Detection of Differential Levels of Proteins in the Urine of Patients with Endometrial Cancer: Analysis Using Two-Dimensional Gel Electrophoresis and O-Glycan Binding Lectin
Abstract
:1. Introduction
2. Results
2.1. Profiling of Urinary Proteins by 2-DE
2.2. Image Analysis of 2-DE Separated Urinary Proteins
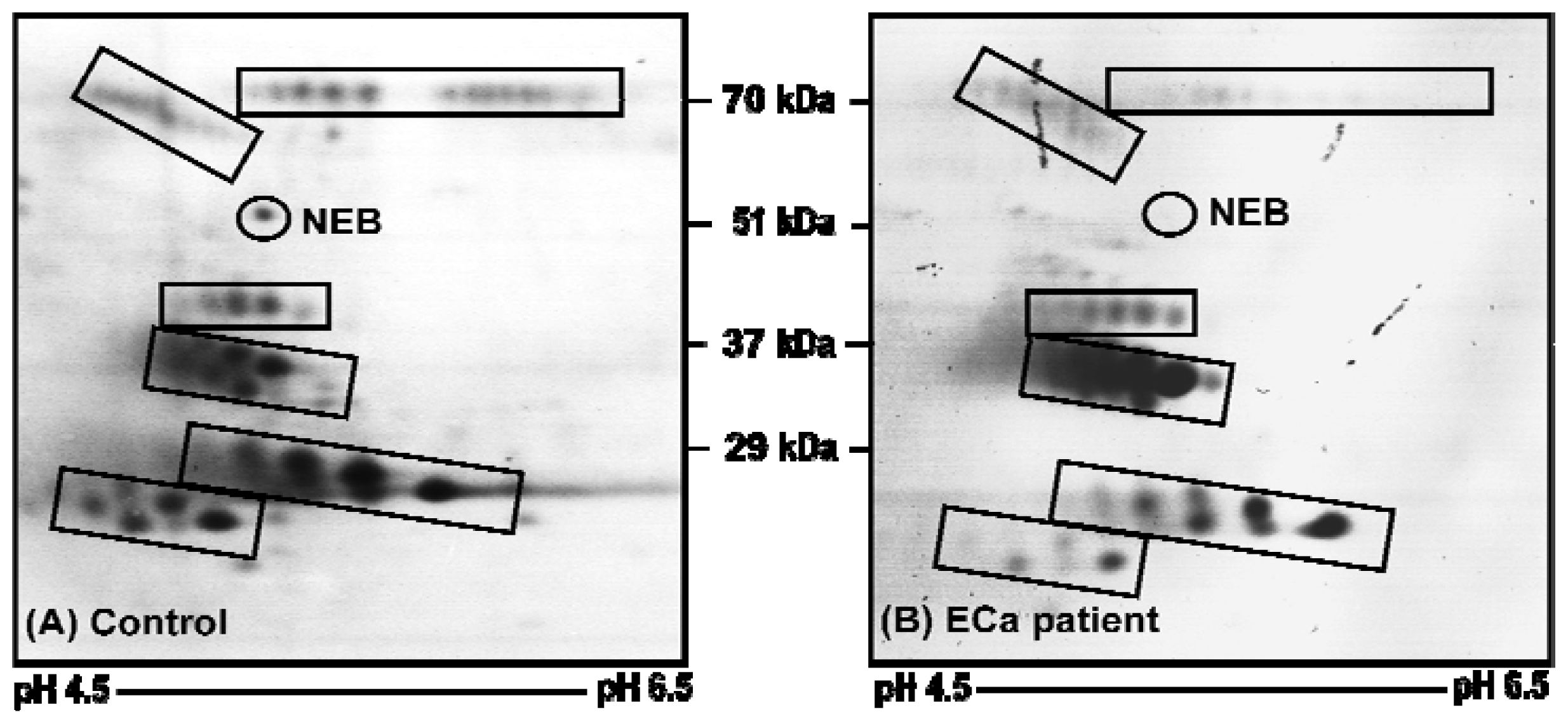
2.3. Profiling of Urinary O-Glycoproteins
2.4. Immobilized CGB Lectin Affinity Chromatography and LC-MS/MS
3. Discussion
4. Experimental Section
4.1. Urine Samples and Processing
4.2. Two-Dimensional Electrophoresis and Silver Staining
4.3. Image Analysis
4.4. On-Membrane Digestion
4.5. MALDI-ToF Mass Spectrometry
4.6. Western Blotting and Detection by CGB Lectin
4.7. CGB Lectin Affinity Separation and LC-MS/MS
5. Conclusions
Acknowledgments
- Conflict of InterestThe authors declare no conflict of interest.
References
- Attarha, S.; Mints, M.; Andersson, S.; Souchelnytskyi, S. Endometrial cancer and application of proteomics. Exp. Oncol 2011, 33, 174–177. [Google Scholar]
- Ueda, Y.; Enomoto, T.; Kimura, T.; Miyatake, T.; Yoshino, K.; Fujita, M.; Kimura, T. Serum biomarkers for early detection of gynecologic cancers. Cancers 2010, 2, 1312–1327. [Google Scholar]
- Barratt, J.; Topham, P. Urine proteomics: The present and future of measuring urinary protein components in disease. CMAJ 2007, 177, 361–368. [Google Scholar]
- Kreunin, P.; Zhao, J.; Rosser, C.; Urquidi, V.; Lubman, D.M.; Goodison, S. Bladder cancer associated glycoprotein signatures revealed by urinary proteomic profiling. J. Proteome Res 2007, 6, 2631–2639. [Google Scholar]
- Abdullah-Soheimi, S.S.; Lim, B.K.; Hashim, O.H.; Shuib, A.S. Patients with ovarian carcinoma excrete different altered levels of urine CD59, kininogen-1 and fragments of inter-alpha-trypsin inhibitor heavy chain H4 and albumin. Proteome Sci 2010, 8, 58. [Google Scholar]
- Brockhausen, I. Mucin-type O-glycans in human colon and breast cancer: Glycodynamics and functions. EMBO Rep 2006, 7, 599–604. [Google Scholar]
- Abdul-Rahman, P.S.; Lim, B.K.; Hashim, O.H. Expression of high abundance proteins in sera of patients with endometrial and cervical cancers: Analysis using two-dimensional gel electrophoresis with silver staining and lectin detection methods. Electrophoresis 2007, 28, 1989–1996. [Google Scholar]
- Mohamed, E.; Abdul-Rahman, P.S.; Doustjalali, S.R.; Chen, Y.; Lim, B.K.; Omar, S.Z.; Bustam, A.Z.; Singh, V.A.; Muhammad-Taib, N.A.; Yip, C.H.; et al. Lectin-based electrophoretic analysis of the expression of the 35 kDa inter-alpha-trypsin inhibitor heavy chain H4 fragment in sera of patients with five different malignancies. Electrophoresis 2008, 29, 2645–2650. [Google Scholar]
- Mukhopadhyay, P.; Chakraborty, S.; Ponnusamy, M.P.; Lakshmanan, I.; Jain, M.; Batra, S.K. Mucins in the pathogenesis of breast cancer: Implications in diagnosis, prognosis and therapy. Biochim. Biophys. Acta 2011, 1815, 224–240. [Google Scholar]
- Hashim, O.H.; Gendeh, G.S.; Nik Jaafar, M.I. Comparative analyses of IgA1 binding lectins from seeds of six distinct clones of Artocarpus integer. Biochem. Mol. Biol. Int 1993, 29, 69–76. [Google Scholar]
- Rahman, M.A.; Karsani, S.A.; Othman, I.; Abdul Rahman, P.S.; Hashim, O.H. Galactose binding lectin from the seeds of champedak (Artocarpus integer): Sequences of its subunits and interactions with human serum O-glycosylated glycoproteins. Biochem. Biophys. Res. Commun 2002, 295, 1007–1013. [Google Scholar]
- UniProtKB. Available online: http://www.uniprot.org accessed on 22 February 2012.
- Matrix Science; Matrix Science Inc.: Boston, MA, USA, 2012.
- Julenius, K.; Mølgaard, A.R.; Gupta, R.; Brunak, S. Prediction, conservation analysis and structural characterization of mammalian mucin-type O-glycosylation sites. Glycobiology 2005, 15, 153–164. [Google Scholar]
- Hassan, M.I.; Waheed, A.; Yadav, S.; Singh, T.P.; Ahmad, F. Zinc alpha 2-glycoprotein: A multidisciplinary protein. Mol. Cancer Res 2008, 6, 892–906. [Google Scholar]
- Hale, L.P.; Price, D.T.; Sanchez, L.M.; Demark-Wahnefried, W.; Madden, J.F. Zinc alpha-2-glycoprotein is expressed by malignant prostatic epithelium and may serve as a potential serum marker for prostate cancer. Clin. Cancer Res 2001, 7, 846–853. [Google Scholar]
- Hu, S.; Matha, A.; Boontheung, P.; Wang, J.; Zhou, H.; Jiang, J.; Elashoff, D.; Wei, R.; Loo, J.A.; Wong, D.T. Salivary proteomics for oral cancer biomarker discovery. Clin. Cancer Res 2008, 4, 6246–6252. [Google Scholar]
- Tantipaiboonwong, P.; Sinchaikul, S.; Sriyam, S.; Phutrukul, S.; Chen, S. Different techniques for urinary protein analysis of normal and lung cancer patients. Proteomics 2005, 5, 1140–1149. [Google Scholar]
- Murray, K.P.; Mathure, S.; Kaul, R.; Khan, S.; Carson, L.F.; Twiggs, L.B.; Martens, M.G.; Kaul, A. Expression of complement regulatory proteins—CD 35, CD 46, CD 55, and CD 59— in benign and malignant endometrial tissue. Gynecol. Oncol 2000, 76, 176–182. [Google Scholar]
- Dubois, V.; Delort, L.; Mishellany, F.; Jarde, T.; Billard, H.; Lequeux, C.; Damour, O.; Penault-Llorca, F.; Vasson, M.P.; Caldefie-Chezet, F. Zinc-alpha2-glycoprotein: A new biomarker of breast cancer? Anticancer Res 2010, 30, 2919–2925. [Google Scholar]
- Huang, Y.; Li, L.Z.; Zhang, C.Z.Y.; Yi, C.; Liu, L.L.; Zhou, X.; Xie, G.B.; Cai, M.Y.; Li, Y.; Yun, J.P. Decreased expression of zinc-alpha2-glycoprotein in hepatocellular carcinoma associates with poor prognosis. J. Transl. Med 2012, 10, 106. [Google Scholar]
- Alexander, H.; Stegner, A.L.; Wagner-Mann, C.; Du Bois, G.C.; Alexander, S.; Sauter, E.R. Proteomic analysis to identify breast cancer biomarkers in nipple aspirate fluid. Clin. Cancer Res 2004, 10, 7500–7510. [Google Scholar]
- Zhang, X.; Xiao, Z.; Liu, X.; Du, L.; Wang, L.; Wang, S.; Zheng, N.; Zheng, G.; Li, W.; Zhang, X.; et al. The potential role of ORM2 in the development of colorectal cancer. PLoS One 2012, 7, e31868. [Google Scholar]
- Bachtiar, I.; Santoso, J.M.; Atmanegara, B.; Gani, R.A.; Hasan, I.; Lesmana, L.A.; Sulaiman, A.; Gu, J.; Tai, S. Combination of alpha-1-acid glycoprotein and alpha-fetoprotein as an improved diagnostic tool for hepatocellular carcinoma. Clin. Chim. Acta 2009, 399, 97–101. [Google Scholar]
- Boylan, K.L.M.; Andersen, J.D.; Andersen, L.B.; Higgins, L.A.; Skubitz, A.P.N. Quantitative proteomic analysis by iTRAQ® for the identification of candidate biomarkers in ovarian cancer serum. Proteome Sci 2010, 8, 31. [Google Scholar]
- Wang, Y.S.; Cao, R.; Jin, H.; Huang, Y.P.; Zhang, X.Y.; Cong, Q.; He, Y.F.; Xu, C.J. Altered protein expression in serum from endometrial hyperplasia and carcinoma patients. J. Hematol. Oncol 2011, 4, 15. [Google Scholar]
- Park, J.Y.; Kim, S.A.; Chung, J.W.; Bang, S.; Park, S.W.; Paik, Y.K.; Song, S.Y. Proteomic analysis of pancreatic juice for the identification of biomarkers of pancreatic cancer. J. Cancer Res. Clin. Oncol 2011, 137, 1229–1238. [Google Scholar]
- Li, W.; Li, J.F.; Qu, Y.; Chen, X.H.; Qin, J.M.; Gu, Q.L.; Yan, M.; Zhu, Z.G.; Liu, B.Y. Comparative proteomics analysis of human gastric cancer. World J. Gastroenterol 2008, 14, 5657–5664. [Google Scholar]
- Tosner, J.; Krejsek, J.; Louda, B. Serum prealbumin, transferrin and alpha-1-acid glycoprotein in patients with gynecological carcinomas. Neoplasma 1988, 35, 403–411. [Google Scholar]
- Weeks, M.E; Hariharan, D.; Petronijevic, L.; Radon, T.P.; Whiteman, H.J.; Kocher, H.M.; Timms, J.F.; Lemoine, N.R.; Crnogorac-Jurcevic, T. Analysis of the urine proteome in patients with pancreatic ductal adenocarcinoma. Proteomics Clin. Appl. 2008, 2, 1047–1057. [Google Scholar]
- Pappas, C.T.; Bliss, K.T.; Zieseniss, A.; Gregorio, C.C. The nebulin family: An actin support group. Trends Cell Biol 2011, 21, 29–37. [Google Scholar]
- Madera, M.; Mechref, Y.; Klouckova, I.; Novotny, M.V. Semiautomated high-sensitivity profiling of human blood serum glycoproteins through lectin preconcentration and multidimensional chromatography/tandem mass spectrometry. J. Proteome Res 2006, 5, 2348–2363. [Google Scholar]
- Ottenheijm, C.A.C.; Granzier, H. Lifting the nebula: Novel insights into skeletal muscle contractility. Physiology 2010, 25, 304–310. [Google Scholar]
- Tanaka, Y.; Akiyama, H.; Kuroda, T.; Jung, G.; Tanahashi, K.; Sugaya, H.; Utsumi, J.; Kawasaki, H.; Hirano, H. A novel approach and protocol for discovering extremely low-abundance proteins in serum. Proteomics 2006, 6, 4845–4855. [Google Scholar]
- Shuib, A.S.; Chua, C.T.; Hashim, O.H. Sera of IgA nephropathy patients contain a heterogeneous population of relatively cationic alpha heavy chains. Nephron 1998, 78, 290–295. [Google Scholar]
- Hashim, O.H.; Shuib, A.S.; Chua, C.T. Neuraminidase treatment abrogates the binding abnormality of IgA1 from IgA nephropathy patients and the differential charge distribution of its alpha-heavy chains. Nephron 2001, 89, 422–425. [Google Scholar]
- Heukeshoven, J.; Dernick, R. Improved silver staining procedure for fast staining in PhastSystem Development Unit. I. Staining of sodium dodecyl sulfate gels. Electrophoresis 1988, 9, 28–32. [Google Scholar]
- Shevchenko, A.; Wilm, M.; Vorm, O.; Mann, M. Mass spectrometric sequencing of proteins from silver-stained polyacrylamide gels. Anal. Chem 1996, 68, 850–858. [Google Scholar]
- Benjamini, Y.; Hochberg, Y. Controlling the false discovery rate: A practical and powerful approach to multiple testing. J. R. Stat. Soc. B 1995, 57, 289–300. [Google Scholar]
- Luque-Garcia, J.L.; Neubert, T.A. On-membrane tryptic digestion of proteins for mass spectrometry analysis. Methods Mol. Biol 2009, 536, 331–341. [Google Scholar]
- Seriramalu, R.; Pang, W.W.; Jayapalan, J.J.; Mohamed, E.; Abdul-Rahman, P.S.; Bustam, A.Z.; Khoo, A.S.B.; Hashim, O.H. Application of champedak mannose-binding lectin in the glycoproteomic profiling of serum samples unmasks reduced expression of alpha-2 macroglobulin and complement factor B in patients with nasopharyngeal carcinoma. Electrophoresis 2010, 31, 2388–2395. [Google Scholar]
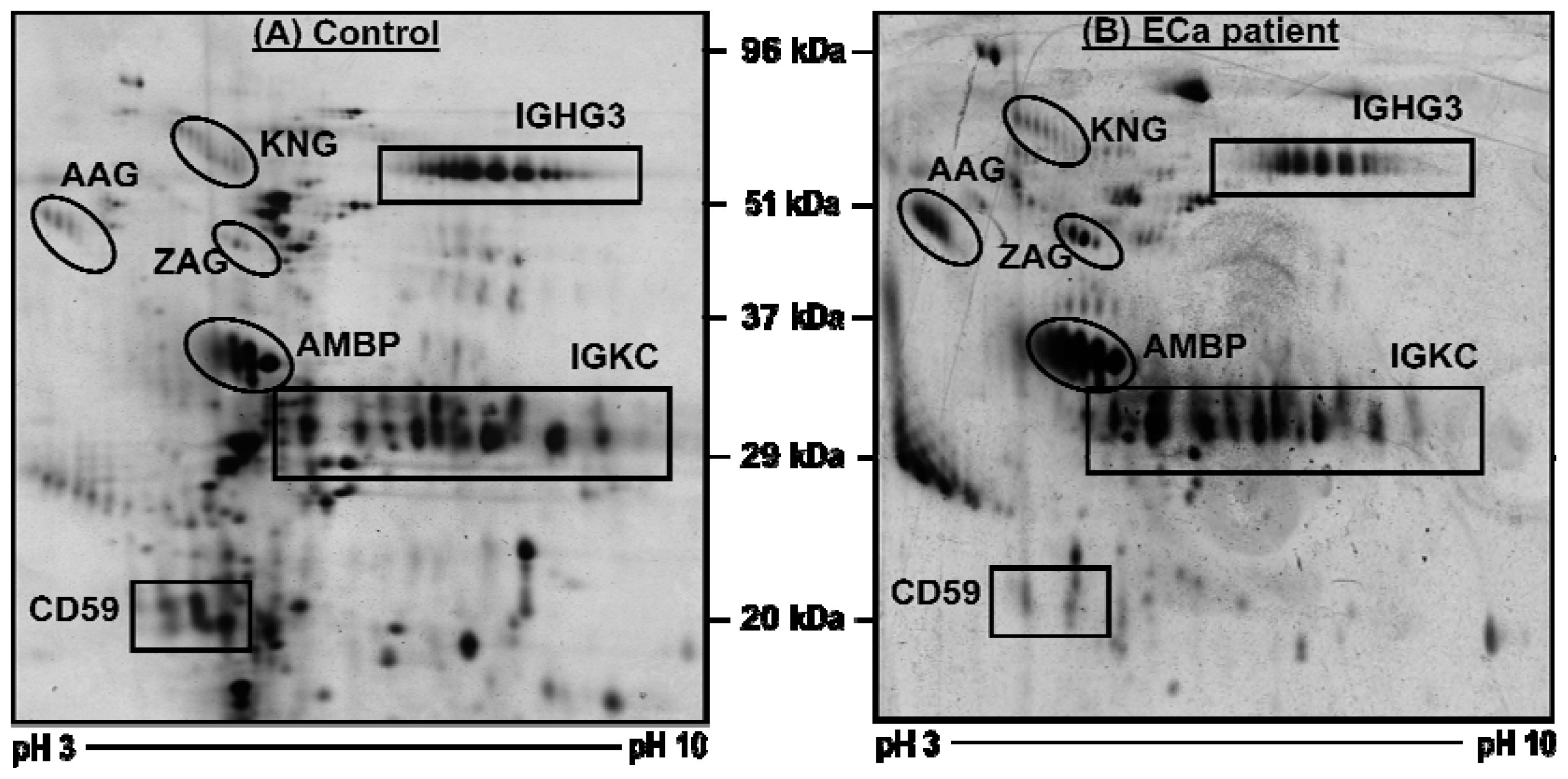
| Protein Entry Name + | Protein Name | Accession Number # | Nominal Mass (kDa)/pI | MOWSE Protein Score | Sequence Coverage (%) |
|---|---|---|---|---|---|
| KNG | Kininogen | P01042 | 71/6.34 | 68 | 7 |
| IGHG3 | Ig g3 chain C region | P01860 | 41/8.46 | 16 | 3 |
| AAG | α1-acid glycoprotein | P19652 | 23/5.03 | 241 | 16 |
| ZAG | Zinc α2 glycoprotein | P25311 | 33/5.57 | 134 | 20 |
| AMBP | Protein AMBP | P02760 | 39/5.95 | 50 | 12 |
| IGKC | Ig l1 chain C region | P01834 | 11/5.58 | 141 | 32 |
| CD59 | CD59 glycoprotein | P13987 | 14/6.02 | 121 | 18 |
| Urinary Proteins | Mean % Vol ± SEM | p | Fold Changes * | |
|---|---|---|---|---|
| Control (n = 11) | ECa (n = 7) | |||
| AAG | 0.161 ± 0.072 | 2.746 ± 0.717 | 0.001 | +17.1 |
| ZAG | 0.175 ± 0.045 | 2.184 ± 0.592 | 0.001 | +12.5 |
| CD59 | 2.575 ± 0.497 | 0.177 ± 0.070 | 0.002 | −14.6 |
| KNG | 5.137 ± 1.826 | 0.945 ± 0.491 | 0.108 | ns |
| IGKC | 14.785 ± 2.197 | 18.840 ± 2.651 | 0.286 | ns |
| AMBP | 8.941 ± 1.706 | 12.837 ± 3.430 | 0.306 | ns |
| IGHG3 | 4.014 ± 1.221 | 4.554 ± 1.706 | 0.807 | ns |
| (A) | Mean % Vol ± SEM | p | Fold Changes | |
|---|---|---|---|---|
| Control (n = 11) | ECa (n = 7) | |||
| 1.08 ± 0.172 | 0.102 ± 0.095 | 0.001 | −10.6 | |
| (B) | Accession Number # | Nominal Mass (kDa)/pI | MOWSE Protein Score | Sequence Coverage (%) |
| P20929 | 775/9.11 | 64 | 1 * | |
| Protein Name | Accession Number | Subcellular Location | Glycan # |
|---|---|---|---|
| Protein AMBP | P02760 | Secreted | O-linked |
| ATP synthase subunit beta, mitochondrial | P24539 | Membrane | Potential O-linked |
| Serotransferrin | P02787 | Secreted | O-linked |
| Transmembrane protein 110 | Q86TLZ | Membrane | Potential O-linked |
| Phosphoinositide-3 kinase interacting protein 1 | Q96FE7 | Membrane | O-linked |
| Ribonuclease pancreatic | P07998 | Secreted | Potential O-linked |
| Prostaglandin-H2-D isomerase | P41222 | Secreted | Potential O-linked |
| Membrane bound transcription factor site-1 protease | Q14703 | Membrane | Potential O-linked |
| Homeobox protein engrailed 2 | P19622 | Nucleus | Potential O-linked |
| Actin, cytoplasmic 1 | P60709 | Cytoplasmic | Potential O-linked |
| Neurofilament medium polypeptide | P07197 | Cytoplasmic | O-linked |
| CD55 decay-accelerating factor splicing variant 4 | Q14UF3 | Membrane | Potential O-linked |
| Leucine rich alpha 2 glycoprotein | P02750 | Secreted | O-linked |
| Spondin-2 | Q9BUD6 | Secreted | Potential O-linked |
| Protein RRP5 homolog | Q14690 | Nucleus | Potential O-linked |
| CD44 antigen | P16070 | Membrane | O-linked |
| Phosphatidylinositol-3,4,5-triphosphate 5-phosphatase-1 | Q92835 | Membrane | Potential O-linked |
| Solute carrier family 12 member 6 | Q9UHW9 | Membrane | Potential O-linked |
| Mucin-5B | Q9HC84 | Secreted | O-linked |
| Major facilitator superfamily domain containing protein 10 | Q14728 | Membrane | Potential O-linked |
| PDZ and LIM domain protein 5 | Q96HC4 | Membrane | Potential O-linked |
| Leukocyte associated immunoglobulin-like receptor 1 | Q6GTX8 | Membrane | Potential O-linked |
| Tumor necrosis receptor superfamily member 16 | P08138 | Membrane | Potential O-linked |
| Lipocalin-1 | P31025 | Secreted | Potential O-linked |
| Amyloid beta A4 | P05067 | Membrane | O-linked |
| Nebulin | P20929 | Cytoplasmic | Potential O-linked |
| Transcription factor HES-2 | Q9Y543 | Nucleus | Potential O-linked |
| Rho GTPase-activating protein 12 | P20936 | Cytoplasmic | Potential O-linked |
| Solute carrier family 13 member 3 | Q8WWT9 | Membrane | Potential O-linked |
| Sodium/bile acid contransporter | Q14973 | Membrane | Potential O-linked |
| Bromodomain-containing protein 3 | Q15059 | Nucleus | Potential O-linked |
| Inter alpha trypsin inhibitor heavy chain H4 | Q14624 | Secreted | O-linked |
| Ig gamma-1 chain C region | P01857 | Secreted | Potential O-linked |
| Ig gamma-2 chain C region | P01859 | Secreted | Potential O-linked |
| Ig alpha-1 chain C region | P01876 | Unknown | O-linked |
| Ig lambda-1 chain C region | P0CG04 | Unknown | Potential O-linked |
| Protein | Type of Cancer | Sample Type | Reference |
|---|---|---|---|
| CD59 | Oral | Saliva | [17] |
| Lung | Urine | [18] | |
| Ovarian | Urine | [5] | |
| Endometrial | Tissue | [19] | |
| ZAG | Breast | Tissue | [20] |
| Hepatocellular | Tissue | [21] | |
| Prostate | Tissue and serum | [16] | |
| Cervical | Serum | [7] | |
| AAG | Breast | Nipple aspirate fluid | [22] |
| Colon | Plasma | [23] | |
| Hepatocellular | Serum | [24] | |
| Ovarian | Serum | [25] | |
| Endometrial | Serum | [26] | |
| NEB | Pancreatic | Pancreatic juice | [27] |
| Gastric | Tissue | [28] | |
| ECa Patient | Stage | Estrogen or Tamoxifen Treatment | Obesity | Late Menopause | Previous Pregnancy | Early Puberty | Blood Creatinine Level |
|---|---|---|---|---|---|---|---|
| 1 | IB | No | No | Yes | Yes | No | Normal |
| 2 | IB | No | No | Yes | Yes | No | Normal |
| 3 | IB | No | No | No | Yes | No | Normal |
| 4 | IB | No | No | No | Yes | No | Normal |
| 5 | IB | No | No | No | Yes | No | Normal |
| 6 | IIA | No | Yes | No | Yes | No | Normal |
| 7 | IIB | No | No | No | No | No | Normal |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mu, A.K.-W.; Lim, B.-K.; Hashim, O.H.; Shuib, A.S. Detection of Differential Levels of Proteins in the Urine of Patients with Endometrial Cancer: Analysis Using Two-Dimensional Gel Electrophoresis and O-Glycan Binding Lectin. Int. J. Mol. Sci. 2012, 13, 9489-9501. https://doi.org/10.3390/ijms13089489
Mu AK-W, Lim B-K, Hashim OH, Shuib AS. Detection of Differential Levels of Proteins in the Urine of Patients with Endometrial Cancer: Analysis Using Two-Dimensional Gel Electrophoresis and O-Glycan Binding Lectin. International Journal of Molecular Sciences. 2012; 13(8):9489-9501. https://doi.org/10.3390/ijms13089489
Chicago/Turabian StyleMu, Alan Kang-Wai, Boon-Kiong Lim, Onn Haji Hashim, and Adawiyah Suriza Shuib. 2012. "Detection of Differential Levels of Proteins in the Urine of Patients with Endometrial Cancer: Analysis Using Two-Dimensional Gel Electrophoresis and O-Glycan Binding Lectin" International Journal of Molecular Sciences 13, no. 8: 9489-9501. https://doi.org/10.3390/ijms13089489




