2.1. Single l-Amino Acid Mediating NZVI Formation
Based on our recent successful results in NZVI formation mediated by Glu [
8], we decided to investigate the effect of another
l-amino acid, namely arginine. This basic
l-amino acid was intentionally chosen for the fact that it (a) bears not only an amino moiety, but also a guanidyl moiety in its structure; and (b) can react with ferrous salts resulting in magnetic iron oxide nanoparticles as claimed in the literature [
27]. However, in the literature mostly XRD is used for nanoparticles characterization and determination of phases included in a sample. The usage of XRD is misleading in many cases (e.g., [
13–
16]). In fact, the results obtained by diffraction techniques are related to crystalline parts of the sample only and cannot detect any amorphous material [
5] or very thin layers on the surface of nanoparticles [
8]. There is, however, an excellent tool for the investigation of iron-containing samples,
i.e., Mössbauer spectroscopy. Since we worked exclusively with iron nanoparticles in our study, we fully exploited the potential of this iron-sensitive technique in order to characterize our samples and correctly evaluate the influence of each
l-amino acid added into the reaction mixture.
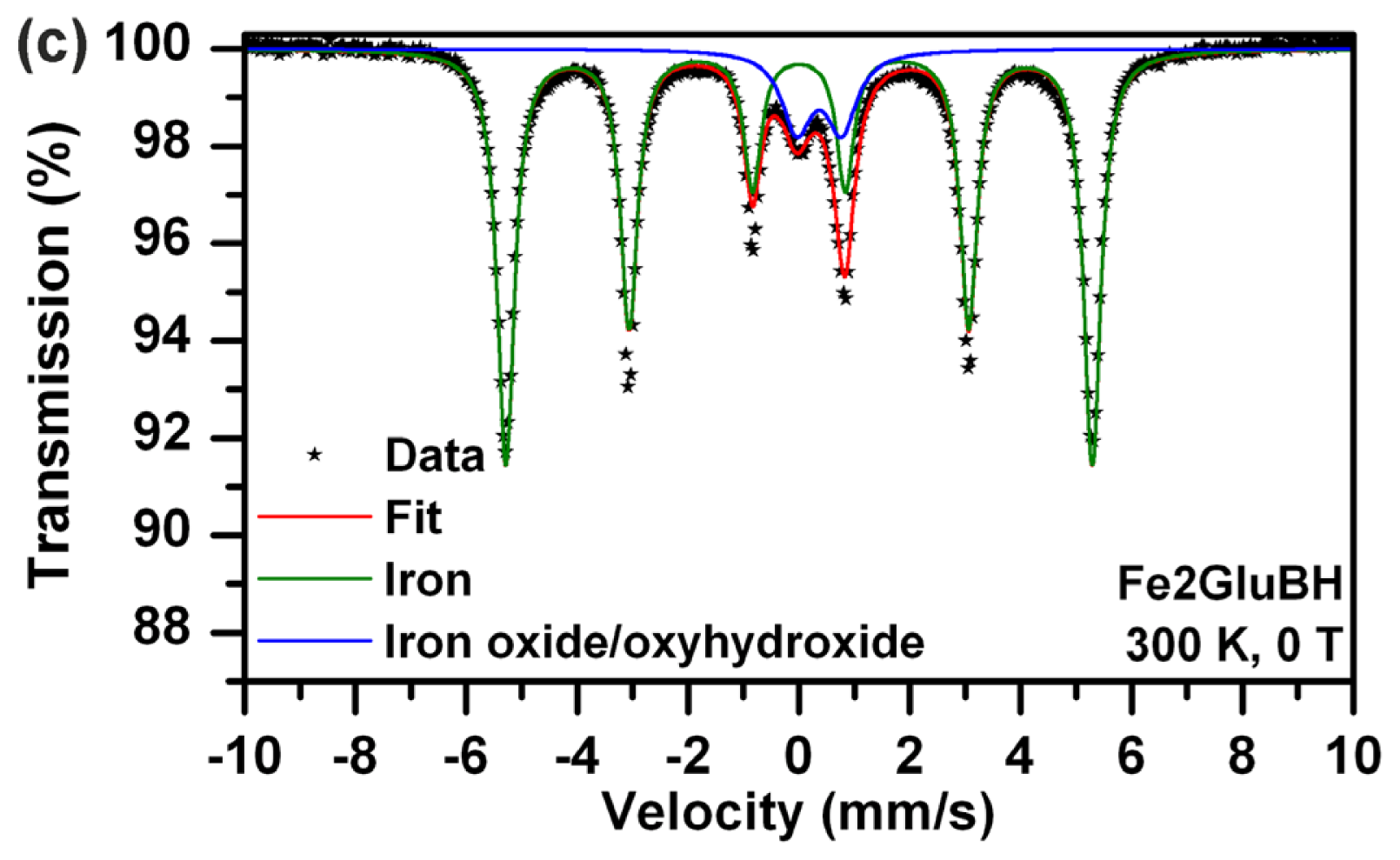
Direct comparison of Mössbauer spectra of Fe2ArgBH (
Figure 1a,b) and Fe2GluBH (
Figure 1c) revealed distinct differences in sample composition (
Table 1). In the case of Fe2ArgBH two spectra of the same sample are shown in
Figure 1; the former is recorded at room temperature (
Figure 1a) and the latter at 5 K (
Figure 1b). The Mössbauer spectrum of Fe2ArgBH measured at room temperature (
Figure 1a) is fitted with two spectral components, one doublet and one sextet. However, the sextet has a very broad line width which points to a distribution of hyperfine magnetic fields. In order to distinguish the exact composition of Fe2ArgBH, a Mössbauer spectrum at 5 K had to be measured (
Figure 1b). The experimental data points in
Figure 1b could be fitted with three components: three sextets with different isomer shifts (IS), quadrupole splitting (QS), and hyperfine magnetic field values. Two of these sextets (IS of 0.48 and 0.39 mm/s, QS of −0.02 and −0.03 mm/s, hyperfine magnetic field of 46.9 and 51.3 T) were attributed to ferric ions in octahedral and tetrahedral positions of γ-Fe
2O
3, respectively (
Table 1) [
7]. The third sextet (IS of 0.10 mm/s, QS of 0.00 mm/s, hyperfine magnetic field of 32.4 T) was assigned to α-Fe [
28]. The areas below each sub-spectrum revealed that 90% of iron atoms in Fe2ArgBH correspond to gamma ferric oxide, while only 10% of iron atoms are found in the zero oxidation state. This is a completely reversed percentual composition from that obtained for Fe2GluBH (
Figure 1c), where ~90% of iron atoms are found in the zero valence state and ~10% of iron atoms in gamma ferric oxide (
Table 1).
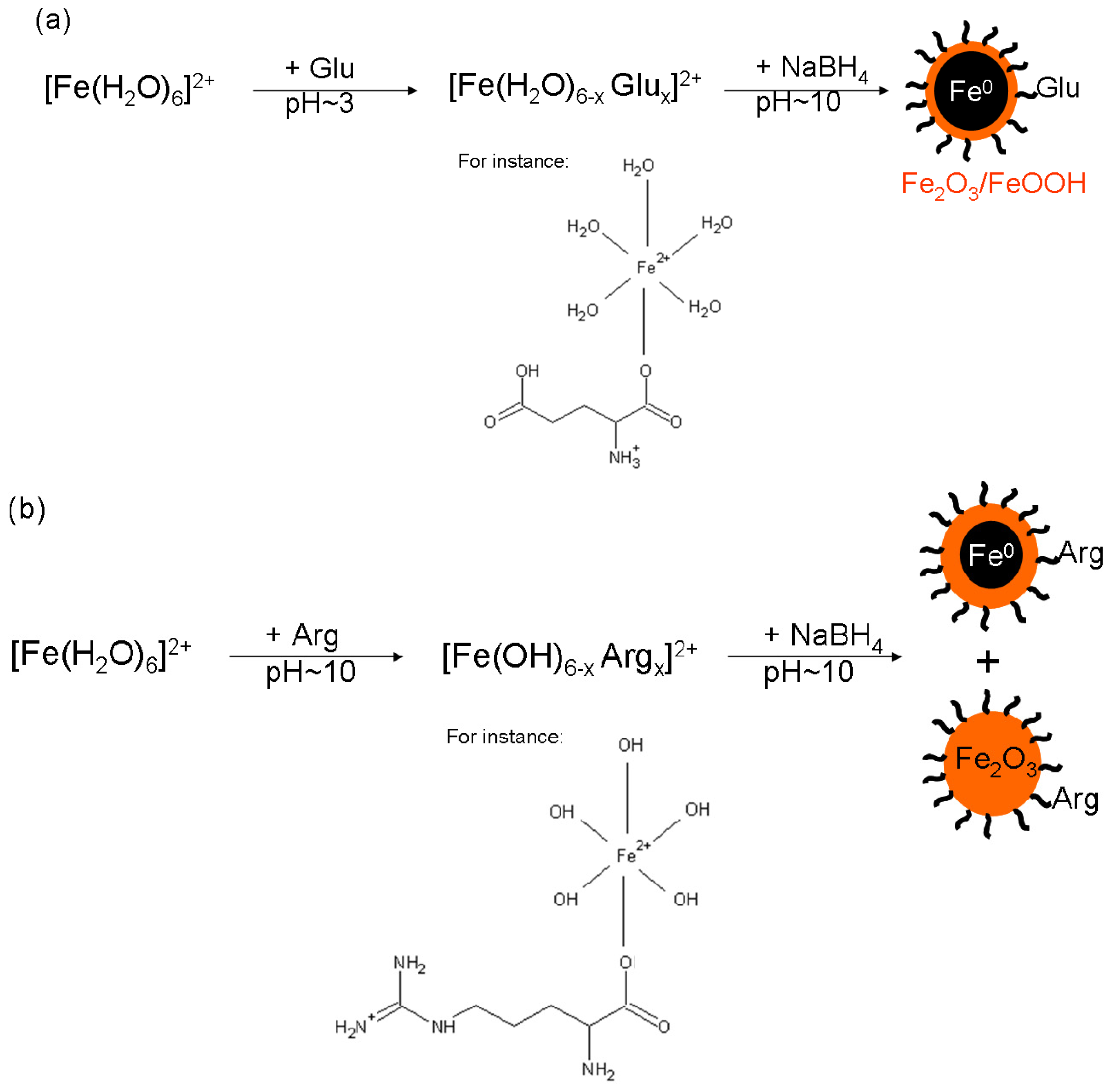
It should be noted that the preparation of both systems was performed in exactly the same way and under the same experimental conditions. Therefore, the only difference lies in the presence of either Arg, or Glu. Arg is known to induce alkaline pH values of the solutions [
27]. Indeed, the pH value of 10 mM Arg solution was measured by us and recorded at 10.02. We consequently prepared a solution of the same pH value employing sodium hydroxide and used it for the dissolution of ferrous sulphate followed by reduction induced by sodium borohydride (sample named Fe2pH10BH). The concentrations of FeSO
4 and NaBH
4 were adjusted to the same as in the case of Fe2ArgBH. The Mössbauer spectrum of the resulting system is shown in
Figure 2 and the parameters of the fit listed in
Table 1. Only a doublet (IS of 0.35 mm/s, QS of 0.70 mm/s) can best fit the experimental data points (
Figure 2). This doublet can be attributed to gamma ferric oxide/oxyhydroxide [
7].
Thus it can be concluded that alkaline pH values around 10 of the mixture of Arg and FeSO
4 provide good conditions for the formation of ferric oxide/oxyhydroxides. Subsequent addition of NaBH
4 (the final concentration of 0.12 M) induced an incomplete reduction of ferric oxide/oxyhydroxides to Fe
0. Although sodium borohydride itself generated pH~10 in each reaction mixture, the dissolution of ferrous sulphate in an alkaline solution (without reduction ability) led to the formation of ferrous hydroxide in the very first step, which was then oxidized to ferric hydroxide within five minutes due to the access of ambient air. This oxidation induced by air access was further supported by a direct visible observation of solution color changes: the addition of FeSO
4 into Arg solution resulted in a green precipitate formation which turned brownish orange within five minutes. Therefore, the formation of Fe
0 was hindered in the case of Fe2ArgBH in comparison to Fe2GluBH due to the generation of ferric oxides/oxyhydroxides which were hardly reduced to Fe
0 when 0.12 M final NaBH
4 concentration used. However, taking into account the resulting compositions of Fe2ArgBH (~90% of ferric oxides and ~10% of Fe
0) and that of the sample presented in
Figure 2 (100% of ferric oxide/oxyhydroxides), there is not only the pure effect of alkaline pH value caused by Arg in Fe2ArgBH. We thus hypothesized that an intermediate complex between a particular
l-amino acid (partially deprotonated at a given pH value) and ferrous sulphate is formed in the solution. The complex can be reduced by sodium borohydride in the next step of the NZVI formation procedure as is schematically expressed in
Scheme 1.
In order to avoid the spontaneous oxidation of iron salt in amino acid solutions with air access, we performed the same type of experiments with the only exception: ferric (instead of ferrous) salt being used as the iron source. The as-prepared Fe3ArgBH and Fe3GluBH were characterized by Mössbauer spectroscopy and for a direct comparison by XRD (
Figure 3). According to Mössbauer spectra evaluation, there are two phases: Fe
0 and Fe
3+. The quantification of each iron phase based on Mössbauer spectra, revealed that the content of Fe
0 is much higher in the case of Fe3GluBH than Fe3ArgBH (
Table 1). This supports the idea of a specific intermediate complex formation between a particular
l-amino acid and iron-containing salt, as well as, the impact of
l-amino acid induced pH value changes.
Similar to Mössbauer spectra, XRD data confirmed the presence of Fe
3+ and Fe
0 phases as shown in
Figure 3. However, the quantification of all phases detectable by XRD revealed 52% of Fe
0 in Fe3ArgBH (in comparison to 43% based on Mössbauer data–
Table 1), with 96% of Fe
0 in Fe3GluBH (80% according to Mössbauer data–
Table 1). It is obvious that there are huge discrepancies between XRD-based (
Figure 3) and Mössbauer-based (
Table 1) quantifications for the same sample. It stems from the fact that any amorphous phase and/or thin layer of ferric oxide/oxyhydroxides around the nanoparticle Fe
0 core are practically invisible to XRD (they contribute to the “XRD”-amorphous background); while detected by Mössbauer spectroscopy (as it is very sensitive in distinguishing iron-containing phases when iron nuclei experience different hyperfine interactions due to different local surroundings). In this regard, Mössbauer spectroscopy is much more accurate in solely characterizing iron-containing particles.
2.3. Effect of Sodium Borohydride on Arg and/or Glu–Verification Based on SERS Measurements
Taking into account the fact that a strong reducing agent was employed during our syntheses of iron nanoparticles, a question concerning a potential destruction of the structure of the
l-amino acids arose. Namely, it is crucial to avoid and/or at least to assess a possible reduction (driven by sodium borohydride) of carboxylic groups in
l-amino acids (amino groups are presumed not to be any further reduced). The carboxylic group is well-known for its ability to bind to an iron oxide nanoparticle surface [
16–
19,
27] and/or core-shell Fe
0-Fe
2O
3/FeOOH nanoparticles [
8]. Therefore, we decided to check whether the carboxylic functional groups of Arg and/or Glu were preserved when they were allowed to react with sodium borohydride. The samples called ArgBH and GluBH were prepared, respectively. Since the concentration of a particular
l-amino acid was low in comparison to that of borate (which arises from sodium borohydride), regular methods of vibrational spectroscopy such as infrared absorption and/or Raman scattering could not be used (borate signal prevailed). Another method, which enables selective and sensitive detection at low concentrations, was chosen,
i.e., SERS [
29–
31]. Due to the possibility of a direct covalent interaction of the amino and/or carboxylic group [
32,
33] of the
l-amino acids with Ag nanoparticle surfaces, it was presumed that the enhancement of the Raman signal would be much stronger for
l-amino acids than for borates interacting through electrostatic bonds with Ag nanoparticle surfaces. Similarly as in the case of our recent study [
34], we thus exploited SERS in conjunction with surface plasmon extinction (SPE) spectroscopy for monitoring changes in
l-amino acids used in this study.
Figure 5 shows the resulting SPE and SERS spectra.
At first, SPE spectra (
Figure 5a,b) were measured in order to determine the most suitable wavelength of a continuous laser for the excitation of Ag nanoparticles interacting with
l-amino acids and/or ArgBH/GluBH. Usually surface plasmon of non-interacting silver nanoparticles in aqueous solution appears at ~400 nm [
29,
30]. However, this characteristic feature of Ag nanoparticles is red-shifted when mutual interactions and consequent aggregation of Ag nanoparticles come into play. In our cases (
Figure 5a,b), the second maximum in the SPE spectra arose at ~660 nm. This can be related to Ag nanoparticles aggregation induced by Arg and/or ArgBH in
Figure 5a, whereas by Glu and/or GluBH in
Figure 5b. The closest excitation wavelength available for us was 633 nm which was thus employed and the resulting SERS spectra recorded (
Figure 5c,d).
For the sake of a direct comparison, the SERS spectra were normalized with respect to the most intensive peak and off set in
Figure 5c,d. It is obvious that positions and relative intensities of all peaks within each SERS spectrum of Arg and/or ArgBH interacting with silver nanoparticles (
Figure 5c) are virtually the same. This gives evidence that all important functionalities of Arg were preserved and no changes caused by sodium borohydride (employed in huge abundance: 60 NaBH
4vs. 1 amino acid in the sample named as ArgBH and/or GluBH). Based on the SERS spectra presented in
Figure 5d, exactly the same statement can be adopted for Glu and GluBH. Now recall that only the Raman signal of Arg and/or Glu molecules close to the silver nanoparticle surface can be enhanced. In other words, NaBH
4 did not reduce the carboxylic groups of the particular
l-amino acids in our systems.
Moreover, SERS measurements revealed the availability of amino and carboxyl groups in ArgBH and GluBH, respectively, for the interaction with the silver nanoparticle surface. Namely, the most intensive peak positioned at ~250 cm
−1 observed in
Figure 5c corresponds most probably to Ag-N interaction [
32]. Conversely, the most intensive peak in
Figure 5d, positioned at ~235 cm
−1, can be assigned to Ag-OOC bond [
33]. This means, in turn, that both functional groups being present in ArgBH and GluBH can be exploited for the interaction either with iron nanoparticle surfaces (in the present work), or with any other type of suitable molecule and/or nanoparticle from solution. This is particularly important for further surface modifications of iron nanoparticles, decorated by
l-amino acids on their surfaces, with the aim to tailor them for a specific application.
2.4. Impact of l-Amino Acids Mixtures on NZVI Generation
Since the effect of each
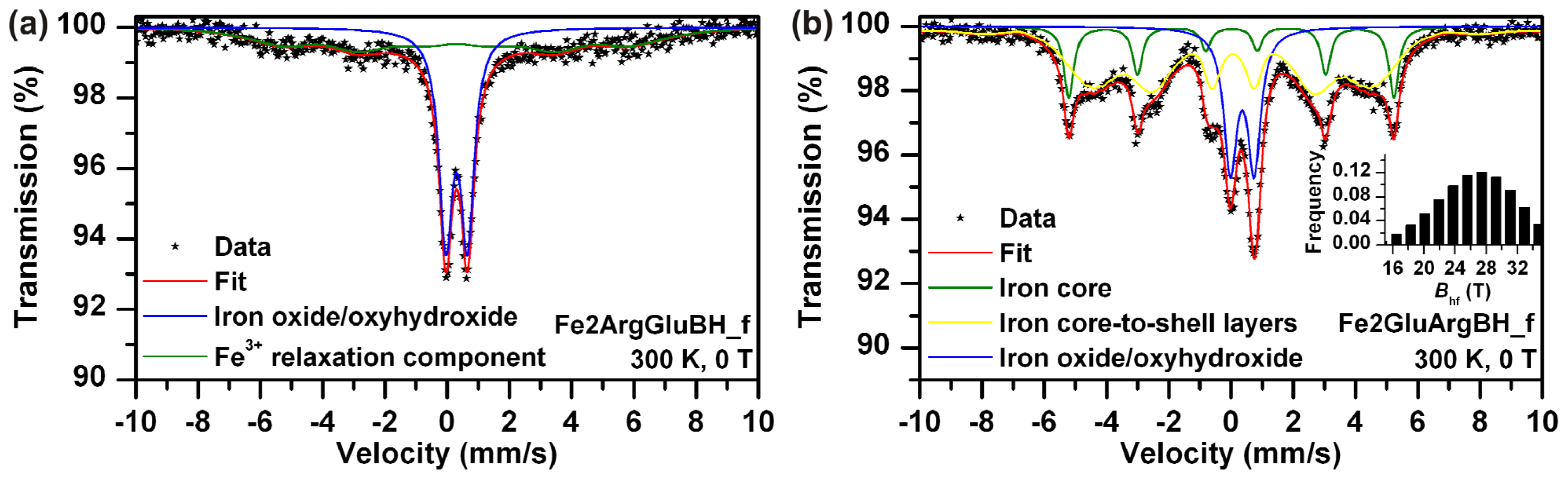
l-amino acid used in this study had already been discussed and evaluated, the impact of addition order was also investigated. Basically two different approaches could be applied: (a) dissolution of ferrous sulphate in Arg solution and subsequent addition of Glu or (b) dissolution of ferrous sulphate in Glu solution and subsequent addition of Arg. We performed both, followed by immediate reduction (within five minutes after mixture preparation), driven by sodium borohydride (the 0.12 M final concentration). The resulting systems were characterized by Mössbauer spectroscopy, TEM imaging, and SQUID magnetometry. Mössbauer spectra are shown in
Figure 6, TEM images in
Figure 7, and SQUID measurements data in
Figure 8.
While Fe2ArgGluBH (
Figure 6a) consists of superparamagnetic and magnetically ordered Fe
2O
3/FeOOH particles, thus only high spin ferric phase (
Table 1); Fe2GluArgBH (
Figure 6b) contains mostly Fe
0 (79 at.% from which 63 at.% corresponded to rather small particles) and minor Fe
2O
3 phase (21 at.%). Therefore, it can be concluded that the choice of the first
l-amino acid interacting with ferrous ions in solution critically influences the resulting phase composition of the whole sample. In other words, it is the determining step of the final iron oxidation state: Glu promotes Fe
0 formation, whereas Arg leads toward superparamagnetic Fe
2O
3/FeOOH particles when subsequent chemical reduction is performed. Furthermore, an intermediate complex between Glu and Fe
II forms (
Scheme 1a) and survives, to some extent, even under alkalization of the system by the dissolution of Arg.
Importantly, both samples (Fe2ArgGluBH and Fe2GluArgBH) manifested themselves by a bimodal distribution of particle sizes as derived from Mössbauer data (
Figure 6a,b) and confirmed by TEM imaging (
Figure 7): (i) big particles of Fe
2O
3/FeOOH being magnetically ordered (
Figure 6a) and/or big particles of Fe
0 providing a well-defined sextet with narrow lines (
Figure 6b); (ii) small particles of Fe
2O
3/FeOOH revealing superparamagnetic behavior (
Figure 6a) and/or distribution of small particles of Fe
0 (
Figure 6b). The bimodal distribution of particle sizes can be related to the simultaneous effect of both
l-amino acids present in reaction mixtures because such a phenomenon (bimodal distribution) was not observed in the cases when each
l-amino acid was applied separately.
Furthermore, TEM images clearly visualized the core-shell structure of nanoparticles in the case of Fe2GluArgBH (
Figure 7a,b). Conversely, very small nanoparticles (with no core-shell structure) together with needle-like structures and/or plates were observed in the case of Fe2ArgGluBH (
Figure 7c,d). While the former is very similar to the characteristic TEM images of Fe2GluBH published in ref. [
8]; the latter resembles TEM features of FeOOH and/or amorphous Fe
2O
3[
5,
27]. There is thus a distinct difference in the order of
l-amino acids addition to ferrous salt on the resulting iron-containing nanoparticulate structures. This conclusion can be made not only by considering TEM images, but also taking into account Mössbauer and SQUID data.
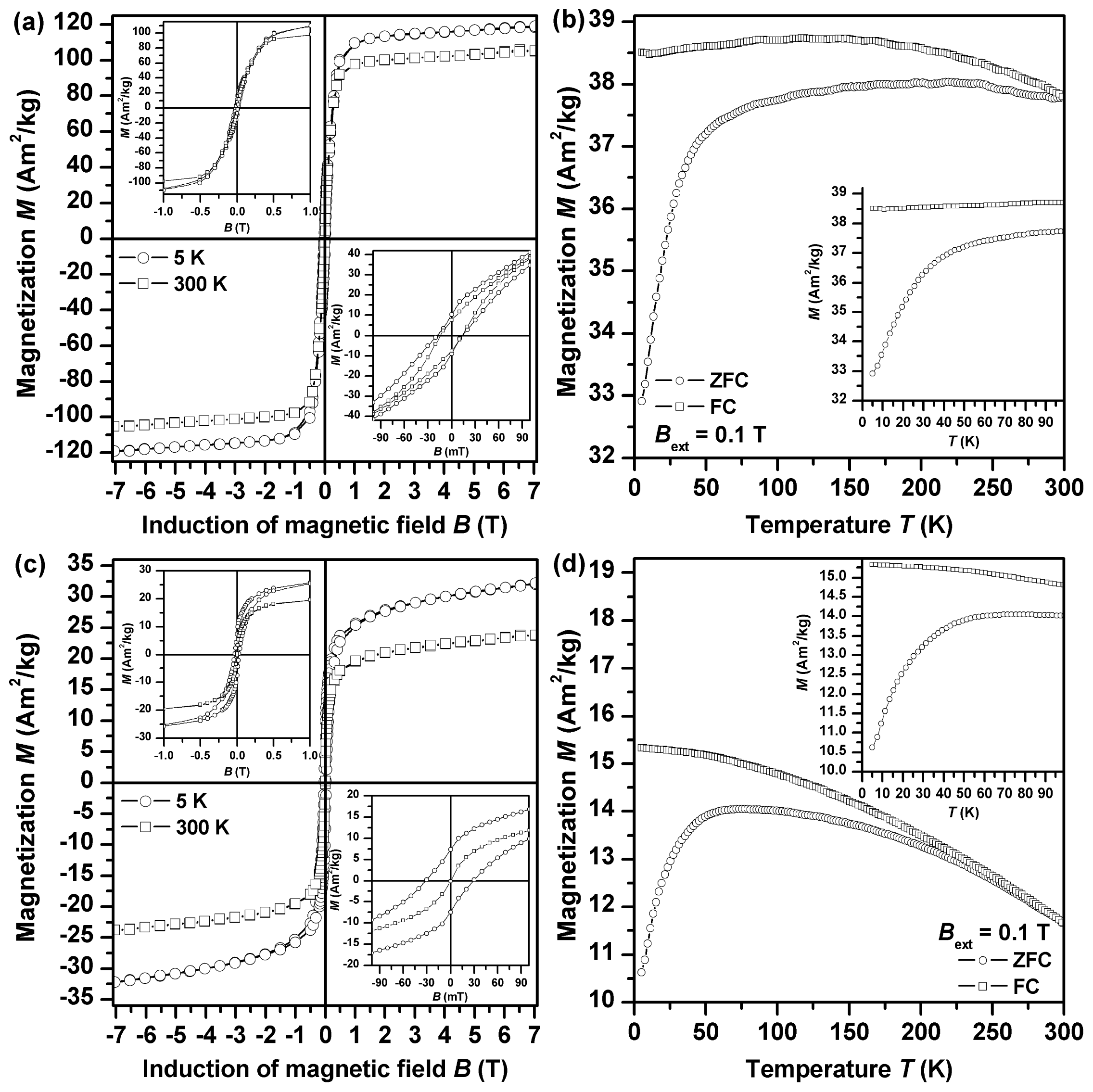
The global magnetic properties of both samples were monitored by measurement of their field- and temperature-dependent magnetization (
Figure 8). In all measuring regimes performed, magnetization values are much higher for Fe2GluArgBH (
Figure 8a,b) than those observed for Fe2ArgGluBH (
Figure 8c,d). This is to be expected as Fe
0 gives a much stronger magnetic response than γ-Fe
2O
3/FeOOH. At 5 K, the isothermal magnetization curve of both Fe2GluArgBH and Fe2ArgGluBH samples shows a hysteresis character (
Figure 8a,c), indicating a magnetically ordered state to which nanoparticles entered on cooling. In the case of Fe
0 nanoparticles, the magnetically ordered state is of a ferromagnetic character whereas for γ-Fe
2O
3 nanoparticles, the blocked state of their particle magnetic moments is supposed to evolve at low temperature. For the Fe2GluArgBH sample, a steep change in magnetization values is observed around the origin of the hysteresis loop (inset in
Figure 8a), implying a presence of some impurity with magnetic response distinct from Fe
0. On the other hand, no such behavior of the hysteresis loop trend is detected for Fe2ArgGluBH (inset in
Figure 8c), confirming, within the experimental error of the SQUID technique, its single-phased nature. This is in accordance with the phase composition of studied samples derived from the analysis of the Mössbauer spectra. In addition, the 5 K hysteresis loop of Fe2GluArgBH (inset in
Figure 8a) shows asymmetry (
i.e., difference in the values of the positive,
BC+, and negative,
BC−, coercivity–
BC+ = 13.6 mT and
BC− = 18.2 mT), a typical feature by which an exchange bias phenomenon is manifested [
35]. Exchange bias phenomenon frequently develops when an interface between two phases with magnetic ordering distinct from each other is formed. Beside other systems, this is commonly observed for nanoparticles having core-shell architecture [
36]. In our case, the particle core is made up of Fe
0 with ferromagnetic ordering whereas the particle shell is of γ-Fe
2O
3/FeOOH nature showing a ferrimagnetic alignment partly distorted by surface effects. The presence of a γ-Fe
2O
3/FeOOH shell is demonstrated by enhancement in coercivity values (for bulk and pure iron, the coercivity values are frequently smaller than 0.1 mT [
37]). A core-shell character of nanoparticles in Fe2GluArgBH is also in part supported by a magnetization value under +7 T (~119 Am
2/kg) [
37], which is reduced compared to the saturation magnetization of bulk iron (~220 Am
2/kg) [
37]. Beside this, the lowering of magnetization values is caused by the presence of diamagnetic
l-amino acids as magnetization values are recalculated on the total weight of the sample measured. On the other hand, for Fe2ArgGluBH, no asymmetric trend of the 5 K hysteresis loop (inset in
Figure 6c) is observed (
BC+ =
BC− = 30.0 mT), indicating no interface between the magnetically distinct phases. Comparing the coercivity value of γ-Fe
2O
3 nanoparticles in the Fe2ArgGluBH sample with that of bulk γ-Fe
2O
3 (~25 mT) [
37], a slight increase can be attributed to the evolution of finite-size and surface effects modifying (strengthening) the particle magnetic anisotropy [
38]. Their presence can also contribute to a reduction of a magnetization value under +7 T when comparing it with the saturation magnetization of bulk Fe
2O
3 (~85 Am
2/kg for γ-Fe
2O
3) [
37]. Similarly, as for Fe2GluArgBH, diamagnetic
l-amino acids play a significant role in lowering the magnetization values.
At 300 K, the profile of the hysteresis loops measured for Fe2GluArgBH and Fe2ArgGluBH markedly differ (
Figure 8a,c, respectively). While hysteretic behavior is still observed for Fe2GluArgBH (inset in
Figure 8a), Fe2ArgGluBH (inset in
Figure 8c) shows no hysteresis. This implies that γ-Fe
2O
3/FeOOH nanoparticles irrespective of their size in the assembly have entered into the superparamagnetic state during the time scale of SQUID measurements (~10 s). On the other hand, Fe
0-Fe
2O
3 core-shell nanoparticles are still in a magnetically stable regime with their magnetic moments fixed along the characteristic easy axis of magnetization. Note that no asymmetry in the hysteresis loop of Fe2GluArgBH (inset in
Figure 8a) is detected; some of the phases present must have entered into a (super)paramagnetic state on warming.
To get a deeper insight into the magnetic behavior of nanoparticles exhibited in both systems studied, zero-field-cooled (ZFC) and field-cooled (FC) magnetization curves were recorded (
Figure 8b,d). It is known that ZFC and FC magnetization curves are very sensitive to magnetic relaxation phenomena such as superparamagnetism. In the case of Fe2GluArgBH (
Figure 8b), the slope of the ZFC magnetization curve changes rapidly above ~50 K, implying the onset of a transition of γ-Fe
2O
3 phase to superparamagnetic regime (superparamagnetism of Fe
0 is expected for much smaller nanoparticles, <5 nm). This disconnects the magnetic interaction between the particle core and shell breaking the exchange bias phenomenon (no asymmetry in the hysteresis loop observed, see above). On the other hand, for Fe2ArgGluBH (
Figure 8d), two features are identified on the ZFC and FC magnetization curve: (i) a maximum in the ZFC magnetization curve (at ~60 K); and (ii) divergence of the ZFC and FC magnetization curve at ~240 K—both features typical for the transition from the blocked to superparamagnetic regime on warming. While the maximum in the ZFC magnetization curve corresponds to the blocking temperature of nanoparticles with the most probable size in the assembly, the temperature at which ZFC and FC magnetization curves separate (the so-called irreversible temperature) reflects the onset of the blocking mechanism of magnetic moments belonging to the biggest nanoparticles in the system. Hence, the difference in the blocking and irreversible temperature can be regarded as a measure of particle size distribution. This is in accordance with the analysis of the Mössbauer spectra, where the coexistence of sextet and doublet components, indicating a particle size distribution, was observed. At 300 K, ZFC and FC magnetization curves overlap as expected in a superparamagnetic state (no hysteresis in the room-temperature isothermal magnetization curve).
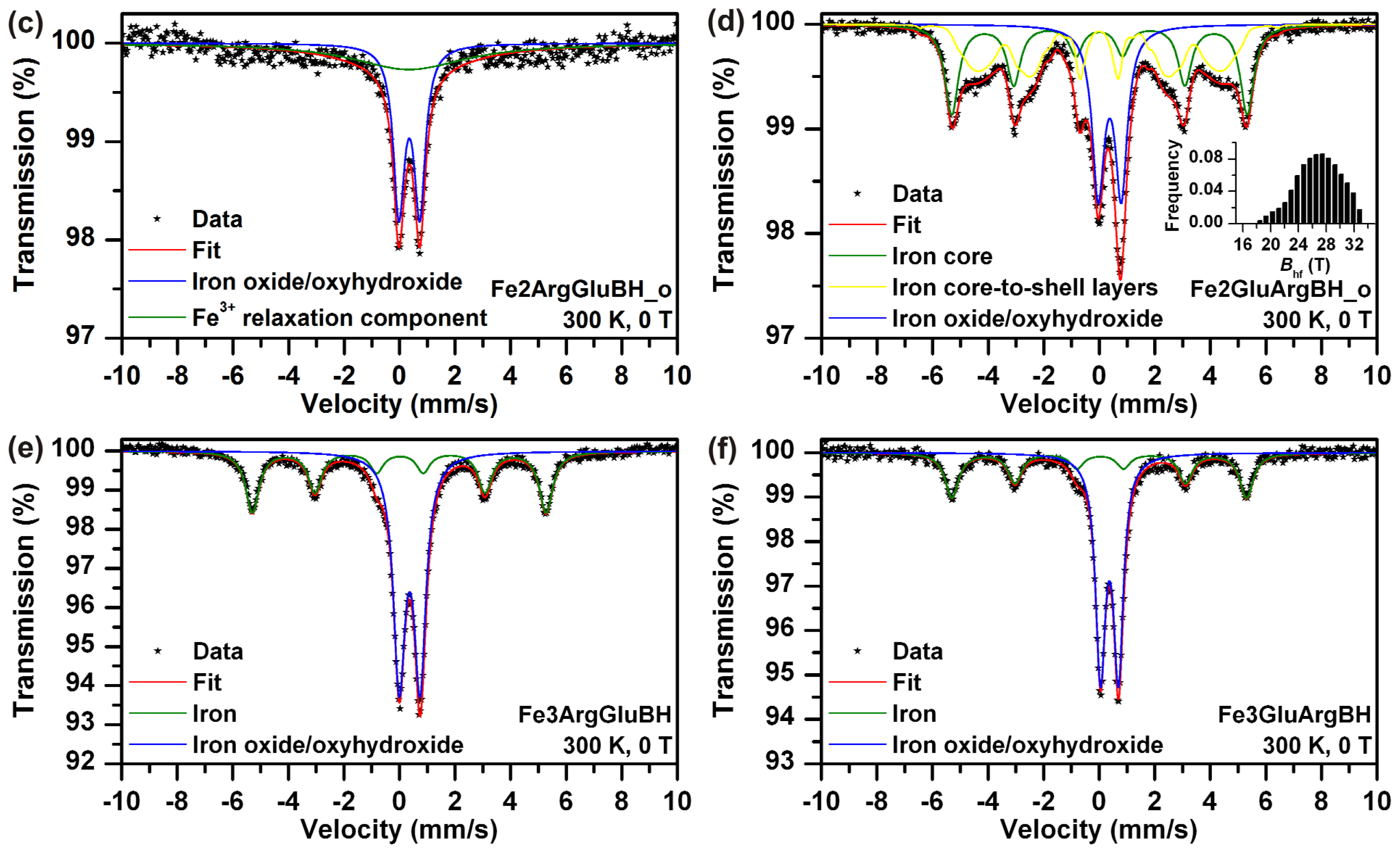
The stability of Fe2ArgGluBH and Fe2GluArgBH stored in closed vials under regular laboratory conditions for two years was investigated. Mössbauer spectra of the samples labeled as Fe2ArgGluBH_o and Fe2GluArgBH_o are presented in
Figure 6c,d, respectively, the parameters derived from the spectra are summarized in
Table 1. Comparing the spectral features of both samples measured as fresh (
Figure 6a,b) and re-measured after two years (
Figure 6c,d), they appear similar. However, regarding the parameters of spectral fits (
Table 1), the samples slightly changed. Fe2ArgGluBH_o contained a singlet corresponding to a ferric relaxation component together with the doublet of ferric oxide/oxyhydroxide shell. The occurrence of the ferric relaxation component can be thus directly related to the sample ageing. Interestingly, the ageing of the other sample revealed slight changes as well: Fe2GluArgBH_o manifested itself by the same phases as Fe2GluArgBH_f, with the only difference being in their ratios. There is a small increase of Fe
3+ content (of about 8 at.%) during two-years ageing, while, more importantly, a decrease in content of iron core-to-shell layers (from 63 at.% to 37 at.%) with a simultaneous increase of iron core component (from 16 at.% to 34 at.%). We hypothesize that the samples recrystallization took place during their ageing and is responsible for the observed results.
Last, but not least, the influence of
l-amino acids addition order to ferric salt and subsequent chemical reduction by NaBH
4 was also looked into. The as-prepared samples, labeled as Fe3ArgGluBH and Fe3GluArgBH, were characterized by Mössbauer spectroscopy (
Figure 6e,f, respectively) revealing the phase composition which is listed in
Table 1. The core-shell structures of Fe
0-Fe
2O
3/FeOOH were generated in both samples. In addition, the Fe
0 content was identical (within the experimental error) in both samples (around 38 at.%). This is quite opposite to the situation when ferrous salt was employed as the iron source (79 at.% of Fe
0 in Fe2GluArgBH and 0 at.% of Fe
0 in Fe2ArgGluBH). It can be thus concluded that an intermediate complex between Glu and ferrous sulphate (suggested in
Scheme 1a), which is subsequently reduced by NaBH
4, leads to the highest yield of Fe
0. A more general declaration based on our results can be formulated as follows: ferrous and ferric salts complexed with two selected, subsequently added
l-amino acids (Arg, Glu) revealed distinctly different behavior and, consequently, resulted in magnetic nanoparticles of various phase compositions. The importance of the choice and the order of the addition of the two selected
l-amino acids are thus supporting evidence.