Microwave-Assisted Method for Simultaneous Extraction and Hydrolysis for Determination of Flavonol Glycosides in Ginkgo Foliage Using Brönsted Acidic Ionic-Liquid [HO3S(CH2)4mim]HSO4 Aqueous Solutions
Abstract
:1. Introduction
2. Results and Discussion
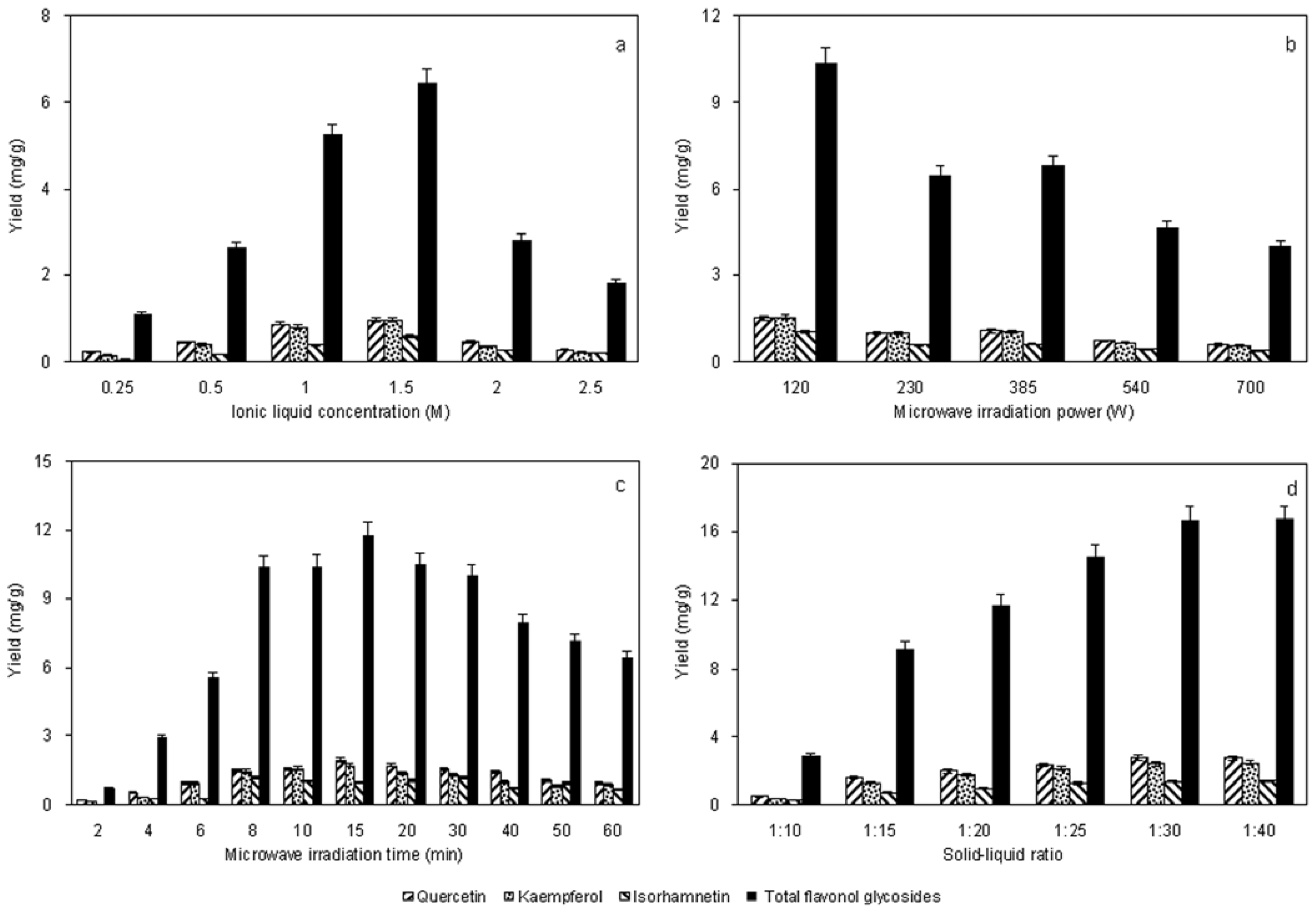
2.1. Effect of Solvent Concentration
2.2. Effect of Microwave-Irradiation Power
2.3. Effect of Microwave-Irradiation Time
2.4. Effect of Solid–Liquid Ratio
2.5. Comparison of ILMASEH Approach with Conventional Methods
2.6. Method Validation
3. Experimental Section
3.1. Materials and Chemicals
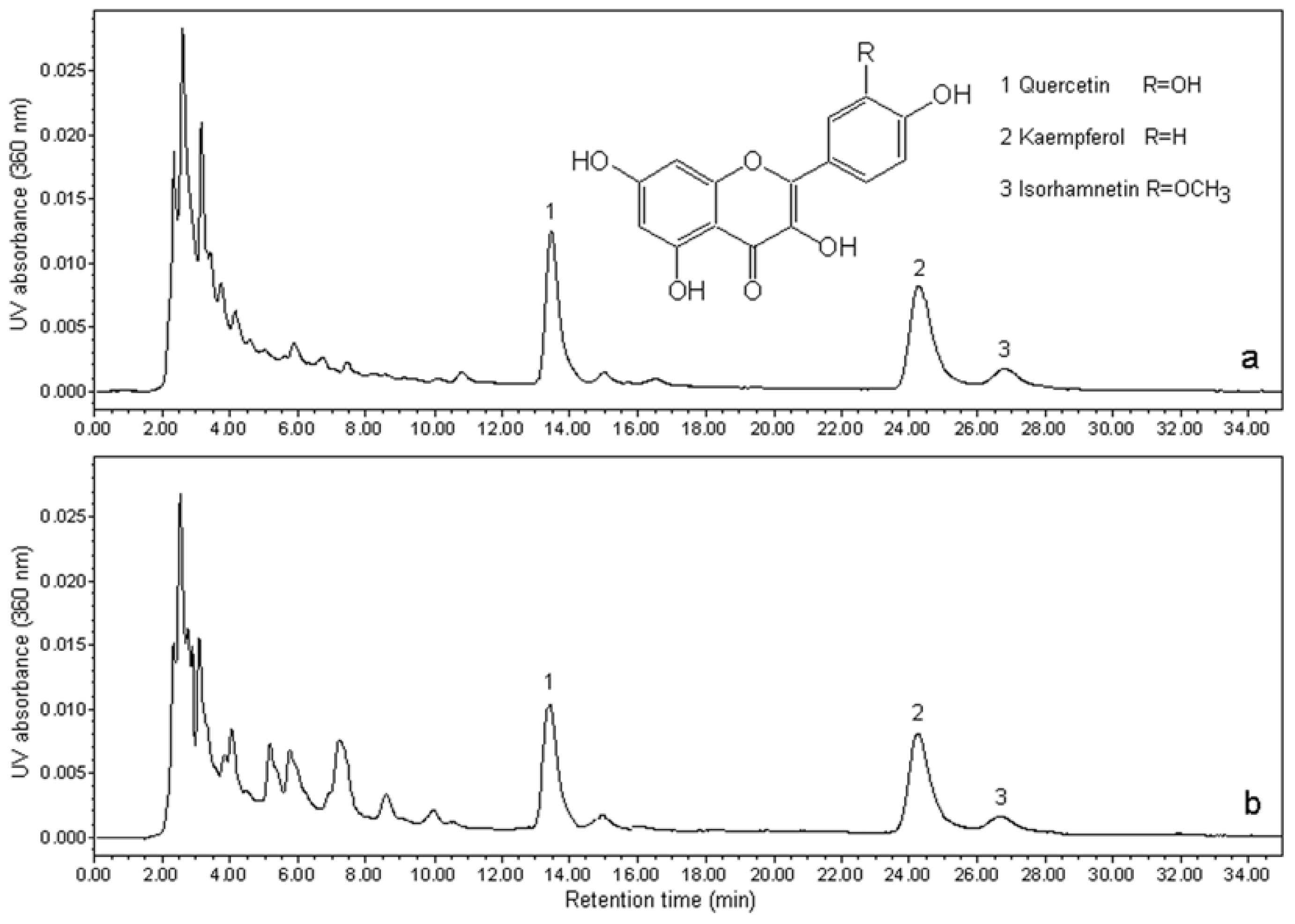
3.2. HPLC Analysis and Quantification
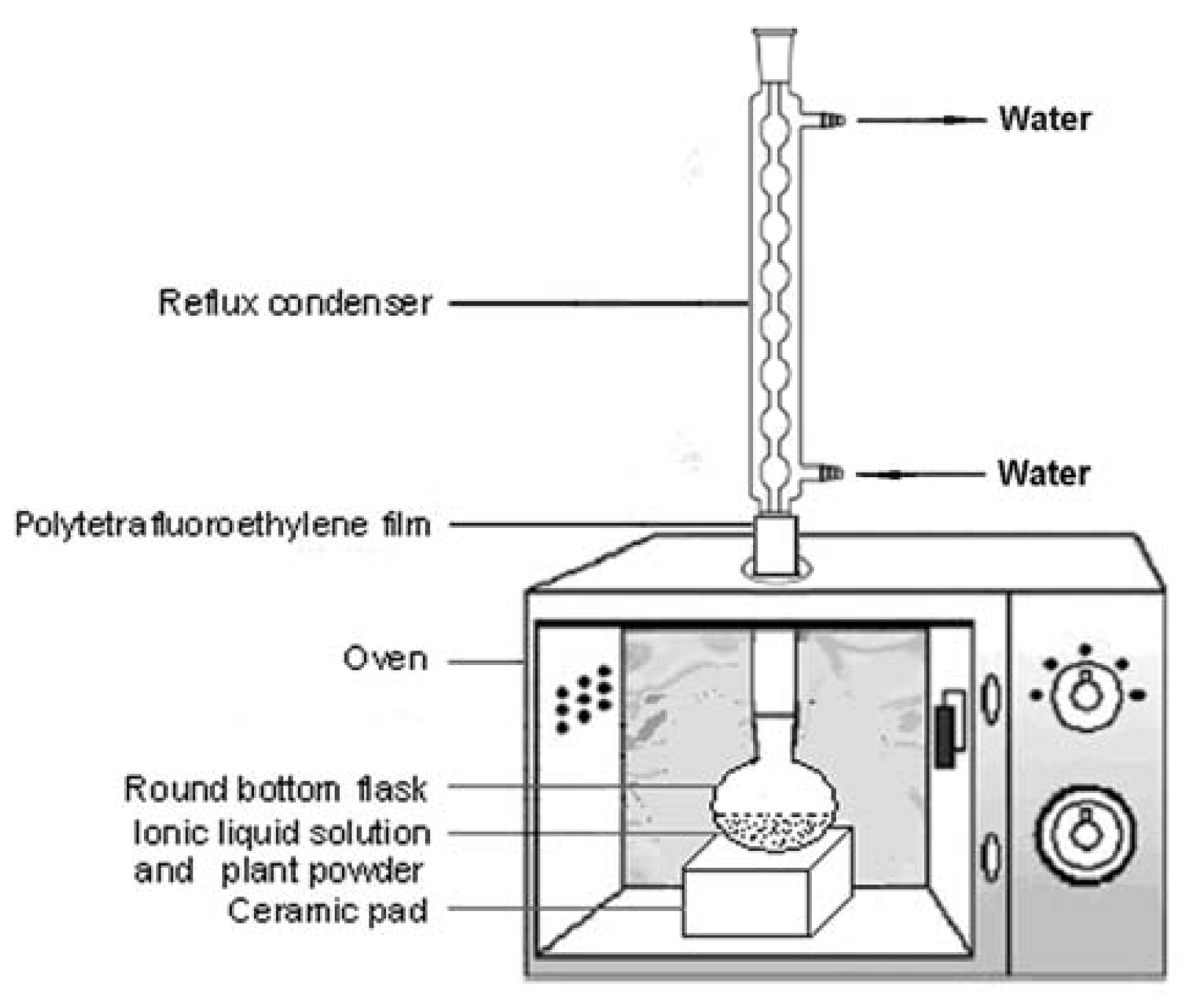
3.3. Ionic Liquid-based Microwave-Assisted Simultaneous Extraction and Hydrolysis (ILMASEH)
3.4. Pharmacopoeia of the People’s Republic of China Sample Preparation Method (PPRCM)
3.5. European Pharmacopoeia Sample Preparation Method (EPM)
3.6. United States Pharmacopeia and National Formulary Sample Preparation Method (USP-NFM)
3.7. Other Reference Sample Preparation Methods
4. Conclusions
Acknowledgment
References
- Kleijnen, J.; Knipschild, P. Ginkgo biloba. Lancet 1992, 340, 1136–1139. [Google Scholar]
- Kudolo, G.B.; Dorsey, S.; Blodgett, J. Effect of the ingestion of Ginkgo biloba extract on platelet aggregation and urinary prostanoid excretion in healthy and Type 2 diabetic subjects. Thromb. Res 2002, 108, 151–160. [Google Scholar]
- Guidetti, C.; Paracchini, S.; Lucchini, S.; Cambieri, M.; Marzatico, F. Prevention of neuronal cell damage induced by oxidative stress in vitro: Effect of different Ginkgo biloba extracts. J. Pharm. Pharm 2001, 53, 387–392. [Google Scholar]
- Van Beek, T.A. Ginkgolides and bilobalide: Their physical, chromatographic and spectroscopic properties. Bioorg. Med. Chem 2005, 13, 5001–5012. [Google Scholar]
- Ahlemeyer, B.; Krieglstein, J. Neuroprotective effects of Ginkgo biloba extract. Cell. Mol. Life Sci 2003, 60, 1779–1792. [Google Scholar]
- Van Beek, T.A. Chemical analysis of Ginkgo biloba leaves and extracts. J. Chromatogr. A 2002, 967, 21–55. [Google Scholar]
- Lai, S.M.; Chen, I.W.; Tsai, M.J. Preparative isolation of terpene trilactones from Ginkgo biloba leaves. J. Chromatogr. A 2005, 1092, 125–134. [Google Scholar]
- Chen, J.X.; Zhang, Y. Rapid microwave-assisted hydrolysis for determination of Ginkgo flavonol glycosides in extracts of Ginkgo biloba leaves. J. Chromatogr. Sci 2008, 46, 117–121. [Google Scholar]
- Zill-e-Huma Vian, M.A.; Fabiano-Tixier, A.; Elmaataoui, M.; Dangles, O.; Chemat, F. A remarkable influence of microwave extraction: Enhancement of antioxidant activity of extracted onion varieties. Food Chem 2011, 127, 1472–1480. [Google Scholar]
- Acquistucci, R.; Panfili, G.; Marconi, E. Application of the microwave hydrolysis to furosine determination in cereal and dairy foods. J. Agric. Food Chem 1996, 44, 3855–3857. [Google Scholar]
- Panfili, G.; Manzi, P.; Compagnone, D.; Scarciglia, L.; Palleschi, G. Rapid assay of choline in foods using microwave hydrolysis and a choline biosensor. J. Agric. Food Chem 2000, 48, 3403–3407. [Google Scholar]
- Quijano, G.; Couvert, A.; Amrane, A. Ionic liquids: Applications and future trends in bioreactor technology. Bioresour. Technol 2010, 101, 8923–8930. [Google Scholar]
- Welton, T. Room-temperature ionic liquids. Solvents for synthesis and catalysis. Chem. Rev 1999, 99, 2071–2084. [Google Scholar]
- Hoffmann, J.; Nüchter, M.; Ondruschka, B.; Wasserscheid, P. Ionic liquids and their heating behaviour during microwave irradiation—A state of the art report and challenge to assessment. Green Chem 2003, 5, 296–299. [Google Scholar]
- Poole, C.F. Chromatographic and spectroscopic methods for the determination of solvent properties of room temperature ionic liquids. J. Chromatogr. A 2004, 1037, 49–82. [Google Scholar]
- Liu, T.; Sui, X.; Zhang, R.; Yang, L.; Zu, Y.; Zhang, L.; Zhang, Y.; Zhang, Z. Application of ionic liquids based microwave-assisted simultaneous extraction of carnosic acid, rosmarinic acid and essential oil from Rosmarinus officinalis. J. Chromatogr. A 2011, 1218, 8480–8489. [Google Scholar]
- Ma, C.; Liu, T.; Yang, L.; Zu, Y.; Chen, X.; Zhang, L.; Zhang, Y.; Zhao, C. Ionic liquid based microwave simultaneous extraction of essential oil and biphenyl cyclooctene lignans from Schisandra chinensis Baill fruits. J. Chromatogr. A 2011, 1218, 8573–8580. [Google Scholar]
- Leadbeater, N.E.; Morenius, T.H. A study of the ionic liquid mediated microwave heating of organic solvents. J. Org. Chem 2002, 67, 3145–3148. [Google Scholar]
- Nara, S.J.; Harjani, J.R.; Salunkhe, M.M. Lipase-catalysed transesterification in ionic liquids and organic solvents: A comparative study. Tetrahedron Lett 2002, 43, 2979–2982. [Google Scholar]
- Ma, C.; Liu, T.; Yang, L.; Zu, Y.; Wang, S.; Zhang, R. Study on ionic liquid-based ultrasonic-assisted extraction of biphenyl cyclooctene lignans from the fruit of Schisandra chinensis Baill. Anal. Chim. Acta 2011, 689, 110–116. [Google Scholar]
- Yang, L.; Wang, H.; Zu, Y.; Zhao, C.; Zhang, L.; Chen, X.; Zhang, Z. Ultrasound-assisted extraction of the three terpenoid indole alkaloids vindoline, catharanthine and vinblastine from Catharanthus roseus using ionic liquid aqueous solutions. Chem. Eng. J 2011, 172, 705–712. [Google Scholar]
- Yang, L.; Liu, Y.; Zu, Y.; Zhao, C.; Zhang, L.; Chen, X.; Zhang, Z. Optimize the process of ionic liquid-based ultrasonic-assisted extraction of aesculin and aesculetin from Cortex Fraxini by response surface methodology. Chem. Eng. J 2011, 175, 539–547. [Google Scholar]
- Wang, S.; Yang, L.; Zu, Y.; Zhao, C.; Sun, X.; Zhang, L.; Zhang, Z. Design and performance evaluation of ionic liquids-microwave based environmental-friendly extraction technique for camptothecin and 10-hydroxycamptothecin from samara of Camptotheca acuminate. Ind. Eng. Chem. Res 2011, 50, 13620–13627. [Google Scholar]
- Wilkes, J.S. A short history of ionic liquids—From molten salts to neoteric solvents. Green Chem 2002, 4, 73–80. [Google Scholar]
- Chiappe, C.; Leandri, E.; Lucchesi, S.; Pieraccini, D.; Hammock, B.D.; Morisseau, C. Biocatalysis in ionic liquids: The stereoconvergent hydrolysis of trans-β-methylstyrene oxide catalyzed by soluble epoxide hydrolase. J. Mol. Catal. B Enzym 2004, 27, 243–248. [Google Scholar]
- Mohile, S.S.; Potdar, M.K.; Harjani, J.R.; Nara, S.J.; Salunkhe, M.M. Ionic liquids: Efficient additives for Candida rugosa lipase-catalysed enantioselective hydrolysis of butyl 2-(4-chlorophenoxy) propionate. J. Mol. Catal. B Enzym 2004, 30, 185–188. [Google Scholar]
- Qiao, K.; Yokoyama, C. Koch carbonylation of tertiary alcohols in the presence of acidic ionic liquids. Catal. Commun 2006, 7, 450–453. [Google Scholar]
- Wasserscheid, P.; Sesing, M.; Korth, W. Hydrogensulfate and tetrakis (hydrogensulfato) borate ionic liquids: Synthesis and catalytic application in highly Brønsted-acidic systems for Friedel–Crafts alkylation. Green Chem 2002, 4, 134–138. [Google Scholar]
- Yang, Q.W.; Wei, Z.J.; Xing, H.B.; Ren, Q.L. Brönsted acidic ionic liquids as novel catalysts for the hydrolyzation of soybean isoflavone glycosides. Catal. Commun 2008, 9, 1307–1311. [Google Scholar]
- Chen, Y.; Xie, M.Y.; Gong, X.F. Microwave-assisted extraction used for the isolation of total triterpenoid saponins from Ganoderma atrum. J. Food Eng 2007, 81, 162–170. [Google Scholar]
- Gray, D.E.; Upton, R.; Chandra, A.; Porter, A.; Harris, R.K. Quantitative analysis of flavonol glycosides in Ginkgo biloba: A comparison of two analytical methods. Phytochem. Anal 2006, 17, 56–62. [Google Scholar]
- Chinese Pharmacopoeia Commission, Pharmacopoeia of the People’s Republic of China I; People’s Medicinal Publishing House: Beijing, China, 2010; pp. 296–297.
- European Pharmacopoeia Commission, European Pharmacopoeia, 7th ed; Council of Europe: Strasbourg, France, 2011; pp. 1828–1829.
- United States Pharmacopeial Convention, United States Pharmacopeia 32 and National Formulary 27; USP-NF 2009; United States Pharmacopeial Convention: Rockville, ML, USA, 2009; p. 1019.
| Methods | Solvents | Solvent volume (mL/g) | Extraction method | Power (W) | Heating time (h) | Yield (mg/g) | |||
|---|---|---|---|---|---|---|---|---|---|
| Quercetin | Kaempferol | Isorhamnetin | Total flavonol glycosides | ||||||
| ILMASEH | 1.5 M [HO3S(CH2)4mim]HSO4 | 35 | Microwave | 120 | 0.25 | 2.75 ± 0.13 | 2.36 ± 0.11 | 1.37 ± 0.06 | 16.33 ± 0.76 |
| PPRCM | Chloroform/Methanol/Hydrochloric acid | 225 | Soxhlet | 500 | 6.50 | 2.45 ± 0.12 | 1.90 ± 0.08 | 1.19 ± 0.06 | 13.94 ± 0.65 |
| EPM | Acetone-H2O (60:40) Hydrochloric acid | Approx. 60 | Reflux | 500 | 2.75 | 2.35 ± 0.14 | 1.95 ± 0.11 | 1.23 ± 0.07 | 13.93 ± 0.81 |
| USP-NFM | Ethanol-H2O-Hydrochloric acid (50:20:8) | 78 | Reflux | 500 | 2.25 | 2.44 ± 0.11 | 2.19 ± 0.12 | 1.35 ± 0.07 | 15.07 ± 0.76 |
| AEMM | Ethanol-H2O-Hydrochloric acid (50:20:8) | 78 | Microwave | 120 | 0.25 | 2.56 ± 0.12 | 2.15 ± 0.10 | 1.20 ± 0.06 | 14.83 ± 0.71 |
| BMM | 1.5 M [Bmim]Br | 78 | Microwave | 120 | 0.25 | 0.57 ± 0.02 | 0.69 ± 0.03 | 0.30 ± 0.01 | 3.92 ± 0.15 |
| ABMM | 1.5 M [Bmim]Br (pH 0.5, adjusted with HCl) | 78 | Microwave | 120 | 0.25 | 2.71 ± 0.14 | 2.45 ± 0.11 | 1.35 ± 0.06 | 16.34 ± 0.78 |
| Compounds | Initial concentration (mg mL−1) | Recovered concentration after ILMASEH (mg mL−1) | RSD% (n = 3) | Average recovery (%) | Recovered concentration after 7 day (mg mL−1) | RSD% (n = 3) | Average recovery (%) |
|---|---|---|---|---|---|---|---|
| Quercetin | 2.00 | 1.97 | 0.96 | 98.5 | 1.94 | 0.97 | 97.0 |
| Kaempferol | 1.50 | 1.48 | 0.97 | 99.3 | 1.39 | 0.99 | 92.7 |
| Isorhamnetin | 1.00 | 0.98 | 1.01 | 98.0 | 0.91 | 1.03 | 91.0 |
| Aglycone content of the sample (mg) | Amount of added aglycone standard (mg) | Amount of the sample determined with added aglycone standard (mg) | Recovery (%) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sample | Quercetin | Kaempferol | Isorhamnetin | Quercetin | Kaempferol | Isorhamnetin | Quercetin | Kaempferol | Isorhamnetin | Quercetin | Kaempferol | Isorhamnetin |
| 1 | 2.80 | 2.46 | 1.39 | 1.40 | 1.30 | 0.50 | 4.23 | 3.72 | 1.91 | 100.71 | 98.94 | 101.06 |
| 2 | 2.80 | 2.46 | 1.39 | 2.80 | 2.60 | 1.00 | 5.57 | 5.1 | 2.41 | 99.46 | 100.79 | 100.84 |
| 3 | 2.80 | 2.46 | 1.39 | 4.20 | 3.90 | 1.50 | 6.88 | 6.33 | 2.86 | 98.29 | 99.53 | 98.96 |
| Average | 99.49 | 99.75 | 100.29 | |||||||||
| Ionic liquid | Cation | Anion | Form (25 °C) | Solubility in H2O (g/100 mL) |
|---|---|---|---|---|
| [HO3S(CH2)4mim] HSO4 | 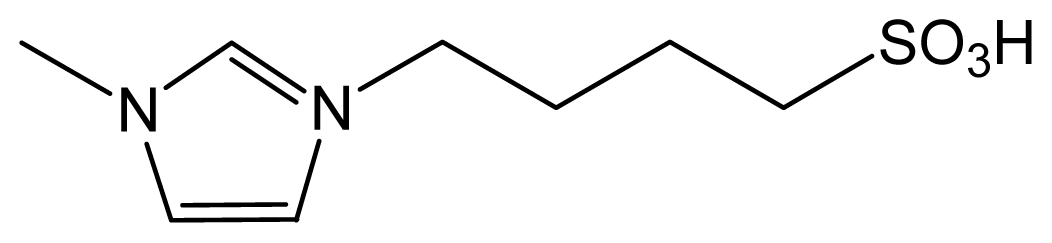 | HSO4− | Liquid | Totally miscible |
| [Bmim] Br | 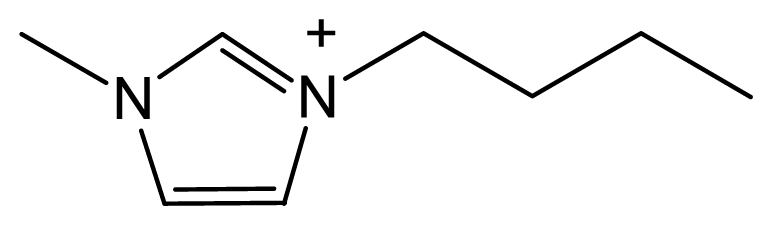 | Br− | Solid | Totally miscible |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yao, H.; Du, X.; Yang, L.; Wang, W.; Yang, F.; Zhao, C.; Meng, X.; Zhang, L.; Zu, Y. Microwave-Assisted Method for Simultaneous Extraction and Hydrolysis for Determination of Flavonol Glycosides in Ginkgo Foliage Using Brönsted Acidic Ionic-Liquid [HO3S(CH2)4mim]HSO4 Aqueous Solutions. Int. J. Mol. Sci. 2012, 13, 8775-8788. https://doi.org/10.3390/ijms13078775
Yao H, Du X, Yang L, Wang W, Yang F, Zhao C, Meng X, Zhang L, Zu Y. Microwave-Assisted Method for Simultaneous Extraction and Hydrolysis for Determination of Flavonol Glycosides in Ginkgo Foliage Using Brönsted Acidic Ionic-Liquid [HO3S(CH2)4mim]HSO4 Aqueous Solutions. International Journal of Molecular Sciences. 2012; 13(7):8775-8788. https://doi.org/10.3390/ijms13078775
Chicago/Turabian StyleYao, Huanhuan, Xinxuan Du, Lei Yang, Wenjie Wang, Fengjian Yang, Chunjian Zhao, Xiangdong Meng, Lin Zhang, and Yuangang Zu. 2012. "Microwave-Assisted Method for Simultaneous Extraction and Hydrolysis for Determination of Flavonol Glycosides in Ginkgo Foliage Using Brönsted Acidic Ionic-Liquid [HO3S(CH2)4mim]HSO4 Aqueous Solutions" International Journal of Molecular Sciences 13, no. 7: 8775-8788. https://doi.org/10.3390/ijms13078775






