Evaluation of Individual and Combined Applications of Serum Biomarkers for Diagnosis of Hepatocellular Carcinoma: A Meta-Analysis
Abstract
:1. Introduction
2. Results and Discussion
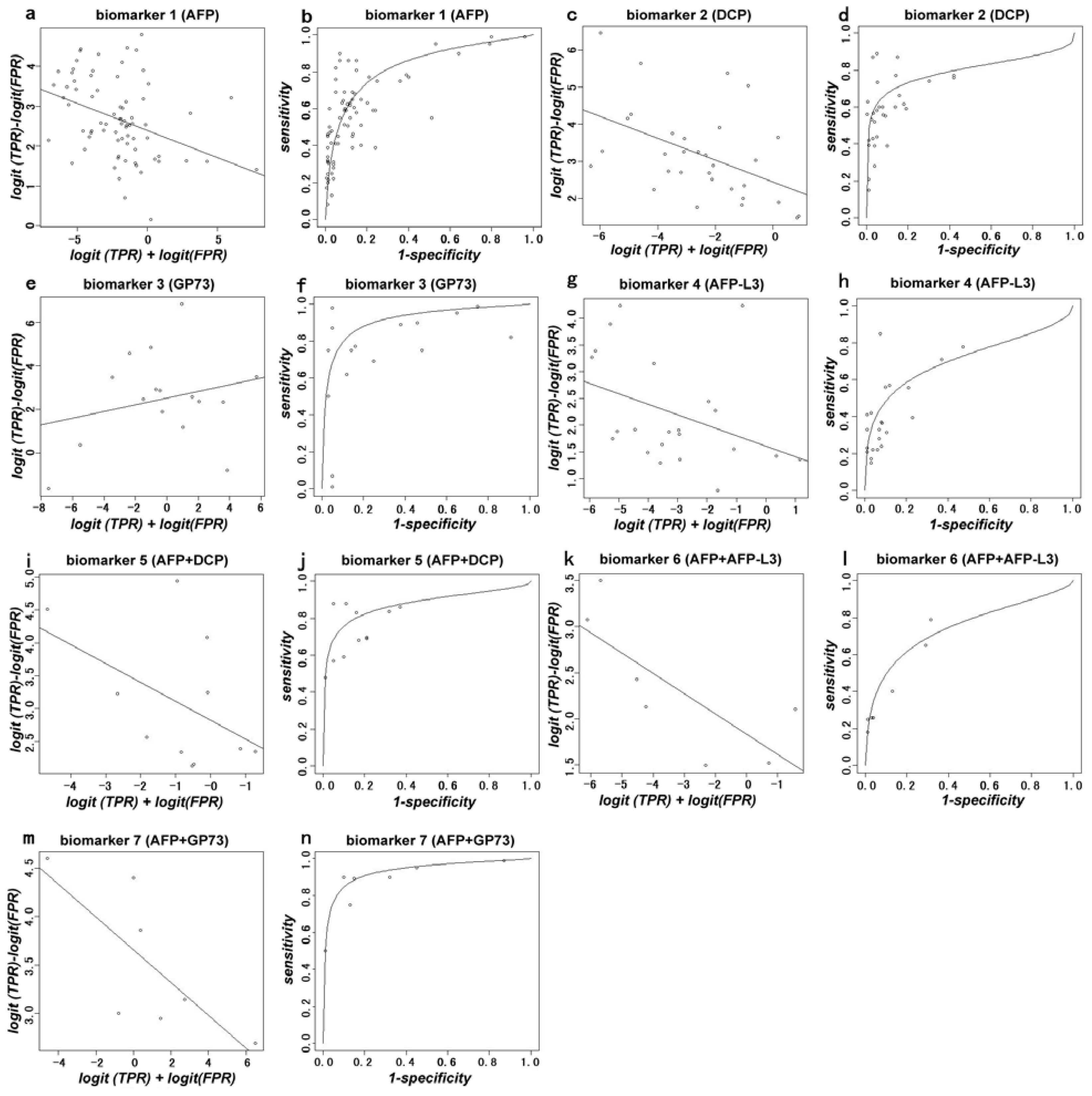
2.1. Results
2.2. Discussion
3. Experimental Section
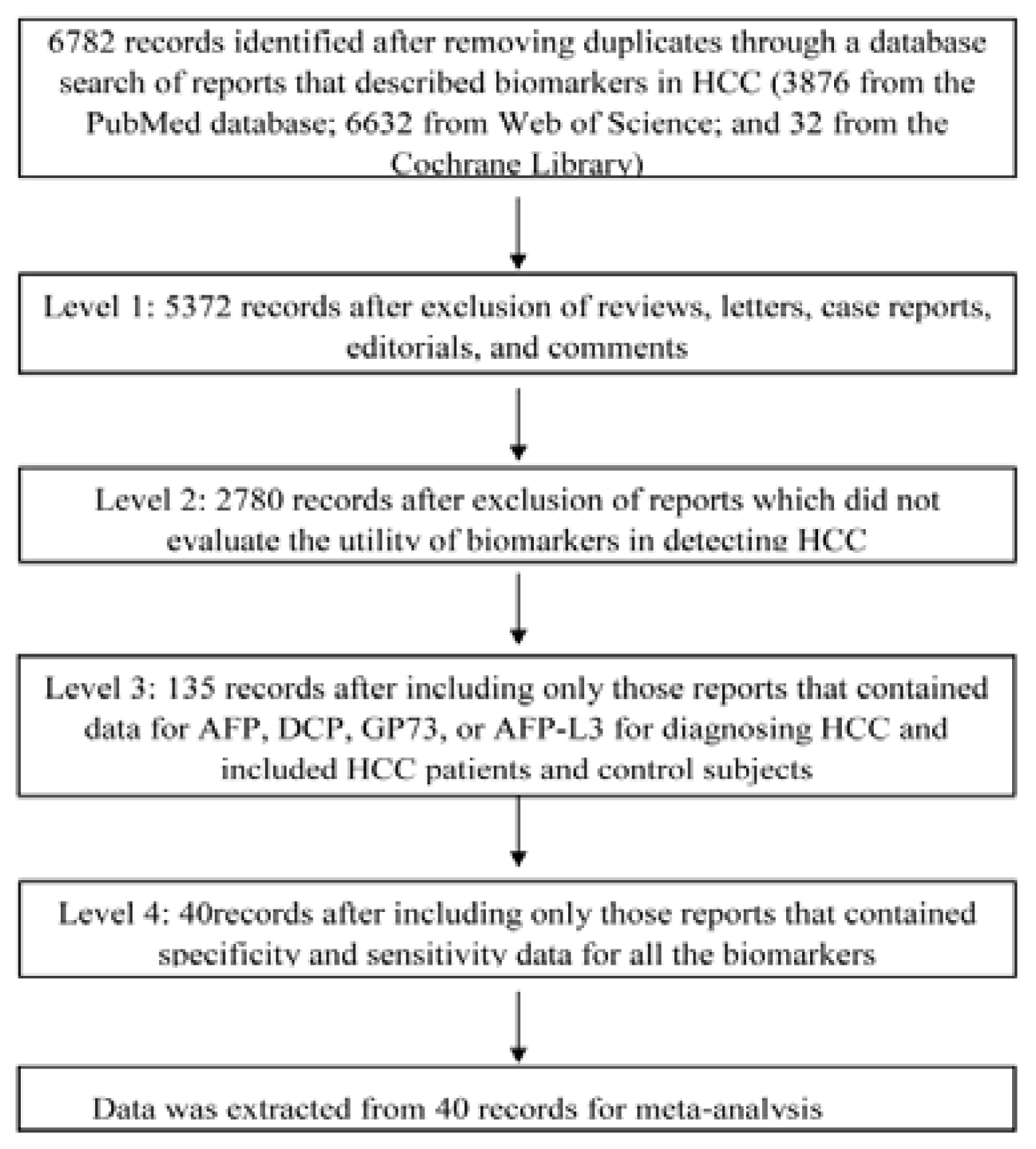
3.1. Identification of Studies
3.2. Literature Screening
3.3. Data Extraction
3.4. Statistical Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Stefaniuk, P.; Cianciara, J.; Wiercinska-Drapalo, A. Present and future possibilities for early diagnosis of hepatocellular carcinoma. World J. Gastroenterol 2010, 16, 418–424. [Google Scholar]
- Oka, H.; Tamori, A.; Kuroki, T.; Kobayashi, K.; Yamamoto, S. Prospective study of alpha-fetoprotein in cirrhotic patients monitored for development of hepatocellular carcinoma. Hepatology 1994, 19, 61–66. [Google Scholar]
- Zoli, M.; Magalotti, D.; Bianchi, G.; Gueli, C.; Marchesini, G.; Pisi, E. Efficacy of a surveillance program for early detection of hepatocellular carcinoma. Cancer 1996, 78, 977–985. [Google Scholar]
- Behne, T.; Copur, M.S. Biomarkers for hepatocellular carcinoma. Int. J. Hepatol 2012. [Google Scholar] [CrossRef]
- Masuzaki, R.; Karp, S.J.; Omata, M. New serum markers of hepatocellular carcinoma. Semin. Oncol 2012, 39, 434–439. [Google Scholar]
- Ba, M.C.; Long, H.; Tang, Y.Q.; Cui, S.Z. GP73 expression and its significance in the diagnosis of hepatocellular carcinoma: A review. Int. J. Clin. Exp. Pathol 2012, 5, 874–881. [Google Scholar]
- Norton, P.A.; Comunale, M.A.; Krakover, J.; Rodemich, L.; Pirog, N.; D’Amelio, A.; Philip, R.; Mehta, A.S.; Block, T.M. N-linked glycosylation of the liver cancer biomarker GP73. J. Cell. Biochem 2008, 104, 136–149. [Google Scholar]
- Leerapun, A.; Suravarapu, S.V.; Bida, J.P.; Clark, R.J.; Sanders, E.L.; Mettler, T.A.; Stadheim, L.M.; Aderca, I.; Moser, C.D.; Nagorney, D.M.; et al. The utility of Lens culinaris agglutinin-reactive alpha-fetoprotein in the diagnosis of hepatocellular carcinoma: Evaluation in a United States referral population. Clin. Gastroenterol. Hepatol 2007, 5, 394–402. [Google Scholar]
- Cedrone, A.; Covino, M.; Caturelli, E.; Pompili, M.; Lorenzelli, G.; Villani, M.R.; Valle, D.; Sperandeo, M.; Rapaccini, G.L.; Gasbarrini, G. Utility of alpha-fetoprotein (AFP) in the screening of patients with virus-related chronic liver disease: Does different viral etiology influence AFP levels in HCC? A study in 350 western patients. Hepato Gastroenterol 2000, 47, 1654–1658. [Google Scholar]
- Wang, C.S.; Lin, C.L.; Lee, H.C.; Chen, K.Y.; Chiang, M.F.; Chen, H.S.; Lin, T.J.; Liao, L.Y. Usefulness of serum des-gamma-carboxy prothrombin in detection of hepatocellular carcinoma. World J. Gastroenterol 2005, 11, 6115–6119. [Google Scholar]
- Durazo, F.A.; Blatt, L.M.; Corey, W.G.; Lin, J.H.; Han, S.; Saab, S.; Busuttil, R.W.; Tong, M.J. Des-gamma-carboxyprothrombin, alpha-fetoprotein and AFP-L3 in patients with chronic hepatitis, cirrhosis and hepatocellular carcinoma. J. Gastroenterol. Hepatol 2008, 23, 1541–1548. [Google Scholar]
- Giannelli, G.; Fransvea, E.; Trerotoli, P.; Beaugrand, M.; Marinosci, F.; Lupo, L.; Nkontchou, G.; Dentico, P.; Antonaci, S. Clinical validation of combined serological biomarkers for improved hepatocellular carcinoma diagnosis in 961 patients. Clin. Chim. Acta 2007, 383, 147–152. [Google Scholar]
- Giannelli, G.; Marinosci, F.; Trerotoli, P.; Volpe, A.; Quaranta, M.; Dentico, P.; Antonaci, S. SCCA antigen combined with alpha-fetoprotein as serologic markers of HCC. Int. J. Cancer 2005, 117, 506–509. [Google Scholar]
- Toyoda, H.; Kumada, T.; Tada, T.; Kaneoka, Y.; Maeda, A.; Kanke, F.; Satomura, S. Clinical utility of highly sensitive Lens culinaris agglutinin-reactive alpha-fetoprotein in hepatocellular carcinoma patients with alpha-fetoprotein <20 ng/mL. Cancer Sci 2011, 102, 1025–1031. [Google Scholar]
- Okuda, H.; Nakanishi, T.; Takatsu, K.; Saito, A.; Hayashi, N.; Watanabe, K.; Magario, N.; Yokoo, T.; Naraki, T. Measurement of serum levels of des-gamma-carboxy prothrombin in patients with hepatocellular carcinoma by a revised enzyme immunoassay kit with increased sensitivity. Cancer 1999, 85, 812–818. [Google Scholar]
- Hsia, C.Y.; Huo, T.I.; Chiang, S.Y.; Lu, M.F.; Sun, C.L.; Wu, J.C.; Lee, P.C.; Chi, C.W.; Lui, W.Y.; Lee, S.D. Evaluation of interleukin-6, interleukin-10 and human hepatocyte growth factor as tumor markers for hepatocellular carcinoma. Eur. J. Surg. Oncol 2007, 33, 208–212. [Google Scholar]
- Hu, J.S.; Wu, D.W.; Liang, S.; Miao, X.Y. GP73, a resident Golgi glycoprotein, is sensibility and specificity for hepatocellular carcinoma of diagnosis in a hepatitis B-endemic Asian population. Med. Oncol 2010, 27, 339–345. [Google Scholar]
- Hussein, M.; Ibrahim, A.; Abdella, H.; Montasser, I.; Hassan, M. Evaluation of serum squamous cell carcinoma antigen as a novel biomarker for diagnosis of hepatocellular carcinoma in Egyptian patients. Indian J. Cancer 2008, 45, 167–172. [Google Scholar]
- Ikoma, J.; Kaito, M.; Ishihara, T.; Nakagawa, N.; Kamei, A.; Fujita, N.; Iwasa, M.; Tamaki, S.; Watanabe, S.; Adachi, Y. Early diagnosis of hepatocellular carcinoma using a sensitive assay for serum des-gamma-carboxy prothrombin: A prospective study. Hepato Gastroenterol 2002, 49, 235–238. [Google Scholar]
- Ertle, J.M.; Wiehert, M.; Küper, R.; Hilgard, P.; Gerken, G.; Schlaak, J. 965 Sensitivity and specificity of Alpha-Feto Protein (Afp), Lens culinaris agglutinin-reactive Afp (Afp-L3) and des-gamma-carboxyprothrombin (Dcp) as tumor markers in patients with hepatocellular carcinoma (Hcc). J. Hepatol 2011, 54, S385. [Google Scholar]
- Yamamoto, K.; Imamura, H.; Matsuyama, Y.; Hasegawa, K.; Beck, Y.; Sugawara, Y.; Makuuchi, M.; Kokudo, N. Significance of alpha-fetoprotein and des-gamma-carboxy prothrombin in patients with hepatocellular carcinoma undergoing hepatectomy. Ann. Surg. Oncol 2009, 16, 2795–2804. [Google Scholar]
- Volk, M.L.; Hernandez, J.C.; Su, G.L.; Lok, A.S.; Marrero, J.A. Risk factors for hepatocellular carcinoma may impair the performance of biomarkers: A comparison of AFP, DCP, and AFP-L3. Cancer Biomark 2007, 3, 79–87. [Google Scholar]
- Mao, Y.; Yang, H.; Xu, H.; Lu, X.; Sang, X.; Du, S.; Zhao, H.; Chen, W.; Xu, Y.; Chi, T.; et al. Golgi protein 73 (GOLPH2) is a valuable serum marker for hepatocellular carcinoma. Gut 2010, 59, 1687–1693. [Google Scholar]
- Yamamoto, K.; Imamura, H.; Matsuyama, Y.; Kume, Y.; Ikeda, H.; Norman, G.L.; Shums, Z.; Aoki, T.; Hasegawa, K.; Beck, Y.; et al. AFP, AFP-L3, DCP, and GP73 as markers for monitoring treatment response and recurrence and as surrogate markers of clinicopathological variables of HCC. J. Gastroenterol 2010, 45, 1272–1282. [Google Scholar]
- Marrero, J.A.; Su, G.L.; Wei, W.; Emick, D.; Conjeevaram, H.S.; Fontana, R.J.; Lok, A.S. Des-gamma carboxyprothrombin can differentiate hepatocellular carcinoma from nonmalignant chronic liver disease in american patients. Hepatology 2003, 37, 1114–1121. [Google Scholar]
- Colombo, M. Screening for cancer in viral hepatitis. Clin. Liver Dis 2001, 5, 109–122. [Google Scholar]
- Nguyen, M.H.; Garcia, R.T.; Simpson, P.W.; Wright, T.L.; Keeffe, E.B. Racial differences in effectiveness of alpha-fetoprotein for diagnosis of hepatocellular carcinoma in hepatitis C virus cirrhosis. Hepatology 2002, 36, 410–417. [Google Scholar]
- Marrero, J.A.; Feng, Z.; Wang, Y.; Nguyen, M.H.; Befeler, A.S.; Roberts, L.R.; Reddy, K.R.; Harnois, D.; Llovet, J.M.; Normolle, D.; et al. Alpha-fetoprotein, des-gamma carboxyprothrombin, and lectin-bound alpha-fetoprotein in early hepatocellular carcinoma. Gastroenterology 2009, 137, 110–118. [Google Scholar]
- El Shafie, M.A.; Fawzy, A.M.; Al Monem, E.A.; Abbass, S.; Zakaria, D.M.; El Baz, S. Golgi Protein 73 (GP73) as a novel serum marker for early detection of hepatocellular carcinoma in egyptian patients. Life Sci. J 2012, 9, 823. [Google Scholar]
- Morota, K.; Nakagawa, M.; Sekiya, R.; Hemken, P.M.; Sokoll, L.J.; Elliott, D.; Chan, D.W.; Dowell, B.L. A comparative evaluation of Golgi protein-73, fucosylated hemopexin, α-fetoprotein, and PIVKA-II in the serum of patients with chronic hepatitis, cirrhosis, and hepatocellular carcinoma. Clin. Chem. Lab. Med 2011, 49, 711–718. [Google Scholar]
- Marrero, J.A.; Romano, P.R.; Nikolaeva, O.; Steel, L.; Mehta, A.; Fimmel, C.J.; Comunale, M.A.; D’Amelio, A.; Lok, A.S.; Block, T.M. GP73, a resident Golgi glycoprotein, is a novel serum marker for hepatocellular carcinoma. J. Hepatol 2005, 43, 1007–1012. [Google Scholar]
- Tong, M.J.; Blatt, L.M.; Kao, V.W. Surveillance for hepatocellular carcinoma in patients with chronic viral hepatitis in the United States of America. J. Gastroenterol. Hepatol 2001, 16, 553–559. [Google Scholar]
- Nomura, F.; Ishijima, M.; Kuwa, K.; Tanaka, N.; Nakai, T.; Ohnishi, K. Serum des-gamma-carboxy prothrombin levels determined by a new generation of sensitive immunoassays in patients with small-sized hepatocellular carcinoma. Am. J. Gastroenterol 1999, 94, 650–654. [Google Scholar]
- Oka, H.; Saito, A.; Ito, K.; Kumada, T.; Satomura, S.; Kasugai, H.; Osaki, Y.; Seki, T.; Kudo, M.; Tanaka, M. Multicenter prospective analysis of newly diagnosed hepatocellular carcinoma with respect to the percentage of Lens culinaris agglutinin-reactive alpha-fetoprotein. J. Gastroenterol. Hepatol 2001, 16, 1378–1383. [Google Scholar]
- Ozkan, H.; Erdal, H.; Tutkak, H.; Karaeren, Z.; Yakut, M.; Yuksel, O.; Koklu, S. Diagnostic and prognostic validity of Golgi protein 73 in hepatocellular carcinoma. Digestion 2011, 83, 83–88. [Google Scholar]
- Peng, Y.C.; Chan, C.S.; Chen, G.H. The effectiveness of serum alpha-fetoprotein level in anti-HCV positive patients for screening hepatocellular carcinoma. Hepato Gastroenterol 1999, 46, 3208–3211. [Google Scholar]
- Porta, C.; de Amici, M.; Quaglini, S.; Paglino, C.; Tagliani, F.; Boncimino, A.; Moratti, R.; Corazza, G.R. Circulating interleukin-6 as a tumor marker for hepatocellular carcinoma. Ann. Oncol 2008, 19, 353–358. [Google Scholar]
- Romeo, R.; Sangiovanni, A.; Iavarone, M.; Vavassori, S.; Della Corte, C.; Colombo, M. Diagnostic value of Lens culinaris agglutinin isoform 3 fraction (AFP-L3%) and des-gamma-carboxy prothrombin (DCP) for the diagnosis of hepatocellular carcinoma in cirrhotic patients. J. Clin. Oncol 2010, 28, 4119. [Google Scholar]
- Sassa, T.; Kumada, T.; Nakano, S.; Uematsu, T. Clinical utility of simultaneous measurement of serum high-sensitivity des-(gamma)-carboxy prothrombin and Lens culinaris agglutinin A-reactive a-fetoprotein in patients with small hepatocellular carcinoma. Eur. J. Gastroenterol. Hepatol 1999, 11, 1387–1392. [Google Scholar]
- Shi, Y.; Chen, J.; Li, L.; Sun, Z.; Zen, L.; Xu, S.; Zhang, Y.; Zhang, L. A study of diagnostic value of Golgi protein GP73 and its genetic assay in primary hepatic carcinoma. Technol. Cancer Res. Treat 2011, 10, 287–294. [Google Scholar]
- Shimauchi, Y.; Tanaka, M.; Kuromatsu, R.; Ogata, R.; Tateishi, Y.; Itano, S.; Ono, N.; Yutani, S.; Nagamatsu, H.; Matsugaki, S. A simultaneous monitoring of Lens culinaris agglutinin A-reactive alpha-fetoprotein and des-gamma-carboxy prothrombin as an early diagnosis of hepatocellular carcinoma in the follow-up of cirrhotic patients. Oncol. Rep 2000, 7, 249–256. [Google Scholar]
- Sterling, R.K.; Jeffers, L.; Gordon, F.; Venook, A.P.; Reddy, K.R.; Satomura, S.; Kanke, F.; Schwartz, M.E.; Sherman, M. Utility of Lens culinaris agglutinin-reactive fraction of alpha-fetoprotein and des-gamma-carboxy prothrombin, alone or in combination, as biomarkers for hepatocellular carcinoma. Clin. Gastroenterol. Hepatol 2009, 7, 104–113. [Google Scholar]
- Sun, Y.; Yang, H.; Mao, Y.; Xu, H.; Zhang, J.; Li, G.; Lu, X.; Sang, X.; Zhao, H.; Zhong, S.; et al. Increased Golgi protein 73 expression in hepatocellular carcinoma tissue correlates with tumor aggression but not survival. J. Gastroenterol. Hepatol 2011, 26, 1207–1212. [Google Scholar]
- Tian, L.; Wang, Y.; Xu, D.; Gui, J.; Jia, X.; Tong, H.; Wen, X.; Dong, Z.; Tian, Y. Serological AFP/Golgi protein 73 could be a new diagnostic parameter of hepatic diseases. Int. J. Cancer 2011, 129, 1923–1931. [Google Scholar]
- Trevisani, F.; D’Intino, P.E.; Morselli-Labate, A.M.; Mazzella, G.; Accogli, E.; Caraceni, P.; Domenicali, M.; de Notariis, S.; Roda, E.; Bernardi, M. Serum alpha-fetoprotein for diagnosis of hepatocellular carcinoma in patients with chronic liver disease: Influence of HBsAg and anti-HCV status. J. Hepatol 2001, 34, 570–575. [Google Scholar]
- Wang, M.; Long, R.E.; Comunale, M.A.; Junaidi, O.; Marrero, J.; di Bisceglie, A.M.; Block, T.M.; Mehta, A.S. Novel fucosylated biomarkers for the early detection of hepatocellular carcinoma. Cancer Epidemiol. Biomark. Prev 2009, 18, 1914–1921. [Google Scholar]
- Li, X.; Wu, K.; Fan, D. Serum Golgi Phosphoprotein 2 level: A better marker than alpha-fetoprotein for diagnosing early hepatocellular carcinoma. Hepatology 2009, 50, 325–325. [Google Scholar]
- Khien, V.V.; Mao, H.V.; Chinh, T.T.; Ha, P.T.; Bang, M.H.; Lac, B.V.; Hop, T.V.; Tuan, N.A.; Don, L.V.; Taketa, K.; et al. Clinical evaluation of lentil lectin-reactive alpha-fetoprotein-L3 in histology-proven hepatocellular carcinoma. Int. J. Biol. Markers 2001, 16, 105–111. [Google Scholar]
- Yoshida, S.; Kurokohchi, K.; Arima, K.; Masaki, T.; Hosomi, N.; Funaki, T.; Murota, M.; Kita, Y.; Watanabe, S.; Kuriyama, S. Clinical significance of Lens culinaris agglutinin-reactive fraction of serum alpha-fetoprotein in patients with hepatocellular carcinoma. Int. J. Oncol 2002, 20, 305–309. [Google Scholar]
- Srivastava, S.; Gopal-Srivastava, R. Biomarkers in cancer screening: A public health perspective. J. Nutr 2002, 132, 2471S–2475S. [Google Scholar]
- Li, D.; Mallory, T.; Satomura, S. AFP-L3: A new generation of tumor marker for hepatocellular carcinoma. Clin. Chim. Acta 2001, 313, 15–19. [Google Scholar]
- Kladney, R.D.; Bulla, G.A.; Guo, L.; Mason, A.L.; Tollefson, A.E.; Simon, D.J.; Koutoubi, Z.; Fimmel, C.J. GP73, a novel Golgi-localized protein upregulated by viral infection. Gene 2000, 249, 53–65. [Google Scholar]
- Taketa, K.; Okada, S.; Win, N.; Hlaing, N.K.; Wind, K.M. Evaluation of tumor markers for the detection of hepatocellular carcinoma in Yangon General Hospital, Myanmar. Acta Med. Okayama 2002, 56, 317–320. [Google Scholar]
- Block, T.M.; Marrero, J.; Gish, R.G.; Sherman, M.; London, W.T.; Srivastava, S.; Wagner, P.D. The degree of readiness of selected biomarkers for the early detection of hepatocellular carcinoma: Notes from a recent workshop. Cancer Biomark 2008, 4, 19–33. [Google Scholar]
- Taketa, K.; Sekiya, C.; Namiki, M.; Akamatsu, K.; Ohta, Y.; Endo, Y.; Kosaka, K. Lectin-reactive profiles of alpha-fetoprotein characterizing hepatocellular carcinoma and related conditions. Gastroenterology 1990, 99, 508–518. [Google Scholar]
- Wang, S.S.; Lu, R.H.; Lee, F.Y.; Chao, Y.; Huang, Y.S.; Chen, C.C.; Lee, S.D. Utility of lentil lectin affinity of alpha-fetoprotein in the diagnosis of hepatocellular carcinoma. J. Hepatol 1996, 25, 166–171. [Google Scholar]
- Sato, Y.; Nakata, K.; Kato, Y.; Shima, M.; Ishii, N.; Koji, T.; Taketa, K.; Endo, Y.; Nagataki, S. Early recognition of hepatocellular carcinoma based on altered profiles of alpha-fetoprotein. N. Engl. J. Med 1993, 328, 1802–1806. [Google Scholar]
- Nouso, K.; Kobayashi, Y.; Nakamura, S.; Kobayashi, S.; Takayama, H.; Toshimori, J.; Kuwaki, K.; Hagihara, H.; Onishi, H.; Miyake, Y.; et al. Prognostic importance of fucosylated alpha-fetoprotein in hepatocellular carcinoma patients with low alpha-fetoprotein. J. Gastroenterol. Hepatol 2011, 26, 1195–1200. [Google Scholar]
- Liebman, H.A.; Furie, B.C.; Tong, M.J.; Blanchard, R.A.; Lo, K.J.; Lee, S.D.; Coleman, M.S.; Furie, B. Des-gamma-carboxy (abnormal) prothrombin as a serum marker of primary hepatocellular carcinoma. N. Engl. J. Med 1984, 310, 1427–1431. [Google Scholar]
- Ishii, M.; Gama, H.; Chida, N.; Ueno, Y.; Shinzawa, H.; Takagi, T.; Toyota, T.; Takahashi, T.; Kasukawa, R. Simultaneous measurements of serum alpha-fetoprotein and protein induced by vitamin K absence for detecting hepatocellular carcinoma. South Tohoku District Study Group. Am. J. Gastroenterol 2000, 95, 1036–1040. [Google Scholar]
- Lamerz, R.; Runge, M.; Stieber, P.; Meissner, E. Use of serum PIVKA-II (DCP) determination for differentiation between benign and malignant liver diseases. Anticancer Res 1999, 19, 2489–2493. [Google Scholar]
- Mita, Y.; Aoyagi, Y.; Yanagi, M.; Suda, T.; Suzuki, Y.; Asakura, H. The usefulness of determining des-gamma-carboxy prothrombin by sensitive enzyme immunoassay in the early diagnosis of patients with hepatocellular carcinoma. Cancer 1998, 82, 1643–1648. [Google Scholar]
- Aoyagi, Y.; Oguro, M.; Yanagi, M.; Mita, Y.; Suda, T.; Suzuki, Y.; Hata, K.; Ichii, K.; Asakura, H. Clinical significance of simultaneous determinations of alpha-fetoprotein and des-gamma-carboxy prothrombin in monitoring recurrence in patients with hepatocellular carcinoma. Cancer 1996, 77, 1781–1786. [Google Scholar]
- Izuno, K.; Fujiyama, S.; Yamasaki, K.; Sato, M.; Sato, T. Early detection of hepatocellular carcinoma associated with cirrhosis by combined assay of des-gamma-carboxy prothrombin and alpha-fetoprotein: A prospective study. Hepato Gastroenterol 1995, 42, 387–393. [Google Scholar]
- Grazi, G.L.; Mazziotti, A.; Legnani, C.; Jovine, E.; Miniero, R.; Gallucci, A.; Palareti, G.; Gozzetti, G. The role of tumor markers in the diagnosis of hepatocellular carcinoma, with special reference to the des-gamma-carboxy prothrombin. Liver Transplant. Surg 1995, 1, 249–255. [Google Scholar]
- Okuda, H.; Nakanishi, T.; Takatsu, K.; Saito, A.; Hayashi, N.; Yamamoto, M.; Takasaki, K.; Nakano, M. Comparison of clinicopathological features of patients with hepatocellular carcinoma seropositive for alpha-fetoprotein alone and those seropositive for des-gamma-carboxy prothrombin alone. J. Gastroenterol. Hepatol 2001, 16, 1290–1296. [Google Scholar]
- Hamamura, K.; Shiratori, Y.; Shiina, S.; Imamura, M.; Obi, S.; Sato, S.; Yoshida, H.; Omata, M. Unique clinical characteristics of patients with hepatocellular carcinoma who present with high plasma des-gamma-carboxy prothrombin and low serum alpha-fetoprotein. Cancer 2000, 88, 1557–1564. [Google Scholar]
- Willyard, C. Researchers look for “sweet” method to diagnose cancer. Nat. Med 2007, 13, 1107–1267. [Google Scholar]
- Zhou, Y.; Yin, X.; Ying, J.; Zhang, B. Golgi protein 73 versus alpha-fetoprotein as a biomarker for hepatocellular carcinoma: A diagnostic meta-analysis. BMC Cancer 2012, 12. [Google Scholar] [CrossRef]
- Littenberg, B.; Moses, L.E. Estimating diagnostic accuracy from multiple conflicting reports: A new meta-analytic method. Med Decis Making 1993, 13, 313–321. [Google Scholar]
| Database | Search criteria | Filters/Limits |
|---|---|---|
| PubMed | Search ((hepatocellular carcinoma [MeSH Terms]) and biological markers [MeSH Terms]) and (“1999” [Date—Publication]: “2012” [Date—Publication]) | Humans; English |
| Web of Science | (hepatoma * or liver cell neoplasm * or hepatocellular neoplasm * or liver cell cancer * or hepatocellular cancer * or liver cell tumo * or hepatocellular tumo * or liver cell carcinom * or hepatocellular carcinom *) and (Biomarker * or Biological Marker * or Biologic Marker * or Biochemical Marker * or Serum Marker * or Clinical Marker *) | Publication date: 1999–present and language: English |
| Cochrane Library | #1 MeSH descriptor: [Carcinoma, Hepatocellular] explode all trees #2 MeSH descriptor: [Biological Markers] explode all trees | Dates: from 1999–2012 |
| Study | Country/District | Publication Year | Number | Biomarker | Test Methods | Sensitivity | Specificity | Cutoff | Journal |
|---|---|---|---|---|---|---|---|---|---|
| Leerapun [8] | USA | 2007 | 52 | AFP-L3 | LiBASys | 0.710 | 0.630 | 10% | Clin. Gastroenterol. Hepatol. |
| 0.330 | 1.000 | 35% | |||||||
| Cedrone [9] | Italy | 2000 | 74 | AFP | ELISA | 0.200 | 0.990 | 200 ug/L | Hepato Gastroenterol. |
| 0.550 | 0.880 | 20 ug/L | |||||||
| Wang [10] | Taiwan, China | 2005 | 61 | AFP | ELISA | 0.590 | 0.770 | 20 ng/mL | World J. Gastroenterol. |
| DCP | EIA | 0.770 | 0.860 | 40 mAU/mL | |||||
| AFP + DCP | ELISA + EIA | 0.836 | 0.682 | 20 ng/mL/40 mAU/mL | |||||
| Durazo [11] | USA | 2008 | 144 | AFP | IAUEC | 0.690 | 0.870 | 25 ng/mL | J. Gastroenterol. Hepatol. |
| DCP | ELISA | 0.870 | 0.850 | 84 mAU/mL | |||||
| AFP-L3 | LAEC | 0.560 | 0.900 | 10% | |||||
| Gianluigi [12] | Italy | 2007 | 499 | AFP | ELISA | 0.410 | 0.940 | 18.8 ng/mL/20.5 IU/mL | Clin. Chim. Acta |
| Giannelli [13] | Italy | 2005 | 120 | AFP | ELISA | 0.450 | 0.870 | 12.6 ng/mL/13.7 IU/mL | Int. J. Cancer |
| Toyoda [14] | Japan | 2011 | 270 | AFP-L3 | μTAS | 0.394 | 0.770 | 7% | Cancer Sci. |
| Okuda [15] | Japan | 1999 | 60 | DCP | EIA | 0.617 | 0.821 | 30 mAU/mL | Cancer |
| 0.600 | 0.863 | 35 mAU/mL | |||||||
| 0.600 | 0.923 | 40 mAU/mL | |||||||
| 0.583 | 0.949 | 45 mAU/mL | |||||||
| 0.567 | 0.966 | 50 mAU/mL | |||||||
| 0.567 | 0.966 | 55 mAU/mL | |||||||
| 0.533 | 0.974 | 60 mAU/mL | |||||||
| Hsia [16] | Taiwan, China | 2007 | 26 | AFP | ELISA | 0.620 | 0.880 | 20 ng/mL | Eur. J. Surg. Oncol. |
| Hu [17] | China | 2010 | 31 | AFP | ELISA | 0.480 | 0.970 | 36 ug/L | Med. Oncol. |
| AFP + GP73 | ELISA/western blotting | 0.770 | 0.840 | 7.4 RU | |||||
| Hussein [18] | Egypt | 2008 | 49 | AFP | ELISA | 0.900 | 0.930 | 7.7 ng/mL | Indian J. Cancer |
| Ikoma [19] | Japan | 2002 | 63 | AFP | ELISA | 0.510 | 0.830 | 20 ng/mL | Hepato Gastroenterol. |
| DCP | ELISA | 0.390 | 0.960 | 16 mAU/mL | |||||
| AFP + DCP | ELISA/ELISA | 0.830 | 0.840 | 20/16 | |||||
| Ertle [20] | Germany | 2011 | 170 | AFP | ELISA | 0.310 | 0.960 | 200 ng/mL | J. Hepatol. |
| DCP | LiBASys | 0.600 | 0.940 | 7.5 ng/mL | |||||
| AFP-L3 | LiBASys | 0.410 | 0.990 | 10% | |||||
| Yamamoto [21] | Japan | 2009 | 714 | AFP | immunometric assay | 0.649 | 0.829 | 11 ng/mL | Ann. Surg. Oncol. |
| 0.608 | 0.861 | 13 ng/mL | |||||||
| 0.513 | 0.908 | 20 ng/mL | |||||||
| 0.304 | 0.986 | 100 ng/mL | |||||||
| 0.247 | 0.990 | 200 ng/mL | |||||||
| DCP | two-step enzyme immunoassay | 0.734 | 0.947 | 20 mAU/mL | |||||
| 0.628 | 0.994 | 30 mAU/mL | |||||||
| 0.559 | 0.998 | 40 mAU/mL | |||||||
| 0.419 | 1.000 | 100 mAU/mL | |||||||
| 0.391 | 1.000 | 125 mAU/mL | |||||||
| Volk [22] | USA | 2007 | 84 | AFP | IAUEC | 0.860 | 0.930 | 150 mAU/mL | Cancer Biomark. |
| 0.690 | 0.910 | 23 ng/mL | |||||||
| AFP-L3 | IAUEC | 0.570 | 0.880 | 3% | |||||
| AFP + DCP | IAUEC/ELISA | 0.880 | 0.890 | 23 ng/mL/150 mAU/mL | |||||
| Mao [23] | China | 2010 | 789 | AFP + GP73 | ELISA/Immunoblot | 0.892 | 0.852 | 35 ng/mL/8.5 RU | Gut |
| GP73 | Immunoblot | 0.750 | 0.970 | 8.5 RU | |||||
| AFP | ELISA/Immunoblot | 0.580 | 0.850 | 35 ug/L | |||||
| Yamamoto [24] | Japan | 2010 | 96 | AFP | ELISA | 0.400 | 0.830 | 15 ng/mL | J. Gastroenterol. |
| 0.390 | 0.870 | 20 ng/mL | |||||||
| 0.280 | 0.960 | 124 ng/mL | |||||||
| 0.220 | 0.960 | 200 ng/mL | |||||||
| DCP | Immunoblot | 0.770 | 0.580 | 20 mAU/mL | |||||
| 0.590 | 0.810 | 30 mAU/mL | |||||||
| 0.550 | 0.910 | 40 mAU/mL | |||||||
| 0.520 | 0.960 | 60 mAU/mL | |||||||
| AFP-L3 | IAUEC | 0.240 | 0.920 | 5% | |||||
| 0.220 | 0.960 | 10% | |||||||
| 0.170 | 0.970 | 15% | |||||||
| 0.150 | 0.970 | 20% | |||||||
| AFP + DCP | Immunoblot | 0.690 | 0.790 | 20 ng/mL/40 mAU/mL | |||||
| 0.680 | 0.830 | 20 ng/mL/60 mAU/mL | |||||||
| 0.590 | 0.900 | 400 ng/mL/40 mAU/mL | |||||||
| 0.570 | 0.950 | 400 ng/mL/60 mAU/mL | |||||||
| AFP + AFP-L3 | Immunoblot/IAUEC | 0.400 | 0.870 | 20 ng/mL/irrespective | |||||
| 0.260 | 0.960 | 20–400 ng/mL/10% | |||||||
| 0.260 | 0.970 | 20–400 ng/mL/15% | |||||||
| 0.180 | 0.990 | 400 ng/mL/irrespective | |||||||
| Marrero [25] | USA | 2003 | 55 | AFP | IAUEC | 0.770 | 0.790 | 11 ng/mL | Hepatology |
| 0.680 | 0.860 | 20 ng/mL | |||||||
| 0.470 | 0.980 | 100 ng/mL | |||||||
| 0.340 | 1.000 | 400 ng/mL | |||||||
| DCP | ELISA | 0.890 | 0.950 | 125 mAU/mL | |||||
| 0.870 | 0.970 | 150 mAU/mL | |||||||
| AFP + DCP | IAUEC/ELISA | 0.880 | 0.950 | logAFP + 4.6 * logDCP | |||||
| Colombo [26] | Italy | 2001 | 55 | AFP | ELISA | 0.390 | 0.760 | 20 ng/ml | Clin. Liver Dis. |
| 0.130 | 0.970 | 100 ng/mL | |||||||
| Nguyen [27] | USA | 2002 | 163 | AFP | ELISA | 0.784 | 0.611 | 10 ng/mL | Hepatology |
| 0.630 | 0.799 | 20 ng/mL | |||||||
| 0.506 | 0.893 | 50 ng/mL | |||||||
| 0.414 | 0.973 | 100 ng/mL | |||||||
| 0.321 | 1.000 | 200 ng/mL | |||||||
| Marrero [28] | USA | 2009 | 419 | AFP | LiBASys | 0.590 | 0.900 | 20 ng/mL | Gastroenterology |
| DCP | ELISA | 0.740 | 0.700 | 150 mAU/mL | |||||
| AFP-L3 | LiBASys | 0.420 | 0.970 | 10% | |||||
| AFP + DCP | ELISA + LiBASys | 0.860 | 0.630 | 20 ng/mL/150 mAU/mL | |||||
| Shafie [29] | Egypt | 2012 | 31 | AFP | ELISA | 0.810 | 0.850 | 32.64 ng/mL | Life Sci. J. |
| 0.770 | 0.600 | 28.51 ng/mL | |||||||
| AFP + GP73 | ELISA | 0.870 | 0.950 | 32.64 ng/mL/7.62 ng/mL | |||||
| 0.900 | 0.900 | 28.51 ng/mL/7.62 ng/mL | |||||||
| Morota [30] | Japan | 2011 | UN | AFP | ELISA | 0.630 | 0.920 | 15.3 ug/L | Clin. Chem. Lab. Med. |
| AFP + GP73 | ELISA | 0.890 | 0.620 | 94.7 ug/L | |||||
| Marrero [31] | USA | 2005 | 144 | AFP | ELISA | 0.300 | 0.960 | 99 ug/L | J. Hepatol. |
| AFP + GP73 | ELISA/Immunoblot | 0.690 | 0.750 | 10.0 RU | |||||
| Tong [32] | USA | 2001 | 31 | AFP | ELISA | 0.410 | 0.950 | 24 ng/mL | J. Gastroenterol. Hepatol. |
| 0.410 | 0.940 | 21 ng/mL | |||||||
| 0.450 | 0.940 | 19 ng/mL | |||||||
| 0.550 | 0.930 | 16 ng/mL | |||||||
| 0.590 | 0.910 | 13 ng/mL | |||||||
| 0.860 | 0.890 | 11 ng/mL | |||||||
| 0.860 | 0.850 | 8 ng/mL | |||||||
| Nomura [33] | Japan | 1999 | 36 | AFP | ELISA | 0.590 | 0.760 | 20 ng/mL | Am. J. Gastroenterol. |
| DCP | Immunoblot | 0.280 | 0.960 | 40 mAU/mL | |||||
| AFP-L3 | ECLIA | 0.220 | 0.940 | 10% | |||||
| Oka [34] | Japan | 2001 | 388 | AFP | ELISA | 0.550 | 0.490 | 20 ng/mL | J. Gastroenterol. Hepatol. |
| AFP-L3 | LAEC | 0.280 | 0.930 | 10% | |||||
| 0.210 | 0.990 | 15% | |||||||
| Ozkan [35] | Turkey | 2011 | 75 | AFP | ELISA | 0.820 | 0.950 | 4.36 ug/L | Digestion |
| 0.690 | 0.950 | 13 ng/mL | |||||||
| 0.760 | 0.950 | 8.46 ng/mL | |||||||
| 0.600 | 0.980 | 20 ng/mL | |||||||
| 0.460 | 1.000 | 100 ng/mL | |||||||
| 0.390 | 1.000 | 200 ng/mL | |||||||
| AFP + GP73 | ELISA | 0.070 | 0.950 | 20 ng/mL/2.36 ug/L | |||||
| 0.820 | 0.090 | 20 ng/mL/0.078 ng/mL | |||||||
| 0.000 | 0.950 | 20 ng/mL/24.43 ng/mL | |||||||
| Peng [36] | Taiwan, China | 1999 | 205 | AFP | ELISA | 0.450 | 1.000 | 200 ug/L | Hepato Gastroenterol. |
| 0.650 | 0.870 | 20 ug/L | |||||||
| Porta [37] | Italy | 2008 | 30 | AFP | ELISA | 0.630 | 0.880 | 12.8 ng/mL | Ann. Oncol. |
| Romeo [38] | Italy | 2010 | 86 | AFP | μ TAS | 0.465 | 0.868 | 20 ng/mL | J. Clin. Oncol. |
| DCP | μ TAS | 0.663 | 0.842 | 0.5 ng/mL | |||||
| 0.558 | 0.921 | 0.75 ng/mL | |||||||
| 0.209 | 1.000 | 2.5 ng/mL | |||||||
| 0.151 | 1.000 | 7.5 ng/mL | |||||||
| AFP-L3 | μ TAS | 0.779 | 0.526 | 5 ng/ml | |||||
| 0.558 | 0.789 | 7 ng/mL | |||||||
| 0.314 | 0.895 | 10 ng/mL | |||||||
| AFP + DCP | μ TAS | 0.698 | 0.789 | 20 ng/mL + 0.75 ng/mL | |||||
| AFP + AFP-L3 | μ TAS | 0.651 | 0.711 | 20 ng/mL + 10 ng/mL | |||||
| 0.791 | 0.684 | 20 ng/mL + 1010 ng/mL | |||||||
| Sassa [39] | Japan | 1999 | 61 | AFP | ELISA | 0.080 | 1.000 | 200 ng/mL | Eur. J. Gastroenterol. Hepatol. |
| DCP | immunoassay | 0.440 | 0.950 | 40 mAU/mL | |||||
| AFP-L3 | LAEC | 0.230 | 0.990 | 10% | |||||
| AFP + DCP | ELISA/immunoassay | 0.480 | 0.990 | 200ng/mL/40 mAU/mL | |||||
| AFP + AFP-L3 | ELISA/LAEC | 0.250 | 0.990 | 200 ng/mL/10% | |||||
| Shi [40] | China | 2011 | 73 | AFP | ELISA | 0.750 | 0.750 | 400 ug/L | Technol. Cancer Res. Treat |
| AFP + GP73 | ELISA | 0.980 | 0.950 | 400 ug/L/100.0 ug/L | |||||
| Shimauch [41] | Japan | 1999 | 21 | AFP-L3 | Immunoblot | 0.330 | 0.930 | 10% | Oncol. Rep. |
| DCP | Immunoblot | 0.430 | 0.970 | 40mAU/mL | |||||
| Sterling [42] | USA | 2009 | 74 | AFP | LiBASys | 0.990 | 0.200 | 200 ng/mL | Clin. Gastroenterol. Hepatol. |
| 0.608 | 0.711 | 20 ng/mL | |||||||
| DCP | LiBASys | 0.760 | 0.580 | 40 mAU/mL | |||||
| LiBASys | 0.390 | 0.900 | 7.5 ng/mL | ||||||
| AFP+DCP | LiBASys | 0.703 | 0.634 | 20 ng/mL/7.5 ng/mL | |||||
| AFP + AFP-L3 | LiBASys | 0.689 | 0.664 | 20 ng/mL/10 | |||||
| AFP-L3 | LiBASys | 0.365 | 0.916 | 10% | |||||
| Sun [43] | China | 2009 | 79 | AFP-L3 | ACSC method | 0.848 | 0.925 | 10 | J. Gastroenterol. Hepatol. |
| Tian [44] | China | 2010 | 153 | AFP | ELISA | 0.950 | 0.470 | 13.6 ug/L | Int. J. Cancer |
| AFP + GP73 | ELISA | 0.750 | 0.520 | 113.8 ug/L | |||||
| Trevisani [45] | Italy | 2001 | 170 | AFP | ELISA | 0.600 | 0.906 | 20 ug/L | J. Hepatol. |
| 0.624 | 0.894 | 16 ug/L | |||||||
| 0.312 | 0.988 | 100 ug/L | |||||||
| 0.224 | 0.994 | 200 ug/L | |||||||
| 0.171 | 0.994 | 400 ug/L | |||||||
| Wang [46] | USA | 2009 | 164 | AFP | ELISA | 0.950 | 0.210 | NK | Cancer Epidemiol. Biomark. Prev. |
| 0.500 | 0.980 | NK | |||||||
| 0.750 | 0.640 | NK | |||||||
| 0.900 | 0.360 | NK | |||||||
| 0.950 | 0.210 | NK | |||||||
| 1.000 | 0.040 | NK | |||||||
| GP73 | Immunoblot | 0.500 | 0.970 | NK | |||||
| 0.750 | 0.860 | NK | |||||||
| 0.900 | 0.540 | NK | |||||||
| 0.950 | 0.350 | NK | |||||||
| 1.000 | 0.250 | NK | |||||||
| AFP + GP73 | ELISA/Immunoblot | 0.500 | 0.990 | NK | |||||
| 0.750 | 0.870 | NK | |||||||
| 0.900 | 0.680 | NK | |||||||
| 0.950 | 0.550 | NK | |||||||
| 1.000 | 0.130 | NK | |||||||
| LI [47] | China | 2009 | 50 | AFP | ELISA | 0.250 | 0.970 | 11.23 ng/mL | Hepatology |
| AFP + GP73 | Immunoblot | 0.620 | 0.880 | 14.37 RU | |||||
| Biomarker | Number of Studies | AUC |
|---|---|---|
| 1 (AFP) | 35 | 0.835 |
| 2 (DCP) | 15 | 0.797 |
| 3 (GP73) | 9 | 0.914 |
| 4 (AFP-L3) | 15 | 0.710 |
| 5 (AFP+DCP) | 8 | 0.874 |
| 6 (AFP+AFP-L3) | 3 | 0.748 |
| 7 (AFP+GP73) | 3 | 0.932 |
| Estimate | Std. Error | t Value | Pr (>|t|) | |
|---|---|---|---|---|
| (Intercept) | 2.39257 | 0.15197 | 15.744 | <2 × 10−16 |
| S.Biomarker.1 | 0.13594 | 0.04532 | 2.999 | 0.00314 |
| S.Biomarker.2 | 0.29219 | 0.10222 | 2.859 | 0.00483 |
| S.Biomarker.3 | 0.15326 | 0.08245 | 1.859 | 0.06492 |
| S.Biomarker.4 | −0.19643 | 0.12226 | −1.607 | 0.11012 |
| S.Biomarker.5 | −0.28662 | 0.20797 | −1.378 | 0.17009 |
| S.Biomarker.6 | 0.21913 | 0.17122 | 1.280 | 0.20250 |
| S.Biomarker.7 | −0.16597 | 0.13200 | 1.257 | 0.21050 |
| Biomarker.2 | 0.08054 | 0.36171 | 0.223 | 0.82409 |
| Biomarker.3 | 0.13195 | 0.31404 | 0.420 | 0.67494 |
| Biomarker.4 | −0.78803 | 0.47604 | −1.655 | 0.09983 |
| Biomarker.5 | 0.43157 | 0.40973 | 1.053 | 0.29381 |
| Biomarker.6 | 0.77644 | 0.70680 | 1.099 | 0.27364 |
| Biomarker.7 | 1.21688 | 0.48657 | 2.501 | 0.01341 |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hu, B.; Tian, X.; Sun, J.; Meng, X. Evaluation of Individual and Combined Applications of Serum Biomarkers for Diagnosis of Hepatocellular Carcinoma: A Meta-Analysis. Int. J. Mol. Sci. 2013, 14, 23559-23580. https://doi.org/10.3390/ijms141223559
Hu B, Tian X, Sun J, Meng X. Evaluation of Individual and Combined Applications of Serum Biomarkers for Diagnosis of Hepatocellular Carcinoma: A Meta-Analysis. International Journal of Molecular Sciences. 2013; 14(12):23559-23580. https://doi.org/10.3390/ijms141223559
Chicago/Turabian StyleHu, Bin, Xiaohui Tian, Jie Sun, and Xiangjun Meng. 2013. "Evaluation of Individual and Combined Applications of Serum Biomarkers for Diagnosis of Hepatocellular Carcinoma: A Meta-Analysis" International Journal of Molecular Sciences 14, no. 12: 23559-23580. https://doi.org/10.3390/ijms141223559




