The Key Enzyme of the Sialic Acid Metabolism Is Involved in Embryoid Body Formation and Expression of Marker Genes of Germ Layer Formation
Abstract
:1. Introduction
2. Results and Discussion
2.1. Characterization of GNE-Deficient Embryonic Stem Cells
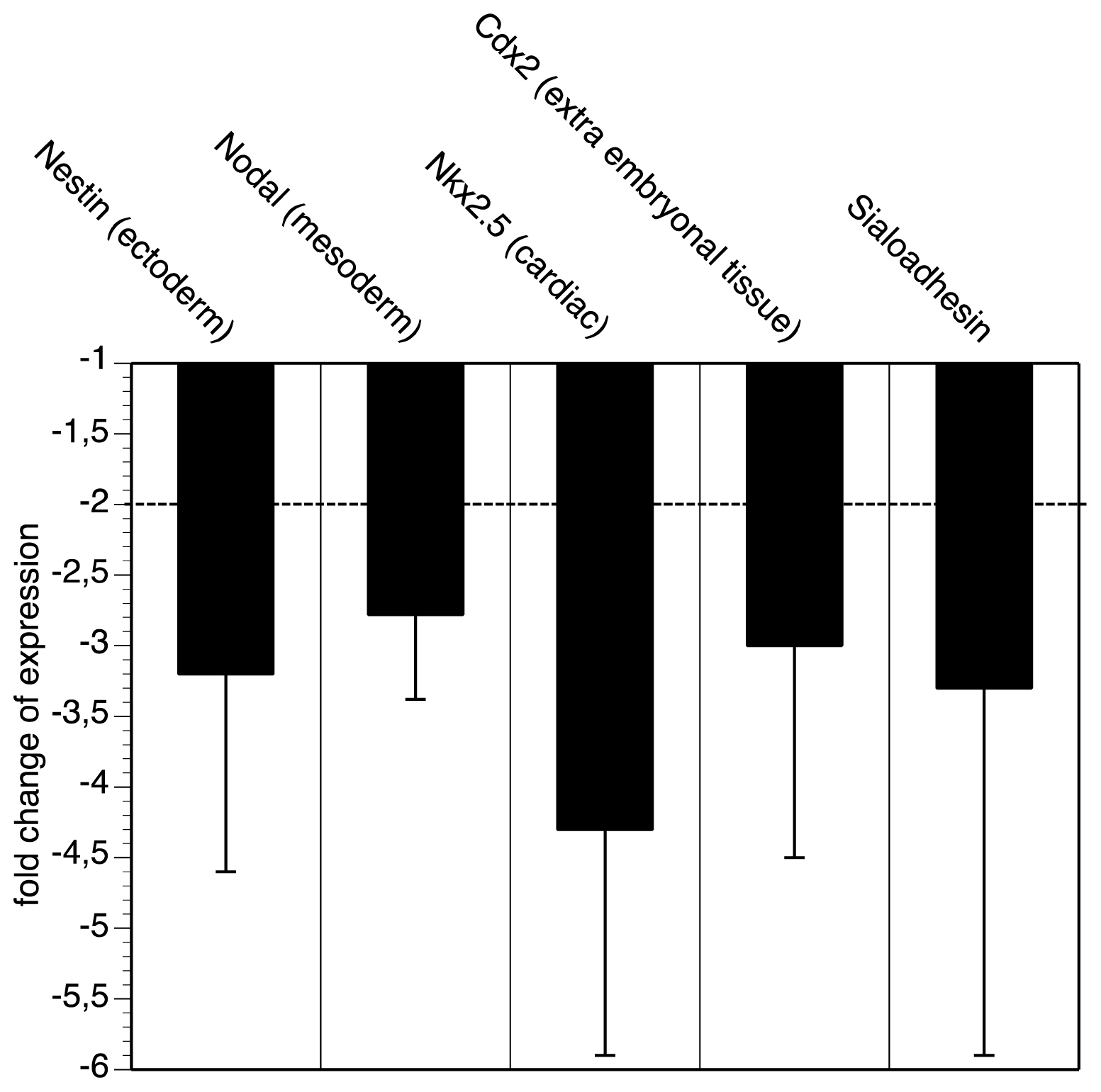
2.2. GNE-Deficient Embryonic Stem Cells Express Less Differentiation Markers
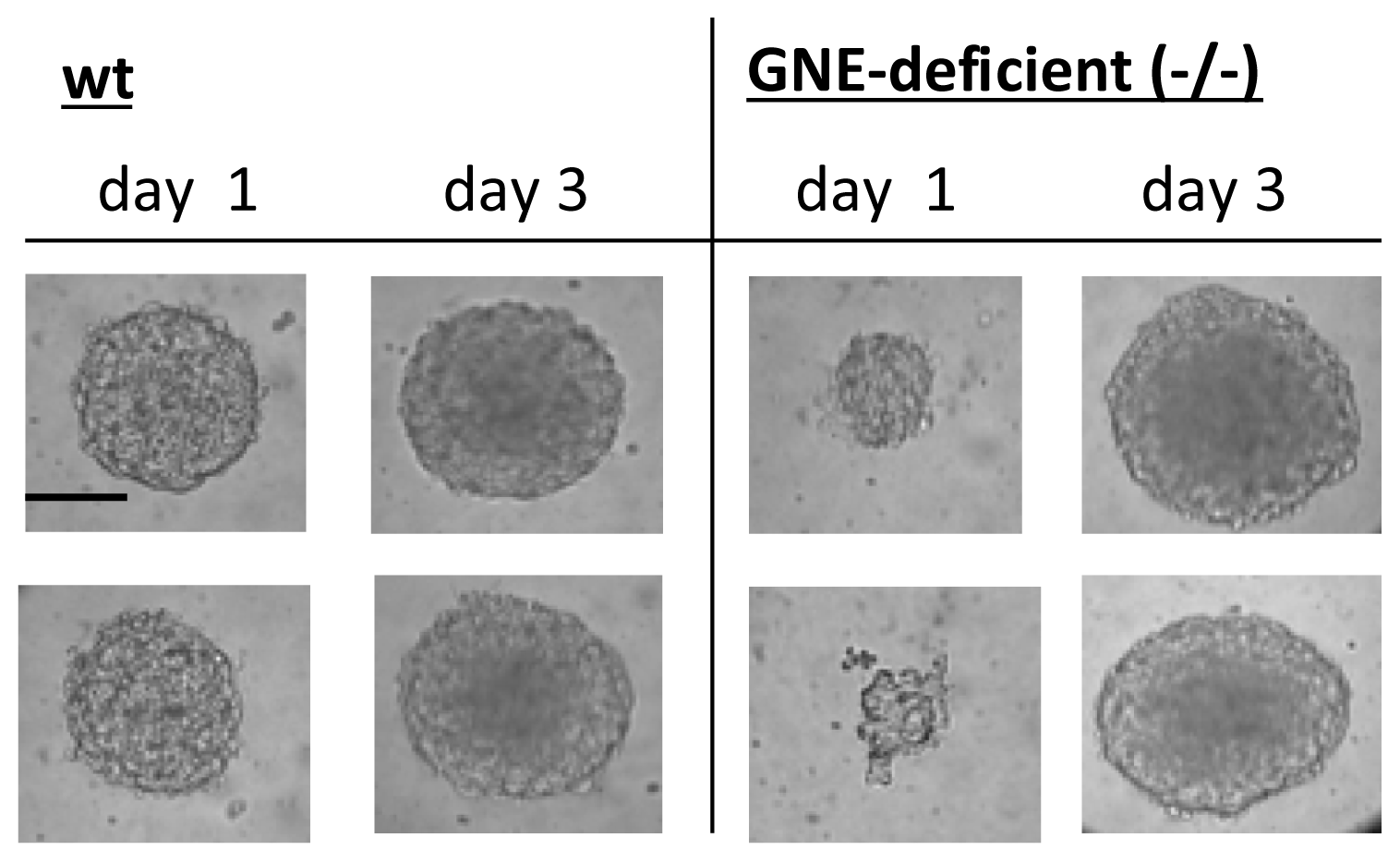
2.3. Embryoid Body Formation of GNE-Deficient Embryonic Stem Cells Is Retarded
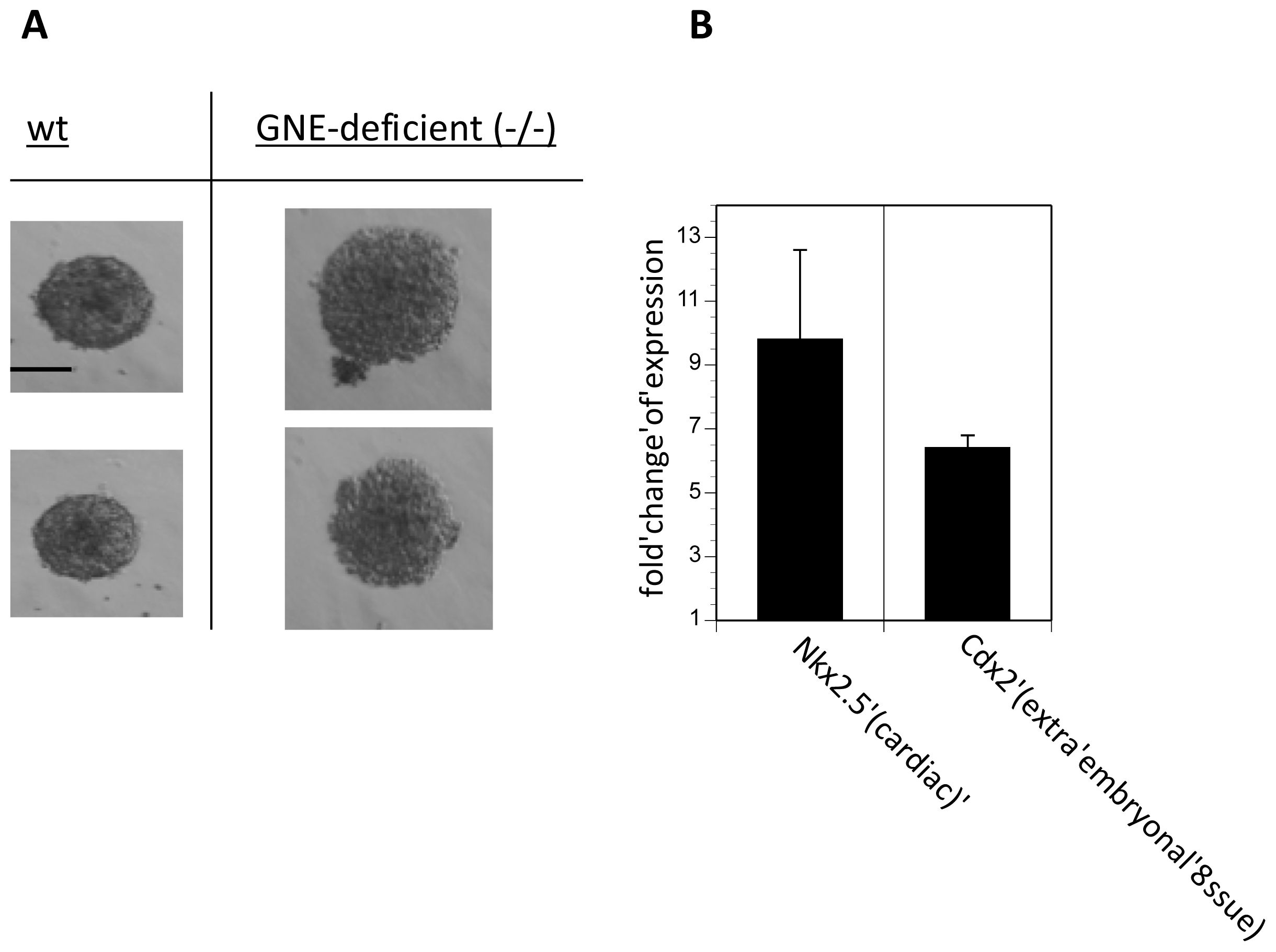
2.4. Embryoid Body Formation of GNE-Deficient Embryonic Stem Cells Is Sialic Acid Dependent
3. Experimental Section
3.1. Cell Cultures
3.2. Embryoid Body Formation
3.3. qRT-PCR
- ΔCtWT = Ctselected gene WT − CtHKG WT and ΔCtKO = Ctselected gene KO − CtHKG KO
- ΔΔCt = ΔCtKO − ΔCtWT
- Ratio = 2−ΔΔCt (maximum efficiency is assumed)
- Ratio = 1, no different gene expression in KO compared to WT
- Ratio > 1, increased gene expression in KO compared to WT
- Ratio < 1, decreased gene expression in KO compared to WT; in this case negative reciprocal value (−1/Ratio) was used for better representation.
4. Conclusions
| Name | Primers | Fragment length |
|---|---|---|
| GAPDH fwd. | 5′-CCT TCA TTG ACC TCA ACT AC-3′ | 259 bp |
| GAPDH rev. | 5′-GAG ATG ATG ACC CTT TTG GC-3′ | |
| Beta1-Integrin fwd. | 5′-TTC AGA CTT CCG CAT TGG CTT TGG-3′ | 117 bp |
| Beta1-Integrin rev. | 5′-TGG GCT GGT GCA GTT TTG TTC AC-3′ | |
| Sialoadhesin fwd. | 5′-GGT GTT GAG GTG GGA GGA GAG-3′ | 50 bp |
| Sialoadhesin rev. | 5′-GAT GGA CTA GCA GAA AGG GGT TAT GAA-3′ | |
| Nestin fwd. | 5′-GAG AAG ACA GTG AGG CAG ATG AGG TTA-3′ | 113 bp |
| Nestin rev. | 5′-GCC TCT GTT CTC CAG CTT GCT-3′ | |
| Nodal fwd. | 5′-GGA GTT TCA TCC TAC CAA CC-3′ | 386 bp |
| Nodal rev. | 5′-TCC TGC CAT GCC ACG GTA GC-3′ | |
| Nkx-2,5 fwd. | 5′-CGA CGG AAG CCA CGC GTG CT-3′ | 181 bp |
| Nkx-2,5 rev. | 5′-CCG CTG TCG CTT GCA CTT G-3′ | |
| Cdx-2 fwd. | 5′-GCA GTC CCT AGG AAG CCA AGT GA-3′ | 162 bp |
| Cdx-2 rev. | 5′-CTC TCG GAG AGC CCA AGT GTG-3′ | |
Acknowledgments
Conflicts of Interest
References
- Schauer, R. Achievements and challenges of sialic acid research. Glycoconj. J 2000, 17, 485–499. [Google Scholar]
- Schauer, R. Chemistry, metabolism, and biological functions of sialic acids. Adv. Carbohydr. Chem. Biochem 1982, 40, 131–234. [Google Scholar]
- Keppler, O.T.; Horstkorte, R.; Pawlita, M.; Schmidt, C.; Reutter, W. Biochemical engineering of the N-acyl side chain of sialic acid: Biological implications. Glycobiology 2001, 11, 11R–18R. [Google Scholar]
- Hinderlich, S.; Stäsche, R.; Zeitler, R.; Reutter, W. A bifunctional enzyme catalyzes the first two steps in N-acetylneuraminic acid biosynthesis of rat liver. Purification and characterization of UDP-N-acetylglucosamine 2-epimerase/N-acetylmannosamine kinase. J. Biol. Chem 1997, 272, 24313–24318. [Google Scholar]
- Stäsche, R.; Hinderlich, S.; Weise, C.; Effertz, K.; Lucka, L.; Moormann, P.; Reutter, W. A bifunctional enzyme catalyzes the first two steps in N-acetylneuraminic acid biosynthesis of rat liver. Molecular cloning and functional expression of UDP-N-acetyl-glucosamine 2-epimerase/N-acetylmannosamine kinase. J. Biol. Chem 1997, 272, 24319–24324. [Google Scholar]
- Horstkorte, R.; Nöhring, S.; Wiechens, N.; Schwarzkopf, M.; Danker, K.; Reutter, W.; Lucka, L. Tissue expression and amino acid sequence of murine UDP-N-acetylglucosamine-2-epimerase/N-acetylmannosamine kinase. Eur. J. Biochem 1999, 260, 923–927. [Google Scholar]
- Lucka, L.; Krause, M.; Danker, K.; Reutter, W.; Horstkorte, R. Primary structure and expression analysis of human UDP-N-acetyl-glucosamine-2-epimerase/N-acetylmannosamine kinase, the bifunctional enzyme in neuraminic acid biosynthesis. FEBS Lett 1999, 454, 341–344. [Google Scholar]
- Schwarzkopf, M.; Knobeloch, K.P.; Rohde, E.; Hinderlich, S.; Wiechens, N.; Lucka, L.; Horak, I.; Reutter, W.; Horstkorte, R. Sialylation is essential for early development in mice. Proc. Natl. Acad. Sci. USA 2002, 99, 5267–5270. [Google Scholar]
- Gagiannis, D.; Orthmann, A.; Danssmann, I.; Schwarzkopf, M.; Weidemann, W.; Horstkorte, R. Reduced sialylation status in UDP-N-acetylglucosamine-2-epimerase/N-acetylmannosamine kinase (GNE)-deficient mice. Glycoconj. J 2007, 24, 125–130. [Google Scholar]
- Wang, Z.; Sun, Z.; Li, A.V.; Yarema, K.J. Roles for UDP-GlcNAc 2-epimerase/ManNAc 6-kinase outside of sialic acid biosynthesis: Modulation of sialyltransferase and BiP expression, GM3 and GD3 biosynthesis, proliferation, and apoptosis, and ERK1/2 phosphorylation. J. Biol. Chem 2006, 281, 27016–27028. [Google Scholar]
- Kontou, M.; Weidemann, W.; Bauer, C.; Reutter, W.; Horstkorte, R. The key enzyme of sialic acid biosynthesis (GNE) promotes neurite outgrowth of PC12 cells. NeuroReport 2008, 19, 1239–1242. [Google Scholar]
- Weidemann, W.; Klukas, C.; Klein, A.; Simm, A.; Schreiber, F.; Horstkorte, R. Lessons from GNE-deficient embryonic stem cells: Sialic acid biosynthesis is involved in proliferation and gene expression. Glycobiology 2010, 20, 107–117. [Google Scholar]
- Milman Krentsis, I.; Sela, I.; Eiges, R.; Blanchard, V.; Berger, M.; Becker Cohen, M.; Mitrani-Rosenbaum, S. GNE is involved in the early development of skeletal and cardiac muscle. PLoS One 2011, 6, e21389. [Google Scholar]
- Eisenberg, I.; Avidan, N.; Potikha, T.; Hochner, H.; Chen, M.; Olender, T.; Barash, M.; Shemesh, M.; Sadeh, M.; Grabov-Nardini, G.; et al. The UDP-N-acetylglucosamine 2-epimerase/N-acetylmannosamine kinase gene is mutated in recessive hereditary inclusion body myopathy. Nat. Genet 2001, 29, 83–87. [Google Scholar]
- Braun, T.; Rudnicki, M.A.; Arnold, H.H.; Jaenisch, R. Targeted inactivation of the muscle regulatory gene Myf-5 results in abnormal rib development and perinatal death. Cell 1992, 71, 369–382. [Google Scholar]
- Tajbakhsh, S.; Buckingham, M.E. Mouse limb muscle is determined in the absence of the earliest myogenic factor myf-5. Proc. Natl. Acad. Sci. USA 1994, 91, 747–751. [Google Scholar]
- Asakura, A.; Komaki, M.; Rudnicki, M. Muscle satellite cells are multipotential stem cells that exhibit myogenic, osteogenic, and adipogenic differentiation. Differentiation 2001, 68, 245–253. [Google Scholar]
- Wiese, C.; Rolletschek, A.; Kania, G.; Blyszczuk, P.; Tarasov, K.V.; Tarasova, Y.; Wersto, R.P.; Boheler, K.R.; Wobus, A.M. Nestin expression—A property of multi-lineage progenitor cells? Cell. Mol. Life Sci 2004, 61, 2510–2522. [Google Scholar]
- Schier, A.F. Nodal morphogens. Cold Spring Harb. Perspect. Biol 2009, 1. [Google Scholar] [CrossRef]
- Scott, I.C. Life before Nkx2.5: Cardiovascular progenitor cells: Embryonic origins and development. Curr. Top. Dev. Biol 2012, 100, 1–31. [Google Scholar]
- Beck, F.; Erler, T.; Russell, A.; James, R. Expression of Cdx-2 in the mouse embryo and placenta: Possible role in patterning of the extra-embryonic membranes. Dev. Dyn 1995, 204, 219–227. [Google Scholar]
- Desbaillets, I.; Ziegler, U.; Groscurth, P.; Gassmann, M. Embryoid bodies: An in vitro model of mouse embryogenesis. Exp. Physiol 2000, 85, 645–651. [Google Scholar]
- Wang, X.; Yang, P. In vitro differentiation of mouse embryonic stem (mES) cells using the hanging drop method. J. Vis. Exp 2008, 23. [Google Scholar] [CrossRef]
- Anisimov, S.V.; Tarasov, K.V.; Riordon, D.; Wobus, A.M.; Boheler, K.R. SAGE identification of differentiation responsive genes in P19 embryonic cells induced to form cardiomyocytes in vitro. Mech. Dev. 2002, 117, 25–74. [Google Scholar]
- Hirsch, E.; Iglesias, A.; Potocnik, A.J.; Hartmann, U.; Fässler, R. Impaired migration but not differentiation of haematopoietic stem cells in the absence of beta1 integrins. Nature 1996, 380, 171–175. [Google Scholar]
- Guan, K.; Rohwedel, J.; Wobus, A.M. Embryonic stem cell differentiation models: Cardiogenesis, myogenesis, neurogenesis, epithelial and vascular smooth muscle cell differentiation in vitro. Cytotechnology 1999, 30, 211–226. [Google Scholar]
- Kurosawa, H. Methods for inducing embryoid body formation: In vitro differentiation system of embryonic stem cells. J. Biosci. Bioeng 2007, 103, 389–398. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Weidemann, W.; Hering, J.; Bennmann, D.; Thate, A.; Horstkorte, R. The Key Enzyme of the Sialic Acid Metabolism Is Involved in Embryoid Body Formation and Expression of Marker Genes of Germ Layer Formation. Int. J. Mol. Sci. 2013, 14, 20555-20563. https://doi.org/10.3390/ijms141020555
Weidemann W, Hering J, Bennmann D, Thate A, Horstkorte R. The Key Enzyme of the Sialic Acid Metabolism Is Involved in Embryoid Body Formation and Expression of Marker Genes of Germ Layer Formation. International Journal of Molecular Sciences. 2013; 14(10):20555-20563. https://doi.org/10.3390/ijms141020555
Chicago/Turabian StyleWeidemann, Wenke, Jessica Hering, Dorit Bennmann, Annett Thate, and Rüdiger Horstkorte. 2013. "The Key Enzyme of the Sialic Acid Metabolism Is Involved in Embryoid Body Formation and Expression of Marker Genes of Germ Layer Formation" International Journal of Molecular Sciences 14, no. 10: 20555-20563. https://doi.org/10.3390/ijms141020555




