Astragalus membranaceus Inhibits Inflammation via Phospho-P38 Mitogen-Activated Protein Kinase (MAPK) and Nuclear Factor (NF)-κB Pathways in Advanced Glycation End Product-Stimulated Macrophages
Abstract
:1. Introduction
2. Results and Discussion
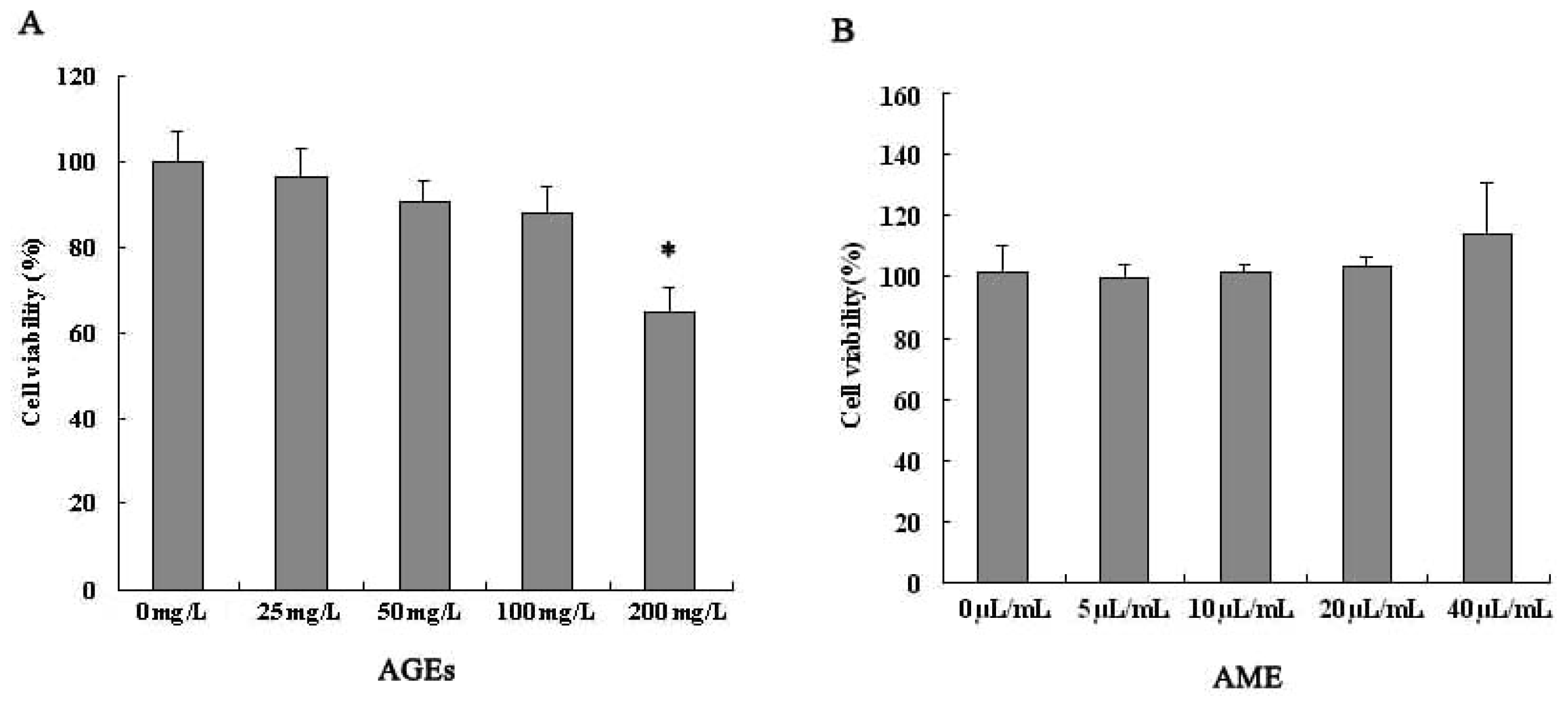
2.1. Effects of AGEs and AME on Cell Viability
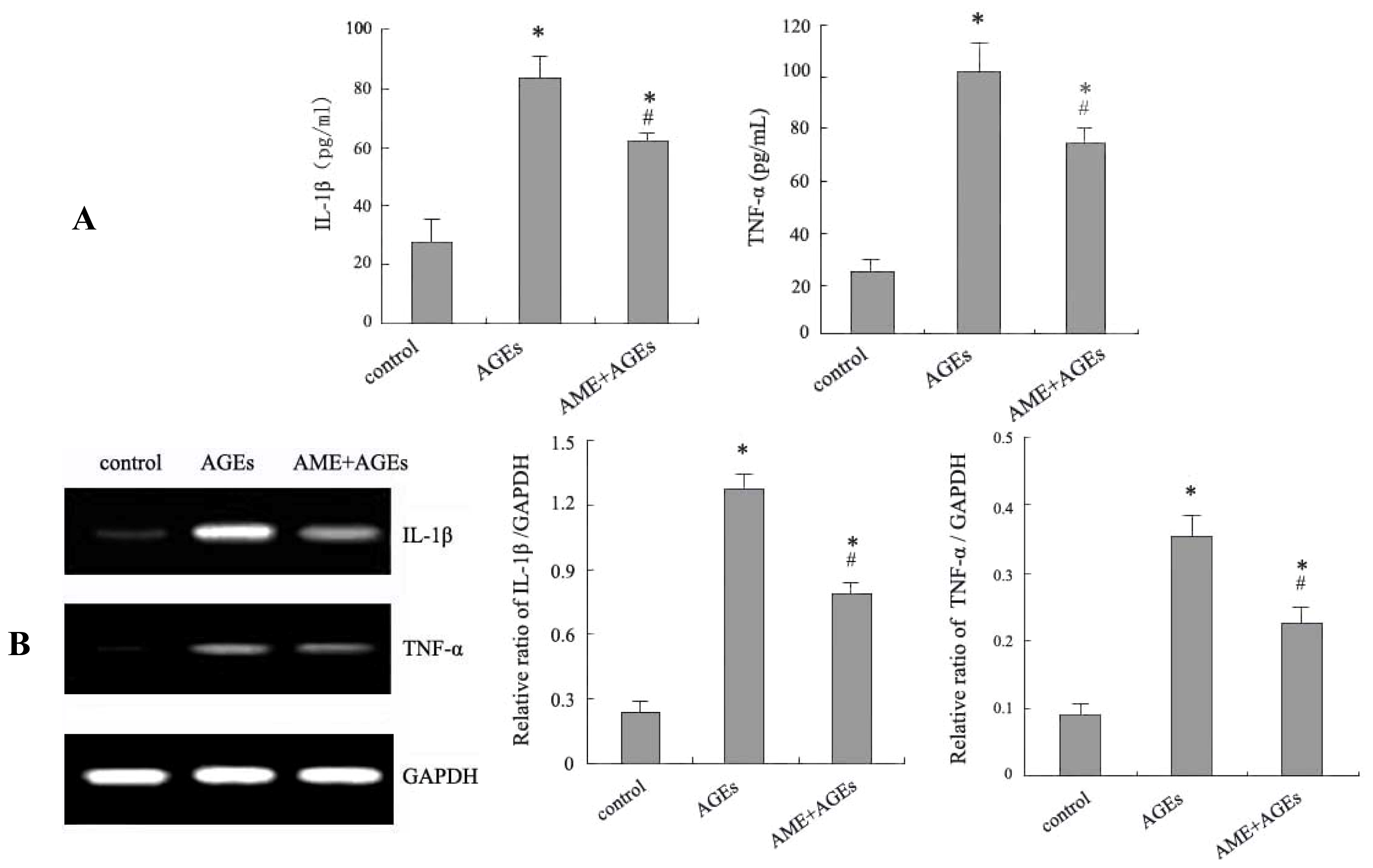
2.2. Effects of AME and AGEs on the mRNA Levels and Secretion of IL-1β and TNF-α in Macrophages
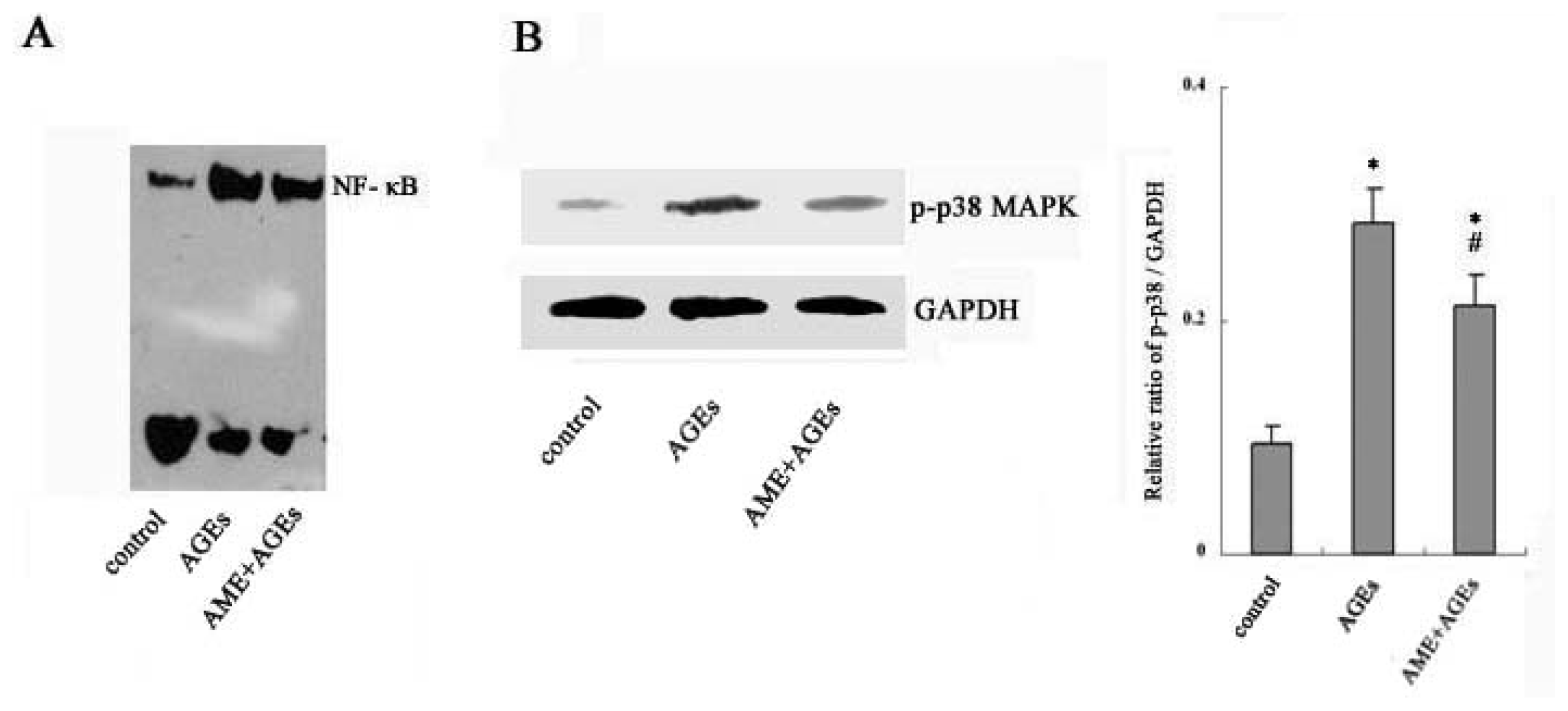
2.3. NF-κB and Phospho-p38 MAPK Mediate the Effects of AGEs and AME on Macrophages
3. Experimental Section
3.1. Materials
3.2. Preparation of AME
3.3. Cell Culture
3.4. Assessment of Cell Viability
3.5. Assessment of TNF-α and IL-1β Secretion
3.6. Detection of mRNAs by RT-PCR
3.7. Western Blot Analysis
3.8. Electrophoretic Mobility Shift Assay (EMSA)
3.9. Statistical Analysis
4. Conclusions
Acknowledgments
- Conflict of InterestThe authors declare no conflict of interest.
References
- Donath, M.Y. Inflammation as a sensor of metabolic stress in obesity and type 2 diabetes. Endocrinology 2011, 152, 4005–4006. [Google Scholar]
- Darisipudi, M.N.; Kulkarni, O.P.; Sayyed, S.G.; Ryu, M.; Migliorini, A.; Sagrinati, C. Dual blockade of the homeostatic chemokine CXCL12 and the proinflammatory chemokine CCL2 has additive protective effects on diabetic kidney disease. Am. J. Pathol 2011, 179, 116–124. [Google Scholar]
- Tesch, G.H. Macrophages and diabetic nephropathy. Semin. Nephrol 2010, 30, 290–301. [Google Scholar]
- Nogi, Y.; Nagashima, M.; Terasaki, M.; Nohtomi, K.; Watanabe, T.; Hirano, T. Glucose-dependent insulinotropic polypeptide prevents the progression of macrophage-driven atherosclerosis in diabetic apolipoprotein e-null mice. PLoS One 2012, 7, e35683. [Google Scholar]
- Qiao, M.; Zhao, Q.; Lee, C.F.; Tannock, L.R.; Smart, E.J.; LeBaron, R.G.; Phelix, C.F.; Rangel, Y.; Asmis, R. Thiol oxidative stress induced by metabolic disorders amplifies macrophage chemotactic responses and accelerates atherogenesis and kidney injury in LDL receptor-deficient mice. Arterioscler. Thromb. Vasc. Biol 2009, 29, 1779–1786. [Google Scholar]
- Ninichuk, V.; Khandoga, A.G.; Segerer, S.; Loetscher, P.; Schlapbach, A.; Revesz, L.; Feifel, R.; Khandoga, A.; Krombach, F.; Nelson, P.J.; et al. The role of interstitial macrophages in nephropathy of type 2 diabetic db/db mice. Am. J. Pathol 2007, 170, 1267–1276. [Google Scholar]
- Usui, H.K.; Shikata, K.; Sasaki, M.; Okada, S.; Matsuda, M.; Shikata, Y. Macrophage scavenger receptor-A-deficient mice are resistant against diabetic nephropathy through amelioration of microinflammation. Diabetes 2007, 56, 363–372. [Google Scholar]
- Reddy, M.A.; Natarajan, R. Epigenetic mechanisms in diabetic vascular complications. Cardiovasc. Res 2011, 90, 421–429. [Google Scholar]
- Bao, W.; Min, D.; Twigg, S.M.; Shackel, N.A.; Warner, F.J.; Yue, D.K. Monocyte CD147 is induced by advanced glycation end products and high glucose concentration: Possible role in diabetic complications. Am. J. Physiol. Cell Physiol 2010, 299, C1212–C1219. [Google Scholar]
- Kaushansky, K.; Broudy, V.C.; Harlan, J.M.; Adamson, J.W. Tumor necrosis factor-alpha and tumor necrosis factor-β (lymphotoxin) stimulate the production of granulocyte-macrophage colony-stimulating factor, macrophage colony-stimulating factor, and IL-1 in vivo. J. Immunol 1988, 141, 3410–3415. [Google Scholar]
- Dinarello, C.A. A clinical perspective of IL-1β as the gatekeeper of inflammation. Eur. J. Immunol 2011, 41, 1203–1217. [Google Scholar]
- Saitoh, T.; Fujita, N.; Jang, M.H.; Uematsu, S.; Yang, B.G.; Satoh, T. Loss of the autophagy protein Atg16 L1 enhances endotoxin-induced IL-1β production. Nature 2008, 456, 264–268. [Google Scholar]
- Ibrahim, A.S.; El-Remessy, A.B.; Matragoon, S.; Zhang, W.; Patel, Y.; Khan, S. Retinal microglial activation and inflammation induced by amadori-glycated albumin in a rat model of diabetes. Diabetes 2011, 60, 1122–1133. [Google Scholar]
- Chen, H.Y.; Huang, X.R.; Wang, W.; Li, J.H.; Heuchel, R.L.; Chung, A.C. The protective role of Smad7 in diabetic kidney disease: Mechanism and therapeutic potential. Diabetes 2011, 60, 590–601. [Google Scholar]
- Ko, J.K.; Chik, C.W. The protective action of radix Astragalus membranaceus against hapten-induced colitis through modulation of cytokines. Cytokine 2009, 47, 85–90. [Google Scholar]
- Zong, H.; Ward, M.; Madden, A.; Yong, P.H.; Limb, G.A.; Curtis, T.M. Hyperglycaemia-induced pro-inflammatory responses by retinal Müller glia are regulated by the receptor for advanced glycation end-products (RAGE). Diabetologia 2010, 53, 2656–2666. [Google Scholar]
- Nin, J.W.; Jorsal, A.; Ferreira, I.; Schalkwijk, C.G.; Prins, M.H.; Parving, H.H. Higher plasma soluble Receptor for Advanced Glycation End Products (sRAGE) levels are associated with incident cardiovascular disease and all-cause mortality in type 1 diabetes: A 12-year follow-up study. Diabetes 2010, 59, 2027–2032. [Google Scholar]
- Aouadi, M.; Tesz, G.J.; Nicoloro, S.M.; Wang, M.; Chouinard, M.; Soto, E. Orally delivered siRNA targeting macrophage Map4k4 suppresses systemic inflammation. Nature 2009, 458, 1180–1184. [Google Scholar]
- Sartippour, M.R.; Renier, G. Upregulation of macrophage lipoprotein lipase in patients with type 2 diabetes: Role of peripheral factors. Diabetes 2000, 49, 597–602. [Google Scholar]
- Zhao, L.H.; Ma, Z.X.; Zhu, J.; Yu, X.H.; Weng, D.P. Characterization of polysaccharide from Astragalus radix as the macrophage stimulator. Cell. Immunol 2011, 271, 329–334. [Google Scholar]
- Van Zoelen, M.A.; Schouten, M.; de Vos, A.F.; Florquin, S.; Meijers, J.C.; Nawroth, P.P. The receptor for advanced glycation end products impairs host defense in pneumococcal pneumonia. J. Immunol 2009, 182, 4349–4356. [Google Scholar]
- Li, Y.; Reddy, M.A.; Miao, F.; Shanmugam, N.; Yee, J.K.; Hawkins, D. Role of the histone H3 lysine 4 methyltransferase, SET7/9, in the regulation of NF-kappaB-dependent inflammatory genes. J. Biol. Chem 2008, 283, 26771–26781. [Google Scholar]
- Dean, J.L.; Brook, M.; Clark, A.R.; Saklatvala, J. p38 mitogen-activated protein kinase regulates cyclooxygenase-2 mRNA stability and transcription in lipopolysaccharidetreated human monocytes. J. Biol. Chem 1999, 274, 264–269. [Google Scholar]
- Zhang, F.; Banker, G.; Liu, X.; Suwanabol, P.A.; Lengfeld, J.; Yamanouchi, D. The Novel Function of Advanced Glycation End Products in Regulation of MMP-9 Production. J. Surg. Res 2011, 171, 871–876. [Google Scholar]
- Weidenbusch, M.; Anders, H.J. Tissue microenvironments define and get reinforced by macrophage phenotypes in homeostasis or during inflammation, repair and fibrosis. J. Innate Immun 2012. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Qin, Q.; Niu, J.; Wang, Z.; Xu, W.; Qiao, Z.; Gu, Y. Astragalus membranaceus Inhibits Inflammation via Phospho-P38 Mitogen-Activated Protein Kinase (MAPK) and Nuclear Factor (NF)-κB Pathways in Advanced Glycation End Product-Stimulated Macrophages. Int. J. Mol. Sci. 2012, 13, 8379-8387. https://doi.org/10.3390/ijms13078379
Qin Q, Niu J, Wang Z, Xu W, Qiao Z, Gu Y. Astragalus membranaceus Inhibits Inflammation via Phospho-P38 Mitogen-Activated Protein Kinase (MAPK) and Nuclear Factor (NF)-κB Pathways in Advanced Glycation End Product-Stimulated Macrophages. International Journal of Molecular Sciences. 2012; 13(7):8379-8387. https://doi.org/10.3390/ijms13078379
Chicago/Turabian StyleQin, Qiaojing, Jianying Niu, Zhaoxia Wang, Wangjie Xu, Zhongdong Qiao, and Yong Gu. 2012. "Astragalus membranaceus Inhibits Inflammation via Phospho-P38 Mitogen-Activated Protein Kinase (MAPK) and Nuclear Factor (NF)-κB Pathways in Advanced Glycation End Product-Stimulated Macrophages" International Journal of Molecular Sciences 13, no. 7: 8379-8387. https://doi.org/10.3390/ijms13078379



