1. Introduction
The ever-increasing number of studies devoted to various aspects of the biological activity of nanoparticles (NPs), including their toxicity, is almost on a par with the pace of developments in the area of nanotechnologies. This inspires hope that a sufficiently reliable evidence base will be obtained in the foreseeable future for assessing health risks, not only associated with the production and broad use of nanomaterials, but also with the contribution of NPs (“ultrafine particles”) to the aerosol contamination of the ambient and workplace air in many traditional industries. Consequently, our team has focused on the effects of metal and metal-oxide NPs, samples of which, with reliably preset dimensional and other relevant characteristics, could be regarded as a model for studying general patterns in the adverse influence on the organism, not only of respective engineered nanomaterials, but also of the ultrafine fraction of “usual” aerosol air pollutants.
Our earlier studies [
1–
6] showed, with reference to iron oxide Fe
3O
4 (magnetite), that, given equal doses by mass, NPs feature considerably higher cytotoxicity for lung macrophages and higher subchronic systemic toxicity than micrometric particles of the same chemical composition while triggering a more active protective response of pulmonary (alveolar) phagocytosis. At the same time, it was demonstrated that, within the conventional nanometric range, the relationship between particle diameter and toxicity is intricate and non-unique, which may be due to differences in the toxicokinetics, which is controlled by both unequally active physiological mechanisms responsible for the elimination and retention of NPs of various diameters, their unequal penetrability through biological barriers and, finally, their unequal solubility.
Among the issues that need to be resolved for developing both the theoretical foundations of comparative nanotoxicology and its regulatory aspects, of paramount importance is the question of which nanomaterial characteristics play the most important role:
Those associated with the nano-dimension of particles of any chemical composition (i.e., a high probability of NP deposition in the naso-pharyngeal region and lower airways; an ability to penetrate through biological barriers, transport with the lymph and blood to remote organs and the retention in them; the penetration into cells and cellular organelles; a large specific surface area; the peculiar character of physical interactions and chemical processes to which the mechanisms of damaging impact at subcellular and cellular levels are related); or,
The chemical nature of the NP-forming substance, which defines important mechanisms of its toxic impact both in the ionic-molecular form and in the form of particles of various sizes.
Within the framework of this problem, we set ourselves the task of comparing the toxic effects of virtually equidimensional silver and gold NPs. The choice of these two metals was determined not only by the theoretical premises set out above, but also by the fact that, in bulk, they are almost inert biologically. Furthermore, the high practical importance of nanosilver (NS) and nanogold (NG), have partly overlapping applications.
A great number of studies have been devoted to assessing the toxicity of various NS samples (e.g., [
7–
17]). A comparison of the results obtained from these studies with the data of the fewer works devoted to the assessment of similar effects evoked by NG (e.g., [
18–
26]) have suggested that the latter is far less toxic, although we are aware of only some studies in which such a comparison was really carried out in parallel testing conditions, and even those studies were performed not on mammals but on
Daphnia magna [
27] or zebrafish (
Danio rerio) embryos [
28].
Special consideration should be given to the issue of possible genotoxicity of even those nanomaterials that do not feature it in an ion-molecular state or in the form of micrometric particles. Recently, experimental evidence has been obtained, confirming that various nanomaterials are capable of producing damaging effects on DNA—namely, NPs of titanium dioxide [
12,
29,
30], zinc oxide [
31], silicon dioxide [
32,
33], carbon black [
34],
etc. It should be noted, however, that not all researchers have received positive results for the same nanomaterials, particularly when testing them
in vivo rather than on cell lines (for example, for nano-titanium dioxide in the crystal form of anatase [
35]).
A large number of papers have been published demonstrating, by means of various tests and on various test objects, the genotoxic effect of nanosilver, again, however, these tests were predominantly
in vitro (e.g., [
14,
32,
36–
44]). A small number of studies using short-term
in vivo tests quite often yielded a negative result (e.g., [
41]). Many authors emphasize the role of the NP-stimulated oxidative stress in the mechanisms of NPs’ damaging effect on DNA. Indeed, some studies not only have shown that NS increases considerably the formation of reactive oxygen species (ROS), but also have revealed significant inhibition of the genotoxic effects of NS in the presence of antioxidants or free radical scavengers [
32,
39,
43,
45].
There is less information concerning the genotoxicity of nanogold, and it is rather contradictory. Some data indicates that NG causes genome instability, also through oxidative stress [
46], but other experimenters have established that neither nano- (2 nm or 20 nm), nor microparticles (200 nm) of gold possess genotoxicity either
in vitro or in rats exposed to three consecutive intravenous [
23] or a single intratracheal [
47] administration.
We have found only one paper in which the genotoxicity of NG and NS was compared in parallel [
48]. The authors established on a human hepatocellular carcinoma cell line (HepG2) that at concentrations (by metal) of up to 100 μM NS provokes greater DNA damage than NG.
On the whole, a review of the above sources, and some additional ones, suggests that NG is not only generally less toxic but also possesses lower genotoxicity as compared with NS. Having stated this, however, we should qualify that:
In too few works has it been possible to find direct comparison data for NS and NG toxicities in parallel testing using any experimental model and equidimensional nanoparticles of these metals under similar conditions of exposure. Moreover, we have not discovered any single chronic or, at least, subchronic, comparative experiment with NG and NS on laboratory mammals in the literature known to us.
We also have not found any papers comparing the phagocytosis response to the pulmonary deposition of nanoparticles of these two metals, this response playing, as is known, a key role in pulmonary clearance. Our own pilot experiments involving intratracheal instillations of NG and NS suspensions have shown that NS provokes a substantially greater increase in the cell count of the bronchoalveolar lavage fluid (BALF), and, judging from the ratio of neutrophil leukocytes to alveolar macrophages in BALF, NS is more cytotoxic for alveolar macrophages than NG [
5]. These newly obtained data, however, need to be reproduced, preferably in experiments with the same NG and NS samples that would be used for comparative assessment of systemic toxicity and genotoxicity.
We have found no information concerning any attempts to enhance the resistance of the whole organism to systemic toxic and genotoxic impacts of these nanomaterials, although the above data on the protective action
in vitro of certain antioxidant and antiradical substances point directly to one of the possible approaches to such “biological prophylaxis.” The meaning of this concept, general principles of bioprophylaxis and numerous examples of their realization have been described by us repeatedly, including in review articles (e.g., [
49]). Beyond any doubt, ours were not the only studies demonstrating a possibility to reduce some metals’ toxicity with this or that innocuous antagonist. For instance, it was recently shown that hepatic copper retention in ram lambs could be reduced by dietary supplementation with molybdenum and zinc [
50]; or that quercetin and especially quercetin in combination with arginine ameliorated nano-zinc oxide’s nephrotoxicity for rats [
51]. Significant reducing of the lead blood levels with the help of calcium dietary supplementation was demonstrated not only in a number of animal experiments, ours included, but also on pregnant women [
52]. An important distinguishing feature of our approach is that we use multicomponent complexes of agents with different mechanisms of protective action rather than isolated bioprotectors [
49].
We decided that the first attempt to ensure such protective effect on the organism should be undertaken in response to the effect of the more toxic of two nanometals under study, which is, presumably, the NS.
3. Experimental Section
All experiments were carried out on outbred white female rats from our own breeding colony. There were 8–14 animals in different exposed and control groups, with the initial body weight of 150 to 220 g. Rats were housed in conventional conditions, breathed unfiltered air, and were fed the standard balanced food. The experiments were planned and implemented in accordance with the “International guiding principles for biomedical research involving animals” developed by the Council for International Organizations of Medical Sciences (1985).
For our study, we have prepared stable suspensions of silver and gold nanoparticles by the method of laser ablation in liquid, including the following main phases: (a) laser ablation of the metal target in water, and, where necessary, (b aser fragmentation for preventing particle aggregation, and (c) concentration of the suspension. In our case, concentration was necessary to enable the administration of effective NP doses to rats in minimal volumes of water.
A plate of silver or gold with a metal content of 99.99% was placed on the bottom of a dish with deionized water. Metal ablation was performed using Fmark-20RL laser material processing system (by Laser Technology Center, Zelenograd, Russia) based on ytterbium-doped pulsed fiber laser (pulse length 100 ns, repetition rate 21 kHz, wavelength 1064 nm). The energy density was 80 J/cm2. The target was Zirradiated in scanning mode with a rate of the laser ray 270 mm/s.
The concentration of the primary suspensions obtained by ablation was 0.10 mg/mL for gold, and 0.12 mg/mL for silver. Fragmentation proved necessary for preventing the aggregation and for enhancing the stability of the nanogold suspension, involving after-treatment with a laser ray focused into the bulk of the suspension. An increase in concentration to 0.5 mg/mL was achieved by drying the suspensions which was not accompanied by nanoparticle aggregation. Particle images were obtained after concentration by scanning electron microscope (SEM), AURIGA CrossBeam Workstation (Carl Zeiss, Oberkochen, Germany), which enabled us to identify their spherical form (
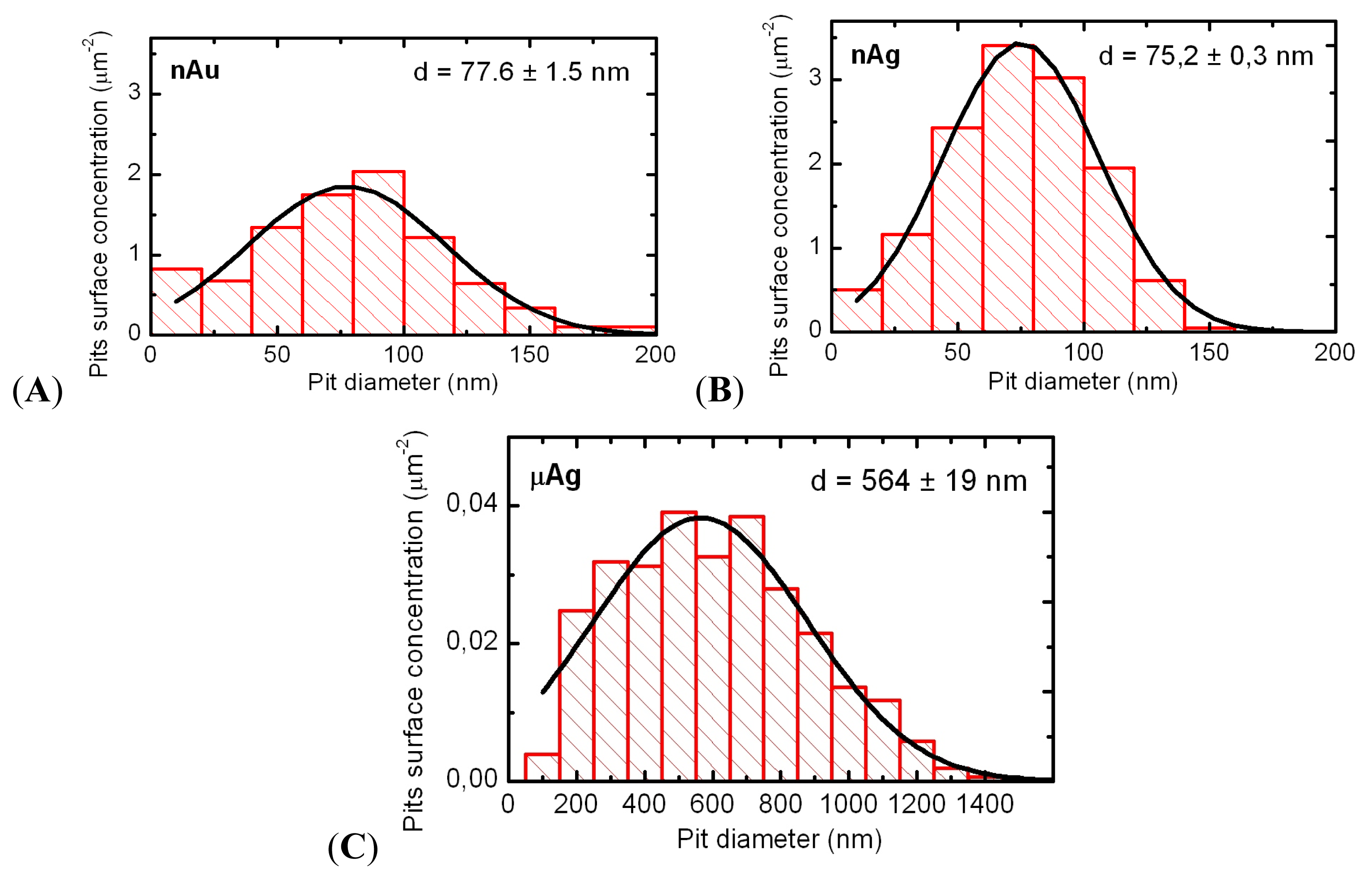
Figure 9). Average NP diameters (± s.d.) obtained through statistical processing of hundreds of SEM images were: 50 ± 10 nm for NG and 49 ± 5 nm for NS, the distribution being symmetrical (
Figure 10). Nanogold average particle size was also measured by the dynamic light scattering method by the Zetasizer Nano ZS analyzer (Malvern Instruments, Worcestershire, UK), and provided a sufficiently close result, 58 ± 19 nm.
No essential changes took place 30 days after the preparation of the suspensions in either zeta potential or the form and position of the plasmon resonance peak, providing evidence of their high stability.
The suspension of silver microparticles with an average diameter of 1.1 μm was obtained by burning ash-free filters impregnated with silver nitrate, with subsequent ultrasonic dispersion in deionized water. All the above described stages and regimes of suspension preparation had been preliminarily optimized in a series of experiments.
Both NS and NG suspensions were administered to rats:
either once intratracheally (i.t.), at a dose of 0.5 mg in 1 mL of the freshly prepared suspensions of NG or NS (in parallel with the same dose of silver microparticles);
or, intraperitoneally, three times a week (up to 20 injections) at a dose of 10 mg/kg in the corresponding volume of the suspension containing 0.5 mg of NS or NG per mL (This concentration was highest for stable nano-suspensions that we were able to obtain with the technique described above, while the biggest tolerable volume of an i.p. injection to a rat was found to be 4 mL. Just these two factors determined the actual dosage of NS and NG, even if we wanted it to be somewhat higher for being more certain with regard to the toxic effects of these metals. However a preliminary short-term (2 weeks) pilot experiment with the presumably least toxic NG demonstrated that even the actually attainable low dosage was not without some adverse effects).
Animals in respective control groups were administered sterile deionized water (from the batch used for preparing suspensions) by the respective route. In the subchronic experiment, an additional group of rats studied in parallel was being injected with the same dosage of NS, but against the background of administration of a bioprotective complex (BPC) described below, and still another group was given the same BPC plus i.p. injections of water.
In the acute intratracheal test, along with optical microscopy of cells sedimented by centrifuging the BALF, obtained as described below, we examined the topography of the BALF cells surface. To this end, we used semi-contact atomic force microscopy (sc-AFM) reputed as a unique technique allowing one to obtain 3D visualizations of the surface topography of biological objects with a nanometric spatial resolution. A 3 μL aliquot of the BALF sediment was precipitated on a fresh cleavage of mica. After 60 s, excessive suspension was removed with a paper filter, and the sample was dried by blowing with clean, dry air or nitrogen for 30 s. It should be noted that the drying of the BALF on a mica surface results in the formation of salt microcrystals, which were removed by washing the sample twice. For washing, the sample was kept for 60 s on the surface of a drop of deionized water (with the working side down). The liquid was then removed with the help of a paper filter. After repeating the washing, the sample was dried by blowing with clean, dry air or nitrogen for 30 s. Investigation of the cell surface morphology was performed by sc-AFM with the help of an NTEGRA Therma scanning probe platform by NT-MDT (Zelenograd, Russia) using semi-contact atomic force microscopy mode with NSG01 probes by NT-MDT (Zelenograd, Russia). The height of the probes was about 15 μm, and the tip curvature radius was less than 10 nm. For statistical processing and analysis of measurement results, we used specialized software, SPIP (Image Metrology, Horsholm, Denmark) and SIAMS Photolab (Ekaterinburg, Russia). The procedures developed made it possible to reveal the pits in the images of cell surfaces and to measure the diameter and depth of each pit. The results of image analysis were used for plotting the histograms of pit dimensions for cells of all groups of rats.
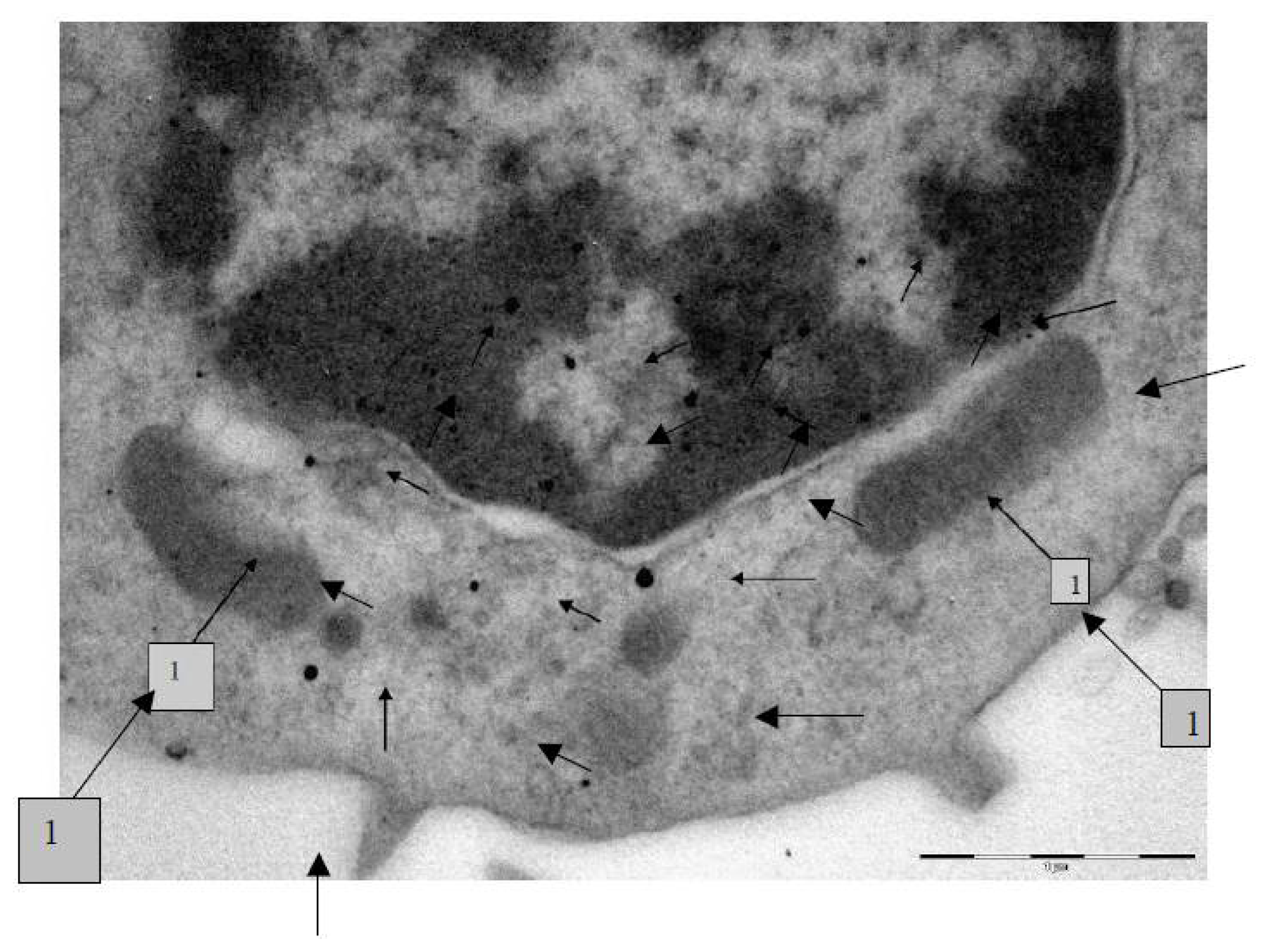
The transmission electron microscopy (TEM) was used to study the localization of different NPs within the BALF phagocytes and to visualize damage to the cells at ultra structural level that may be attributed to the cytotoxic effect of NPs.
Bronchoalveolar lavage was carried out 24 h after the i.t. instillation of suspensions or reference water. A cannula connected to a Lüer’s syringe containing 10 mL of normal saline was inserted into the surgically prepared trachea of a rat under hexenal anesthesia. The fluid entered the lungs slowly under the gravity of the piston, with the animal and syringe positioned vertically. Then the rat and the syringe were turned 180°, and the fluid flowed back into the syringe. The extracted BALF was poured into siliconized refrigerated tubes. An aliquot sample of the BALF was drawn into a WBC count pipette together with 3% acetic acid and methylene blue. Cell count was performed in a standard hemocytometer (the so-called Goryayev’s Chamber).
For cytological examination and semi-contact atomic force microscopy (sc-AFM), the BALF was centrifuged for 4 min at 1000 rpm, before the fluid was decanted, and the sediment was used for preparing (a) sc-AFM samples as described above, and (b) smears on 2 microscope slides. After air drying, the smears were fixed with methyl alcohol and stained with azure eosin. The differential count (under optical microscope with immersion at a magnification of 1000×) for determining the percentage of alveolar macrophages (AM), neutrophil leukocytes (NL) and other cells was conducted up to a total number of 100 counted cells. Allowing for the total number of cells in the BALF, these percentages were recalculated in terms of absolute AM and NL counts.
For performing TEM, BALF was centrifuged for 30 min at 3000 rpm. The cell sediment was fixed in 2.5% solution of glutaraldehyde with subsequent additional fixing in 1% solution of osmium tetroxide for 2 h. Then it was washed in 0.2 M phosphate buffer and passed through alcohols of increasing concentration and through acetone for dehydration. Then the sample was placed for 24 h in a mixture of araldite and acetone at a ratio of 1:1, following which it was polymerized in araldite at 37 °C for 1 day and at 50–60 °C for the following 2–3 days. Ultrathin sections were obtained on a Leica EM UC6 ultramicrotome (Wetzlar, Germany), contrasted with lead citrate and examined on a Morgagni 268 electron microscope (FEI Company, Eindhoven, The Netherlands).
In the subchronic intraperitoneal test, immediately after the subchronic exposure period, the following procedures were performed for all rats:
weighing;
estimation of the CNS ability to the temporal summation of sub-threshold impulses—a variant of withdrawal reflex and its facilitation by repeated electrical stimulations in intact, conscious rat [
68];
recording of the number of head-dips into holes of a hole-board, which is frequently used for studying behavioral effects of toxicants and drugs (e.g., [
69,
70]);
collection of daily urine for analysis of its density, urine output, coproporhyrin, delta-aminolevulinic acid (δ-ALA), hydroxyproline, and creatinine contents;
sampling of capillary blood from a notch on the tail for examining the hemogram, hemoglobin content, and for cytochemical determination of succinate dehydrogenase (SDH) activity in lymphocytes (by the reduction of nitrotetrazolium violet to formasan, the number of granules of which in a cell is counted under immersion microscopy).
Then the rats were killed by decapitation and blood was collected by exsanguination. Biochemical indices determined from serum included total protein, albumin, globulin, bilirubin, ceruloplasmin, malonyldialdehyde (MDA), alkaline phosphatase, alanine- and asparate-transaminases (ALT, AST), catalase, gamma glutamyl transferase. For four rats in each treatment and control group, liver, kidney, and spleen tissue sections were prepared for histological examination by staining with hematoxilin-eosine and with the Van Gieson’s stain.
3.1. Testing of Genotoxicity (the RAPD Test)
In total, we analyzed 96 samples, each sample in three replications. Muscular tissue was separated with a scalpel from the bone tissue. Samples of solid organs were minced with a scalpel. Then for the purpose of obtaining homogeneous mass, the samples were subjected four times to freezing in liquid nitrogen and to thawing in an ultrasonic bath (38 °C) in the Ca2+, Mg2+ free PBS solution (Sigma, St. Louis, MO, USA) with subsequent passing through needles of decreasing diameter. Bone marrow cells were washed out of the femur (after cutting off the epiphyseal and diaphyseal areas) with the same PBS solution with subsequent passing through needles of decreasing diameter. Nucleated peripheral blood cells were isolated from the whole blood of the laboratory animals by centrifuging in the Percoll gradient (Sigma, St. Louis, MO, USA).
To isolate DNA from the cells, we used a GenElute (Sigma, St. Louis, MO, USA) set of reagents in accordance with the manufacturer’s guidelines for use. In the samples obtained, we determined spectrophotometrically with Ultraspec 1100 pro (Biochrom Ltd, Cambridge, UK) the content of DNA and freezed and stored them at −84 °C in a kelvinator Sanyo (Moriguti, Japan) till the beginning of the analysis.
The Random Amplification of Polymorphic DNA (RAPD) test was performed as described earlier [
65,
66]. This technique allows one to define quantitatively the degree of DNA fragmentation as an estimate for the genotoxicity of harmful agents and for the protective effects of the complex of bioprotectors studied. The method is based on the fact that, unlike a fragmented DNA, which forms the so-called “comet tail” in the agarose gel in electrophoresis, a non-fragmented DNA has a very low degree of migration and virtually stays in the same place (“comet head”), and the degree of migration is directly related to the degree of DNA fragmentation. To characterize the degree of damage to DNA we used “coefficient of fragmentation”,
i.e., the ratio of total radio-activity of all “tail” fractions to the radioactivity of the “head”.
The gold or silver content of liver, spleen and kidneys was determined by atomic emission spectrometer with inductively coupled plasma iCAP-6500 Duo (Thermo Scientific, Billerica, MA, USA). Samples of freeze-dried homogenized tissue were subjected to acid digestion with the help of a MARS 5 microwave accelerated reaction system.
3.2. Choice of Bioprotectors
A review of the literature data on the mechanisms of toxic and genotoxic action of silver, combined with our experience in the testing of various bioprotectors for other intoxications (summarized in [
49]), allowed us to choose, for estimating the possible protective action at subchronic intoxication with nanosilver, a complex that included the following biologically active substances:
Glutamate as an effective cell membrane stabilizer through the intensification of ATP synthesis under exposure to the damaging action of various cytotoxic particles (e.g., [
64–
67,
71]) and, at the same time, as one of the three precursors of glutathione, a powerful cell protector against free radicals.
The other two precursors of glutathione: glycine and cysteine (the latter in a highly active and metabolically easily available form of N-acetylcysteine).
Other components of the organism’s antioxidant system (vitamins A, E and C, and selenium).
Trace elements that are physiological and toxicological antagonists of silver (selenium, copper, calcium) [
16,
72].
Omega-3 polyunsaturated fatty acids, whose intracellular derivatives are eicosanoids that activate DNA replication and thus play an important part in its repair, which was demonstrated under exposure to various genotoxic agents (e.g., [
64–
67]).
Pectine enterosorbent as an agent that prevents the reabsorption of the metal excreted into the intestines with bile.
Based on the experience of our earlier experiments ([
64–
67,
71] and many others), we administered these bioprotectors in the following ways (see
Table 7). We gave glutamate to rats as a 1.5% solution instead of drinking water
ad libitum. “Eicosovitol” (by Farmavit Ltd., Tyumen, Russia), a fish oil preparation rich in PUFA pertaining mainly to the omega-3 group was administered through gavage at a dose of 1 mL per rat. The apple pectin enterosorbent (by Promavtomatika Ltd, Belgorod, Russia) was added to the rats’ fodder in a quantity corresponding to a dose
ca. 1000 mg/kg body weight. Other commercial preparations of amino acids, vitamins and minerals available as tablets were crushed and added to another portion of the fodder in quantities corresponding to the recommended daily intake of these micronutrients by rats. (If such recommendations were known only for humans, a recalculation to the rat’s nutritional requirement was made based on the species’ standard metabolisms ratio.)
Taking into account that the standard balanced food presumably meets the normal nutritional requirements of a rat, we assumed that additional intake of the above-listed bioactive substances would meet the increased needs connected with molecular mechanisms of silver toxicity. Nevertheless, it had to be checked whether or not such “overloading” with them would engender any unfavorable effects. For this reason, in our subchronic experiment, one group of rats was administered the same BPC but not exposed to NS.