulin, V.A. Synthesis and properties of multiarmed geminis. J. Org. Chem 1999, 64, 8916–8921. [Google Scholar]
- Yoshimura, T.; Yoshida, H.; Ohno, A.; Esumi, K. Physicochemical properties of quaternary ammonium bromide-type trimeric surfactants. J. Colloid Interface Sci 2003, 267, 167–172. [Google Scholar]
- In, M.; Bec, V.; Aguerre-Chariol, O.; Zana, R. Quaternary ammonium bromide surfactant oligomers in aqueous solution: Self-association and microstructure. Langmuir 2000, 16, 141–148. [Google Scholar]
- Laschewsky, A.; Wattebled, L.; Arotçaréna, M.; Habib-Jiwan, J.-L.; Rakotoaly, R.H. Synthesis and properties of cationic oligomeric surfactants. Langmuir 2005, 21, 7170–7179. [Google Scholar]
- Murguía, M.C.; Cabrera, M.I.; Guastavino, J.F.; Grau, R.J. New oligomeric surfactants with multiple-ring spacers: Synthesis and tensioactive properties. Colloids Surf. A Physicochem. Eng. Asp 2005, 262, 1–7. [Google Scholar]
- Hou, Y.; Han, Y.; Deng, M.; Xiang, J.; Wang, Y. Aggregation behavior of a tetrameric cationic surfactant in aqueous solution. Langmuir 2010, 26, 28–33. [Google Scholar]
- Zhang, X.; Wang, C. Supramolecular amphiphiles. Chem. Soc. Rev 2011, 40, 94–101. [Google Scholar]
- Shinkai, S.; Mori, S.; Tsubaki, T.; Sone, T.; Manabe, O. New water-soluble host molecules derived from calix[6]arene. Tetrahedron Lett 1984, 25, 5315–5318. [Google Scholar]
- Shinkai, S.; Mori, S.; Koreishi, H.; Tsubaki, T.; Manabe, O. Hexasulfonated calix[6]arene derivatives: A new class of catalysts, surfactants, and host molecules. J. Am. Chem. Soc 1986, 108, 2409–2416. [Google Scholar]
- Matsuoka, H.; Tsurumi, M.; Ise, N. Small-angle X-ray scattering study of aqueous solutions of sulfonated calix[6]arene derivatives. Phys. Rev. B 1988, 38, 6279–6286. [Google Scholar]
- Shinkai, S.; Araki, K.; Manabe, O. Does the calixarene cavity recognise the size of guest molecules? On the “hole-size selectivity” in water-soluble calixarenes. J. Chem. Soc. Chem. Commun. 1988, 187–189. [Google Scholar]
- Shinkai, S.; Arimura, T.; Araki, K.; Kawabata, H.; Satoh, H.; Tsubaki, T.; Manabe, O.; Sunamoto, J. Syntheses and aggregation properties of new water-soluble calixarenes. J. Chem. Soc. Perkin Trans. 1 1989, 2039–2045. [Google Scholar]
- Shinkai, S.; Koreishi, H.; Mori, S.; Sone, T.; Manabe, O. Host-guest-type acid catalysts derived from calix[6]arene. Chem. Lett. 1985, 1033–1036. [Google Scholar]
- Shinkai, S.; Mori, S.; Araki, K.; Manabe, O. Influence of hexasulfonated calix[6]arenes on the reactivities of arenediazonium ions in an aqueous system. Bull. Chem. Soc. Jpn 1987, 60, 3679–3685. [Google Scholar]
- Shinkai, S.; Mori, S.; Arimura, T.; Manabe, O. Stabilization of arenediazonium ions by complexation with hexasulphonated calix[6]arenes in an aqueous system. J. Chem. Soc. Chem. Commun. 1987, 238–240. [Google Scholar]
- Shinkai, S.; Kawabata, H.; Arimura, T.; Matsuda, T.; Satoh, H.; Manabe, O. New water-soluble calixarenes bearing sulphonate groups on the “lower rim”: The relation between calixarene shape and binding ability. J. Chem. Soc. Perkin Trans. 1 1989, 1073–1074. [Google Scholar]
- Bernardo, A.R.; Lu, T.; Cordova, E.; Zhang, L.; Gokel, G.W.; Kaifer, A.E. Host-guest complexation at the electrode/solution interface: The inclusion of an amphiphilic viologen guest by an amphiphilic calix[6]arene host. Chem. Commun. 1994, 529–530. [Google Scholar]
- Zhang, L.; Godínez, L.A.; Lu, T.; Gokel, G.W.; Kaifer, A.E. Molecular recognition at an interface: Binding of monolayer-anchored ferrocenyl groups by an amphiphilic calixarene host. Angew. Chem. Int. Ed 1995, 34, 235–237. [Google Scholar]
- Tao, W.; Barra, M. Thermodynamic study of p-sulfonated calixarene complexes in aqueous solution. J. Chem. Soc. Perkin Trans. 1 1998, 1957–1960. [Google Scholar]
- Liu, Y.; Han, B.-H.; Chen, Y.-T. Inclusion complexation of acridine red dye by calixarenesulfonates and cyclodextrins: Opposite fluorescent behavior. J. Org. Chem 2000, 65, 6227–6230. [Google Scholar]
- Tian, H.; Chen, Y.; Wang, D.; Zeng, C.; Li, C. Calix[6]arene derivatives bearing sulfonate and alkyl groups as surfactants in Sc(OTf)3-catalyzed Mukaiyama aldol reactions in water. Tetrahedron Lett 2000, 41, 2529–2532. [Google Scholar]
- Zhang, Y.; Cao, W. Self-assembly of small molecules: An approach combining electrostatic self-assembly technology with host–guest chemistry. New J. Chem 2001, 25, 483–486. [Google Scholar]
- Liu, Y.; Han, B.-H.; Chen, Y.-T. Molecular recognition and complexation thermodynamics of dye guest molecules by modified cyclodextrins and calixarenesulfonates. J. Phys. Chem. B 2002, 106, 4678–4687. [Google Scholar]
- Tian, H.-Y.; Li, H.-J.; Chen, Y.-J.; Wang, D.; Li, C.-J. Development of highly effective encapsulating surfactants for mukaiyama aldol reactions in water. Ind. Eng. Chem. Res 2002, 41, 4523–4527. [Google Scholar]
- Jin, T.; Fujii, F.; Sakata, H.; Tamura, M.; Kinjo, M. Amphiphilic p-sulfonatocalix[4]arene-coated CdSe/ZnS quantum dots for the optical detection of the neurotransmitter acetylcholine. Chem. Commun. 2005, 4300–4302. [Google Scholar]
- Lodi, A.; Caselli, M.; Casnati, A.; Momicchioli, F.; Sansone, F.; Vanossi, D.; Ponterini, G. Solvent-dependent host–guest complexation of two homologous merocyanines by a water-soluble calix[8]arene: Spectroscopic analysis and structural calculations. J. Mol. Struct 2007, 846, 49–54. [Google Scholar]
- Lobach, A.S.; Ryzhkina, I.S.; Spitsina, N.G.; Obraztsova, E.D. Solubilization of single-wall carbon nanotubes in aqueous solutions of calixarenes. Phys. Status Solidi B 2007, 244, 4030–4034. [Google Scholar]
- Cui, J.; Uzunova, V.D.; Guo, D.-S.; Wang, K.; Nau, W.M.; Liu, Y. Effect of lower-RIM alkylation of p-sulfonatocalix[4]arene on the thermodynamics of host–guest complexation. Eur. J. Org. Chem. 2010, 1704–1710. [Google Scholar]
- Basilio, N; García-Río, L.; Martín-Pastor, M. NMR evidence of slow monomer–micelle exchange in a calixarene-based surfactant. J. Phys. Chem. B 2010, 114, 4816–4820. [Google Scholar]
- Basílio, N.; Garcia-Rio, L. Calixarene-based surfactants: Conformational-dependent solvation shells for the alkyl chains. ChemPhysChem 2012, 13, 2368–2376. [Google Scholar]
- Basílio, N.; Garcia-Rio, L.; Martín-Pastor, M. Calixarene-based surfactants: Evidence of structural reorganization upon micellization. Langmuir 2012, 28, 2404–2414. [Google Scholar]
- Sansone, F.; Dudic, M.; Donofrio, G.; Rivetti, C.; Baldini, L.; Casnati, A.; Cellai, S.; Ungaro, R. DNA condensation and cell transfection properties of guanidinium calixarenes: Dependence on macrocycle lipophilicity, size, and conformation. J. Am. Chem. Soc 2006, 128, 14528–14536. [Google Scholar]
- Consoli, G.M.; Granata, G.; Lo Nigro, R.; Malandrino, G.; Geraci, C. Spontaneous self-assembly of water-soluble nucleotide-calixarene conjugates in small micelles coalescing to microspheres. Langmuir 2008, 24, 6194–6200. [Google Scholar]
- Zana, R. Critical micellization concentration of surfactants in aqueous solution and free energy of micellization. Langmuir 1996, 12, 1208–1211. [Google Scholar]
- Basilio, N.; García-Río, L. Sulfonated Calix[6]arene host–guest complexes induce surfactant self-assembly. Chem. Eur. J 2009, 15, 9315–9319. [Google Scholar]
- Basilio, N.; Martín-Pastor, M.; García-Río, L. Insights into the structure of the supramolecular amphiphile formed by a sulfonated calix[6]arene and alkyltrimethylammonium surfactants. Langmuir 2012, 28, 6561–6568. [Google Scholar]
- Francisco, V.; Basilio, N.; Garcia-Rio, L.; Leis, J.R.; Maques, E.F.; Vázquez-Vázquez, C. Novel catanionic vesicles from calixarene and single-chain surfactant. Chem. Commun 2010, 46, 6551–6553. [Google Scholar]
- Mitchell, D.J.; Ninham, B.W.; Israelachvili, J.N. Theory of self-assembly of hydrocarbon amphiphiles into micelles and bilayers. J. Chem. Soc. Dalton Trans. 2 1976, 72, 1525–1568. [Google Scholar]
- Israelachvili, J.N.; Ninham, B.W.; Mitchell, D.J. Theory of the self-assembly of lipid bilayers and vesicles. Biochim. Biophys. Acta 1977, 470, 185–201. [Google Scholar]
- Nagarajan, R. Molecular packing parameter and surfactant self-assembly: The neglected role of the surfactant tail. Langmuir 2002, 18, 31–38. [Google Scholar]
- Rosen, M.J. Surfactants and Interfacial Phenomena, 3rd ed; John Wiley & Sons Ltd: Hoboken, NJ, USA, 2004. [Google Scholar]
- Holmberg, K.; Jönsson, B.; Kronberg, B.; Lindman, B. Surfactants and Polymers in Aqueous Solution, 3rd ed; John Wiley & Sons Ltd: Chichester, UK, 2002. [Google Scholar]
- Guo, D.-S.; Wang, K.; Wang, Y.-X.; Liu, Y. Cholinesterase-responsive supramolecular vesicle. J. Am. Chem. Soc 2012, 134, 10244–10250. [Google Scholar]
- Wang, K.; Guo, D.-S.; Wang, X.; Liu, Y. Multistimuli responsive supramolecular vesicles based on the recognition of p-sulfonatocalixarene and its controllable release of doxorubicin. ACS Nano 2011, 5, 2880–2894. [Google Scholar]
- Wang, K.; Guo, D.-S.; Liu, Y. Temperature-controlled supramolecular vesicles modulated by p-sulfonatocalix[5]arene with pyrene. Chem. Eur. J 2010, 16, 8006–8011. [Google Scholar]
- Li, Z.; Hu, C.; Cheng, Y.; Xu, H.; Cao, X.; Song, X.; Zhang, H.; Liu, Y. Supramolecular vesicles of cationic gemini surfactants modulated by p-sulfonatocalix[4]arene. Sci. China Chem 2012, 55, 2063–2068. [Google Scholar]
- Rehm, M.; Frank, M.; Schatz, J. Water-soluble calixarenes—Self-aggregation and complexation of noncharged aromatic guests in buffered aqueous solution. Tetrahedron Lett 2009, 50, 93–96. [Google Scholar]
- Segota, S.; Tezak, D. Spontaneous formation of vesicles. Adv. Colloid Interface Sci 2006, 121, 51–75. [Google Scholar]
- Whitesides, G.M.; Boncheva, M. Beyond molecules: Self-assembly of mesoscopic and macroscopic components. Proc. Natl. Acad. Sci. USA 2002, 99, 4769–4774. [Google Scholar]
- Lee, M.; Lee, S.-J.; Jiang, L.-H. Stimuli-responsive supramolecular nanocapsules from amphiphilic calixarene assembly. J. Am. Chem. Soc 2004, 126, 12724–12725. [Google Scholar]
- Chécot, F.; Lecommandoux, S.; Gnanou, Y.; Klok, H.-A. Water-soluble stimuli-responsive vesicles from peptide-based diblock copolymers. Angew. Chem. Int. Ed 2002, 41, 1339–1343. [Google Scholar]
- Sumida, Y.; Masuyama, A.; Takasu, M.; Kida, T.; Nakatsuji, Y.; Ikeda, I.; Nojima, M. New pH-sensitive vesicles. Release control of trapped materials from the inner aqueous phase of vesicles made from triple-chain amphiphiles bearing two carboxylate groups. Langmuir 2001, 17, 609–612. [Google Scholar]
- Johnsson, M.; Wagenaar, A.; Engberts, J.B. Sugar-based gemini surfactant with a vesicle-to-micelle transition at acidic pH and a reversible vesicle flocculation near neutral pH. J. Am. Chem. Soc 2003, 125, 757–760. [Google Scholar]
- Zhu, J.; Munn, R.J.; Nantz, M.H. Self-cleaving ortho ester lipids: A new class of pH-vulnerable amphiphiles. J. Am. Chem. Soc 2000, 122, 2645–2646. [Google Scholar]
- Orihara, Y.; Matsumura, A.; Saito, Y.; Ogawa, N.; Saji, T.; Yamaguchi, A.; Sakai, H.; Abe, M. Reversible release control of an oily substance using photoresponsive micelles. Langmuir 2001, 17, 6072–6076. [Google Scholar]
- Mynar, J.L.; Goodwin, A.P.; Cohen, J.A.; Ma, Y.; Fleming, G.R.; Fréchet, J.M.J. Two-photon degradable supramolecular assemblies of linear-dendritic copolymers. Chem. Commun. 2007, 2081–2082. [Google Scholar]
- Power-Billard, K.N.; Spontak, R.J.; Manners, I. Redox-active organometallic vesicles: Aqueous self-assembly of a diblock copolymer with a hydrophilic polyferrocenylsilane polyelectrolyte block. Angew. Chem. Int. Ed 2004, 43, 1260–1264. [Google Scholar]
- Chen, Z.; Stepanenko, V.; Dehm, V.; Prins, P.; Siebbeles, L.D.A.; Seibt, J.; Marquetand, P.; Engel, V.; Würthner, F. Photoluminescence and conductivity of self-assembled pi-pi stacks of perylene bisimide dyes. Chem. Eur. J 2007, 13, 436–449. [Google Scholar]
- Chen, Z.; Lohr, A.; Saha-Möller, C.R.; Würthner, F. Self-assembled pi-stacks of functional dyes in solution: Structural and thermodynamic features. Chem. Soc. Rev 2009, 38, 564–584. [Google Scholar]
- Varga, O.; Kubinyi, M.; Vidóczy, T.; Baranyai, P.; Bitter, I.; Kállay, M. Methylene blue–calixarenesulfonate supramolecular complexes and aggregates in aqueous solutions. J. Photochem. Photobiol. A 2009, 207, 167–172. [Google Scholar]



© 2013 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Basilio, N.; Francisco, V.; Garcia-Rio, L. Aggregation of p-Sulfonatocalixarene-Based Amphiphiles and Supra-Amphiphiles. Int. J. Mol. Sci. 2013, 14, 3140-3157. https://doi.org/10.3390/ijms14023140
Basilio N, Francisco V, Garcia-Rio L. Aggregation of p-Sulfonatocalixarene-Based Amphiphiles and Supra-Amphiphiles. International Journal of Molecular Sciences. 2013; 14(2):3140-3157. https://doi.org/10.3390/ijms14023140
Chicago/Turabian StyleBasilio, Nuno, Vitor Francisco, and Luis Garcia-Rio. 2013. "Aggregation of p-Sulfonatocalixarene-Based Amphiphiles and Supra-Amphiphiles" International Journal of Molecular Sciences 14, no. 2: 3140-3157. https://doi.org/10.3390/ijms14023140
APA StyleBasilio, N., Francisco, V., & Garcia-Rio, L. (2013). Aggregation of p-Sulfonatocalixarene-Based Amphiphiles and Supra-Amphiphiles. International Journal of Molecular Sciences, 14(2), 3140-3157. https://doi.org/10.3390/ijms14023140



