Pre-Ischemic Treadmill Training for Prevention of Ischemic Brain Injury via Regulation of Glutamate and Its Transporter GLT-1
Abstract
:1. Introduction
2. Results and Discussion
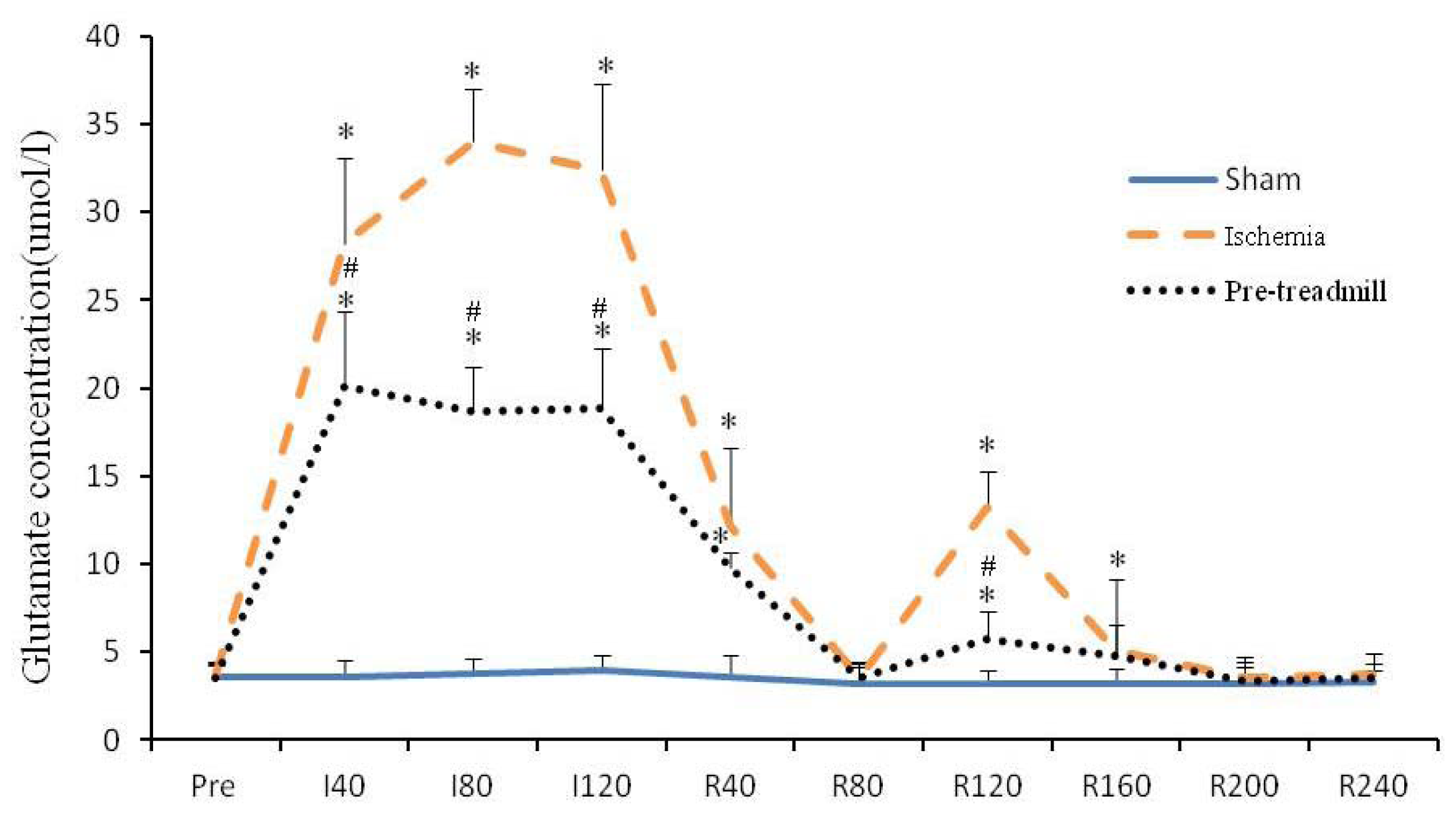
2.1. Extracellular Glutamate Concentration
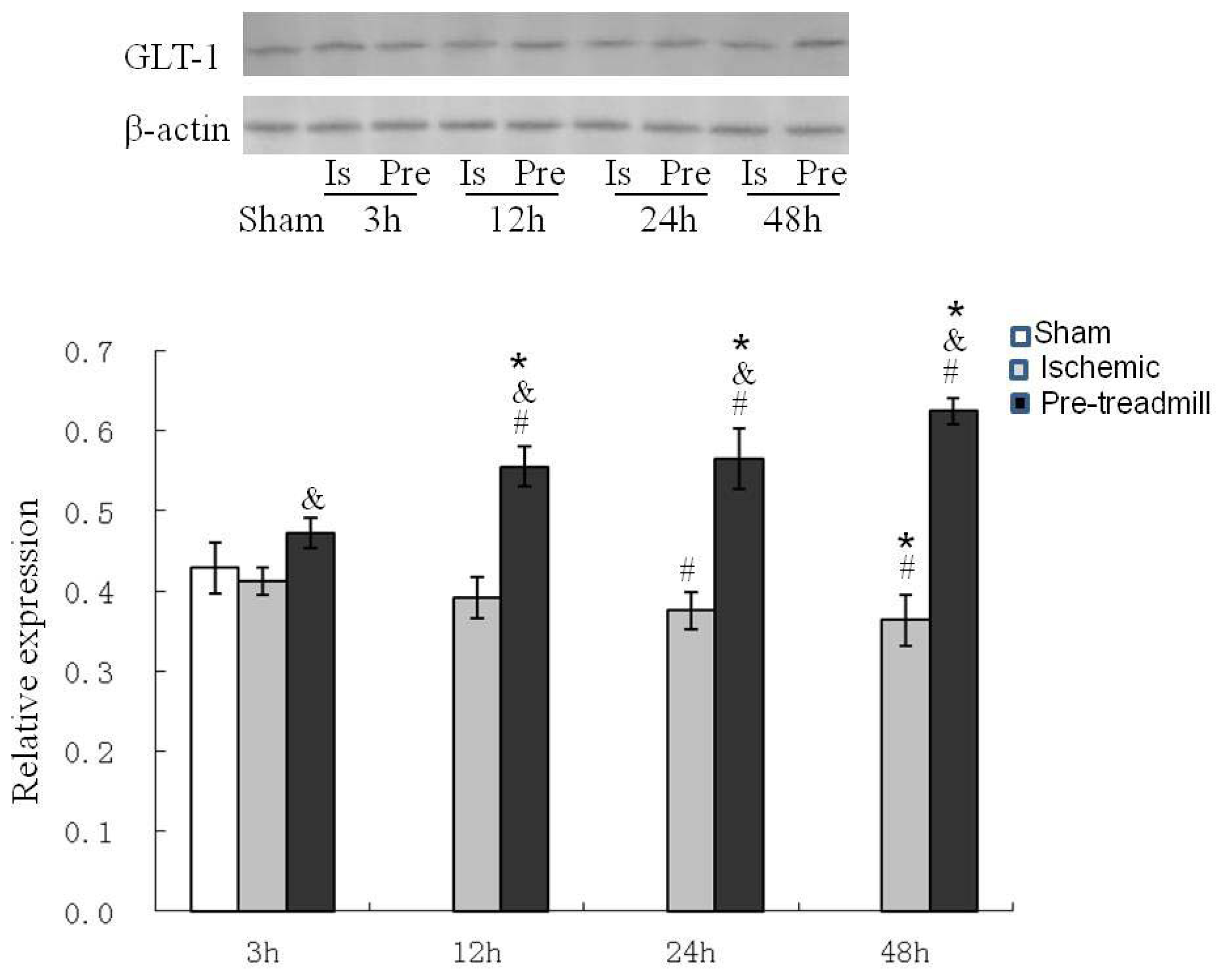
2.2. GLT-1 Protein Expression
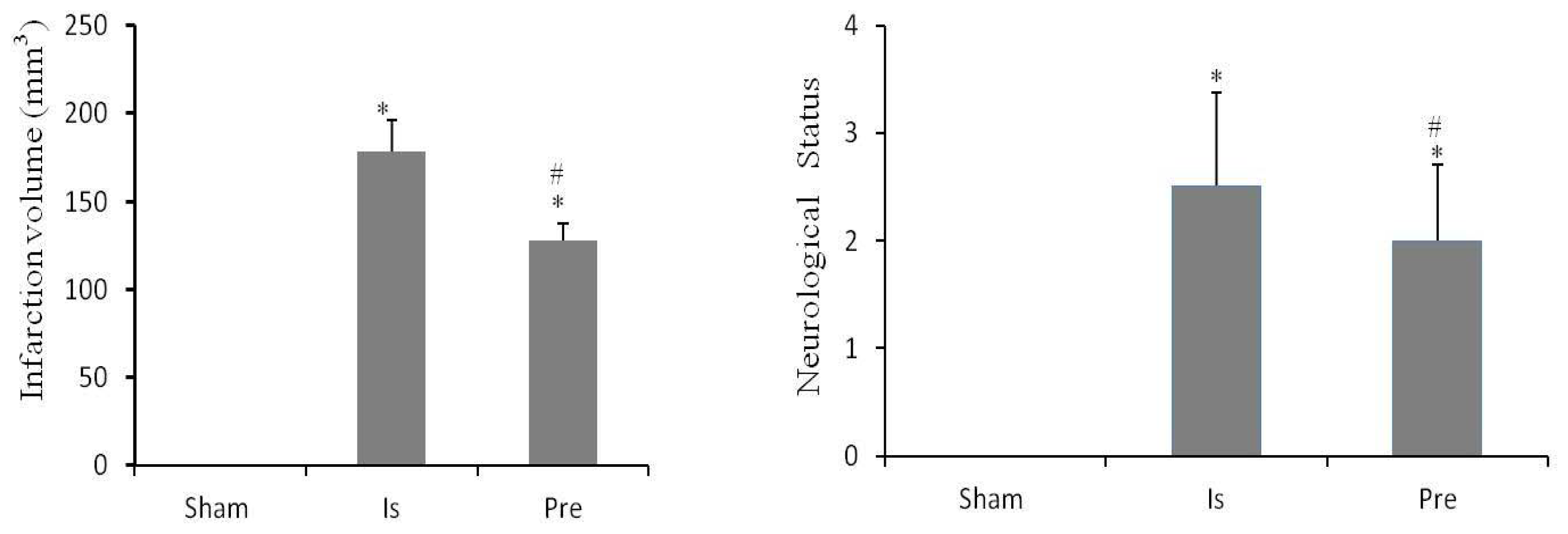
2.3. Neurobehavioral Score
2.4. Cerebral Infarct Volume
2.5. Discussion
3. Experimental Section
3.1. Animals and Groups
3.2. Establishment of Rat Model of Middle Cerebral Artery Occlusion (MCAO)
3.3. Microdialysis Sampling
3.4. Determination of Glutamate Concentration
3.5. Western Blot Analysis
3.6. Neurobehavioral Score
3.7. Measurement of Cerebral Infarct Volume
3.8. Statistical Analysis
4. Conclusions
Acknowledgments
References
- Goldstein, L.B.; Bushnell, C.D.; Adams, R.J.; Appel, L.J.; Braun, L.T.; Chaturvedi, S.; Creager, M.A.; Culebras, A.; Eckel, R.H.; Hart, R.G.; et al. Guidelines for the primary prevention of stroke: A guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2011, 42, 517–584. [Google Scholar]
- Cotman, C.W.; Berchtold, N.C.; Christie, L.A. Exercise builds brain health: Key roles of growth factor cascades and inflammation. Trends Neurosci 2007, 30, 464–472. [Google Scholar]
- Yang, Y.R.; Wang, R.Y.; Wang, P.S.; Yu, S.M. Treadmill training effects on neurological outcome after middle cerebral artery occlusion in rats. Can. J. Neurol. Sci 2003, 30, 252–258. [Google Scholar]
- Ding, Y.H.; Ding, Y.; Li, J.; Bessert, D.A.; Rafols, J.A. Exercise pre-conditioning strengthens brain microvascular integrity in a rat stroke model. Neurol. Res 2006, 28, 184–189. [Google Scholar]
- Guo, M.; Cox, B.; Mahale, S.; Davis, W.; Carranza, A.; Hayes, K.; Sprague, S.; Jimenez, D.; Ding, Y. Pre-ischemic exercise reduces matrix metalloproteinase-9 expression and ameliorates blood-brain barrier dysfunction in stroke. Neuroscience 2008, 151, 340–351. [Google Scholar]
- Curry, A.; Guo, M.; Patel, R.; Liebelt, B.; Sprague, S.; Lai, Q.; Zwagerman, N.; Cao, F.X.; Jimenez, D.; Ding, Y. Exercise pre-conditioning reduces brain inflammation in stroke via tumor necrosis factor-alpha, extracellular signal-regulated kinase 1/2 and matrix metalloproteinase-9 activity. Neurol. Res 2010, 32, 756–762. [Google Scholar]
- Guyot, L.L.; Diaz, F.G.; O’Regan, M.H.; McLeod, S.; Park, H.; Phillis, J.W. Real-time measurement of glutamate release from the ischemic penumbra of the rat cerebral cortex using a focal middle cerebral artery occlusion model. Neurosci. Lett 2001, 299, 37–40. [Google Scholar]
- Hinzman, J.M.; Thomas, T.C.; Quintero, J.E.; Gerhardt, G.A.; Lifshitz, J. Disruptions in the regulation of extracellular glutamate by neurons and glia in the rat striatum two days after diffuse brain injury. J. Neurotrauma 2012, 29, 1197–1208. [Google Scholar]
- Beart, P.M.; O’Shea, R.D. Transporters for l-glutamate: An update on their molecular pharmacology and pathological involvement. Br. J. Pharmacol 2007, 150, 5–17. [Google Scholar]
- Sims, K.D.; Robinson, M.B. Expression patterns and regulation of glutamate transporters in the developing and adult nervous system. Crit. Rev. Neurobiol 1999, 13, 169–197. [Google Scholar]
- Suchak, S.K.; Baloyianni, N.V.; Perkinton, M.S.; Williams, R.J.; Meldrum, B.S.; Rattray, M. The ‘glial’ glutamate transporter, EAAT2 (Glt-1) accounts for high affinity glutamate uptake into adult rodent nerve endings. J. Neurochem 2003, 84, 522–532. [Google Scholar]
- Jia, J.; Hu, Y.S.; Wu, Y.; Liu, G.; Yu, H.X.; Zheng, Q.P.; Zhu, D.N.; Xia, C.M.; Cao, Z.J. Pre-ischemic treadmill training affects glutamate and gamma aminobutyric acid levels in the striatal dialysate of a rat model of cerebral ischemia. Life Sci 2009, 84, 505–511. [Google Scholar]
- Zhang, F.; Jia, J.; Wu, Y.; Hu, Y.; Wang, Y. The effect of treadmill training pre-exercise on glutamate receptor expression in rats after cerebral ischemia. Int. J. Mol. Sci 2010, 11, 2658–2669. [Google Scholar]
- Liu, A.J.; Hu, Y.Y.; Li, W.B.; Xu, J.; Zhang, M. Cerebral ischemic pre-conditioning enhances the binding characteristics and glutamate uptake of glial glutamate transporter-1 in hippocampal CA1 subfield of rats. J. Neurochem 2011, 119, 202–209. [Google Scholar]
- Gong, S.J.; Chen, L.Y.; Zhang, M.; Gong, J.X.; Ma, Y.X.; Zhang, J.M.; Wang, Y.J.; Hu, Y.Y.; Sun, X.C.; Li, W.B.; Zhang, Y. Intermittent hypobaric hypoxia preconditioning induced brain ischemic tolerance by up-regulating glial glutamate transporter-1 in rats. Neurochem. Res 2012, 37, 527–537. [Google Scholar]
- Kanai, Y.; Hediger, M.A. The glutamate/neutral amino acid transporter family SLC1: Molecular, physiological and pharmacological aspects. Pflugers Arch 2004, 447, 469–479. [Google Scholar]
- Verma, R.; Mishra, V.; Sasmal, D.; Raghubir, R. Pharmacological evaluation of glutamate transporter 1 (GLT-1) mediated neuroprotection following cerebral ischemia/reperfusion injury. Eur. J. Pharmacol 2010, 638, 65–71. [Google Scholar]
- Tanaka, K.; Watase, K.; Manabe, T.; Yamada, K.; Watanabe, M.; Takahashi, K.; Iwama, H.; Nishikawa, T.; Ichihara, N.; Kikuchi, T.; et al. Epilepsy and exacerbation of brain injury in mice lacking the glutamate transporter GLT-1. Science 1997, 276, 1699–1702. [Google Scholar]
- Mimura, K.; Tomimatsu, T.; Minato, K.; Jugder, O.; Kinugasa-Taniguchi, Y.; Kanagawa, T.; Nozaki, M.; Yanagihara, I.; Kimura, T. Ceftriaxone preconditioning confers neuroprotection in neonatal rats through glutamate transporter 1 upregulation. Reprod. Sci 2011, 18, 1193–1201. [Google Scholar]
- Kawahara, K.; Kosugi, T.; Tanaka, M.; Nakajima, T.; Yamada, T. Reversed operation of glutamate transporter GLT-1 is crucial to the development of preconditioning-induced ischemic tolerance of neurons in neuron/astrocyte co-cultures. Glia 2005, 49, 349–359. [Google Scholar]
- Torp, R.; Lekieffre, D.; Levy, L.M.; Haug, F.M.; Danbolt, N.C.; Meldrum, B.S.; Ottersen, O.P. Reduced postischemic expression of a glial glutamate transporter, GLT1, in the rat hippocampus. Exp. Brain Res 1995, 103, 51–58. [Google Scholar]
- Zhang, M.; Li, W.B.; Geng, J.X.; Li, Q.J.; Sun, X.C.; Xian, X.H.; Qi, J.; Li, S.Q. The upregulation of glial glutamate transporter-1 participates in the induction of brain ischemic tolerance in rats. J. Cereb Blood Flow Metab 2007, 27, 1352–1368. [Google Scholar]
- Münch, C.; Zhu, B.G.; Leven, A.; Stamm, S.; Einkörn, H.; Schwalenstöcker, B.; Ludolph, A.C.; Riepe, M.W.; Meyer, T. Differential regulation of 5′ splice variants of the glutamate transporter EAAT2 in an in vivo model of chemical hypoxia induced by 3-nitropropionic acid. J. Neurosci. Res 2003, 71, 819–825. [Google Scholar]
- Pow, D.V.; Naidoo, T.; Lingwood, B.E.; Healy, G.N.; Williams, S.M.; Sullivan, R.K.P.; O’Driscoll, S.; Colditz, P.B. Loss of glial glutamate transporters and induction of neuronal expression of GLT-1B in the hypoxic neonatal pig brain. Dev. Brain Res 2004, 153, 1–11. [Google Scholar]
- Rauen, T.; Wiessner, M. Fine tuning of glutamate uptake and degradation in glial cells: Common transcriptional regulation of GLAST1 and GS. Neurochem. Int 2000, 37, 179–189. [Google Scholar]
- Genda, E.N.; Jackson, J.G.; Sheldon, A.L.; Locke, S.F.; Greco, T.M.; O’Donnell, J.C.; Spruce, L.A.; Xiao, R.; Guo, W.; Putt, M.; et al. Co-compartmentalization of the astroglial glutamate transporter, GLT-1, with glycolytic enzymes and mitochondria. J. Neurosci 2011, 31, 18275–18288. [Google Scholar]
- Zhang, Q.; Wu, Y.; Zhang, P.; Sha, H.; Jia, J.; Hu, Y.; Zhu, J. Exercise induces mitochondrial biogenesis after brain ischemia in rats. Neuroscience 2012, 205, 10–17. [Google Scholar]
- Swain, R.A.; Harris, A.B.; Wiener, E.C.; Dutka, M.V.; Morris, H.D.; Theien, B.E.; Konda, S.; Engberg, K.; Lauterbur, P.C.; Greenough, W.T. Prolonged exercise induces angiogenesis and increases cerebral blood volume in primary motor cortex of the rat. Neuroscience 2003, 117, 1037–1046. [Google Scholar]
- Bullitt, E.; Rahman, F.N.; Smith, J.K.; Kim, E.; Zeng, D.; Katz, L.M.; Marks, B.L. The effect of exercise on the cerebral vasculature of healthy aged subjects as visualized by MR angiography. AJNR Am. J. Neuroradiol 2009, 30, 1857–1863. [Google Scholar]
- Benedek, A.; Móricz, K.; Jurányi, Z.; Gigler, G.; Lévay, G.; Hársing, L.G., Jr; Mátyus, P.; Szénási, G.; Albert, M. Use of TTC staining for the evaluation of tissue injury in the early phases of reperfusion after focal cerebral ischemia in rats. Brain Res. 2006, 1116, 159–165. [Google Scholar]
- Jia, J.; Hu, Y.S.; Wu, Y.; Yu, H.X.; Liu, G.; Zhu, D.N.; Xia, C.M.; Cao, Z.J.; Zhang, X.; Guo, Q.C. Treadmill pre-training suppresses the release of glutamate resulting from cerebral ischemia in rats. Exp. Brain Res 2010, 204, 173–179. [Google Scholar]
- Longa, E.Z.; Weinstein, P.R.; Carlson, S.; Cummins, R. Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke 1989, 20, 84–91. [Google Scholar]
- Huang, Z.; Huang, P.L.; Panahian, N.; Dalkara, T.; Fishman, M.C.; Moskowitz, M.A. Effects of cerebral ischemia in mice deficient in neuronal nitric oxide synthase. Science 1994, 265, 1883–1885. [Google Scholar]
- Zhang, Q.; Wu, Y.; Sha, H.; Zhang, P.; Jia, J.; Hu, Y.; Zhu, J. Early exercise affects mitochondrial transcription factors expression after cerebral ischemia in rats. Int. J. Mol. Sci 2012, 13, 1670–1679. [Google Scholar]
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yang, X.; He, Z.; Zhang, Q.; Wu, Y.; Hu, Y.; Wang, X.; Li, M.; Wu, Z.; Guo, Z.; Guo, J.; et al. Pre-Ischemic Treadmill Training for Prevention of Ischemic Brain Injury via Regulation of Glutamate and Its Transporter GLT-1. Int. J. Mol. Sci. 2012, 13, 9447-9459. https://doi.org/10.3390/ijms13089447
Yang X, He Z, Zhang Q, Wu Y, Hu Y, Wang X, Li M, Wu Z, Guo Z, Guo J, et al. Pre-Ischemic Treadmill Training for Prevention of Ischemic Brain Injury via Regulation of Glutamate and Its Transporter GLT-1. International Journal of Molecular Sciences. 2012; 13(8):9447-9459. https://doi.org/10.3390/ijms13089447
Chicago/Turabian StyleYang, Xiaojiao, Zhijie He, Qi Zhang, Yi Wu, Yongshan Hu, Xiaolou Wang, Mingfen Li, Zhiyuan Wu, Zhenzhen Guo, Jingchun Guo, and et al. 2012. "Pre-Ischemic Treadmill Training for Prevention of Ischemic Brain Injury via Regulation of Glutamate and Its Transporter GLT-1" International Journal of Molecular Sciences 13, no. 8: 9447-9459. https://doi.org/10.3390/ijms13089447




