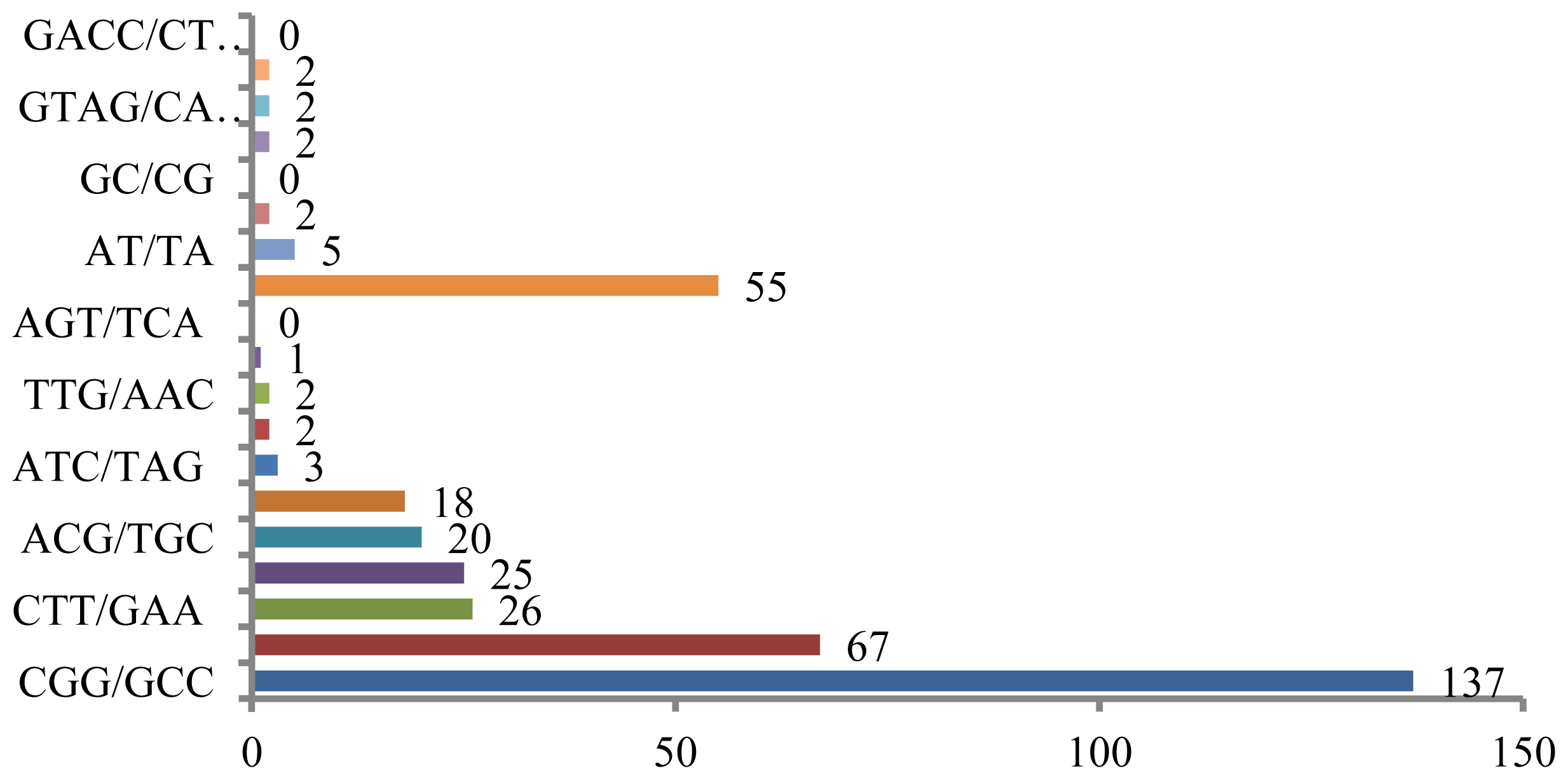1. Introduction
Rice blast, which is caused by
Pyricularia grisea (Cooke) Sacc., the anamorphous state of
Magnaporthe grisea (T.T. Hebert) Barr [
1], is the primary limiting biotic factor for rice production throughout the world. The use of resistant cultivars is the most effective and economical way to control rice blast disease, and therefore, breeding efforts to develop new resistant cultivars continue to be a priority for rice breeding programs. One of the challenges facing breeders during the development of improved rice cultivars, be they for conventional or organic agriculture, is the incorporation of disease resistance. Since the idea of indirect selection using genetic markers was first reported by Sax [
2] over 80 years ago, and particularly in the last few decades, new technologies have emerged that allow breeders to more easily select changes at the DNA level. Much of the progress to date has centered on marker-assisted backcrossing or the pyramiding of genes against rice blast [
3]. Presently, the integration of genomics and molecular-based breeding strategies for developing disease resistance, with gene-based marker assisted selection (MAS) being particularly effective, is a powerful method for efficient selection. In this context, pyramiding several major resistance genes into a valuable genetic background is simplified by the use of marker-based selection [
4]. In short, the development and use of DNA markers has irrevocably changed the fields of rice genetics and breeding.
Molecular markers are now widely used to track loci and genome regions in crop breeding programs, as large numbers of molecular markers that are tightly linked to disease resistance traits are available in most major crop species [
5–
7]. The majority of molecular markers have been isolated from genomic DNA libraries or from libraries of randomly amplified PCR fragments. Molecular markers are essential for mapping genes of interest, marker-assisted breeding, and cloning genes using mapping-based cloning strategies [
8]. Other uses of molecular markers include gene introgression through backcrossing, germplasm characterization and phylogenetic analysis [
6]. Of the various classes of existing markers, microsatellites have emerged as the markers of choice for plant breeding applications [
9]. Restriction fragment length polymorphism (RFLP) analyses are not easily scalable to high-throughput methods, and random amplification of polymorphic DNA (RAPD) assays are often not reproducible or transferable between laboratories. Although both microsatellites and amplified fragment length polymorphisms (AFLPs) can both be used to efficiently identify polymorphisms, microsatellite-based methods are more readily automated [
10]. In addition, AFLP analysis is not always straightforward, as seemingly individual bands may actually be composed of multiple fragments [
11], particularly when using large genomic templates. Furthermore, molecular markers can be used to estimate overall genetic variability, determine the proportion of a genome that has been introgressed from a donor, identify genes that are phenotypically related to a particular analyzed trait, and select for traits during multiple rounds of introgression [
12].
Identifying resistance genes using molecular markers is the basic prerequisite for performing MAS in resistance breeding programs [
13]. PCR-based microsatellite markers have attracted a great deal of attention due to several key advantages. Most importantly, they are hypervariable, abundant and well distributed throughout the rice genome. Furthermore, these markers are readily accessible through published linkage maps and public databases, and they permit the differentiation between homozygous and heterozygous individuals [
14,
15]. Overall, the speed, reliability and cost-effectiveness of acrylamide gel-based microsatellite analysis make this method an attractive tool for MAS in blast resistance breeding programs [
16].
In recent years, microsatellite markers have been widely used to screen, characterize and evaluate genetic diversity in cereal species [
17]. In particular, microsatellite-based methods offer an attractive high-throughput and non-labor-intensive way to tag blast resistance genes in breeding programs. A number of microsatellite markers have been developed from publicly available databases (
http://www.gramene.org) that are tightly linked with the
Pi-ta2,
Pi-kh and
Pi-b resistance genes [
18,
19]. Furthermore, the development of molecular methods to efficiently identify novel resistance genes has the potential to greatly improve modern cultivars, and such methods would help accelerate the application of MAS and marker-assisted backcross (MAB) breeding in rice improvement programs. Use of these markers should also facilitate the development of multiline cultivars that carry one or more blast resistance genes, without the need for pathogenic inoculation or phenotyping. Microsatellite markers have been integrated into the molecular genetic maps of a number of plant species, and they have been successfully used to perform gene-mapping, population and evolutionary studies for the purpose of cultivar development. In this review, we summarize the known microsatellite markers that are linked to rice blast disease resistance. Furthermore, we investigate the organization of microsatellites within the rice genome and evaluate their usefulness as genetic markers. In particular, this review focuses on the availability of DNA markers linked to blast resistance in rice improvement programs and their potential use in MAS. The main objectives of this review are the following: (i) to help increase the efficiency of MAS in breeding-program crosses, thus improving the chances of developing new lines for commercial release; (ii) to review the basic principles and characteristics of commonly used microsatellite markers; (iii) to outline the advantages and limitations of these markers; and (iv) to provide examples of how microsatellites have been used in molecular breeding programs for blast resistance.
6. Utility of Microsatellite Markers
A significant advance in the practical utilization of molecular markers was the development of microsatellite markers [
38]. Microsatellites are extremely informative markers that can be used for a variety of population genetics studies. Microsatellites are also considered ideal markers for genetic mapping studies [
36,
39,
40] and germplasm evolution. For instance, Sakai
et al. [
41] used microsatellite markers to investigate the genomic evolution of the African cultivated rice
Oryza glaberrima and the Asian cultivated rice
Oryza sativa, and they identified 2451 microsatellite from these two genomes. Of these 2451 microsatellites, 883 were identical among the three studied genomes (
Oryza glaberrima,
Oryza sativa indica and
Oryza sativa japonica), whereas the remaining 1568 markers were variable. Microsatellite markers show a higher degree of polymorphism in rice compared with restriction fragment length polymorphisms (RFLPs), and they are also suitable for evaluating genetic diversity among closely related rice cultivars [
42]. For MAS applications, microsatellite markers with high polymorphism information content (PIC) values are generally more useful. Parida
et al. [
43] observed higher PIC in
indica strains compared with aromatic,
japonica and aus/wild strains, which agreed well with earlier observations using microsatellite and SNP markers [
44–
51]. Using their model, they were also able to identify four major genetically distinct groups within rice—
indica,
japonica, aromatics and aus/wild—based on population-structure analyses using microsatellites and SNP markers [
45,
48–
50]. The estimated genetic diversity among the rice subpopulations in their study was significantly higher than previously observed using microsatellite and SNP markers [
47,
49] but was comparable to the diversity detected within a larger group of rice genotypes analyzed using microsatellite markers [
51]. There exist several other examples of using microsatellites for these types of studies. Another interesting application of microsatellites in rice breeding was described by Liu and Wu [
52]. In a study by Edwards
et al. [
53], genotyping microarrays utilizing single feature polymorphisms (SFPs) were used to assess the genetic variability across 20 diverse
O. sativa varieties representing five different subpopulations, as determined using STRUCTURE [
54] analysis with 169 microsatellite markers [
49]. The highest level of polymorphism (66.2%) was found between the
temperate japonica and
indica subpopulations. The lowest levels of polymorphism were found within the
temperate japonica sub-population (10.4%), which is also the least diverse subpopulation according to microsatellite markers [
49].
As the order of markers along chromosomes is conserved within species and generally conserved between related species, this information can be used to create linkage maps, which are becoming increasingly available and detailed for a large number of species. This information is available for many marker types, and microsatellite-based maps are particularly well suited for genotyping [
55]. For example, by analyzing such mapping information, it is possible to determine the fates of distinct parts of the genome over the course of developing multi-line varieties and composite-crosses [
56]. Summaries of the various applications of microsatellite-based markers in plants are presented in
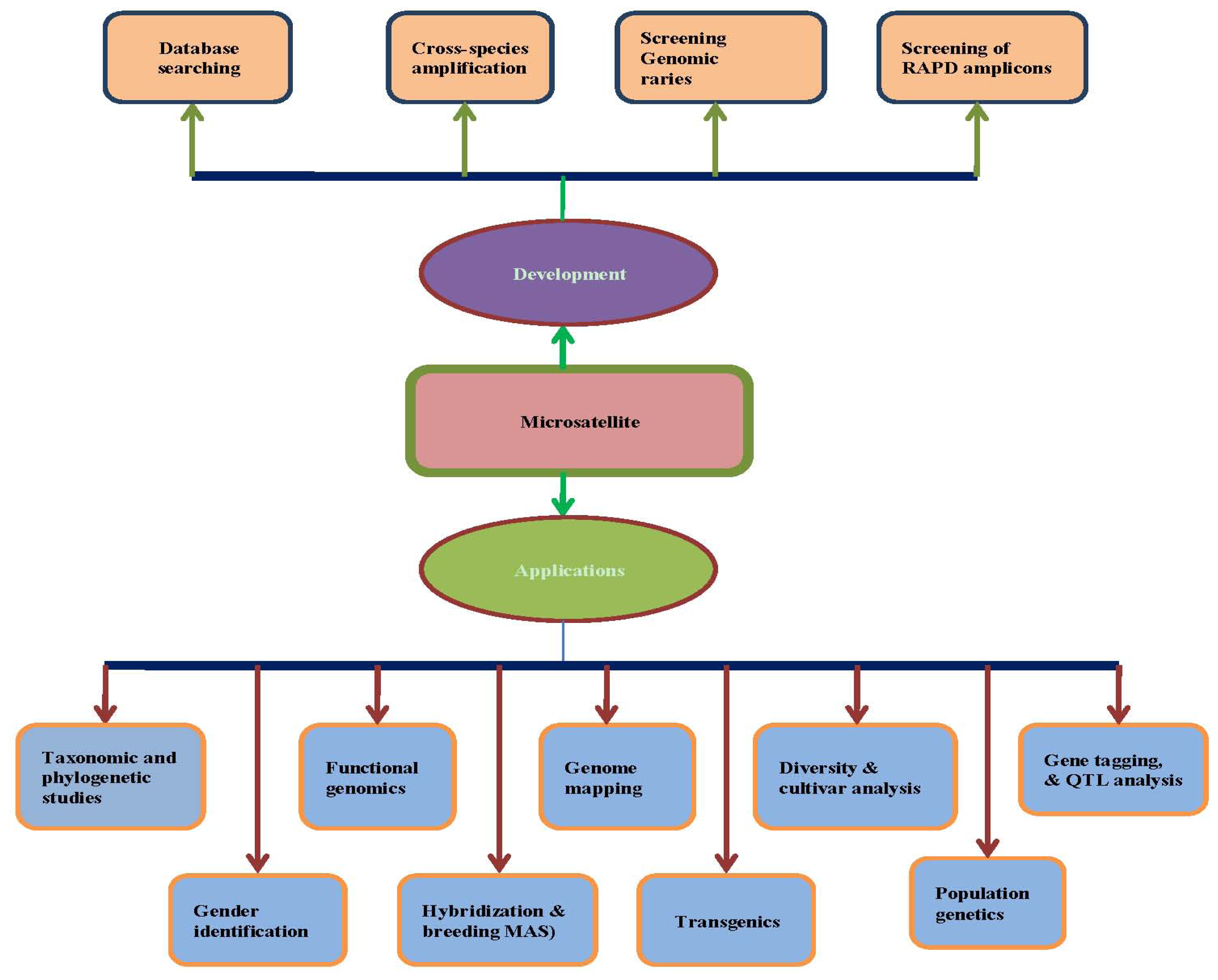
Table 4 and
Figure 1. However, it should be noted that each type of microsatellite-based marker possesses its own set of advantages and disadvantages based on mode of inheritance, informativeness, reproducibility, or procedural complexity, as well as economic concerns, such as cost and labor time. Therefore, the decision concerning which type of marker to use should be carefully considered based on the nature of the particular research project.
7. Microsatellite Marker Development
Completely sequenced genomes provide the basis upon which to design a large number of gene-based microsatellite markers. For example, rice (
Oryza sativa L.) was the first cereal to have its genome completely sequenced, which has enabled the development of a large number of microsatellite markers [
58]. Recently, Zhang
et al. [
59] developed 52,485 microsatellite markers that are polymorphic between
indica and
japonica. However, the difficulty now lies in choosing the most useful and informative microsatellite markers from such large datasets to use in rice genotyping applications. This problem can be overcome by constructing smaller, informative microsatellite marker databases composed of markers located in potentially functional genic sequences with relatively high polymorphic potential. Considering the excellent genetic attributes and higher predicted informativeness of genic non-coding microsatellite (GNMS) markers, Parida
et al. [
60] identified 19,555 perfect GNMS repeats on chromosomes 1 and 12 in rice. With the entire rice genome now sequenced, microsatellite markers can be developed within a few thousand base pairs of any gene. For example, a study by Goff
et al. [
61] suggests the presence, on average, of one microsatellite repeat (defined as at least eight repeats of a 2–4 bp motif) every 8 kb, yielding a total of 48,351 markers in the entire genome. Of course, not all these repeats can be developed into microsatellite markers [
37], which can also be identified by screening database sequences or by screening libraries of clones (
Figure 1).
PCR primers for amplifying microsatellite markers were designed by Rozen and Skaletsky [
62] using the online program Primer3 by subjectively choosing primers flanking the repeat regions. The primers were then ordered from various commercial vendors and tested for their ability to amplify the microsatellites and differentiate polymorphisms among the parental lines used in the
Pi-z mapping studies. Markers RM527 and RM6836, which were previously localized near the
Pi-z locus [
63], were obtained from the gramene website (
http://www.gramene.org/) [
64] and also included in primer testing [
65].
Next-generation sequencing technologies (HighSSR, Roche 454 GS FLX) are now being used for microsatellite discovery with significant savings in cost and time [
66–
70]. Rapid progress in DNA sequencing technologies has substantially reduced costs while exponentially increasing throughput and accuracy. Currently, the most cost effective next-generation sequencing platform is the IlluminaHiSeq2000 [
71], which can reduce costs 3400-fold relative to traditional sequencing methods; it is reasonable to expect that continued improvements will lead to even lower costs [
72].
Fjellstrom
et al. [
73] designed DNA markers using four different methods. Five of these markers (RM101, RM138, RM144, RM155, and RM166) were based on an earlier set of microsatellites identified at Texas A & M University identified by screening the NCBI public DNA sequence database for repeated sequences, as described in Temnykh
et al. [
42]. The genomic locations of
Pi-b,
Pi-k, and
Pi-ta2 and their actual linkages were confirmed by mapping these genes in several of the populations described by Conaway
et al. [
19]. By mapping these initial markers relative to microsatellite markers developed using traditional methods at Cornell University [
42], three additional tightly linked markers were subsequently identified: RM208, RM224, and RM266. After identifying candidate microsatellite markers from public database resources, the authors mapped these markers near to the blast resistance genes
Pi-b,
Pi-k, and
Pi-ta2 on rice chromosomes 2, 11 and 12, respectively. The public release of the Monsanto rice microsatellite database then allowed for the development of two additional linked microsatellite markers, which released as RM1233 and RM7102 [
14]. Although DNA markers for rice blast resistance have been developed, most are not suitable for routine use in the MAS program involving large numbers of progeny. A dominant marker for the
Pi-b gene,
Pibdom, has also been developed based on the sequence of the cloned
Pi-b gene [
74] (GenBank accession AB013448). These markers should facilitate the introgression and pyramiding of these three blast resistance genes into new rice cultivars and elite lines [
73]. In addition, a high-density microsatellite map with a genome coverage of approximately one microsatellite per 0.5 cM has been developed by the International Rice Microsatellite Initiative (IRMI) [
75], which can be used for developing tightly linked markers for a variety agronomic traits, including blast resistance. Accessibility to the complete genomic sequences of the rice subspecies
indica and
japonica under public domain (
http://rgp.dna.affrc.go.jp;
http://www.genomics.org.cn) has enabled rice researchers to generate additional markers for the fine-scale mapping of targeted genes. For the purposes of obtaining a high-density linkage map for fine-scale mapping within their target region, new SSR, InDel, and CAPS markers were developed [
76] using the publicly available rice genome sequence (
http://rgp.dna.affrc.go.jp).
In species with large genomes, the conversion of microsatellite-containing sequences into useful markers can be difficult [
77–
80]. The recovery percentage of functional microsatellite primers in such genomes is usually low, which can be due to (i) the amplification of complex, weak or nonspecific patterns; (ii) lack of amplification; or (iii) non-polymorphic PCR products. Therefore, researchers often choose to use tri- and tetra-nucleotide repeat motifs, as opposed to di-nucleotide motifs, as these generally yield fewer “stutter bands” [
40,
81]. However, di-nucleotide motifs are more common than tri- or tetra-nucleotide motifs and are therefore easier to use in combinatorial screens. The wide variety of DNA markers discovered and developed in rice is shown in
Table 5.
Although microsatellite marker are considered to be the most practical genetic markers, their application remains somewhat limited due to the time and effort needed to develop them. Two general strategies are used to identify and create microsatellite markers: (i) searching for sequences containing microsatellites within databases; and (ii) constructing and screening genomic libraries with probes complementary to microsatellite sequences. Two websites recommended by Romero
et al. [
82] are shown in
Table 6.
8. Abundance of Microsatellite Motifs in Rice
The frequency of microsatellite motifs varies significantly among different organisms [
39,
83,
84]. The most abundant microsatellite motif reported in plants is (AT)
n, whereas (AC)
n is most abundant motif in the human genome. Currently, there are no reliable estimates of the number of (AT)
n or (GC)
n sites in rice due to the difficulty of the hybridization-based screening methods used to detect these motifs [
85,
86]. The size of the rice genome is ~0.45 × 10
9 bp [
87], whereas the size of the human genome is ~6.6 times larger (3.0 × 10
9 bp) [
88]. These figures suggest that there should be one (AC)
n site approximately every 360–450 kb in rice, compared with one every 40–80 kb in humans; similarly, it is estimated that there is one (GA)
n motif every 225–330 kb in rice [
85,
86]. Three hundred and twenty three microsatellite markers identified by library screening [
42,
86,
89,
90] and GenBank searches of rice sequences [
42,
83,
86] have been localized on the rice genetic map. The vast majority of these markers contain di- and tri-nucleotide motifs, with only seven loci containing tetra-nucleotide repeats, such as (AATT)
n, (TTCC)
n, (GATG)
n, (ATGT)
n, (GTAT)
n, (ATTT)
n, and (TTTG)
n [
42,
86,
89,
90]. (GATA)
n sequences, which to date have not been mapped in rice, are the most common tetra-nucleotide repeat, although only 270 of these motifs are found in the entire rice genome [
91]. However, it should be kept in mind that it is the degree of polymorphism shown by microsatellites, rather than their abundance within the genome, that ultimately determines their usefulness in genomic analyses [
92].
The motif (CGG)
n has been reported to be very abundant in rice and is interspersed throughout the genome [
89]. Several studies based on GenBank searches of rice sequences identified a variety of microsatellites motifs [
83,
86,
90]. In a study by Panaud
et al. [
85], 34-bp oligonucleotides (representing two di-nucleotide, seven tri-nucleotide, and four tetra-nucleotide motifs) were synthesized and used as probes for library screening. Screening of a 15-kb insert genomic library suggested that the relative frequency of the various microsatellite motifs was inversely correlated with the size of the repeat motif [
38]. This finding was consistent with previous reports based on GenBank searches in a range of plant species [
39]. The microsatellite (GATA)
n has also been frequently used for DNA fingerprinting. For example, Davierwala
et al. [
92] identified three polymorphic (GATA)
n-harboring loci (OS1A6, OS1H10 and OS2E7) that contained 7–13 repeat motifs by probing a genomic library from the cultivated rice strain
Oryza sativa var. Basmati-370 with a oligonucleotide (GATA)4 probe.
Microsatellites can also be screened for using published rice sequences in the DDBJ databank, as numerous sequencing datasets, including those for EST sites, are available in this database. Homology searches can be performed to identify all di- and tri-nucleotide motifs, as well as four types of tetra-nucleotide repeats, and several of these are shown in
Figure 2. Out of 11,798 total sequences, 369 contained complete repeats, and most types of repeat sequences, with the exception of (GC/CG)
n, (AGT/TCA)
n and (GACC/CTGG)
n, were found. Sequences containing (CGG/GCC)
n were most frequently identified in the published rice sequences, followed by sequences containing (GAG/CTC)
n (
Figure 2). Poly (CGG) loci were also abundant and were found throughout the rice genome [
89], and the results presented here are likely representative of this feature of the rice genome. However, only a limited number of published rice sequences contain (AT/TA) sequences, although this motif is generally abundant in other plant genomes [
83,
84].
9. Microsatellites for Tracking Blast Resistance in Rice
Many
Pi genes confer resistance to overlapping spectra of blast pathotypes, and it is often difficult to monitor for the presence of individual resistance genes and pyramid these in breeding lines using traditional phenotypic screening. Therefore, DNA markers provide a straightforward and rapid means to select for multiple blast resistance genes without performing extensive progeny testing or disease screening. DNA markers linked to several of the
Pi genes have been localized on rice chromosomes, as well as markers for
Pi-ta [
93–
95] and
Pi-b [
96]. Unfortunately, the majority of DNA markers for blast resistance are RFLPs, which are relatively labor intensive to analyze for use in breeding programs. Markers that can be analyzed by PCR are more amenable for breeding purposes, such as the ones developed for
Pi-2 [
94] and
Pi-ta [
93].
The rice blast resistance gene
Pi-z, which is present in the rice genotypes Zenith and Fukunishiki, represents a potential source of blast resistance for the northwestern Himalayan region of India. Sharma
et al. [
97] and Rathour
et al. [
98] both tested the reliability of microsatellite markers linked to
Pi-z for assessing the blast resistance phenotype in commercially important crosses. A new set of microsatellite markers linked to
Pi-z was developed by exploiting publicly available marker and genomic resources in rice. Of the three previously reported markers for
Pi-z, only MRG5836 was found to be suitable for MAS. Among 17 microsatellites selected from the putative
Pi-z locus, two (RM8225 and RM8226) co-segregated with MRG5836, and they were located at distance of 1.2–4.5 cM from the gene. In addition, a new microsatellite marker, SSR236, was derived from the (CT)16 repeat within the PAC clone P0502B12, and it showed even closer linkage to
Pi-z. A survey of the allelic diversity at the loci containing the
Pi-z-linked microsatellite markers revealed that the Fukunishiki- and Zenith-type alleles were not present in a majority of the local
indica rice genotypes. Therefore, as these markers are polymorphic between the
Pi-z donors and the great majority of local
indica rice strains that were tested, they can be used as selection tools in rice breeding programs aimed at improving blast resistance in local rice varieties [
99]. Conaway-Bormans
et al. [
63] identified three microsatellite markers that mapped at a distance of 0.0–11.5 cM from
Pi-z in several different crosses segregating for the gene. Genetic distances between markers often vary in different mapping populations due to differences in the genetic backgrounds of the parental genotypes [
100]. The gene
Pi-z has been reported to be allelic with, or at least closely linked to, three other blast resistance genes,
Pi-2,
Pi-zt and
Pi-9, which map close to the centromere of chromosome 6 [
8,
101]. Furthermore, the SSR236 marker has now bridged the 7.5-cM gap between the microsatellite markers RM8226 (54.1 cM) and RM3330 (61.6 cM) in the current version of the IRMI SSR map [
99]. In addition, RM208 has been linked to
Pi-b resistance, YL155 and YL183 have been linked to
Pi-ta resistance in
indica, and AP5659-1 has been linked to
Pi-z resistance [
65,
73,
102].
The
Pi20(t) gene was identified from 160 Chinese
Magnaporthe oryzae isolates, and among these, isolate 98095 can specifically differentiate the
Pi20(t) gene present in cv. IR24. Two flanking and three co-segregating microsatellite markers for
Pi20(t), which is located near the centromeric region of chromosome 12, were identified using 526 highly susceptible F
2 plants derived from a cross between Asominori (highly susceptible) and IR24 (resistant). The microsatellite OSR32 was mapped at a distance of 0.2 cM from
Pi20(t), and the microsatellite RM28050 was mapped to the other side of
Pi20(t) at a distance of 0.4 cM. The other three microsatellite markers, RM1337, RM5364 and RM7102, were observed to co-segregate with
Pi20(t). In particular, RM1337 and RM5364 were found to be reliable markers of the resistance mediated by
Pi20(t) in a wide range of elite rice germplasms from China. Therefore, these markers are useful tags for use in marker-assisted rice breeding programs aimed at incorporating
Pi20(t) into advanced rice breeding lines [
123]. Sharma
et al. [
127] concluded that RM25 and RM310 are two microsatellite markers linked to blast resistance in the Laxmi cultivar. These two markers are located 4.5 cM apart on chromosome 8 in rice [
42]. An advanced backcross population of BC
3F
3 lines derived from the rice varieties Vandana and Moroberekan was analyzed for blast resistance by Wu
et al. [
128], and they identified four microsatellite markers (RM21, RM168, RM215 and RM250) that were significantly associated with the resistance gene.
This locus was mapped by Liu
et al. [
119] to a 5.8-cM interval bounded by RM5647 and RM8018 on the short arm of chromosome 8. This novel resistance gene has been tentatively designated as
Pi36(t). According to a report by Liu,
Pi-1 is located 6.8 cM away from the RM144 microsatellite; their results indicate that the physical distance between these two loci was between 57 and 72 kb [
129]. Chen
et al. [
114] determined that the genetic distance between
Pi-1 and the MRG4766 microsatellite marker was 1.3 cM. More recently, there has been a report concerning the identification of rice blast resistance using RM144 [
130], indicating that it may be possible to identify this resistance gene using microsatellite markers. The known blast resistance genes and their linked microsatellite markers are shown in
Table 7.
The Korean cultivar Suweon 365 carries three major resistance genes,
Pi18,
Pi21(t) and
Pi22(t), that are effective against the Korean isolates KI-313, KJ-101, and KJ-201, respectively, and these genes have been tracked using microsatellite markers in an F
2 population derived from a cross of the Suweon and Chucheongbyeo cultivars [
131]. Microsatellite markers were used to map the gene
Pi-kh, which confers resistance to blast races in the Himalayan region of Northeast India [
121]. Pinheiro
et al. [
13] developed the cultivar Cica-8 by crossing the susceptible cultivar Metica-1 to the resistant cultivar Cica-8, and they found that one microsatellite marker, RM7102, was closely linked to the resistant allele. Yang
et al. [
132], while working on identifying and mapping the
Pi41 gene, utilized resistant cultivar 93-11 and susceptible cultivar Nipponbare, as well as an F
2 population derived from a cross of these two cultivars. They studied 180 microsatellite markers and identified seven markers that were linked to
Pi41, one of which was RM7102. The markers RM144, RM224 and yca72 are linked to the resistance genes
Pi-ks,
Pi1 and
Pia, respectively [
133,
134]. Koide
et al. [
135] identified four microsatellite markers, RM7419, RM1268, RM6648 and RM5811 that were linked to
Pish. With respect to
Pib, both a gene-specific marker,
Pibdom, and a co-segregation marker, RM208, have been reported [
73]. Gouda
et al. [
136] found that the two microsatellite markers RM5926 and AP5659-5 were tightly linked to the
Pi-1 and
Piz-5 genes, respectively, in PRR78. Pan
et al. reported that
Pii(t),
Pi3(t),
Pi5(t) and
Pi15(t) are all located within the same interval on chromosome 9 in rice [
124]. Moumeni and Leung reported that three microsatellite markers, RM224, RM179 and RM 277, on chromosomes 11 and 12 were tightly linked to components of rice blast resistance [
137]. In addition, Liu
et al. [
109] revealed that RM247 and RM463 were located on chromosome 12 and were linked to the
Pi39 resistance gene. Abedi
et al. [
138] also identified four microsatellite markers, RM224, RM277, RM463 and RM179, which are linked to resistance genes on rice chromosomes in Iranian rice genotypes. This suggests there exists at least one race-specific resistance gene among the genetic sources of genotypes that confers resistance functions to the blast races. The marker AP5930, which is linked to
Piz-5 [
65], and RM206, which is linked to
Pi54 [
121], were used for foreground selection in both backcrossed and “selfed” generations. Finally, Singh
et al. [
139] also used a microsatellite marker (RM6100) when incorporating blast resistance into PRR78, an elite Basmati rice restorer line, through marker-assisted backcross breeding.
A large number of databases are available for selecting molecular markers linked to the
Pi genes (
http://www.gramene.org). Most of the known blast resistance genes have been mapped to chromosomes 12, 11 and 6 [
140–
143] through the work of a large number of researchers. For example,
Pi-1(t), Pi-2(t) and
Pi-4(t) were mapped to chromosomes 11, 6 and 12, respectively [
144]. The microsatellite markers RM168, RM8225, RM1233, RM6836, RM5961 and RM413 were analyzed by Ashkani
et al. [
145], and they were linked to blast resistance genes specific to pathotype P7.2. Furthermore, molecular marker-assisted rice breeding programs have been developed with the aim of developing durable blast resistance in rice cultivars by pyramiding the resistance genes
Pi-1(t),
Pi-2(t) and
Pi-33(t), which shows potential for controlling blast pathogen populations in Latin America [
146].