Infrared Study of Er3+/Yb3+ Co-Doped GeO2-PbO-Bi2O3 Glass
Abstract
:1. Introduction
2. Results and Discussion
FTIR
3. Experimental Section
4. Conclusions
References
- Lezal, D. Chalcogenide glasses-survey and progress. J. Optoelectron. Adv. Mater. 2003, 5, 23–34. [Google Scholar]
- Li, X.; Nie, Q.; Dai, S.; Xu, T.; Lu, L.; Zhang, X. Energy transfer and frequency upconversion in Ho3+/Yb3+ co-doped bismuth-germanate glasses. J. Alloy Compd 2008, 454, 510–514. [Google Scholar]
- Kassab, L.; Hora, W.; Lozano, W.; Oliveira, M.; Maciel, G. Optical properties of Er3+ doped GeO2–PbO glass: Effect of doping with Bi2O3. Opt. Commun 2007, 269, 356–361. [Google Scholar]
- Cacho, V.; Kassab, L.; Oliveira, S.; Mansano, R.; Verdonck, P. Blue cooperative luminescence properties in Yb3+ doped GeO2 PbO Bi2O3 vitreous system for the production of thin films. Thin Solid Films 2006, 515, 764–767. [Google Scholar]
- Baia, L.; Iliescu, T.; Simon, S.; Kiefer, W. Raman and IR spectroscopic studies of manganese doped GeO2-Bi2O3 glass. J. Mol. Struct 2001, 599, 9–13. [Google Scholar]
- Ribeiro, J.L.; Dexpert-Ghy, J.; Piriou, B.; Mastelaro, R. Structural studies in lead germanate glasses: EXAFS and vibrational spectroscopy. J. Non-Cryst. Solids 1993, 159, 213–221. [Google Scholar]
- Vasantharani, P.; Shailajha, S. Acoustic properties of borate glasses doped with Ag2O. Glob. J. Mol. Sci 2009, 4, 29–33. [Google Scholar]
- Hwa, L.G.; Chao, W.C.; Szu, S.P. Temperature dependence of elastic moduli of lanthanum gallogermanate glasses. J. Mater. Sci 2002, 37, 3423–3427. [Google Scholar] [Green Version]
- Kim, Y.; Saienga, J.; Martin, S.W. Glass formation in and structural investigation of Li2S + GeS2 + GeO2 composition using Raman and IR spectroscopy. J. Non-Cryst. Solids 2005, 351, 3716–3724. [Google Scholar]
- Saddeek, Y.B.; Abousehly, A.M.; Hussien, S.I. Synthesis and several features of the Na2O-B2O3-Bi2O3-MoO3 glasses. J. Phys. D Appl. Phys 2007, 40, 4674–4681. [Google Scholar]
- Rath, S.; Kabiraj, D.; Avasthi, D.K.; Tripathi, A.; Jain, K.P.; Kumar, M.; Mavi, H.S.; Shukla, A.K. Evidence of nanostructure formation in Ge oxide by crystallization induced by swift heavy ion irradiation. Nucl. Instrum. Meth. Phys. Res. B 2007, 263, 419–423. [Google Scholar]
- Pascuta, P.; Culea, E. FTIR spectroscopic study of some bismuth germanate glasses containing gadolinium ions. Mater. Lett 2008, 62, 4127–4129. [Google Scholar]
- Baia, L.; Stefan, R.; Popp, J.; Simon, S.; Kiefer, W. Vibrational investigations of highly iron doped B2O3–Bi2O3 glass systems. J. Non-Cryst. Solids 2003, 324, 109–117. [Google Scholar]
- Dachille, F.; Roy, R. The use of infra-red absorption and molar refractivities to check coordination. Z. Kristallogr 1959, 111, 462–470. [Google Scholar]
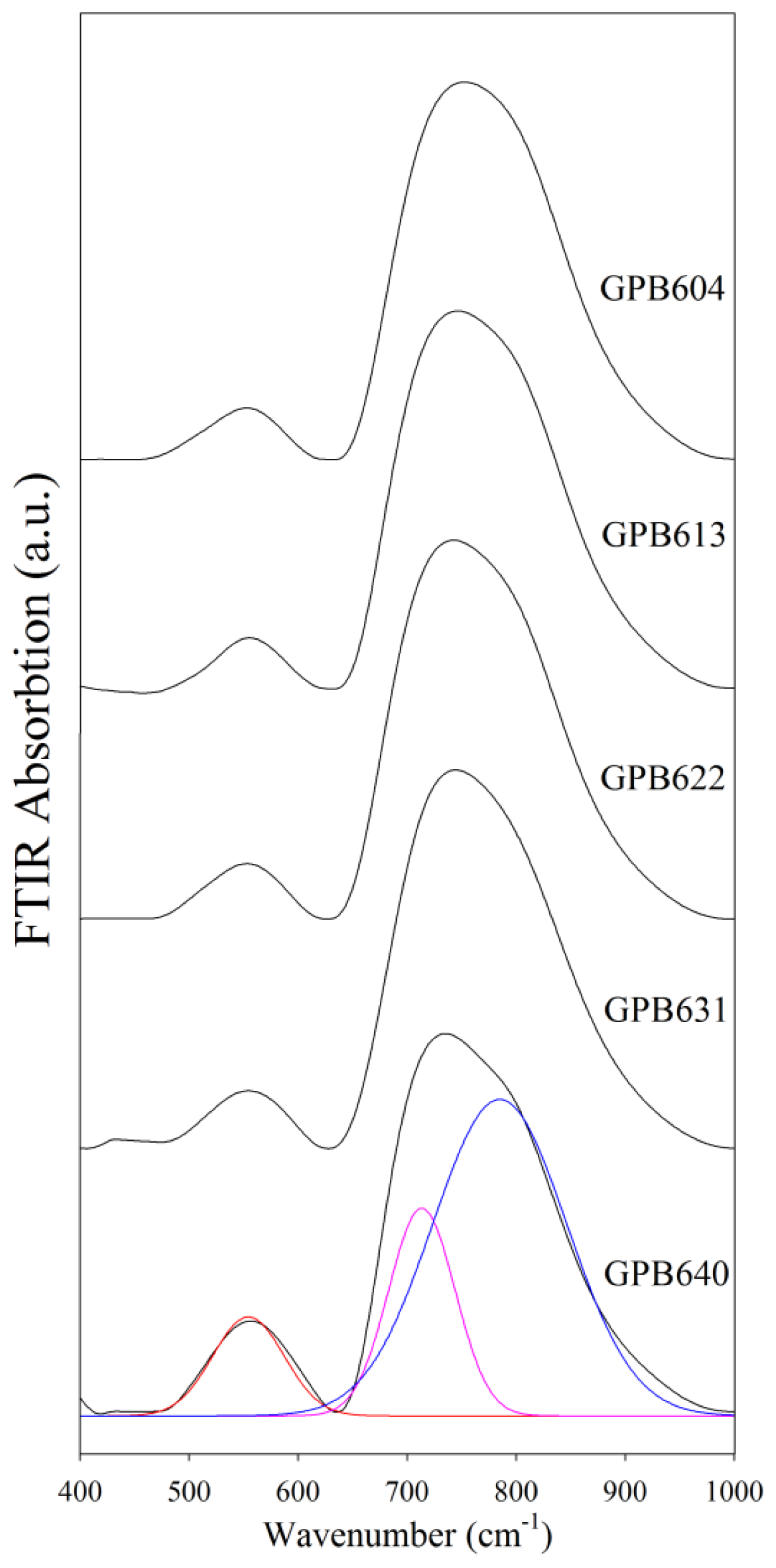
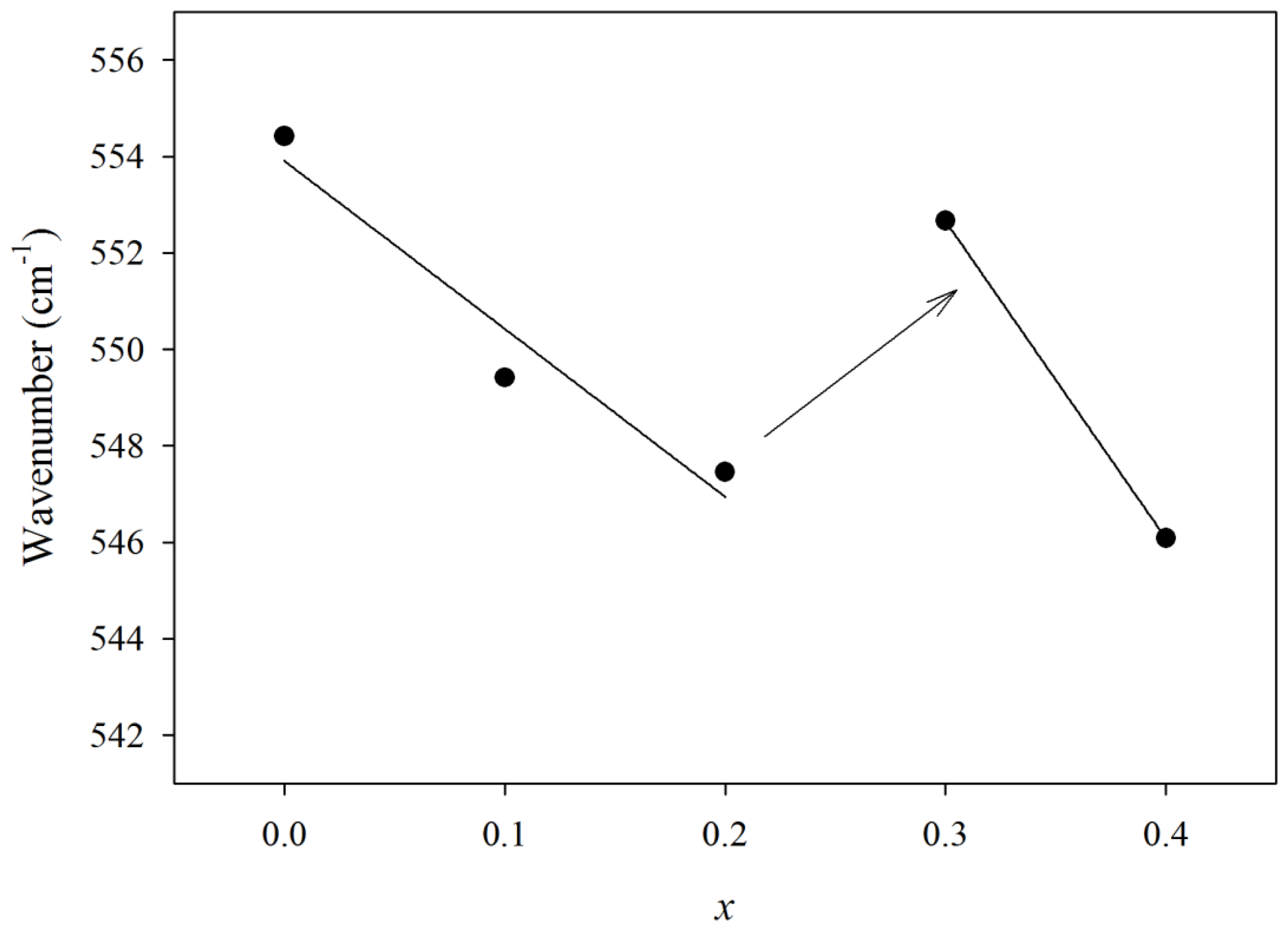
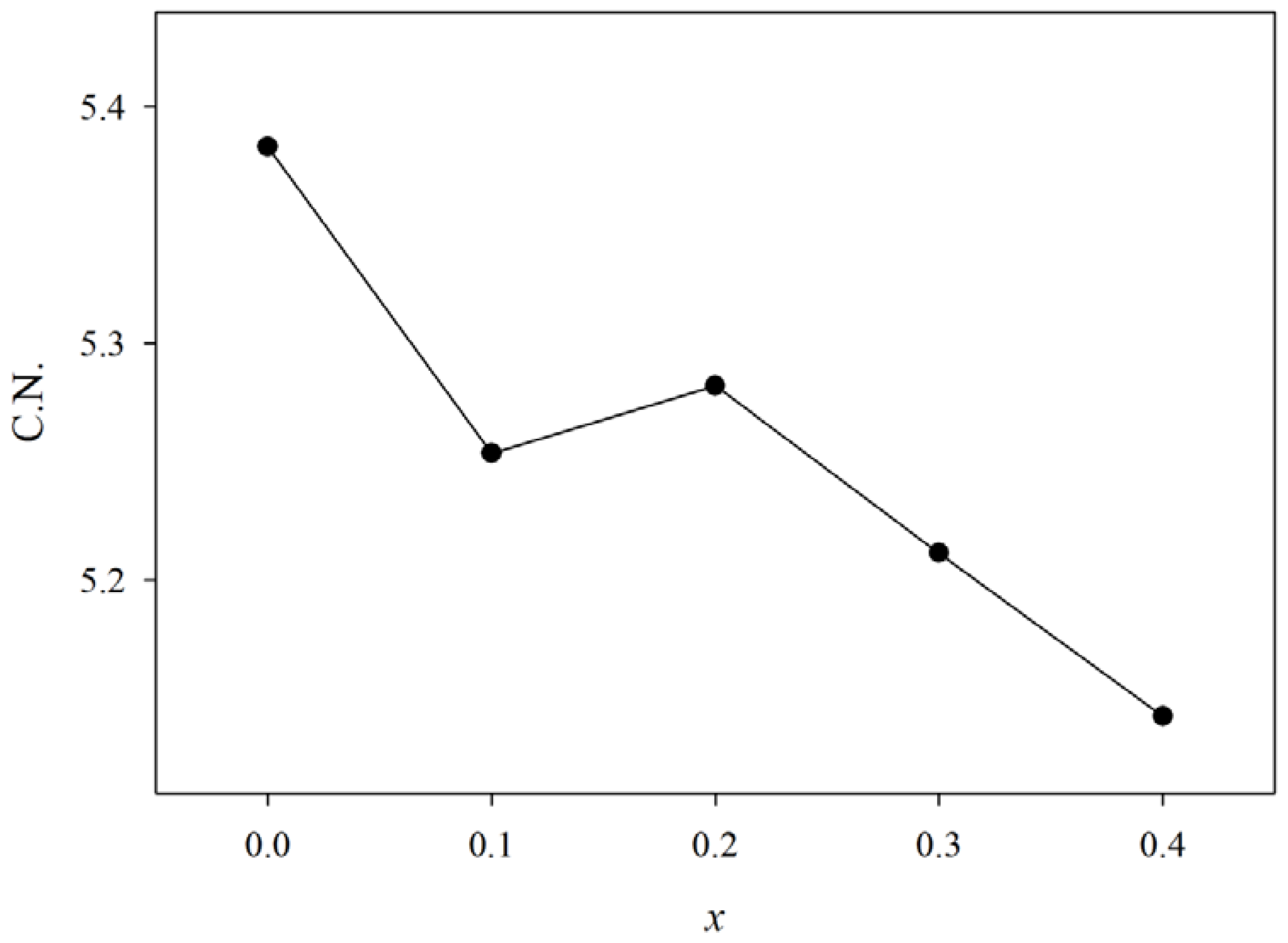
| x | Composition (mol%) | Composition (wt%) | Sample Code | f1 (cm−1) | f2 (cm−1) | f3 (cm−1) | Coordination number | |||
|---|---|---|---|---|---|---|---|---|---|---|
| GeO2 | PbO | Bi2O3 | Er2O3 | Yb2O3 | ||||||
| 0 | 60 | 40 | 0 | 0.5 | 1.5 | GPB640 | 554 | 714 | 785 | 5.38 |
| 0.1 | 60 | 30 | 10 | 0.5 | 1.5 | GPB631 | 549 | 720 | 791 | 5.25 |
| 0.2 | 60 | 20 | 20 | 0.5 | 1.5 | GPB622 | 547 | 714 | 785 | 5.28 |
| 0.3 | 60 | 10 | 30 | 0.5 | 1.5 | GPB613 | 553 | 717 | 788 | 5.21 |
| 0.4 | 60 | 0 | 40 | 0.5 | 1.5 | GPB604 | 546 | 719 | 790 | 5.14 |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bahari, H.-R.; Sidek, H.A.A.; Adikan, F.R.M.; Yunus, W.M.M.; Halimah, M.K. Infrared Study of Er3+/Yb3+ Co-Doped GeO2-PbO-Bi2O3 Glass. Int. J. Mol. Sci. 2012, 13, 8609-8614. https://doi.org/10.3390/ijms13078609
Bahari H-R, Sidek HAA, Adikan FRM, Yunus WMM, Halimah MK. Infrared Study of Er3+/Yb3+ Co-Doped GeO2-PbO-Bi2O3 Glass. International Journal of Molecular Sciences. 2012; 13(7):8609-8614. https://doi.org/10.3390/ijms13078609
Chicago/Turabian StyleBahari, Hamid-Reza, Hj. A. A. Sidek, Faisal Rafiq M. Adikan, Wan M. M. Yunus, and Mohamed K. Halimah. 2012. "Infrared Study of Er3+/Yb3+ Co-Doped GeO2-PbO-Bi2O3 Glass" International Journal of Molecular Sciences 13, no. 7: 8609-8614. https://doi.org/10.3390/ijms13078609




