Differential Expression Analysis of a Subset of Drought-Responsive GmNAC Genes in Two Soybean Cultivars Differing in Drought Tolerance
Abstract
:1. Introduction
2. Results and Discussion
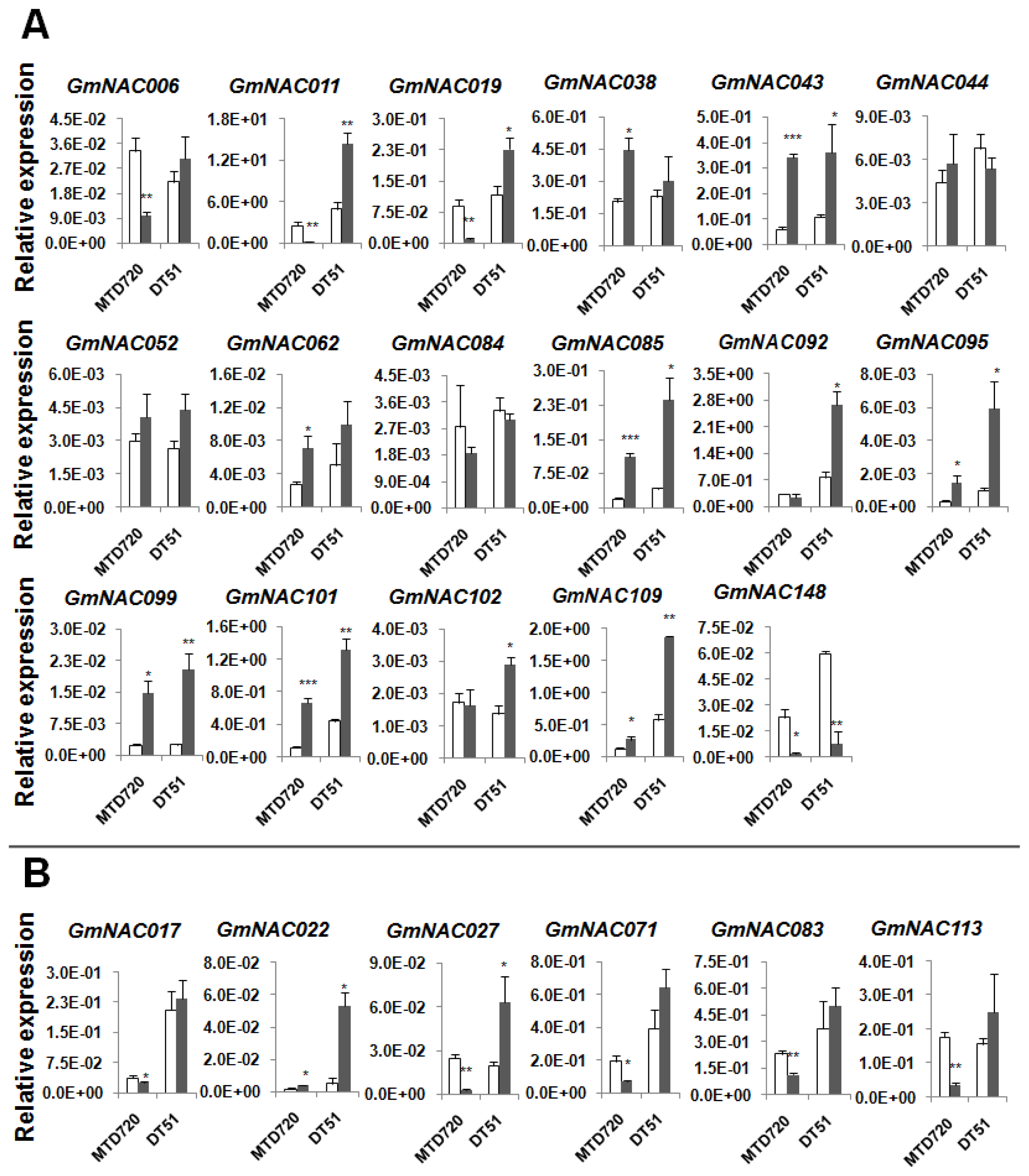
2.1. Differential Expression of a Subset of Drought-Responsive GmNAC Genes in the Roots of DT51 and MTD720
2.2. Potential Drought-Responsive Genes for Genetic Engineering
3. Experimental Section
3.1. Plant Growth, Drought Treatment and Collection of Root Tissues
3.2. Total RNA Isolation and cDNA Synthesis
3.3. Real-Time Quantitative PCR
3.4. Discovery of cis-Regulatory Motifs in Promoter Regions of GmNACs
3.5. Statistical Analysis of the Data
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Thao, N.P.; Tran, L.-S.P. Potentials toward genetic engineering of drought-tolerant soybean. Crit. Rev. Biotechnol 2012, 32, 349–362. [Google Scholar]
- Shinozaki, K.; Yamaguchi-Shinozaki, K. Gene networks involved in drought stress response and tolerance. J. Exp. Bot 2007, 58, 221–227. [Google Scholar]
- Nishiyama, R.; Watanabe, Y.; Leyva-Gonzalez, M.A.; van Ha, C.; Fujita, Y.; Tanaka, M.; Seki, M.; Yamaguchi-Shinozaki, K.; Shinozaki, K.; Herrera-Estrella, L. Arabidopsis AHP2, AHP3, and AHP5 histidine phosphotransfer proteins function as redundant negative regulators of drought stress response. Proc. Natl. Acad. Sci. USA 2013, 110, 4840–4845. [Google Scholar]
- Yang, S.; Vanderbeld, B.; Wan, J.; Huang, Y. Narrowing down the targets: Towards successful genetic engineering of drought-tolerant crops. Mol. Plant 2010, 3, 469–490. [Google Scholar]
- Yamaguchi-Shinozaki, K.; Shinozaki, K. Transcriptional regulatory networks in cellular responses and tolerance to dehydration and cold stresses. Annu. Rev. Plant Biol 2006, 57, 781–803. [Google Scholar]
- Zhang, G.; Chen, M.; Li, L.; Xu, Z.; Chen, X.; Guo, J.; Ma, Y. Overexpression of the soybean GmERF3 gene, an AP2/ERF type transcription factor for increased tolerances to salt, drought, and diseases in transgenic tobacco. J. Exp. Bot 2009, 60, 3781–3796. [Google Scholar]
- Lu, G.; Gao, C.; Zheng, X.; Han, B. Identification of OsbZIP72 as a positive regulator of ABA response and drought tolerance in rice. Planta 2009, 229, 605–615. [Google Scholar]
- Abe, H.; Urao, T.; Ito, T.; Seki, M.; Shinozaki, K.; Yamaguchi-Shinozaki, K. Arabidopsis AtMYC2 (bHLH) and AtMYB2 (MYB) function as transcriptional activators in abscisic acid signaling. Plant Cell 2003, 15, 63–78. [Google Scholar]
- Luo, X.; Bai, X.; Sun, X.; Zhu, D.; Liu, B.; Ji, W.; Cai, H.; Cao, L.; Wu, J.; Hu, M. Expression of wild soybean WRKY20 in Arabidopsis enhances drought tolerance and regulates ABA signalling. J. Exp. Bot 2013, 64, 2155–2169. [Google Scholar]
- Tran, L.-S.P.; Nakashima, K.; Sakuma, Y.; Simpson, S.D.; Fujita, Y.; Maruyama, K.; Fujita, M.; Seki, M.; Shinozaki, K.; Yamaguchi-Shinozaki, K. Isolation and functional analysis of Arabidopsis stress-inducible NAC transcription factors that bind to a drought-responsive cis-element in the early responsive to dehydration stress 1 promoter. Plant Cell 2004, 16, 2481–2498. [Google Scholar]
- Souer, E.; van Houwelingen, A.; Kloos, D.; Mol, J.; Koes, R. The no apical meristem gene of petunia is required for pattern formation in embryos and flowers and is expressed at meristem and primordia boundaries. Cell 1996, 85, 159–170. [Google Scholar]
- Nuruzzaman, M.; Manimekalai, R.; Sharoni, A.M.; Satoh, K.; Kondoh, H.; Ooka, H.; Kikuchi, S. Genome-wide analysis of NAC transcription factor family in rice. Gene 2010, 465, 30–44. [Google Scholar]
- Hu, R.; Qi, G.; Kong, Y.; Kong, D.; Gao, Q.; Zhou, G. Comprehensive analysis of NAC domain transcription factor gene family in Populus trichocarpa. BMC Plant Biol 2010, 10, 145. [Google Scholar]
- Rushton, P.J.; Bokowiec, M.T.; Han, S.; Zhang, H.; Brannock, J.F.; Chen, X.; Laudeman, T.W.; Timko, M.P. Tobacco transcription factors: Novel insights into transcriptional regulation in the Solanaceae. Plant Physiol 2008, 147, 280–295. [Google Scholar]
- Mochida, K.; Yoshida, T.; Sakurai, T.; Yamaguchi-Shinozaki, K.; Shinozaki, K.; Tran, L.S.P. In silico analysis of transcription factor repertoire and prediction of stress responsive transcription factors in soybean. DNA Res 2009, 16, 353–369. [Google Scholar]
- Xie, Q.; Frugis, G.; Colgan, D.; Chua, N.-H. Arabidopsis NAC1 transduces auxin signal downstream of TIR1 to promote lateral root development. Genes Dev 2000, 14, 3024–3036. [Google Scholar]
- Duval, M.; Hsieh, T.-F.; Kim, S.Y.; Thomas, T.L. Molecular characterization of AtNAM: A member of the Arabidopsis NAC domain superfamily. Plant Mol. Biol 2002, 50, 237–248. [Google Scholar]
- Yoo, S.Y.; Kim, Y.; Kim, S.Y.; Lee, J.S.; Ahn, J.H. Control of flowering time and cold response by a NAC-domain protein in Arabidopsis. PLoS One 2007, 2, e642. [Google Scholar]
- Zhong, R.; Richardson, E.A.; Ye, Z.-H. Two NAC domain transcription factors, SND1 and NST1, function redundantly in regulation of secondary wall synthesis in fibers of Arabidopsis. Planta 2007, 225, 1603–1611. [Google Scholar]
- Olsen, A.N.; Ernst, H.A.; Leggio, L.L.; Skriver, K. NAC transcription factors: Structurally distinct, functionally diverse. Trends Plant Sci 2005, 10, 79–87. [Google Scholar]
- Puranik, S.; Sahu, P.P.; Srivastava, P.S.; Prasad, M. NAC proteins: Regulation and role in stress tolerance. Trends Plant Sci 2012, 17, 369–381. [Google Scholar]
- Hao, Y.J.; Wei, W.; Song, Q.X.; Chen, H.W.; Zhang, Y.Q.; Wang, F.; Zou, H.F.; Lei, G.; Tian, A.G.; Zhang, W.K. Soybean NAC transcription factors promote abiotic stress tolerance and lateral root formation in transgenic plants. Plant J 2011, 68, 302–313. [Google Scholar]
- Riechmann, J.L.; Ratcliffe, O.J. A genomic perspective on plant transcription factors. Curr. Opin. Plant Biol 2000, 3, 423–434. [Google Scholar]
- Fang, Y.; You, J.; Xie, K.; Xie, W.; Xiong, L. Systematic sequence analysis and identification of tissue-specific or stress-responsive genes of NAC transcription factor family in rice. Mol. Genet. Genomics 2008, 280, 547–563. [Google Scholar]
- Nakashima, K.; Tran, L.S.P.; van Nguyen, D.; Fujita, M.; Maruyama, K.; Todaka, D.; Ito, Y.; Hayashi, N.; Shinozaki, K.; Yamaguchi-Shinozaki, K. Functional analysis of a NAC-type transcription factor OsNAC6 involved in abiotic and biotic stress-responsive gene expression in rice. Plant J 2007, 51, 617–630. [Google Scholar]
- Hu, H.; Dai, M.; Yao, J.; Xiao, B.; Li, X.; Zhang, Q.; Xiong, L. Overexpressing a NAM, ATAF, CUC (NAC) transcription factor enhances drought resistance and salt tolerance in rice. Proc. Natl. Acad. Sci. USA 2006, 103, 12987–12992. [Google Scholar]
- Zheng, X.; Chen, B.; Lu, G.; Han, B. Overexpression of a NAC transcription factor enhances rice drought and salt tolerance. Biochem. Bioph. Res. Commun 2009, 379, 985–989. [Google Scholar]
- Xue, G.-P.; Way, H.M.; Richardson, T.; Drenth, J.; Joyce, P.A.; McIntyre, C.L. Overexpression of TaNAC69 leads to enhanced transcript levels of stress up-regulated genes and dehydration tolerance in bread wheat. Mol. Plant 2011, 4, 697–712. [Google Scholar]
- Tang, Y.; Liu, M.; Gao, S.; Zhang, Z.; Zhao, X.; Zhao, C.; Zhang, F.; Chen, X. Molecular characterization of novel TaNAC genes in wheat and overexpression of TaNAC2a confers drought tolerance in tobacco. Physiol. Plant 2012, 144, 210–224. [Google Scholar]
- Liu, X.; Liu, S.; Wu, J.; Zhang, B.; Li, X.; Yan, Y.; Li, L. Overexpression of Arachis hypogaea NAC3 in tobacco enhances dehydration and drought tolerance by increasing superoxide scavenging. Plant Physiol. Bioch 2013, 70, 354–359. [Google Scholar]
- Tran, L.S.P.; Quach, T.N.; Guttikonda, S.K.; Aldrich, D.L.; Kumar, R.; Neelakandan, A.; Valliyodan, B.; Nguyen, H.T. Molecular characterization of stress-inducible GmNAC genes in soybean. Mol. Genet. Genomics 2009, 281, 647–664. [Google Scholar]
- Soybean Production (Food and Agriculture Organization of the United Nations—FAO). Available online: http://faostat3.fao.org/faostat-gateway/go/to/download/Q/QC/E (accessed on 15 October 2013).
- Sadeghipour, O.; Abbasi, S. Soybean response to drought and seed inoculation. World Appl. Sci. J 2012, 17, 55–60. [Google Scholar]
- Ku, Y.-S.; Au-Yeung, W.-K.; Yung, Y.-L.; Li, M.-W.; Wen, C.-Q.; Liu, X.; Lam, H.-M. Drought Stress and Tolerance in Soybean. In A Comprehensive Survey of Internaitonal Soybean Research—Genetics, Physiology, Agronomy and Nitrogen Relationships; Board, J.E., Ed.; InTech: New York, NY, USA, 2013; pp. 209–237. [Google Scholar]
- Stolf-Moreira, R.; Lemos, E.G.M.; Carareto-Alves, L.; Marcondes, J.; Pereira, S.S.; Rolla, A.A.P.; Pereira, R.M.; Neumaier, N.; Binneck, E.; Abdelnoor, R.V. Transcriptional profiles of roots of different soybean genotypes subjected to drought stress. Plant Mol. Biol. Rep 2011, 29, 19–34. [Google Scholar]
- Le, D.T.; Tarrago, L.; Watanabe, Y.; Kaya, A.; Lee, B.C.; Tran, U.; Nishiyama, R.; Fomenko, D.E.; Gladyshev, V.N.; Tran, L.-S.P. Diversity of plant methionine sulfoxide reductases b and evolution of a form specific for free methionine sulfoxide. PLoS One 2013, 8, e65637. [Google Scholar]
- Van Ha, C.; Le, D.T.; Nishiyama, R.; Watanabe, Y.; Sulieman, S.; Tran, U.T.; Mochida, K.; van Dong, N.; Yamaguchi-Shinozaki, K.; Shinozaki, K. The auxin response factor transcription factor family in soybean: Genome-wide identification and expression analyses during development and water stress. DNA Res 2013, 20, 511–524. [Google Scholar]
- Hossain, Z.; Khatoon, A.; Komatsu, S. Soybean proteomics for unraveling abiotic stress response mechanism. J. Proteome Res 2013, 12, 4670–4684. [Google Scholar]
- Wang, Z.; Libault, M.; Joshi, T.; Valliyodan, B.; Nguyen, H.; Xu, D.; Stacey, G.; Cheng, J. Soydb: A knowledge database of soybean transcription factors. BMC Plant Biol 2010, 10, 14. [Google Scholar]
- Schmutz, J.; Cannon, S.B.; Schlueter, J.; Ma, J.; Mitros, T.; Nelson, W.; Hyten, D.L.; Song, Q.; Thelen, J.J.; Cheng, J. Genome sequence of the palaeopolyploid soybean. Nature 2010, 463, 178–183. [Google Scholar]
- Meng, Q.; Zhang, C.; Gai, J.; Yu, D. Molecular cloning, sequence characterization and tissue-specific expression of six NAC-like genes in soybean (Glycine max (L.) Merr.). J. Plant Physiol 2007, 164, 1002–1012. [Google Scholar]
- Pinheiro, G.L.; Marques, C.S.; Costa, M.D.B.L.; Reis, P.A.B.; Alves, M.S.; Carvalho, C.M.; Fietto, L.G.; Fontes, E.P.B. Complete inventory of soybean NAC transcription factors: Sequence conservation and expression analysis uncover their distinct roles in stress response. Gene 2009, 444, 10–23. [Google Scholar]
- Le, D.T.; Nishiyama, R.; Watanabe, Y.; Mochida, K.; Yamaguchi-Shinozaki, K.; Shinozaki, K.; Tran, L.S.P. Genome-wide survey and expression analysis of the plant-specific NAC transcription factor family in soybean during development and dehydration stress. DNA Res 2011, 18, 263–276. [Google Scholar]
- Le, D.T.; Nishiyama, R.; Watanabe, Y.; Tanaka, M.; Seki, M.; Yamaguchi-Shinozaki, K.; Shinozaki, K.; Tran, L.-S.P. Differential gene expression in soybean leaf tissues at late developmental stages under drought stress revealed by genome-wide transcriptome analysis. PLoS One 2012, 7, e49522. [Google Scholar]
- Manavalan, L.P.; Guttikonda, S.K.; Tran, L.S.P.; Nguyen, H.T. Physiological and molecular approaches to improve drought resistance in soybean. Plant Cell Physiol 2009, 50, 1260–1276. [Google Scholar]
- Gowda, V.R.; Henry, A.; Yamauchi, A.; Shashidhar, H.; Serraj, R. Root biology and genetic improvement for drought avoidance in rice. Field Crop. Res 2011, 122, 1–13. [Google Scholar]
- Abd Allah, A.; Shimaa, A.; Zayed, B.; Gohary, A.E. The role of root system traits in the drought tolerance of rice (Oryza sativa L.). Int. J. Agric. Biol. Sci 2010, 1, 83–87. [Google Scholar]
- Guo, P.; Baum, M.; Grando, S.; Ceccarelli, S.; Bai, G.; Li, R.; von Korff, M.; Varshney, R.K.; Graner, A.; Valkoun, J. Differentially expressed genes between drought-tolerant and drought-sensitive barley genotypes in response to drought stress during the reproductive stage. J. Exp. Bot 2009, 60, 3531–3544. [Google Scholar]
- Kasuga, M.; Liu, Q.; Miura, S.; Yamaguchi-Shinozaki, K.; Shinozaki, K. Improving plant drought, salt, and freezing tolerance by gene transfer of a single stress-inducible transcription factor. Nat. Biotechnol 1999, 17, 287–291. [Google Scholar]
- Rivero, R.M.; Kojima, M.; Gepstein, A.; Sakakibara, H.; Mittler, R.; Gepstein, S.; Blumwald, E. Delayed leaf senescence induces extreme drought tolerance in a flowering plant. Proc. Natl. Acad. Sci. USA 2007, 104, 19631–19636. [Google Scholar]
- Yamaguchi-Shinozaki, K.; Shinozaki, K. Organization of cis-acting regulatory elements in osmotic-and cold-stress-responsive promoters. Trends Plant Sci 2005, 10, 88–94. [Google Scholar]
- Behnam, B.; Iuchi, S.; Fujita, M.; Fujita, Y.; Takasaki, H.; Osakabe, Y.; Yamaguchi-Shinozaki, K.; Kobayashi, M.; Shinozaki, K. Characterization of the promoter region of an Arabidopsis gene for 9-cis-epoxycarotenoid dioxygenase involved in dehydration-inducible transcription. DNA Res 2013, 20, 315–324. [Google Scholar]
- Ramya, M.; Raveendran, M.; Ramalingam, S.J. In silico analysis of drought tolerant genes in rice. Int. J. Biol. Med. Res 2010, 3, 110–114. [Google Scholar]
- Maruyama, K.; Todaka, D.; Mizoi, J.; Yoshida, T.; Kidokoro, S.; Matsukura, S.; Takasaki, H.; Sakurai, T.; Yamamoto, Y.Y.; Yoshiwara, K. Identification of cis-acting promoter elements in cold-and dehydration-induced transcriptional pathways in Arabidopsis, rice, and soybean. DNA Res 2012, 19, 37–49. [Google Scholar]
- Tran, L.S.P.; Nakashima, K.; Sakuma, Y.; Osakabe, Y.; Qin, F.; Simpson, S.D.; Maruyama, K.; Fujita, Y.; Shinozaki, K.; Yamaguchi-Shinozaki, K. Co-expression of the stress-inducible zinc finger homeodomain ZFHD1 and NAC transcription factors enhances expression of the ERD 1 gene in Arabidopsis. Plant J 2007, 49, 46–63. [Google Scholar]
- Thu, N.B.A.; Nguyen, Q.T.; Hoang, X.L.T.; Thao, N.P.; Tran, L.S.P. Evaluation of drought tolerance of the Vietnamese soybean cultivars provides potential resources for soybean production and genetic engineering. Unpublished Observation 2013. [Google Scholar]
- Le, D.T.; Aldrich, D.L.; Valliyodan, B.; Watanabe, Y.; van Ha, C.; Nishiyama, R.; Guttikonda, S.K.; Quach, T.N.; Gutierrez-Gonzalez, J.J.; Tran, L.-S.P. Evaluation of candidate reference genes for normalization of quantitative RT-PCR in soybean tissues under various abiotic stress conditions. PLoS One 2012, 7, e46487. [Google Scholar]
- Le, D.T.; Nishiyama, R.; Watanabe, Y.; Mochida, K.; Yamaguchi-Shinozaki, K.; Shinozaki, K.; Tran, L.S.P. Genome-wide expression profiling of soybean two-component system genes in soybean root and shoot tissues under dehydration stress. DNA Res 2011, 18, 17–29. [Google Scholar]
| Nomenclature | Glyma ID | DT51 | p-value | Regulation | MTD720 | p-value | Regulation | Regulation in roots of W82 * |
|---|---|---|---|---|---|---|---|---|
| GmNAC006 | Glyma02g07700.1 | 1.4 | 0.1884 | Unaltered | 3.3 | 0.0085 | Down | Unaltered |
| GmNAC011 | Glyma02g26480.1 | 2.9 | 0.0074 | Up | 13.3 | 0.0098 | Down | Unaltered |
| GmNAC019 | Glyma04g38990.1 | 1.9 | 0.0299 | Unaltered | 9.1 | 0.0089 | Down | Up |
| GmNAC038 | Glyma06g15990.1 | 1.3 | 0.2121 | Unaltered | 2.2 | 0.0121 | Up | Up |
| GmNAC043 | Glyma06g38410.1 | 3.4 | 0.0478 | Up | 5.8 | 0.0001 | Up | Up |
| GmNAC062 | Glyma08g19300.1 | 1.9 | 0.2011 | Unaltered | 2.6 | 0.0340 | Up | Up |
| GmNAC085 | Glyma12g22880.1 | 5.5 | 0.0402 | Up | 5.9 | 0.0008 | Up | Up |
| GmNAC092 | Glyma12g35000.1 | 3.5 | 0.0126 | Up | 1.3 | 0.3609 | Unaltered | Up |
| GmNAC095 | Glyma13g05540.1 | 6.0 | 0.0289 | Up | 4.7 | 0.0447 | Up | Unaltered |
| GmNAC099 | Glyma13g31660.1 | 7.8 | 0.0097 | Up | 5.9 | 0.0114 | Up | Unaltered |
| GmNAC101 | Glyma13g35550.1 | 3.0 | 0.0026 | Up | 5.7 | 0.0009 | Up | Up |
| GmNAC102 | Glyma13g35560.1 | 2.1 | 0.0143 | Up | 1.0 | 0.4799 | Unaltered | Up |
| GmNAC109 | Glyma14g24220.1 | 3.3 | 0.0016 | Up | 2.3 | 0.0315 | Up | Up |
| GmNAC148 | Glyma20g04400.1 | 7.6 | 0.0027 | Down | 12.0 | 0.0151 | Down | Up |
| GmNAC022 | Glyma04g42800.1 | 10.5 | 0.0203 | Up | 2.1 | 0.0450 | Up | Unaltered |
| GmNAC027 | Glyma05g24910.1 | 3.2 | 0.0434 | Up | 7.9 | 0.0013 | Down | Unaltered |
| GmNAC071 | Glyma10g04350.1 | 1.6 | 0.1411 | Unaltered | 2.7 | 0.0207 | Down | Unaltered |
| GmNAC083 | Glyma12g13710.1 | 1.3 | 0.3577 | Unaltered | 2.0 | 0.0093 | Down | Unaltered |
| GmNAC113 | Glyma15g07620.1 | 1.6 | 0.1812 | Unaltered | 5.2 | 0.0016 | Down | Unaltered |
| Nomenclature | Glyma ID | Roots | Regulation | ||||
|---|---|---|---|---|---|---|---|
| Normal | p-value | Drought | p-value | DT51 | MTD720 | ||
| GmNAC006 | Glyma02g07700.1 | −1.5 | 0.1098 | 3.0 | 0.0386 | Unaltered | Down |
| GmNAC011 | Glyma02g26480.1 | 2.0 | 0.0707 | 76.6 | 0.0008 | Up | Down |
| GmNAC019 | Glyma04g38990.1 | 1.3 | 0.2279 | 23.3 | 0.0016 | Unaltered | Down |
| GmNAC085 | Glyma12g22880.1 | 2.3 | 0.0119 | 2.1 | 0.0467 | Up | Up |
| GmNAC092 | Glyma12g35000.1 | 2.4 | 0.0349 | 11.3 | 0.0045 | Up | Unaltered |
| GmNAC095 | Glyma13g05540.1 | 3.3 | 0.0129 | 4.2 | 0.0438 | Up | Up |
| GmNAC101 | Glyma13g35550.1 | 3.8 | 0.0002 | 2.0 | 0.0101 | Up | Up |
| GmNAC109 | Glyma14g24220.1 | 4.8 | 0.0075 | 6.9 | 0.0001 | Up | Up |
| GmNAC148 | Glyma20g04400.1 | 2.6 | 0.0039 | 4.2 | 0.1324 | Down | Down |
| GmNAC017 | Glyma04g33270.1 | 5.4 | 0.0443 | 9.8 | 0.0085 | Unaltered | Unaltered |
| GmNAC022 | Glyma04g42800.1 | 2.9 | 0.1947 | 14.3 | 0.0040 | Up | Up |
| GmNAC027 | Glyma05g24910.1 | −1.3 | 0.1492 | 20.2 | 0.0194 | Up | Down |
| GmNAC071 | Glyma10g04350.1 | 2.0 | 0.0952 | 9.1 | 0.0061 | Unaltered | Down |
| GmNAC083 | Glyma12g13710.1 | 1.6 | 0.1714 | 4.4 | 0.0161 | Unaltered | Down |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Thao, N.P.; Thu, N.B.A.; Hoang, X.L.T.; Ha, C.V.; Tran, L.-S.P. Differential Expression Analysis of a Subset of Drought-Responsive GmNAC Genes in Two Soybean Cultivars Differing in Drought Tolerance. Int. J. Mol. Sci. 2013, 14, 23828-23841. https://doi.org/10.3390/ijms141223828
Thao NP, Thu NBA, Hoang XLT, Ha CV, Tran L-SP. Differential Expression Analysis of a Subset of Drought-Responsive GmNAC Genes in Two Soybean Cultivars Differing in Drought Tolerance. International Journal of Molecular Sciences. 2013; 14(12):23828-23841. https://doi.org/10.3390/ijms141223828
Chicago/Turabian StyleThao, Nguyen Phuong, Nguyen Binh Anh Thu, Xuan Lan Thi Hoang, Chien Van Ha, and Lam-Son Phan Tran. 2013. "Differential Expression Analysis of a Subset of Drought-Responsive GmNAC Genes in Two Soybean Cultivars Differing in Drought Tolerance" International Journal of Molecular Sciences 14, no. 12: 23828-23841. https://doi.org/10.3390/ijms141223828
APA StyleThao, N. P., Thu, N. B. A., Hoang, X. L. T., Ha, C. V., & Tran, L.-S. P. (2013). Differential Expression Analysis of a Subset of Drought-Responsive GmNAC Genes in Two Soybean Cultivars Differing in Drought Tolerance. International Journal of Molecular Sciences, 14(12), 23828-23841. https://doi.org/10.3390/ijms141223828






