Antitumor Effects of Rapamycin in Pancreatic Cancer Cells by Inducing Apoptosis and Autophagy
Abstract
:1. Introduction
2. Results and Discussions
2.1. Effect of Rapa on Proliferation of PC-2 Cells
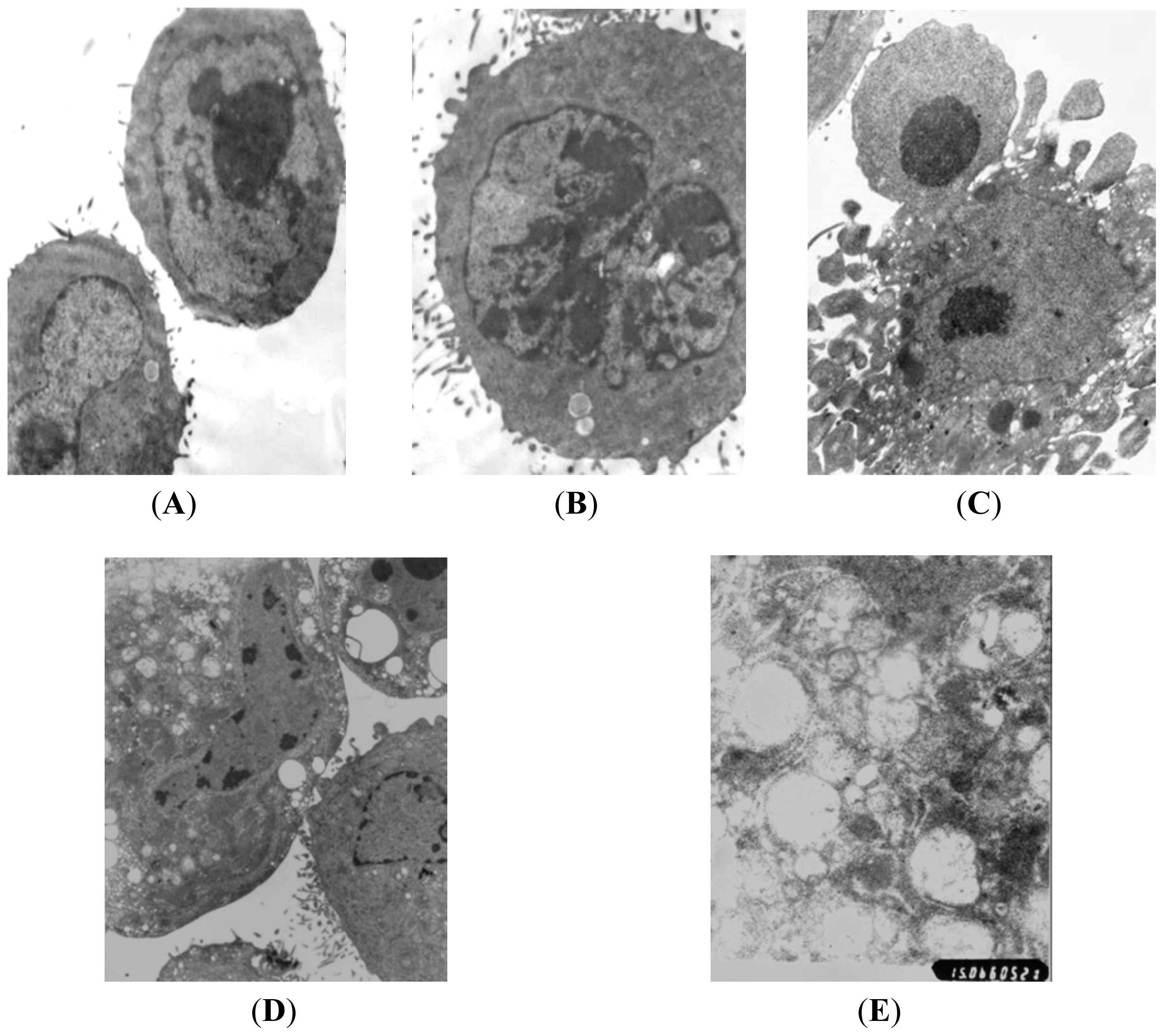
2.2. Morphological Observation of Apoptosis and Autophagy of PC-2 Cells Induced by Rapa
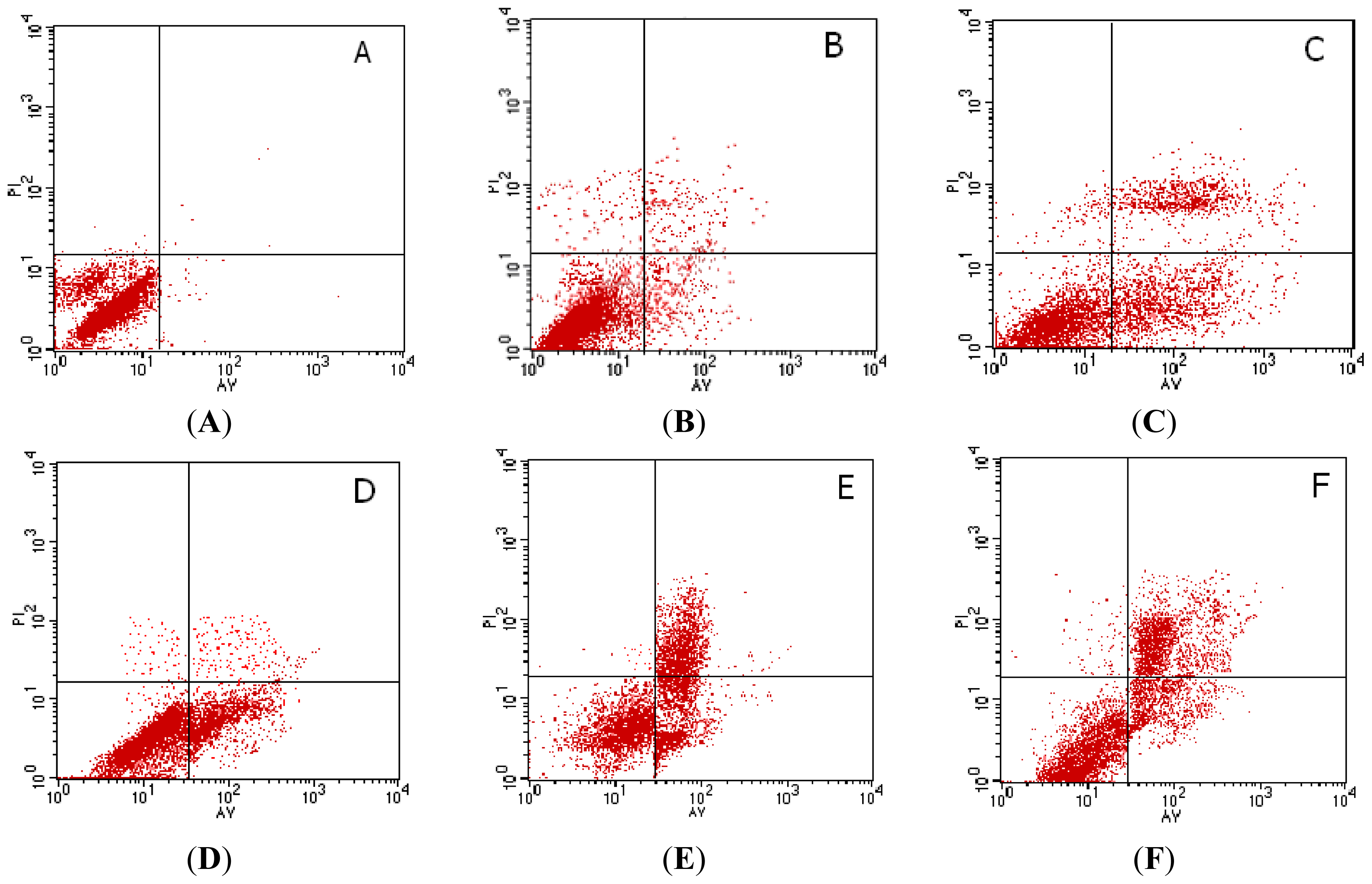
2.3. Flow Cytometry(FCM) Analysis of Cell Apoptosis Induced by Rapa
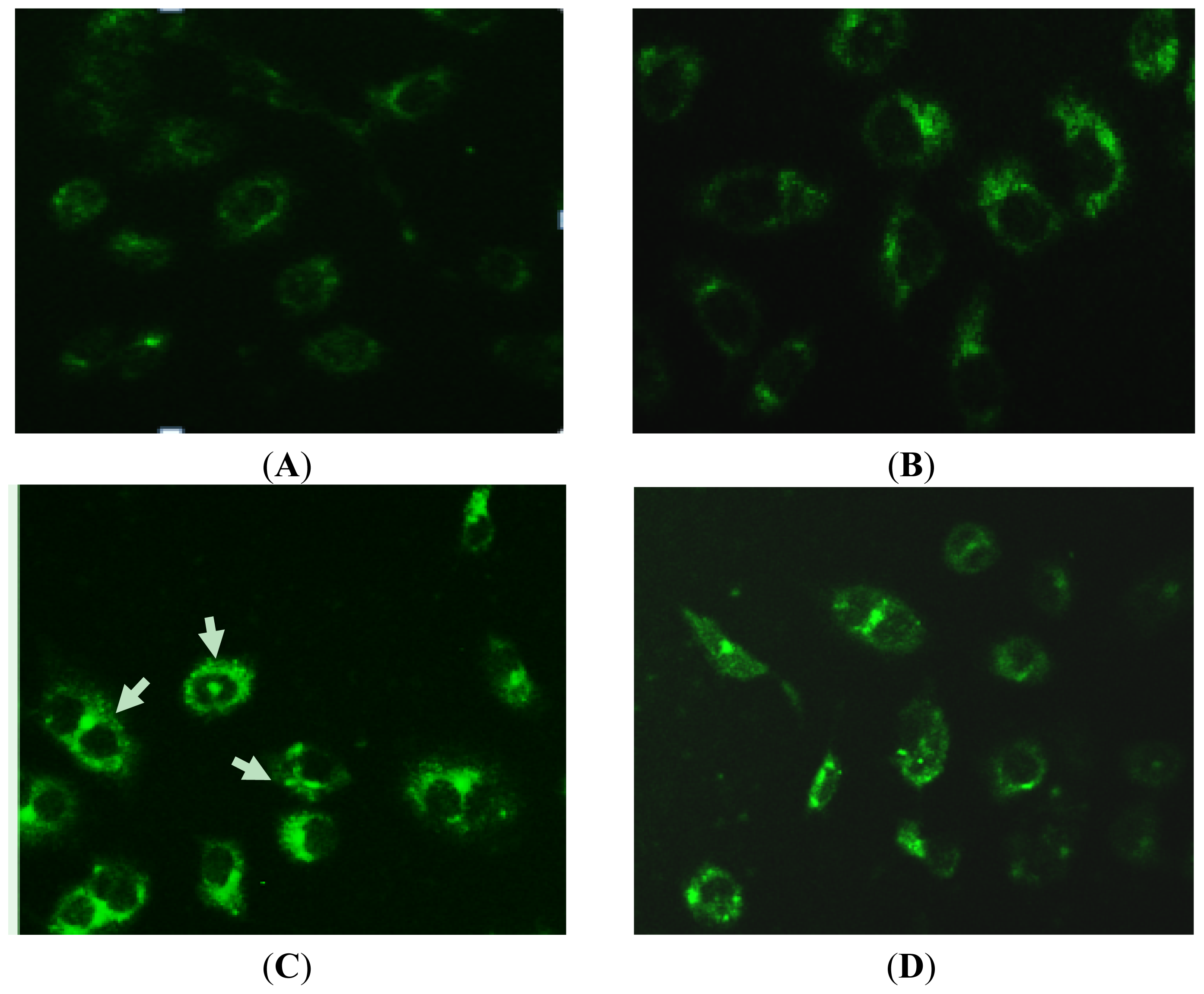
2.4. MDC-Labeled Vacuoles in Rapa-Treated PC-2 Cells
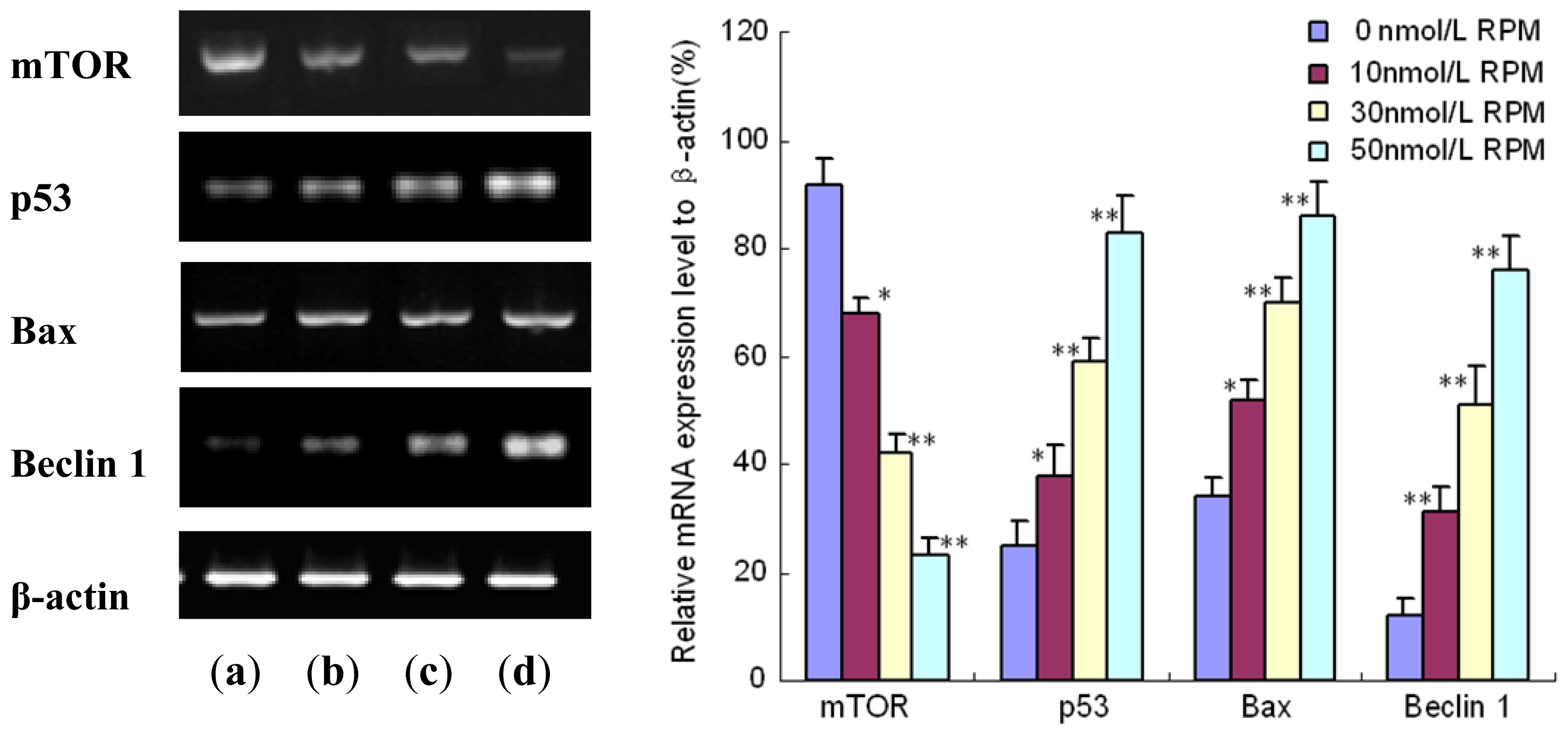
2.5. Expression of mTOR, p53, Bax and Beclin 1 Detected by Reverse-Transcription PCR(RT-PCR)
3. Experimental Section
3.1. Reagents
3.2. Cell Line and Cell Culture
3.3. MTT Assay for the Proliferation of Pancreatic Cancer Cells
3.4. Detection of Morphological Change by Transmission Electron Microscope
3.5. Apoptosis Detection by FCM
3.6. MDC Staining of Autophagic Vacuoles
3.7. Semi-Quantitative Reverse Transcription Polymerase Chain Reaction (RT-PCR) Assay
3.8. Statistical Analysis
4. Conclusions
Acknowledgments
References
- Stathis, A.; Moore, M.J. Advanced pancreatic carcinoma: Current treatment and future challenges. Nat. Rev. Clin. Oncol 2010, 7, 163–172. [Google Scholar]
- Hidalgo, M. Pancreatic cancer. N. Engl. J. Med 2010, 362, 1605–1617. [Google Scholar]
- Vincent, A.; Herman, J.; Schulick, R.; Hruban, R.H.; Goggins, M. Pancreatic cancer. Lancet 2011, 378, 607–620. [Google Scholar]
- Morris, R.E. Rapamycin: Antifungal, antitumor, antiproliferative and immunosuppressive macrolide. Transpl. Rev 1992, 16, 39–45. [Google Scholar]
- Svirshchevskaya, E.V.; Mariotti, J.; Wright, M.H.; Viskova, N.Y.; Telford, W.; Fowler, D.H.; Varticovski, L. Rapamycin delays growth of Wnt-1 tumors in spite of suppression of host immunity. BMC Cancer 2008, 8. [Google Scholar] [CrossRef]
- Wu, Q.; Kiguchi, K.; Kawamoto, T.; Ajiki, T.; Traag, J.; Carbajal, S.; Ruffino, L.; Thames, H.; Wistuba, I.; Thomas, M.; et al. Therapeutic effect of rapamycin on gallbladder cancer in a transgenic mouse model. Cancer Res 2007, 67, 3794–3800. [Google Scholar]
- Butzal, M.; Loges, S.; Schweizer, M.; Fischer, U.; Gehling, U.M.; Hossfeld, D.K.; Fiedler, W. Rapamycin inhibits proliferation and differentiation of human endothelial progenitor cells in vitro. Exp. Cell Res 2004, 300, 65–71. [Google Scholar]
- Morikawa, Y.; Koike, H.; Sekine, Y.; Matsui, H.; Shibata, Y.; Ito, K.; Suzuki, K. Rapamycin enhances docetaxel-induced cytotoxicity in a androgen-independent prostate cancer xenograft model by survivin downregulation. Biochem. Biophys. Res. Commun 2012, 419, 584–589. [Google Scholar]
- Li, R.; Wang, R.; Zhai, R.; Dong, Z. Targeted inhibition of mammalian target of rapamycin (mTOR) signaling pathway inhibits proliferation and induces apoptosis of laryngeal carcinoma cells in vitro. Tumori 2011, 97, 781–786. [Google Scholar]
- Di Paolo, S.; Teutonico, A.; Ranieri, E.; Gesualdo, L.; Schena, P.F. Monitoring antitumor efficacy of rapamycin in Kaposi sarcoma. Am. J. Kidney Dis 2007, 49, 462–470. [Google Scholar]
- Guba, M.; von Breitenbuch, P.; Steinbauer, M.; Koehl, G.; Flegel, S.; Hornung, M.; Bruns, C.J.; Zuelke, C.; Farkas, S.; Anthuber, M.; et al. Rapamycin inhibits primary and metastatic tumor growth by antiangiogenesis: Involvement of vascular endothelial growth factor. Nat. Med 2002, 8, 128–135. [Google Scholar]
- Huang, S.; Houghton, P.J. Targeting mTOR signaling for cancer therapy. Curr. Opin. Pharmacol 2003, 3, 371–377. [Google Scholar]
- Asnaghi, L.; Bruno, P.; Priulla, M.; Nicolin, A. mTOR: A protein kinase switching between life and death. Pharmacol. Res 2004, 50, 545–549. [Google Scholar]
- Ito, D.; Fujimoto, K.; Mori, T.; Kami, K.; Koizumi, M.; Toyoda, E.; Kawaguchi, Y.; Doi, R. In vivo antitumor effect of the mTOR inhibitor CCI-779 and gemcitabine in xenograft models of human pancreatic cancer. Int. J. Cancer 2006, 118, 2337–2343. [Google Scholar]
- Lang, S.A.; Gaumann, A.; Koehl, G.E.; Seidel, U.; Bataille, F.; Klein, D.; Ellis, L.M.; Bolder, U.; Hofstaedter, F.; Schlitt, H.J.; et al. Mammalian target of rapamycin is activated in human gastric cancer and serves as a target for therapy in an experimental model. Int. J. Cancer 2007, 120, 1803–1810. [Google Scholar]
- Shafer, A.; Zhou, C.; Gehrig, P.A.; Boggess, J.F.; Bae-Jump, V.L. Rapamycin potentiates the effects of paclitaxel in endometrial cancer cells through inhibition of cell proliferation and induction of apoptosis. Int. J. Cancer 2010, 126, 1144–1154. [Google Scholar]
- Makhlin, I.; Zhang, J.; Long, C.J.; Devarajan, K.; Zhou, Y.; Klein-Szanto, A.J.; Huang, M.; Chernoff, J.; Boorjian, S.A. The mTOR pathway affects proliferation and chemosensitivity of urothelial carcinoma cells and is upregulated in a subset of human bladder cancers. BJU Int 2011, 108, E84–E90. [Google Scholar]
- Kobayashi, S.; Kishimoto, T.; Kamata, S.; Otsuka, M.; Miyazaki, M.; Ishikura, H. Rapamycin, a specific inhibitor of the mammalian target of rapamycin, suppresses lymphangiogenesis and lymphatic metastasis. Cancer Sci 2007, 98, 726–733. [Google Scholar]
- Garrido-Laguna, I.; Tan, A.C.; Uson, M.; Angenendt, M.; Ma, W.W.; Villaroel, M.C.; Zhao, M.; Rajeshkumar, N.V.; Jimeno, A.; Donehower, R.; et al. Integrated preclinical and clinical development of mTOR inhibitors in pancreatic cancer. Br. J. Cancer 2010, 103, 649–655. [Google Scholar]
- Wolpin, B.M.; Hezel, A.F.; Abrams, T.; Blaszkowsky, L.S.; Meyerhardt, J.A.; Chan, J.A.; Enzinger, P.C.; Allen, B.; Clark, J.W.; Ryan, D.P.; et al. Oral mTOR inhibitor everolimus in patients with gemcitabine-refractory metastatic pancreatic cancer. J. Clin. Oncol 2009, 27, 193–198. [Google Scholar]
- Vo, K.; Amarasinghe, B.; Washington, K.; Gonzalez, A.; Berlin, J.; Dang, T.P. Targeting notch pathway enhances rapamycin antitumor activity in pancreas cancers through PTEN phosphorylation. Mol. Cancer 2011, 10. [Google Scholar] [CrossRef]
- Shen, Y.; Wang, X.; Xia, W.; Wang, C.; Cai, M.; Xie, H.; Zhou, L.; Zheng, S. Antiproliferative and overadditive effects of rapamycin and FTY720 in pancreatic cancer cells in vitro. Transplant. Proc 2008, 40, 1727–1733. [Google Scholar]
- Baehrecke, E.H. How death shapes life during development. Nat. Rev. Mol. Cell Biol 2002, 3, 779–787. [Google Scholar]
- Klionsky, D.J. The molecular machinery of autophagy: Unanswered questions. J. Cell Sci 2005, 118, 7–18. [Google Scholar]
- Kondo, Y.; Kondo, S. Autophagy and cancer therapy. Autophagy 2006, 2, 85–90. [Google Scholar]
- Yang, C.; Kaushal, V.; Shah, S.V.; Kaushal, G.P. Autophagy is associated with apoptosis in cisplatin injury to renal tubular epithelial cells. Am. J. Physiol. Renal Physiol 2008, 294, F777–F787. [Google Scholar]
- Morselli, E.; Galluzzi, L.; Kepp, O.; Vicencio, J.M.; Criollo, A.; Maiuri, M.C.; Kroemer, G. Anti- and pro-tumor functions of autophagy. Biochim. Biophys. Acta 2009, 1793, 1524–1532. [Google Scholar]
- Liang, X.H.; Jackson, S.; Seaman, M.; Brown, K.; Kempkes, B.; Hibshoosh, H.; Levine, B. Induction of autophagy and inhibition of tumorigenesis by beclin 1. Nature 1999, 402, 672–676. [Google Scholar]
- Yue, Z.; Jin, S.; Yang, C.; Levine, A.J.; Heintz, N. Beclin 1, an autophagy gene essential for early embryonic development, is a haploinsufficient tumor suppressor. Proc. Natl. Acad. Sci. USA 2003, 100, 15077–15082. [Google Scholar]
- Zhou, H.; Huang, S. Role of mTOR signaling in tumor cell motility, invasion and metastasis. Curr. Protein Pept. Sci 2011, 12, 30–42. [Google Scholar]
- Liu, J.; Stevens, P.D.; Gao, T. mTOR-dependent regulation of PHLPP expression controls the rapamycin sensitivity in cancer cells. J. Biol. Chem 2011, 286, 6510–6520. [Google Scholar]
- Garcia-Echeverria, C. Blocking the mTOR pathway: A drug discovery perspective. Biochem. Soc. Trans 2011, 39, 451–455. [Google Scholar]
- Ghayad, S.E.; Bieche, I.; Vendrell, J.A.; Keime, C.; Lidereau, R.; Dumontet, C.; Cohen, P.A. mTOR inhibition reverses acquired endocrine therapy resistance of breast cancer cells at the cell proliferation and gene-expression levels. Cancer Sci 2008, 99, 1992–2003. [Google Scholar]
- Dasanu, C.A.; Clark, B.A., 3rd; Alexandrescu, D.T. mTOR-blocking agents in advanced renal cancer: An emerging therapeutic option. Expert Opin. Investig. Drugs 2009, 18, 175–187. [Google Scholar]
- Eum, K.H.; Lee, M. Targeting the autophagy pathway using ectopic expression of Beclin 1 in combination with rapamycin in drug-resistant v-Ha-ras-transformed NIH 3T3 cells. Mol. Cells 2011, 31, 231–238. [Google Scholar]
- Biederbick, A.; Kern, H.F.; Elsässer, H.P. Monodansylcadaverine (MDC) is a specific in vivo marker for autophagic vacuoles. Eur. J. Cell Biol 1995, 66, 3–14. [Google Scholar]
- Haupt, S.; Berger, M.; Goldberg, Z.; Haupt, Y. Apoptosis—The p53 network. J. Cell Sci 2003, 116, 4077–4085. [Google Scholar]
- Miyake, N.; Chikumi, H.; Takata, M.; Nakamoto, M.; Igishi, T.; Shimizu, E. Rapamycin induces p53-independent apoptosis through the mitochondrial pathway in non-small cell lung cancer cells. Oncol. Rep 2012, 28, 848–854. [Google Scholar]
- Thornborrow, E.C.; Patel, S.; Mastropietro, A.E.; Schwartzfarb, E.M.; Manfredi, J.J. A conserved intronic response element mediates direct p53-dependent transcriptional activation of both the human and murine bax genes. Oncogene 2002, 21, 990–999. [Google Scholar]
- Wu, X.; Deng, Y. Bax and BH3 domain only proteins in P53 mediated apoptosis. Front. BioSci 2002, 7, 151–156. [Google Scholar]
- Kihara, A.; Kabeya, Y.; Ohsumi, Y.; Yoshimori, T. Beclin-phosphatidylinositol 3-kinase complex functions at the transgolgi network. EMBO Rep 2001, 2, 330–335. [Google Scholar]
- Donohue, T.M., Jr. Autophagy and ethanol-induced liver injury. World J. Gastroenterol. 2009, 15, 1178–1185. [Google Scholar]
- Dai, Z.J.; Gao, J.; Ma, X.B.; Yan, K.; Liu, X.X.; Kang, H.F.; Ji, Z.Z.; Guan, H.T.; Wang, X.J. Up-regulation of hypoxia inducible factor-1á by cobalt chloride correlates with proliferation and apoptosis in PC-2 cells. J. Exp. Clin. Cancer Res. 2012, 31. [Google Scholar] [CrossRef]
- Ma, G.; Yang, C.L.; Qu, Y.; Wei, H.Y.; Zhang, T.T.; Zhang, N.J. The flavonoid component isorhamnetin in vitro inhibits proliferation and induces apoptosis in Eca-109 cells. Chem. Biol. Interact 2007, 167, 153–160. [Google Scholar]
- Dai, Z.J.; Gao, J.; Ji, Z.Z.; Wang, X.J.; Ren, H.T.; Liu, X.X.; Wu, W.Y.; Kang, H.F.; Guan, H.T. Matrine Induces Apoptosis in Gastric Carcinoma Cells via Alteration of Fas/FasL and Activation of Caspase-3. J. Ethnopharmacol 2009, 123, 91–96. [Google Scholar]
- Zhang, J.Q.; Li, Y.M.; Liu, T.; He, W.T.; Chen, Y.T.; Chen, X.H.; Li, X.; Zhou, W.C.; Yi, J.F.; Ren, Z.J. Antitumor effect of matrine in human hepatoma G2 cells by inducing apoptosis and autophagy. World J. Gastroenterol. 2010, 16, 4281–4290. [Google Scholar]
- Guan, H.T.; Xue, X.H.; Dai, Z.J.; Wang, X.J.; Li, A.; Qin, Z.Y. Downregulation of survivin expression by small interfering RNA induces pancreatic cancer cell apoptosis and enhances its radiosensitivity. World J. Gastroenterol 2006, 12, 2901–2907. [Google Scholar]


© 2013 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Dai, Z.-J.; Gao, J.; Ma, X.-B.; Kang, H.-F.; Wang, B.-F.; Lu, W.-F.; Lin, S.; Wang, X.-J.; Wu, W.-Y. Antitumor Effects of Rapamycin in Pancreatic Cancer Cells by Inducing Apoptosis and Autophagy. Int. J. Mol. Sci. 2013, 14, 273-285. https://doi.org/10.3390/ijms14010273
Dai Z-J, Gao J, Ma X-B, Kang H-F, Wang B-F, Lu W-F, Lin S, Wang X-J, Wu W-Y. Antitumor Effects of Rapamycin in Pancreatic Cancer Cells by Inducing Apoptosis and Autophagy. International Journal of Molecular Sciences. 2013; 14(1):273-285. https://doi.org/10.3390/ijms14010273
Chicago/Turabian StyleDai, Zhi-Jun, Jie Gao, Xiao-Bin Ma, Hua-Feng Kang, Bao-Feng Wang, Wang-Feng Lu, Shuai Lin, Xi-Jing Wang, and Wen-Ying Wu. 2013. "Antitumor Effects of Rapamycin in Pancreatic Cancer Cells by Inducing Apoptosis and Autophagy" International Journal of Molecular Sciences 14, no. 1: 273-285. https://doi.org/10.3390/ijms14010273



