The Changes in mGluR2 and mGluR7 Expression in Rat Medial Vestibular Nucleus and Flocculus Following Unilateral Labyrinthectomy
Abstract
:1. Introduction
2. Results and Discussion
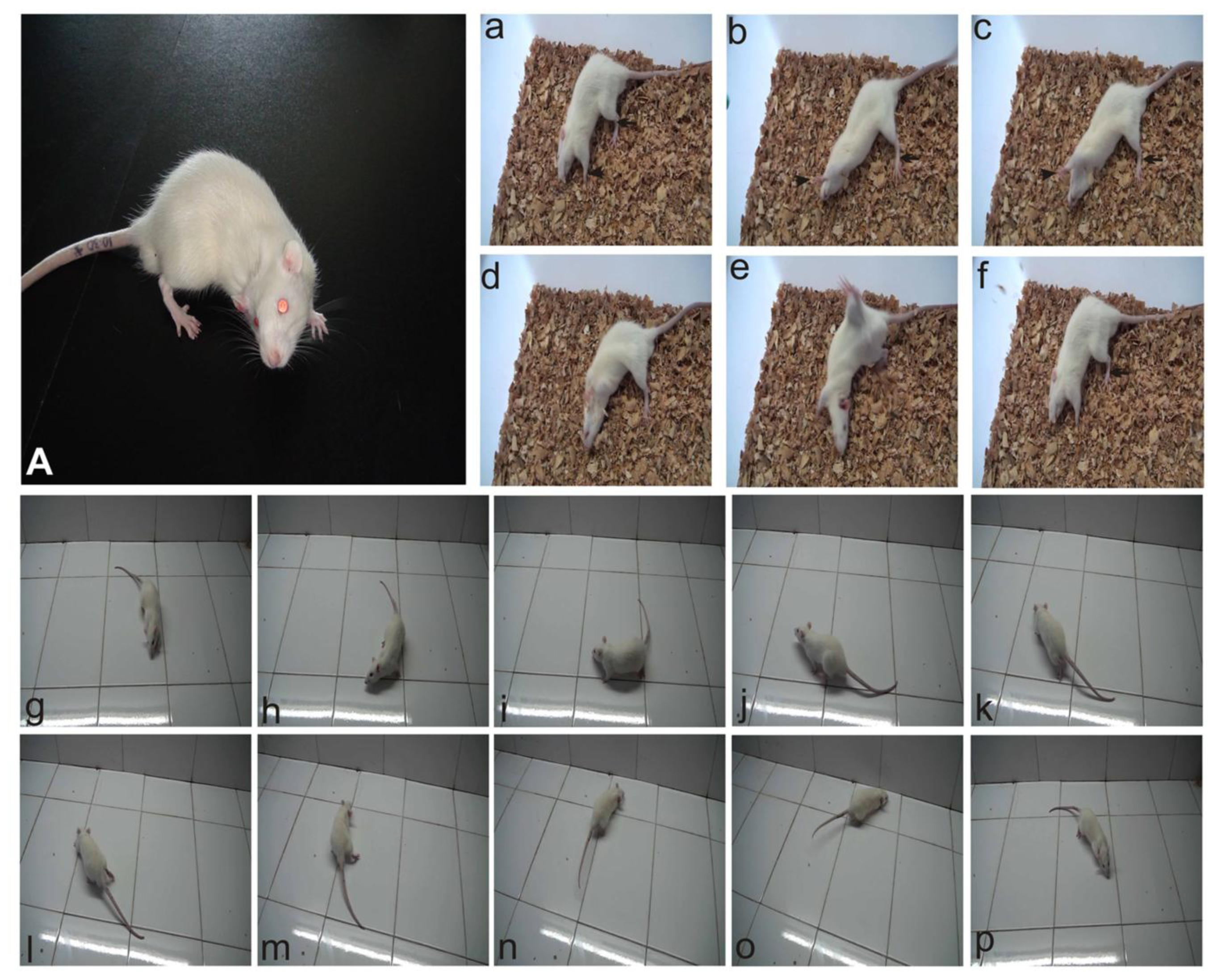
2.1. Behavior Observation after UL
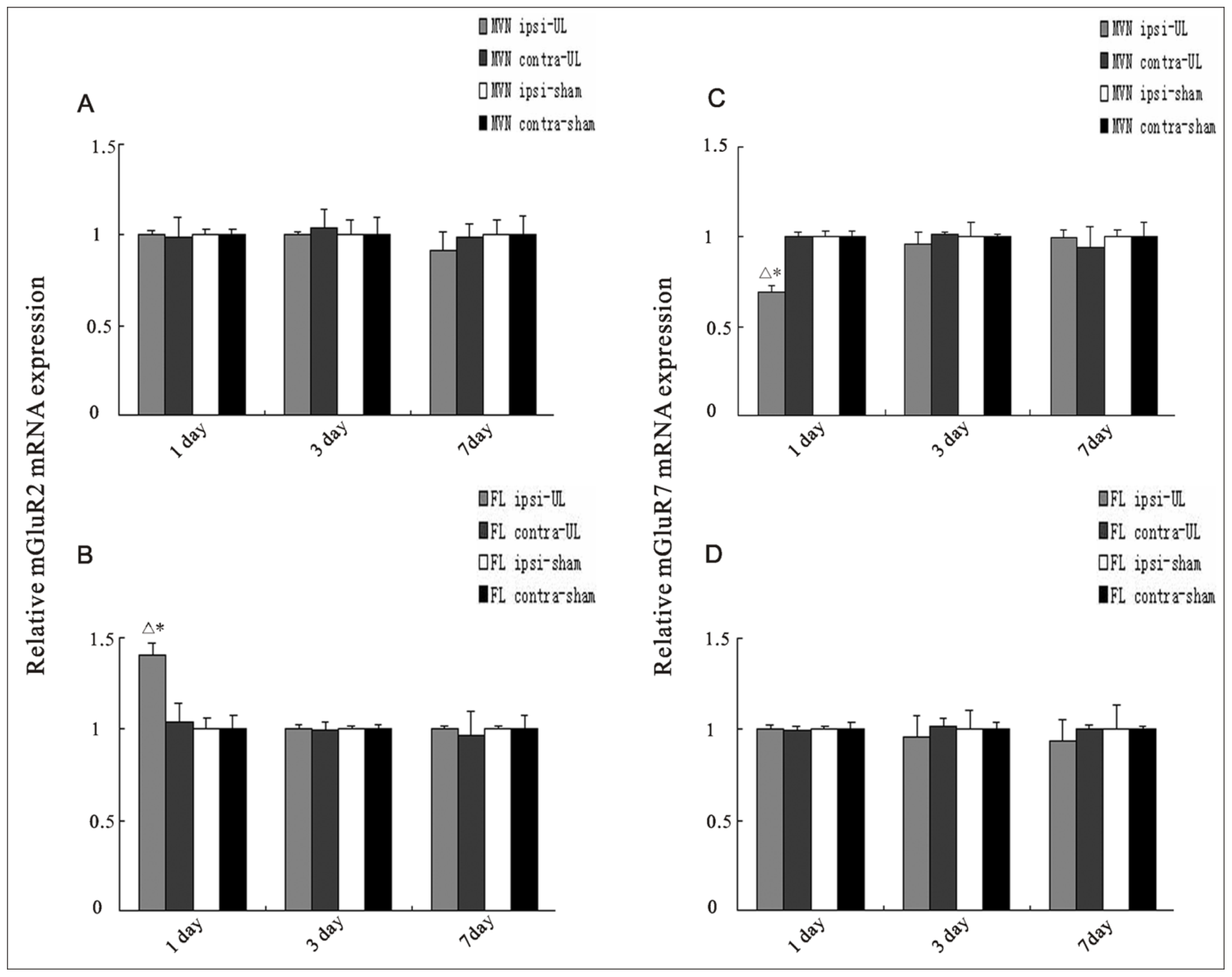
2.2. UL Induces the Changes of mRNA Levels of mGluR2 and mGluR7 in the MVN and Flocculus
2.3. UL Induces the Changes of Protein Levels of mGluR2 and mGluR7 in the MVN and Flocculus
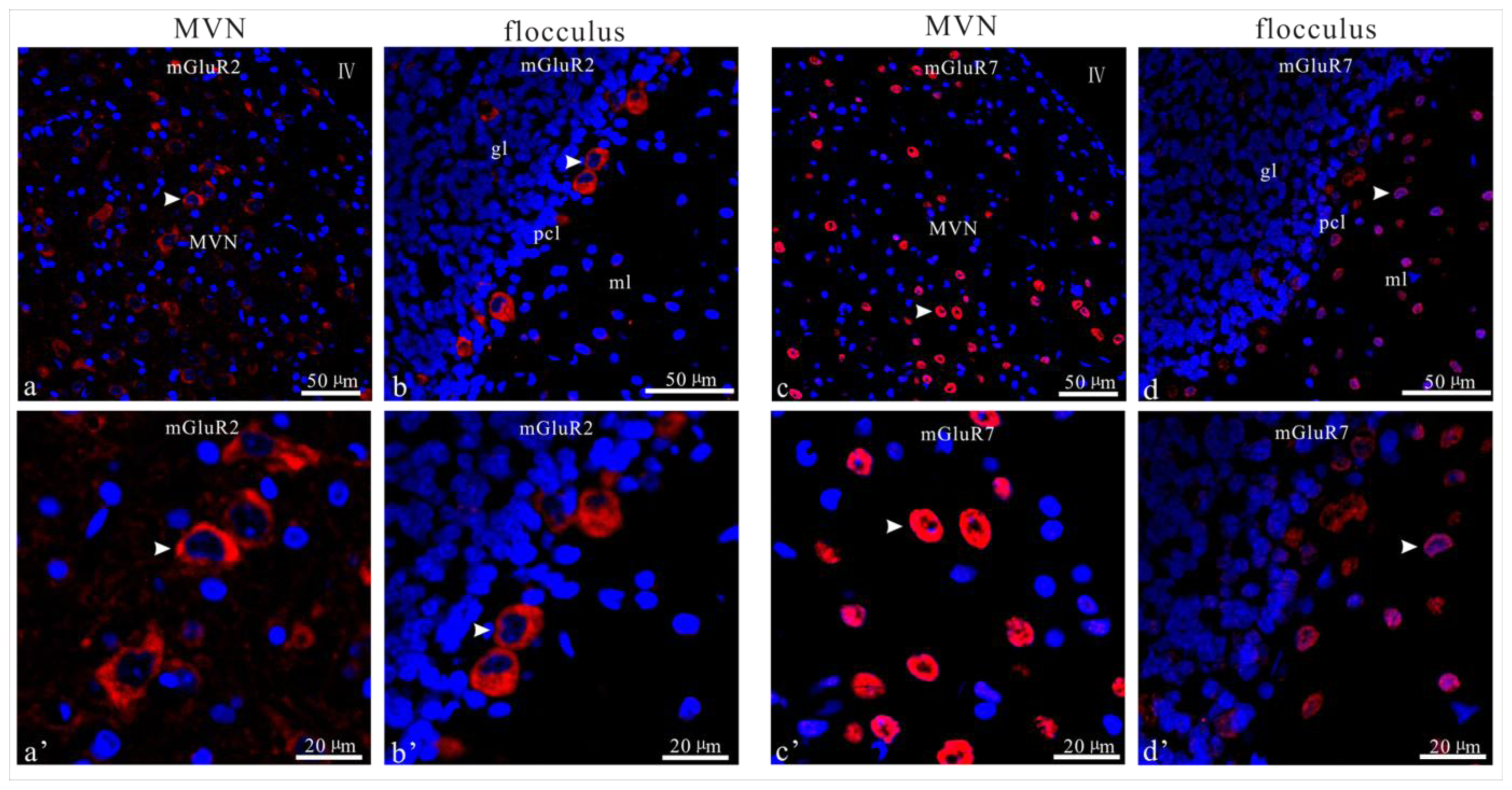
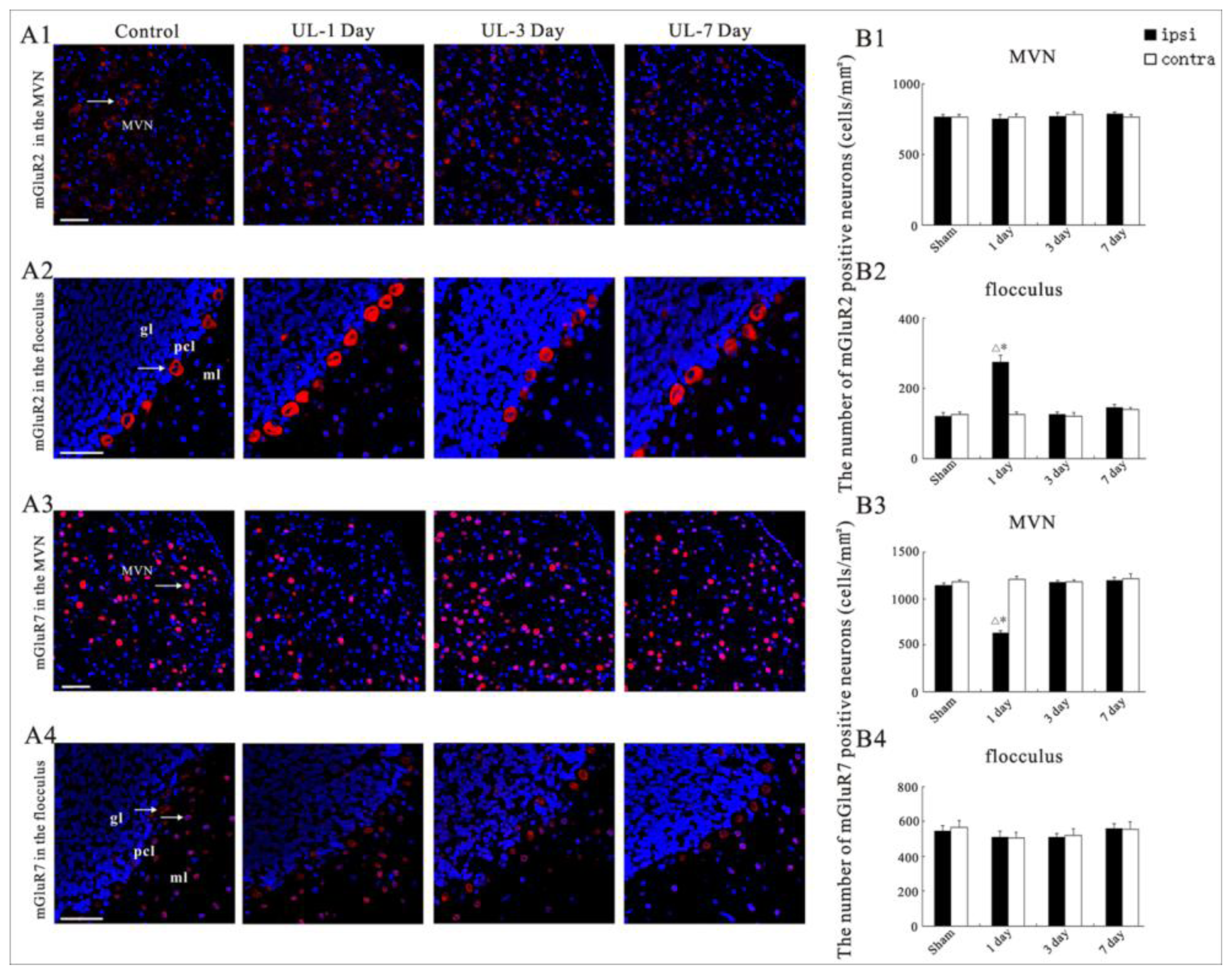
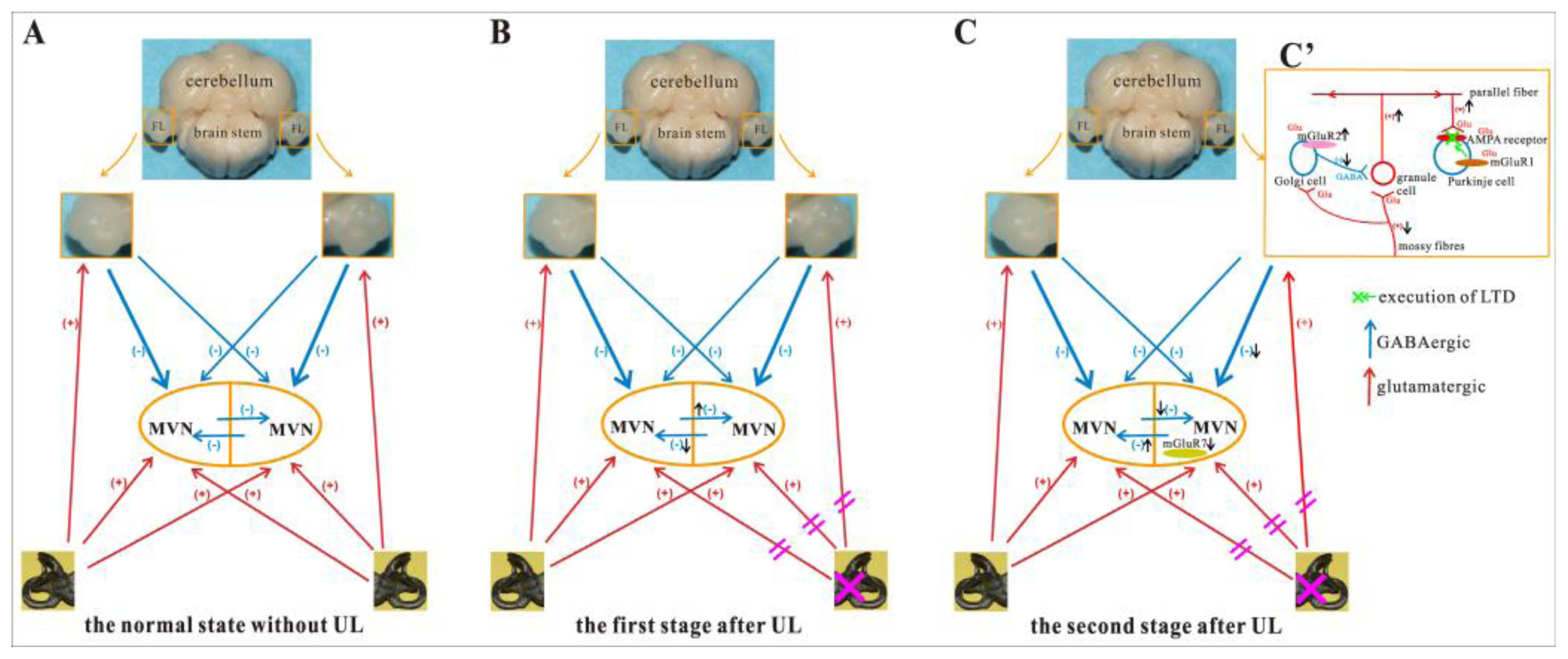
2.4. UL Induces the Asymmetrical Distribution of mGluR2 and mGluR7 in Bilateral MVN and Flocculus
2.5. Discussion
2.5.1. Medial Vestibular Nucleus
2.5.2. Flocculus
3. Experimental Section
3.1. Animal Procedures
3.2. Unilateral Labyrinthectomy
3.3. Quantitative RT-PCR
3.4. Western Blotting
3.5. Immunohistochemistry
3.6. Statistical Analysis
4. Conclusions


Acknowledgments
Conflicts of Interest
References
- Cass, S.P.; Goshgarian, H.G. Vestibular compensation after labyrinthectomy and vestibular neurectomy in cats. Otolaryngol. Head Neck Surg 1991, 104, 14–19. [Google Scholar]
- Smith, P.F.; Curthoys, I.S. Mechanisms of recovery following unilateral labyrinthectomy: A review. Brain Res. Brain Res. Rev 1989, 14, 155–180. [Google Scholar]
- Smith, P.F.; Curthoys, I.S. Neuronal activity in the contralateral medial vestibular nucleus of the guinea pig following unilateral labyrinthectomy. Brain Res 1988, 444, 295–307. [Google Scholar]
- Smith, P.F.; Curthoys, I.S. Neuronal activity in the ipsilateral medial vestibular nucleus of the guinea pig following unilateral labyrinthectomy. Brain Res 1988, 444, 308–319. [Google Scholar]
- Grassi, S.; Pettorossi, V.E. Synaptic plasticity in the medial vestibular nuclei: Role of glutamate receptors and retrograde messengers in rat brainstem slices. Prog. Neurobiol 2001, 64, 527–553. [Google Scholar]
- Smith, P.F.; Darlington, C.L. Neurochemical mechanisms of recovery from peripheral vestibular lesions (vestibular compensation). Brain Res. Brain Res. Rev 1991, 16, 117–133. [Google Scholar]
- Paterson, J.M.; Short, D.; Flatman, P.W.; Seckl, J.R.; Aitken, A. Changes in protein expression in the rat medial vestibular nuclei during vestibular compensation. J. Physiol 2006, 575, 777–788. [Google Scholar]
- Olabi, B.; Bergquist, F.; Dutia, M.B. Rebalancing the commissural system: Mechanisms of vestibular compensation. J. Vestib. Res 2009, 19, 201–207. [Google Scholar]
- Bergquist, F.; Ludwig, M.; Dutia, M.B. Role of the commissural inhibitory system in vestibular compensation in the rat. J. Physiol 2008, 586, 4441–4452. [Google Scholar]
- Azzena, G.B.; Mameli, O.; Tolu, E. Cerebellar contribution in compensating the vestibular function. Prog. Brain Res 1979, 50, 599–606. [Google Scholar]
- Johnston, A.R.; Seckl, J.R.; Dutia, M.B. Role of the flocculus in mediating vestibular nucleus neuron plasticity during vestibular compensation in the rat. J. Physiol 2002, 545, 903–911. [Google Scholar]
- Kitahara, T.; Takeda, N.; Saika, T.; Kubo, T.; Kiyama, H. Role of the flocculus in the development of vestibular compensation: Immunohistochemical studies with retrograde tracing and flocculectomy using Fos expression as a marker in the rat brainstem. Neuroscience 1997, 76, 571–580. [Google Scholar] [Green Version]
- Courjon, J.H.; Flandrin, J.M.; Jeannerod, M.; Schmid, R. The role of the flocculus in vestibular compensation after hemilabyrinthectomy. Brain Res 1982, 239, 251–257. [Google Scholar]
- Darlington, C.L.; Smith, P.F. Molecular mechanisms of recovery from vestibular damage in mammals: Recent advances. Prog. Neurobiol 2000, 62, 313–325. [Google Scholar]
- Ito, M. Cerebellar control of the vestibulo-ocular reflex—Around the flocculus hypothesis. Annu. Rev. Neurosci 1982, 5, 275–296. [Google Scholar]
- Miles, F.A.; Lisberger, S.G. Plasticity in the vestibulo-ocular reflex: A new hypothesis. Annu. Rev. Neurosci 1981, 4, 273–299. [Google Scholar]
- Kawato, M.; Gomi, H. The cerebellum and VOR/OKR learning models. Trends Neurosci 1992, 15, 445–453. [Google Scholar]
- Anwyl, R. Metabotropic glutamate receptors: Electrophysiological properties and role in plasticity. Brain Res. Brain Res. Rev 1999, 29, 83–120. [Google Scholar]
- De Waele, C.; Muhlethaler, M.; Vidal, P.P. Neurochemistry of the central vestibular pathways. Brain Res. Brain Res. Rev 1995, 20, 24–46. [Google Scholar]
- Horii, A.; Smith, P.F.; Darlington, C.L. Quantitative changes in gene expression of glutamate receptor subunits/subtypes in the vestibular nucleus, inferior olive and flocculus before and following unilateral labyrinthectomy in the rat: Real-time quantitative PCR method. Exp. Brain Res 2001, 139, 188–200. [Google Scholar]
- Darlington, C.L.; Smith, P.F. Metabotropic glutamate receptors in the guinea-pig medial vestibular nucleus in vitro. Neuroreport 1995, 6, 1799–1802. [Google Scholar]
- Grassi, S.; Malfagia, C.; Pettorossi, V.E. Effects of metabotropic glutamate receptor block on the synaptic transmission and plasticity in the rat medial vestibular nuclei. Neuroscience 1998, 87, 159–169. [Google Scholar]
- Cartmell, J.; Schoepp, D.D. Regulation of neurotransmitter release by metabotropic glutamate receptors. J. Neurochem 2000, 75, 889–907. [Google Scholar]
- Kinney, G.A.; Slater, N.T. Potentiation of NMDA receptor-mediated transmission in turtle cerebellar granule cells by activation of metabotropic glutamate receptors. J. Neurophysiol 1993, 69, 585–594. [Google Scholar]
- Shigemoto, R.; Abe, T.; Nomura, S.; Nakanishi, S.; Hirano, T. Antibodies inactivating mGluR1 metabotropic glutamate receptor block long-term depression in cultured Purkinje cells. Neuron 1994, 12, 1245–1255. [Google Scholar]
- Schoepp, D.D. Unveiling the functions of presynaptic metabotropic glutamate receptors in the central nervous system. J. Pharmacol. Exp. Ther 2001, 299, 12–20. [Google Scholar]
- Gu, Z.; Liu, W.; Wei, J.; Yan, Z. Regulation of N-methyl-d-aspartic acid (NMDA) receptors by metabotropic glutamate receptor 7. J. Biol. Chem 2012, 287, 10265–10275. [Google Scholar]
- Ohishi, H.; Shigemoto, R.; Nakanishi, S.; Mizuno, N. Distribution of the messenger RNA for a metabotropic glutamate receptor, mGluR2, in the central nervous system of the rat. Neuroscience 1993, 53, 1009–1018. [Google Scholar]
- Kinoshita, A.; Shigemoto, R.; Ohishi, H.; van der Putten, H.; Mizuno, N. Immunohistochemical localization of metabotropic glutamate receptors, mGluR7a and mGluR7b, in the central nervous system of the adult rat and mouse: A light and electron microscopic study. J. Comp. Neurol 1998, 393, 332–352. [Google Scholar]
- Beraneck, M.; Hachemaoui, M.; Idoux, E.; Ris, L.; Uno, A.; Godaux, E.; Vidal, P.P.; Moore, L.E.; Vibert, N. Long-term plasticity of ipsilesional medial vestibular nucleus neurons after unilateral labyrinthectomy. J. Neurophysiol 2003, 90, 184–203. [Google Scholar]
- Ris, L.; Capron, B.; de Waele, C.; Vidal, P.P.; Godaux, E. Dissociations between behavioural recovery and restoration of vestibular activity in the unilabyrinthectomized guinea-pig. J. Physiol 1997, 500, 509–522. [Google Scholar]
- Ris, L.; de Waele, C.; Serafin, M.; Vidal, P.P.; Godaux, E. Neuronal activity in the ipsilateral vestibular nucleus following unilateral labyrinthectomy in the alert guinea pig. J. Neurophysiol 1995, 74, 2087–2099. [Google Scholar]
- Zhang, R.; Smith, P.F.; Darlington, C.L. Immunocytochemical and stereological study of glucocorticoid receptors in rat medial vestibular nucleus neurons and the effects of unilateral vestibular deafferentation. Acta Otolaryngol 2005, 125, 1258–1264. [Google Scholar]
- Kosinski, C.M.; Risso, B.S.; Conn, P.J.; Levey, A.I.; Landwehrmeyer, G.B. Localization of metabotropic glutamate receptor 7 mRNA and mGluR7a protein in the rat basal ganglia. J. Comp. Neurol 1999, 415, 266–284. [Google Scholar]
- Shigemoto, R.; Kinoshita, A.; Wada, E.; Nomura, S.; Ohishi, H. Differential presynaptic localization of metabotropic glutamate receptor subtypes in the rat hippocampus. J. Neurosci 1997, 17, 7503–7522. [Google Scholar]
- Mitchell, S.J.; Silver, R.A. Glutamate spillover suppresses inhibition by activating presynaptic mGluRs. Nature 2000, 404, 498–502. [Google Scholar]
- Dutia, M.B. Mechanisms of vestibular compensation: Recent advances. Curr. Opin. Otolaryngol. Head Neck Surg 2010, 18, 420–424. [Google Scholar]
- Beraneck, M.; McKee, J.L.; Aleisa, M.; Cullen, K.E. Asymmetric recovery in cerebellar-deficient mice following unilateral labyrinthectomy. J. Neurophysiol 2008, 100, 945–958. [Google Scholar]
- Faulstich, M.; van Alphen, A.M.; Luo, C.; du Lac, S.; de Zeeuw, C.I. Oculomotor plasticity during vestibular compensation does not depend on cerebellar LTD. J. Neurophysiol 2006, 96, 1187–1195. [Google Scholar]
- Nelson, A.B.; Krispel, C.M.; Sekirnjak, C.; du Lac, S. Long-lasting increases in intrinsic excitability triggered by inhibition. Neuron 2003, 40, 609–620. [Google Scholar]
- Girardi, E.S.; Canitrot, J.; Antonelli, M.; Gonzalez, N.N.; Coirini, H. Differential expression of cerebellar metabotropic glutamate receptors mGLUR2/3 and mGLUR4a after the administration of a convulsant drug and the adenosine analogue cyclopentyladenosine. Neurochem. Res 2007, 32, 1120–1128. [Google Scholar]
- Ohishi, H.; Neki, A.; Mizuno, N. Distribution of a metabotropic glutamate receptor, mGluR2, in the central nervous system of the rat and mouse: An immunohistochemical study with a monoclonal antibody. Neurosci. Res 1998, 30, 65–82. [Google Scholar]
- Mitchell, S.J.; Silver, R.A. GABA spillover from single inhibitory axons suppresses low-frequency excitatory transmission at the cerebellar glomerulus. J. Neurosci 2000, 20, 8651–8658. [Google Scholar]
- Knopfel, T.; Grandes, P. Metabotropic glutamate receptors in the cerebellum with a focus on their function in Purkinje cells. Cerebellum 2002, 1, 19–26. [Google Scholar]
- Hartell, N.A. Induction of cerebellar long-term depression requires activation of glutamate metabotropic receptors. Neuroreport 1994, 5, 913–916. [Google Scholar]
- Obata, K.; Takeda, K. Release of gamma-aminobutyric acid into the fourth ventricle induced by stimulation of the cat’s cerebellum. J. Neurochem 1969, 16, 1043–1047. [Google Scholar]
- Curthoys, I.S.; Halmagyi, G.M. Vestibular compensation. Adv. Otorhinolaryngol 1999, 55, 82–110. [Google Scholar]
- Dieringer, N. Vestibular compensation: Neural plasticity and its relations to functional recovery after labyrinthine lesions in frogs and other vertebrates. Prog. Neurobiol 1995, 46, 97–129. [Google Scholar]
- Kitahara, T.; Takeda, N.; Emson, P.C.; Kubo, T.; Kiyama, H. Changes in nitric oxide synthase-like immunoreactivities in unipolar brush cells in the rat cerebellar flocculus after unilateral labyrinthectomy. Brain Res 1997, 765, 1–6. [Google Scholar]
- Paxinos, G.; Watson, C. The Rat Brain in Stereotaxic Coordinates, 6th ed; Academic Press: New York, NY, USA, 2007; pp. 56–69. [Google Scholar]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 2001, 25, 402–408. [Google Scholar]
- Zheng, Y.; Horii, A.; Appleton, I.; Darlington, C.L.; Smith, P.F. Damage to the vestibular inner ear causes long-term changes in neuronal nitric oxide synthase expression in the rat hippocampus. Neuroscience 2001, 105, 1–5. [Google Scholar]
- Ashton, J.C.; Little, E.; Muir, M.; Smith, P.F.; Darlington, C.L. Mitochondrial ultrastructure and apoptotic protein expression in the vestibular nucleus complex following unilateral labyrinthectomy. Brain Res 2005, 1055, 165–170. [Google Scholar]
- Liu, P.; Zheng, Y.; King, J.; Darlington, C.L.; Smith, P.F. Long-term changes in hippocampal n-methyl-d-aspartate receptor subunits following unilateral vestibular damage in rat. Neuroscience 2003, 117, 965–970. [Google Scholar]
- Mizukoshi, A.; Kitama, T.; Omata, T.; Ueno, T.; Kawato, M. Motor dynamics encoding in the rostral zone of the cat cerebellar flocculus during vertical optokinetic eye movements. Exp. Brain Res 2000, 132, 260–268. [Google Scholar]
- Sato, Y.; Kawasaki, T. Identification of the Purkinje cell/climbing fiber zone and its target neurons responsible for eye-movement control by the cerebellar flocculus. Brain Res. Brain Res. Rev 1991, 16, 39–64. [Google Scholar]
- Kitama, T.; Omata, T.; Mizukoshi, A.; Ueno, T.; Sato, Y. Motor dynamics encoding in cat cerebellar flocculus middle zone during optokinetic eye movements. J. Neurophysiol 1999, 82, 2235–2248. [Google Scholar]
- Sato, Y.; Kawasaki, T.; Mizukoshi, K. Eye movement control by Purkinje cell/climbing fiber zones of cerebellar flocculus in cat. Acta Otolaryngol. Suppl 1991, 481, 237–241. [Google Scholar]
- Deak, A.; Bacskai, T.; Gaal, B.; Racz, E.; Matesz, K. Effect of unilateral labyrinthectomy on the molecular composition of perineuronal nets in the lateral vestibular nucleus of the rat. Neurosci. Lett 2012, 513, 1–5. [Google Scholar]
- Hong, S.M.; Cha, C.I.; Park, B.R. Changes in calbindin expression within the flocculus after unilateral labyrinthectomy in rats. Neurosci. Lett 2009, 460, 52–55. [Google Scholar]
- Fan, G.R.; Yin, Z.D.; Sun, Y.; Chen, S.; Zhang, W.J.; Kong, W.J. Reversible neurotoxicity of kanamycin on dorsal cochlear nucleus. Brain Res 2013, 1502, 30–46. [Google Scholar]
- Zhou, L.Q.; Zhou, W.; Zhang, S.L.; Liu, B.; Leng, Y.M.; Zhou, R.H.; Kong, W.J. Changes in histamine receptors (H1, H2, and H3) expression in rat medial vestibular nucleus and flocculus after unilateral labyrinthectomy: Histamine receptors in vestibular compensation. PLoS One 2013, 8, e66684. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhou, W.; Zhou, L.-Q.; Zhang, S.-L.; Liu, B.; Leng, Y.-M.; Zhou, R.-H.; Kong, W.-J. The Changes in mGluR2 and mGluR7 Expression in Rat Medial Vestibular Nucleus and Flocculus Following Unilateral Labyrinthectomy. Int. J. Mol. Sci. 2013, 14, 22857-22875. https://doi.org/10.3390/ijms141122857
Zhou W, Zhou L-Q, Zhang S-L, Liu B, Leng Y-M, Zhou R-H, Kong W-J. The Changes in mGluR2 and mGluR7 Expression in Rat Medial Vestibular Nucleus and Flocculus Following Unilateral Labyrinthectomy. International Journal of Molecular Sciences. 2013; 14(11):22857-22875. https://doi.org/10.3390/ijms141122857
Chicago/Turabian StyleZhou, Wen, Liu-Qing Zhou, Su-Lin Zhang, Bo Liu, Yang-Ming Leng, Ren-Hong Zhou, and Wei-Jia Kong. 2013. "The Changes in mGluR2 and mGluR7 Expression in Rat Medial Vestibular Nucleus and Flocculus Following Unilateral Labyrinthectomy" International Journal of Molecular Sciences 14, no. 11: 22857-22875. https://doi.org/10.3390/ijms141122857





