Less Expression of Prohibitin Is Associated with Increased Paired Box 2 (PAX2) in Renal Interstitial Fibrosis Rats
Abstract
:1. Introduction
2. Results
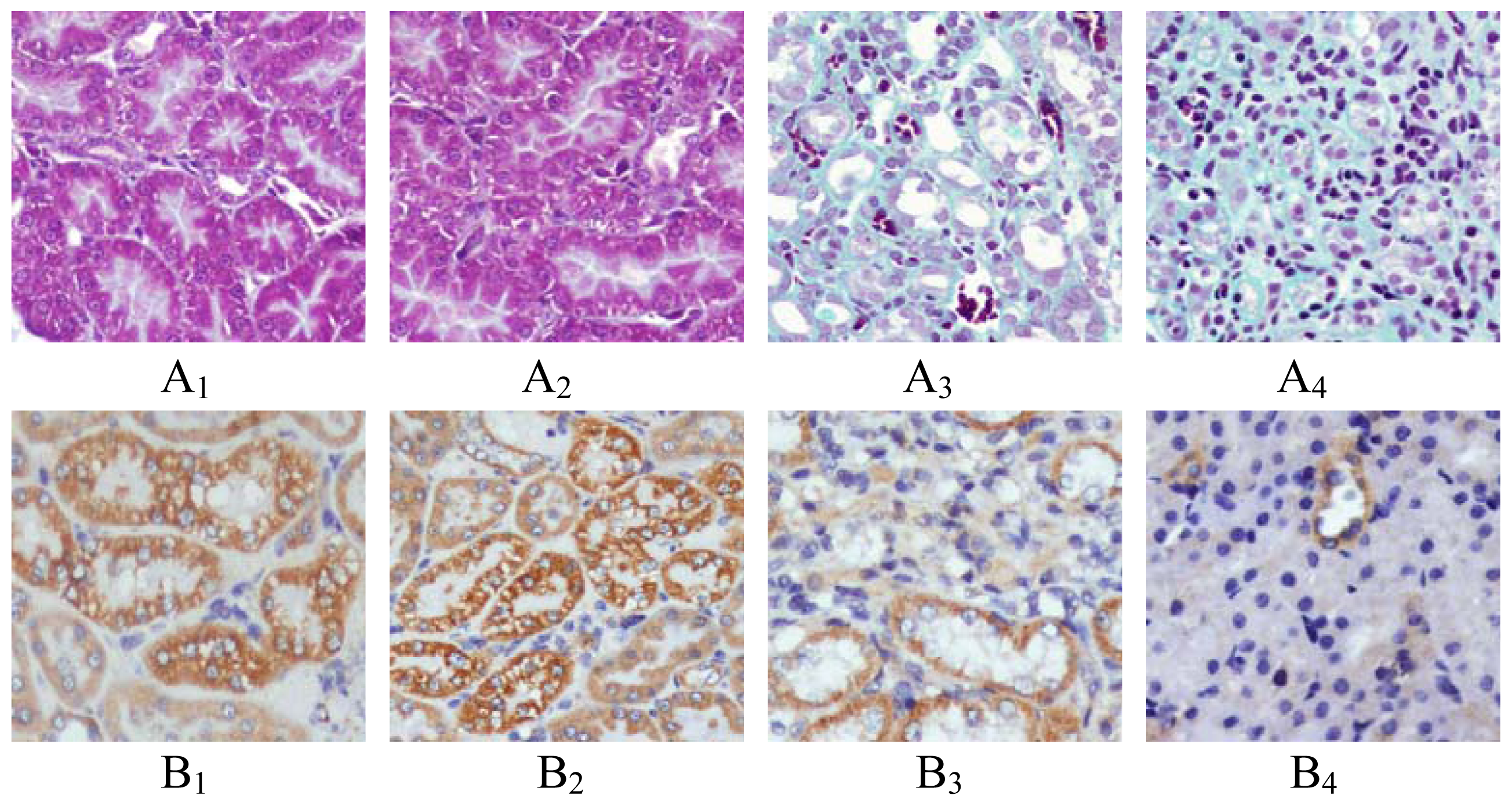
2.1. Renal Morphology
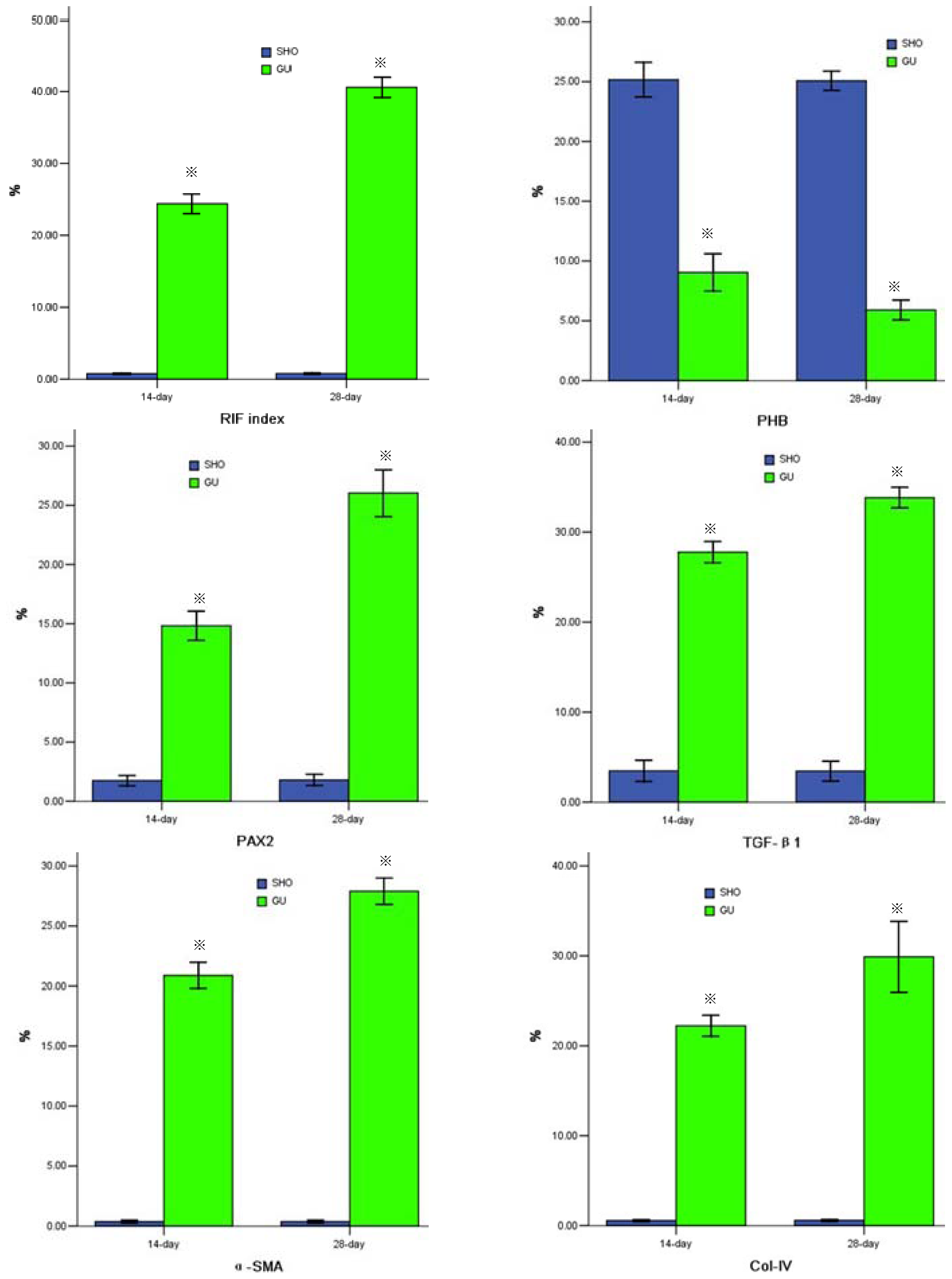
2.2. Protein Expression of PHB, PAX2, TGF-βl, α-SMA, Col-IV, FN or Cleaved Caspase-3
2.3. Cell Apoptosis
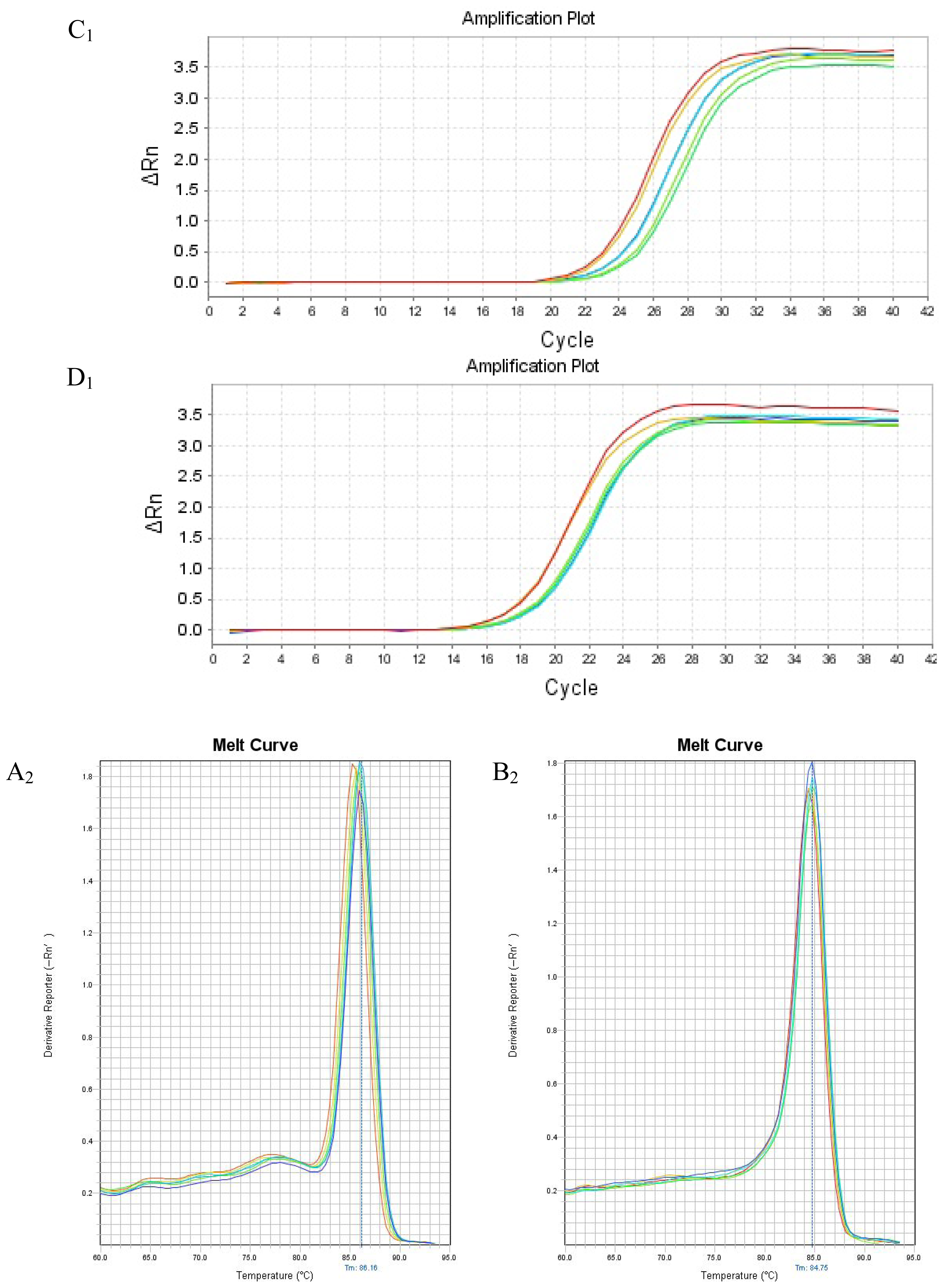
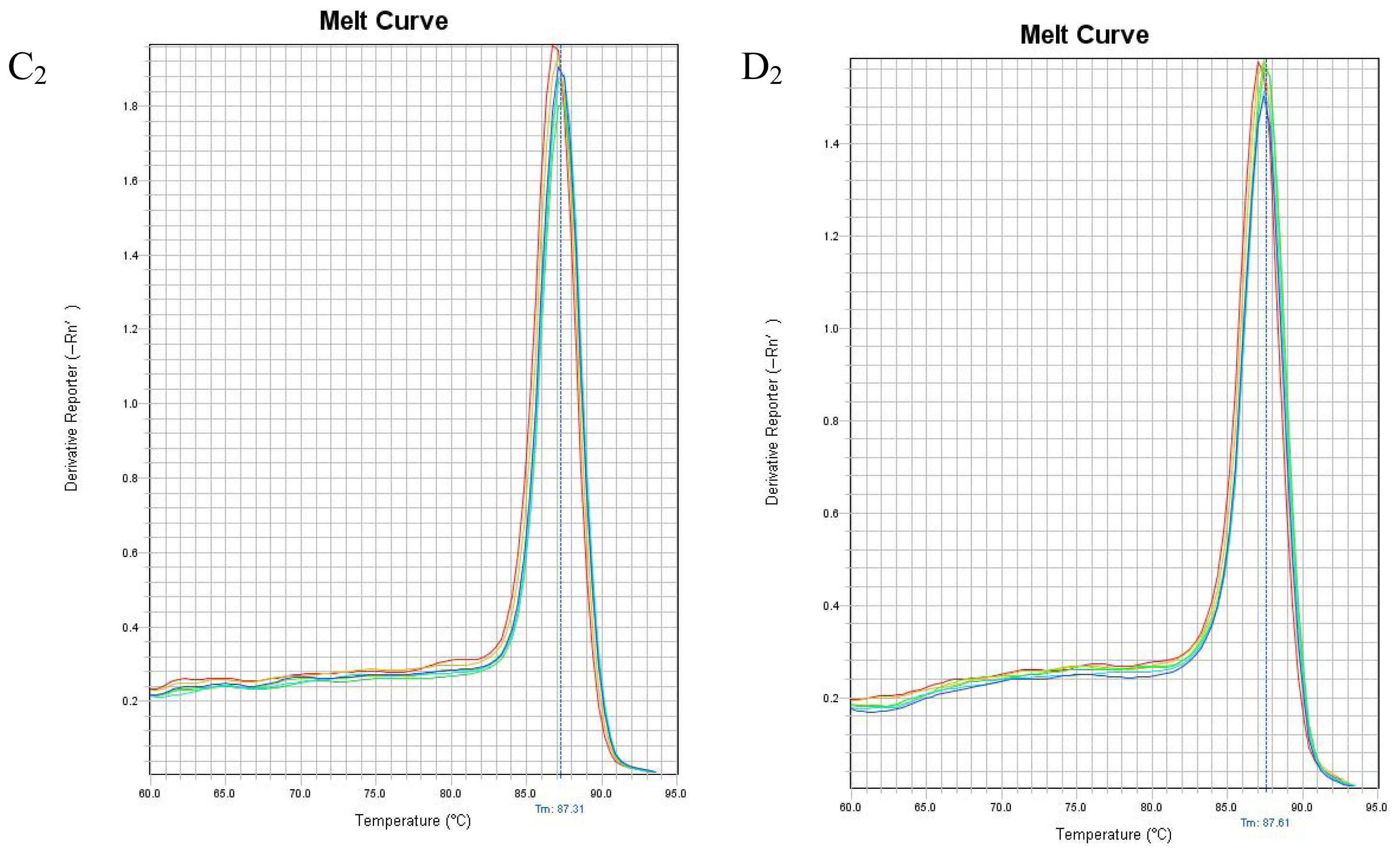
2.4. mRNA Expression of PHB, PAX2 or TGF-βl
2.5. Correlation Analysis
3. Discussion
4. Materials and Methods
4.1. Animal Model
4.2. Renal Morphology
4.3. Immunohistochemical Analysis of the Protein Expressions of PHB, TGF-β1, α-SMA, Collagen-IV (Col-IV), Fibronectin (FN) and Cleaved Caspase-3
4.4. Apoptosis Assay
4.5. Real Time Reverse Transcription Polymerase Chain Reaction to Detect PHB, PAX2 and TGF-β1 mRNA Expressions in Renal Tissue
4.6. Statistical Analysis
5. Conclusions
Acknowledgments
- Conflict of InterestThe authors declare no conflict of interest.
References
- Xie, X.S.; Liu, H.C.; Wang, F.P.; Zhang, C.L.; Zuo, C.; Deng, Y.; Fan, J.M. Ginsenoside Rg1 modulation on thrombospondin-1 and vascular endothelial growth factor expression in early renal fibrogenesis in unilateral obstruction. Phytother Res 2010, 24, 1581–1587. [Google Scholar]
- Correa-Costa, M.; Semedo, P.; Monteiro, A.P.; Silva, R.C.; Pereira, R.L.; Goncalves, G.M.; Marques, G.D.; Cenedeze, M.A.; Faleiros, A.C.; Keller, A.C.; et al. Induction of heme oxygenase-1 can halt and even reverse renal tubule-interstitial fibrosis. PLoS One 2010, 5, 14298. [Google Scholar]
- Mishra, S.; Ande, S.R.; Nyomba, B.L. The role of prohibitin in cell signaling. FEBS. J 2010, 277, 3937–3946. [Google Scholar]
- Li, Q.F.; Liang, Y.; Shi, S.L.; Liu, Q.R.; Xu, D.H.; Jing, G.J.; Wang, S.Y.; Kong, H.Y. Localization of prohibitin in the nuclear matrix and alteration of its expression during differentiation of human neuroblastoma SK-N-SH cells induced by retinoic acid. Cell Mol. Neurobiol 2011, 31, 203–211. [Google Scholar]
- Liu, X.; Ren, Z.; Zhan, R.; Wang, X.; Wang, X.; Zhang, Z.; Leng, X.; Yang, Z.; Qian, L. Prohibitin protects against oxidative stress-induced cell injury in cultured neonatal cardiomyocyte. Cell Stress Chaperones 2009, 14, 311–319. [Google Scholar]
- Popova, E.N.; Pletjushkina, O.Y.; Dugina, V.B.; Domnina, L.V.; Ivanova, O.Y.; Izyumov, D.S.; Skulachev, V.P.; Chernyak, B.V. Scavenging of reactive oxygen species in mitochondria induces myofibroblast differentiation. Antioxid. Redox. Signal 2010, 13, 1297–1307. [Google Scholar]
- Guo, W.; Xu, H.; Chen, J.; Yang, Y.; Jin, J.W.; Fu, R.; Liu, H.M.; Zha, X.L.; Zhang, Z.G.; Huang, W.Y. Prohibitin suppresses renal interstitial fibroblasts proliferation and phenotypic change induced by transforming growth factor-beta1. Mol. Cell Biochem 2007, 295, 167–177. [Google Scholar]
- Luu, V.D.; Boysen, G.; Struckmann, K.; Casagrande, S.; Von Teichman, A.; Wild, P.J.; Sulser, T.; Schraml, P.; Moch, H. Loss of VHL and hypoxia provokes PAX2 up-regulation in clear cell renal cell carcinoma. Clin. Cancer Res 2009, 15, 3297–3304. [Google Scholar]
- Zhou, T.B.; Qin, Y.H.; Lei, F.Y.; Zhao, Y.J.; Huang, W.F. Association of PAX2 with cell apoptosis in unilateral ureteral obstruction rats. Ren. Fail 2012, 34, 194–202. [Google Scholar]
- Sehgal, R.; Sheibani, N.; Rhodes, S.J.; Belecky, A.T. BMP7 and SHH regulate PAX2 in mouse retinal astrocytes by relieving TLX repression. Dev. Biol 2009, 332, 429–443. [Google Scholar]
- Busse, A.; Rietz, A.; Schwartz, S.; Thiel, E.; Keilholz, U. An intron 9 containing splice variant of PAX2. J. Transl. Med 2009, 7, 36. [Google Scholar]
- Warchol, M.E.; Richardson, G.P. Expression of the PAX2 transcription factor is associated with vestibular phenotype in the avian inner ear. Dev. Neurobiol 2009, 69, 191–202. [Google Scholar]
- Bose, S.K.; Gibson, W.; Bullard, R.S.; Donald, C.D. PAX2 oncogene negatively regulates the expression of the host defense peptide human beta defensin-1 in prostate cancer. Mol. Immunol 2009, 46, 1140–1148. [Google Scholar]
- Chen, Y.W.; Liu, F.; Tran, S.; Zhu, Y.; Hebert, M.J.; Ingelfinger, J.R.; Zhang, S.L. Reactive oxygen species and nuclear factor-kappa B pathway mediate high glucose-induced PAX2 gene expression in mouse embryonic mesenchymal epithelial cells and kidney explants. Kidney Int 2006, 70, 1607–1615. [Google Scholar]
- Zhang, S.L.; Chen, Y.W.; Tran, S.; Chenier, I.; Hebert, M.J.; Ingelfinger, J.R. Reactive oxygen species in the presence of high glucose alter ureteric bud morphogenesis. J. Am. Soc. Nephrol 2007, 18, 2105–2115. [Google Scholar]
- Murer, L.; Caridi, G.; Della, V.M.; Montini, G.; Carasi, C.; Ghiggeri, G.; Zacchello, G. Expression of nuclear transcription factor PAX2 in renal biopsies of juvenile nephronophthisis. Nephron 2002, 91, 588–593. [Google Scholar]
- Meng, L.; Qu, L.; Tang, J.; Cai, S.Q.; Wang, H.; Li, X. A combination of Chinese herbs, Astragalus membranaceus var. mongholicus and Angelica sinensis, enhanced nitric oxide production in obstructed rat kidney. Vascul. Pharmacol 2007, 47, 174–183. [Google Scholar]
- Kim, D.S.; Kim, B.; Tahk, H.; Kim, D.H.; Ahn, E.R.; Choi, C.; Jeon, Y.; Park, S.Y.; Lee, H.; Oh, S.H.; Kim, S.Y. Transglutaminase 2 gene ablation protects against renal ischemic injury by blocking constant NF-kappaB activation. Biochem. Biophys. Res. Commun 2010, 403, 479–484. [Google Scholar]
- Kaneyama, T.; Kobayashi, S.; Aoyagi, D.; Ehara, T. Tranilast modulates fibrosis, epithelial-mesenchymal transition and peritubular capillary injury in unilateral ureteral obstruction rats. Pathology 2010, 42, 564–573. [Google Scholar]
- Cheng, J.; Truong, L.D.; Wu, X.; Kuhl, D.; Lang, F.; Du, J. Serum- and glucocorticoid-regulated kinase 1 is upregulated following unilateral ureteral obstruction causing epithelial-mesenchymal transition. Kidney Int 2010, 78, 668–678. [Google Scholar]
- Tang, R.; Yang, C.; Tao, J.L.; You, Y.K.; An, N.; Li, S.M.; Wu, H.L.; Liu, H.F. Epithelial-mesenchymal transdifferentiation of renal tubular epithelial cells induced by urinary proteins requires the activation of PKC-alpha and betaI isozymes. Cell Biol. Int 2011, 35, 953–959. [Google Scholar]
- Wang, Q.L.; Tao, Y.Y.; Yuan, J.L.; Shen, L.; Liu, C.H. Salvianolic acid B prevents epithelial-to-mesenchymal transition through the TGF-beta1 signal transduction pathway in vivo and in vitro. BMC. Cell Biol 2010, 11, 31. [Google Scholar]
- Minz, R.W.; Bakshi, A.; Chhabra, S.; Joshi, K.; Sakhuja, V. Role of myofibroblasts and collagen type IV in patients of IgA nephropathy as markers of renal dysfunction. Indian J. Nephrol 2010, 20, 34–39. [Google Scholar]
- Ma, F.Y.; Tesch, G.H.; Ozols, E.; Xie, M.; Schneider, M.D.; Nikolic-Paterson, D.J. TGF-β1-activated kinase-1 (TAK1) regulates inflammation and fibrosis in the obstructed kidney. Am. J. Physiol. Renal. Physiol 2011, 300, F1410–F1421. [Google Scholar]
- Barnes, J.L.; Gorin, Y. Myofibroblast differentiation during fibrosis: Role of NAD(P)H oxidases. Kidney Int 2011, 79, 944–956. [Google Scholar]
- Djamali, A.; Vidyasagar, A.; Yagci, G.; Huang, L.J.; Reese, S. Mycophenolic acid may delay allograft fibrosis by inhibiting transforming growth factor-beta1-induced activation of Nox-2 through the nuclear factor-kappaB pathway. Transplantation 2010, 90, 387–393. [Google Scholar]
- Zhou, G.; Li, C.; Cai, L. Advanced glycation end-products induce connective tissue growth factor-mediated renal fibrosis predominantly through transforming growth factor beta-independent pathway. Am. J. Pathol 2004, 165, 2033–2043. [Google Scholar]
- Kelley, R.; Werdin, E.S.; Bruce, A.T.; Choudhury, S.; Wallace, S.M.; Ilagan, R.M.; Cox, B.R.; Tatsumi-Ficht, P.; Rivera, E.A.; Spencer, T.; et al. Tubular cell-enriched subpopulation of primary renal cells improves survival and augments kidney function in rodent model of chronic kidney disease. Am. J. Physiol. Renal Physiol 2010, 299, 1026–1039. [Google Scholar]
- Mohamed, S.A.; Misfeld, M.; Hanke, T.; Charitos, E.I.; Bullerdiek, J.; Belge, G.; Kuehnel, W.; Sievers, H.H. Inhibition of caspase-3 differentially affects vascular smooth muscle cell apoptosis in the concave versus convex aortic sites in ascending aneurysms with a bicuspid aortic valve. Ann. Anat 2010, 192, 145–150. [Google Scholar]
- Cai, H.; Gu, Y.; Sun, Q.; Zeng, J.; Dong, N.; Zhao, G. Effect of hematoporphyrin monomethyl ether-mediated photodynamic therapy on hypertrophic scar fibroblasts. Photodermatol. Photoimmunol. Photomed 2011, 27, 90–96. [Google Scholar]
- Yue, Z.; She, R.P.; Bao, H.H.; Tian, J.; Yu, P.; Zhu, J.; Chang, L.; Ding, Y.; Sun, Q. Necrosis and apoptosis of renal tubular epithelial cells in rats exposed to 3-methyl-4-nitrophenol. Environ. Toxicol 2011. [Google Scholar] [CrossRef]
- He, W.; Wang, Y.; Zhang, M.Z.; You, L.; Davis, L.S.; Fan, H.; Yang, H.C.; Fogo, A.B.; Zent, R.; Harris, R.C.; et al. Sirt1 activation protects the mouse renal medulla from oxidative injury. J. Clin. Invest 2010, 120, 1056–1068. [Google Scholar]
- Li, L.; Zepeda-Orozco, D.; Black, R.; Lin, F. Autophagy is a component of epithelial cell fate in obstructive uropathy. Am. J. Pathol 2010, 176, 1767–1778. [Google Scholar]
- Mandache, E.; Gherghiceanu, M.; Serafinceanu, C.; Penescu, M.; Mircescu, G. Myofibroblast involvement in tubular basement membrane remodeling in type II diabetic nephropathy. Rom. J. Morphol. Embryol 2011, 52, 75–79. [Google Scholar]
- Ko, K.S.; Tomasi, M.L.; Iglesias-Ara, A.; French, B.A.; French, S.W.; Ramani, K.; Lozano, J.J.; Oh, P.; He, L.; Stiles, B.L.; et al. Liver-specific deletion of prohibitin 1 results in spontaneous liver injury, fibrosis, and hepatocellular carcinoma in mice. Hepatology 2010, 52, 2096–2108. [Google Scholar]
- Sato, S.; Murata, A.; Orihara, T.; Shirakawa, T.; Suenaga, K.; Kigoshi, H.; Uesugi, M. Marine natural product aurilide activates the OPA1-mediated apoptosis by binding to prohibitin. Chem. Biol 2011, 18, 131–139. [Google Scholar]
- Muraguchi, T.; Kawawa, A.; Kubota, S. Prohibitin protects against hypoxia-induced H9c2 cardiomyocyte cell death. Biomed. Res 2010, 31, 113–122. [Google Scholar]
- Wu, H.Z.; Guo, L.; Mak, Y.F.; Liu, N.; Poon, W.T.; Chan, Y.W.; Cai, Z. Proteomics investigation on aristolochic acid nephropathy: A case study on rat kidney tissues. Anal. Bioanal. Chem 2011, 399, 3431–3439. [Google Scholar]
- Quan, H.; Peng, X.; Liu, S.; Bo, F.; Yang, L.; Huang, Z.; Li, H.; Chen, X.; Di, W. Differentially expressed protein profile of renal tubule cell stimulated by elevated uric acid using SILAC coupled to LC-MS. Cell Physiol. Biochem 2011, 27, 91–98. [Google Scholar]
- Sievers, C.; Billig, G.; Gottschalk, K.; Rudel, T. Prohibitins are required for cancer cell proliferation and adhesion. PLoS One 2010, 5, 12735. [Google Scholar]
- Dressler, G.R. Patterning and early cell lineage decisions in the developing kidney: The role of PAX genes. Pediatr. Nephrol 2011, 26, 1387–1394. [Google Scholar]
- Negrisolo, S.; Benetti, E.; Centi, S.; Vella, M.D.; Ghirardo, G.; Zanon, G.; Murer, L.; Artifoni, L. PAX2 gene mutations in pediatric and young adult transplant recipients: Kidney and urinary tract malformations without ocular anomalies. Clin. Genet 2011, 80, 581–585. [Google Scholar]
- Raca, G.; Jackson, C.A.; Kucinskas, L.; Warman, B.; Shieh, J.T.; Schneider, A.; Bardakjian, T.M.; Schimmenti, L.A. Array comparative genomic hybridization analysis in patients with anophthalmia, microphthalmia, and coloboma. Genet. Med 2011, 13, 437–442. [Google Scholar]
- Li, L.; Wu, Y.; Zhang, W. PAX2 re-expression in renal tubular epithelial cells and correlation with renal interstitial fibrosis of rats with obstructive nephropathy. Ren. Fail 2010, 32, 603–611. [Google Scholar]
- Benetti, E.; Caridi, G.; Vella, M.D.; Rampoldi, L.; Ghiggeri, G.M.; Artifoni, L.; Murer, L. Immature renal structures associated with a novel UMOD sequence variant. Am. J. Kidney Dis 2009, 53, 327–331. [Google Scholar]
- Mure, P.Y.; Gelas, T.; Dijoud, F.; Guerret, S.; Benchaib, M.; Hartmann, D.J.; Mouriquand, P. Complete unilateral ureteral obstruction in the fetal lamb. Part II: Long-term outcomes of renal tissue development. J. Urol 2006, 175, 1548–1558. [Google Scholar]
- Huang, B.; Pi, L.; Chen, C.; Yuan, F.; Zhou, Q.; Teng, J.; Jiang, T. WT1 and PAX2 re-expression is required for epithelial-mesenchymal transition in 5/6 nephrectomized rats and cultured kidney tubular epithelial cells. Cells Tissue Organs 2012, 195, 296–312. [Google Scholar]
- Fujita, H.; Hida, M.; Kanemoto, K.; Fukuda, K.; Nagata, M.; Awazu, M. Cyclic stretch induces proliferation and TGF-beta1-mediated apoptosis via p38 and ERK in ureteric bud cells. Am. J. Physiol. Renal Physiol 2010, 299, 648–655. [Google Scholar]
- Liu, S.; Cieslinski, D.A.; Funke, A.J.; Humes, H.D. Transforming growth factor-beta 1 regulates the expression of PAX2, a developmental control gene, in renal tubule cells. Exp. Nephrol 1997, 5, 295–300. [Google Scholar]
- Huang, S.; Zhang, F.; Miao, L.; Zhang, H.; Fan, Z.; Wang, X.; Ji, G. Lentiviral-mediated Smad4 RNAi induced anti-proliferation by p16 up-regulation and apoptosis by caspase 3 down-regulation in hepatoma SMMC-7721 cells. Oncol. Rep 2008, 20, 1053–1059. [Google Scholar]
- Van der Heide, L.P.; Van Dinther, M.; Moustakas, A.; Ten, D.P. TGFbeta activates mitogen- and stress-activated protein kinase-1 (MSK1) to attenuate cell death. J. Biol. Chem 2011, 286, 5003–5011. [Google Scholar]
- Zheng, X.; Boerboom, D.; Carriere, P.D. Transforming growth factor-beta1 inhibits luteinization and promotes apoptosis in bovine granulosa cells. Reproduction 2009, 137, 969–977. [Google Scholar]
- Xie, P.; Sun, L.; Nayak, B.; Haruna, Y.; Liu, F.Y.; Kashihara, N.; Kanwar, Y.S. C/EBP-beta modulates transcription of tubulointerstitial nephritis antigen in obstructive uropathy. J. Am. Soc. Nephrol 2009, 20, 807–819. [Google Scholar]
- Zhang, D.; Sun, L.; Xian, W.; Liu, F.; Ling, G.; Xiao, L.; Liu, Y.; Peng, Y.; Haruna, Y.; Kanwar, Y.S. Low-dose paclitaxel ameliorates renal fibrosis in rat UUO model by inhibition of TGF-beta/Smad activity. Lab. Invest 2010, 90, 436–447. [Google Scholar]
- Zhou, T.B.; Qin, Y.H.; Zhou, C.; Lei, F.Y.; Zhao, Y.J.; Chen, J.; Su, L.N.; Huang, W.F. Less expression of prohibitin is associated with increased Caspase-3 expression and cell apoptosis in renal interstitial fibrosis rats. Nephrology (Carlton) 2012, 17, 189–196. [Google Scholar]
- Hu, X.; Zhou, X.; He, B.; Xu, C.; Wu, L.; Cui, B.; Wen, H.; Lu, Z.; Jiang, H. Minocycline protects against myocardial ischemia and reperfusion injury by inhibiting high mobility group box 1 protein in rats. Eur. J. Pharmacol 2010, 638, 84–89. [Google Scholar]
- Ha, T.; Hua, F.; Liu, X.; Ma, J.; McMullen, J.R.; Shioi, T.; Izumo, S.; Kelley, J.; Gao, X.; Browder, W.; et al. Lipopolysaccharide-induced myocardial protection against ischaemia/reperfusion injury is mediated through a PI3K/Akt-dependent mechanism. Cardiovasc. Res 2008, 78, 546–553. [Google Scholar]
- Li, W.; Wang, J.; Jiang, H.R.; Xu, X.L.; Zhang, J.; Liu, M.L.; Zhai, L.Y. Combined effects of cyclooxygenase-1 and cyclooxygenase-2 selective inhibitors on ovarian carcinoma in vivo. Int. J. Mol. Sci 2011, 12, 668–681. [Google Scholar]
- Lai, J.Y.; Li, Y.T.; Wang, T.P. In vitro response of retinal pigment epithelial cells exposed to chitosan materials prepared with different cross-linkers. Int. J. Mol. Sci 2010, 11, 5256–5272. [Google Scholar]
- Zhou, T.B.; Qin, Y.H.; Li, Z.Y.; Xu, H.L.; Zhao, Y.J.; Lei, F.Y. All-trans retinoic Acid treatment is associated with prohibitin expression in renal interstitial fibrosis rats. Int. J. Mol. Sci 2012, 13, 2769–2782. [Google Scholar]
- Capozzi, V.; Arena, M.P.; Crisetti, E.; Spano, G.; Fiocco, D. The hsp 16 gene of the probiotic lactobacillus acidophilus is differently regulated by salt, high temperature and acidic stresses, as revealed by reverse transcription quantitative PCR (qRT-PCR) analysis. Int. J. Mol. Sci 2011, 12, 5390–5405. [Google Scholar]
- Liu, X.D.; Fan, R.F.; Zhang, Y.; Yang, H.Z.; Fang, Z.G.; Guan, W.B.; Lin, D.J.; Xiao, R.Z.; Huang, R.W.; Huang, H.Q.; et al. Down-regulation of telomerase activity and activation of caspase-3 are responsible for tanshinone I-induced apoptosis in monocyte leukemia cells in vitro. Int. J. Mol. Sci 2010, 11, 2267–2280. [Google Scholar]
- Zhou, T.B.; Qin, Y.H.; Ou, C.; Lei, F.Y.; Su, L.N.; Huang, W.F.; Zhao, Y.J. All-trans retinoic acid can regulate the expressions of gelatinases and apolipoprotein E in glomerulosclerosis rats. Vascul. Pharmacol 2011, 55, 169–177. [Google Scholar]
- Zhou, T.B.; Qin, Y.H.; Lei, F.Y.; Su, L.N.; Zhao, Y.J.; Huang, W.F. All-trans retinoic acid regulates the expression of apolipoprotein E in rats with glomerulosclerosis induced by Adriamycin. Exp. Mol. Pathol 2011, 90, 287–294. [Google Scholar]
- Zhou, T.B.; Qin, Y.H.; Lei, F.Y.; Su, L.N.; Zhao, Y.J.; Huang, W.F. Less gelatinases is associated with apolipoprotein E accumulation in glomerulosclerosis rats. Histol. Histopathol 2012, 27, 249–256. [Google Scholar]
- Zhou, T.B.; Qin, Y.H.; Lei, F.Y.; Su, L.N.; Zhao, Y.J.; Huang, W.F. apoE expression in glomerulus and correlation with glomerulosclerosis induced by adriamycin in rats. Ren. Fail 2011, 33, 348–354. [Google Scholar]



© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhou, T.-B.; Zeng, Z.-Y.; Qin, Y.-H.; Zhao, Y.-J. Less Expression of Prohibitin Is Associated with Increased Paired Box 2 (PAX2) in Renal Interstitial Fibrosis Rats. Int. J. Mol. Sci. 2012, 13, 9808-9825. https://doi.org/10.3390/ijms13089808
Zhou T-B, Zeng Z-Y, Qin Y-H, Zhao Y-J. Less Expression of Prohibitin Is Associated with Increased Paired Box 2 (PAX2) in Renal Interstitial Fibrosis Rats. International Journal of Molecular Sciences. 2012; 13(8):9808-9825. https://doi.org/10.3390/ijms13089808
Chicago/Turabian StyleZhou, Tian-Biao, Zhi-Yu Zeng, Yuan-Han Qin, and Yan-Jun Zhao. 2012. "Less Expression of Prohibitin Is Associated with Increased Paired Box 2 (PAX2) in Renal Interstitial Fibrosis Rats" International Journal of Molecular Sciences 13, no. 8: 9808-9825. https://doi.org/10.3390/ijms13089808



