A Novel Preparation Method for 5-Aminosalicylic Acid Loaded Eudragit S100 Nanoparticles
Abstract
:1. Introduction
2. Results and Discussion
2.1. Effect of Process Parameters
2.1.1. Pressure Effect
2.1.2. Temperature Effect
2.1.3. Effect of 5-ASA Concentration
2.1.4. Effect of Solution Flow Rate
2.2. Physicochemical Properties of Nanoparticles
2.2.1. XRD Analysis
2.2.2. DSC Analysis
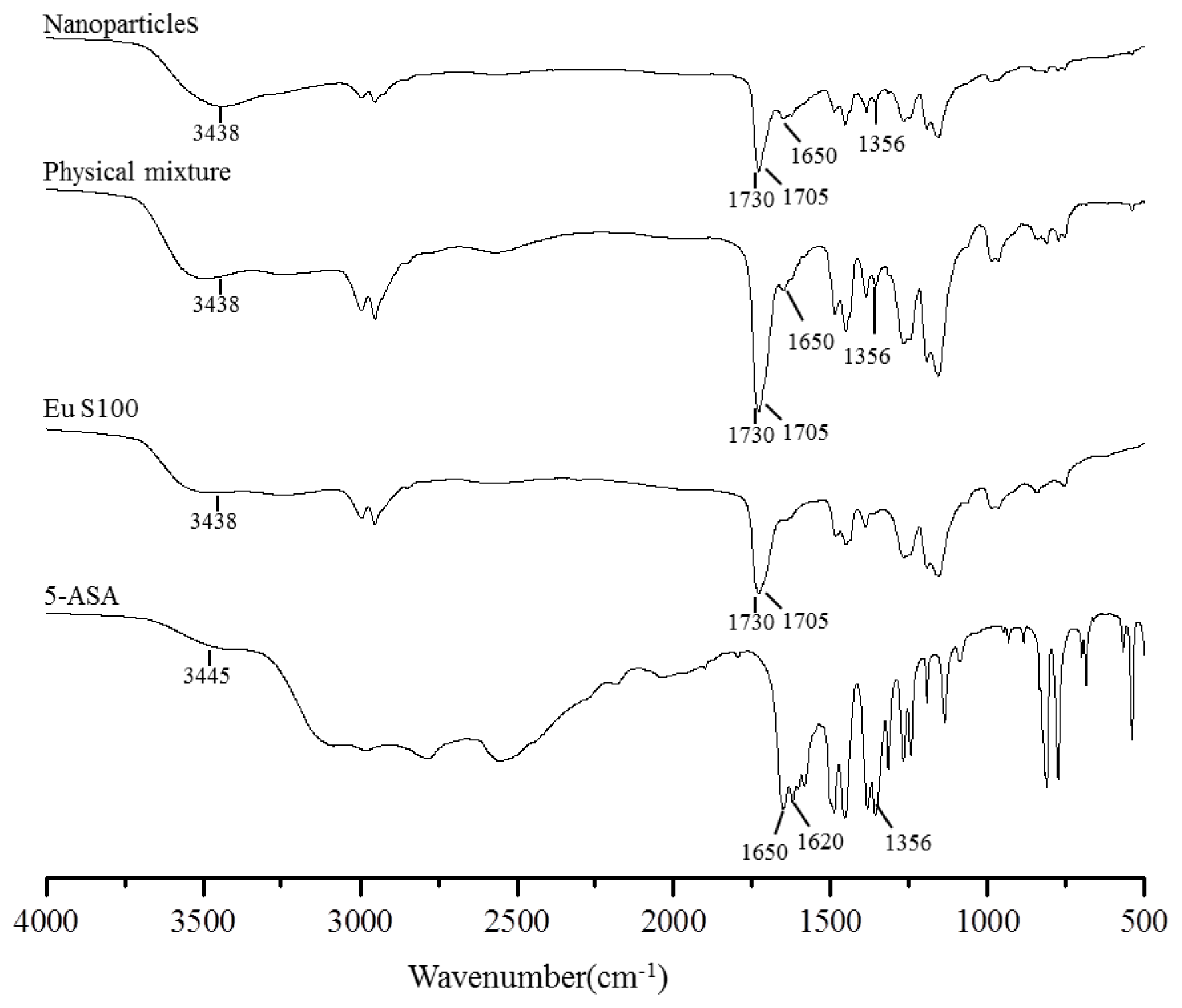
2.2.3. FTIR Spectroscopy Analysis
3. Experimental Section
3.1. Materials
3.2. Experimental Apparatus and Procedure
3.3. Preparation Conditions of 5-ASA/EU S100 Nanoparticles
3.4. Nanoparticle Characterization
3.4.1. SEM Analysis
3.4.2. Particle Size and Particle Size Distribution Analysis
3.4.3. 5-ASA Loading and Entrapment Efficiency Measurement
3.4.4. XRD Analysis
3.4.5. DSC Analysis
3.4.6. FTIR Spectroscopy Analysis
4. Conclusions
Acknowledgments
- Conflict of InterestThe authors declare that there are no conflicts of interest.
References
- Chourasia, M.K.; Jain, S.K. Pharmaceutical approaches to colon targeted drug delivery systems. J. Pharm. Pharm. Sci 2003, 6, 33–66. [Google Scholar]
- Kumar, P.; Mishra, B. Colon targeted drug delivery systems-an overview. Curr. Drug Deliv 2008, 5, 186–198. [Google Scholar]
- Pinto, J.F. Site-specific drug delivery systems within the gastro-intestinal tract: From the mouth to the colon. Int. J. Pharm 2010, 16, 44–52. [Google Scholar]
- Shah, N.; Shah, T.; Amin, A. Polysaccharides: A targeting strategy for colonic drug delivery. Expert Opin. Drug Deliv 2011, 8, 779–796. [Google Scholar]
- Williams, C.; Panaccione, R.; Ghosh, S.; Rioux, K. Optimizing clinical use of mesalazine (5-aminosalicylic acid) in inflammatory bowel disease. Ther. Adv. Gastroenterol 2011, 4, 237–248. [Google Scholar]
- Pertuit, D.; Moulari, B.; Betz, T.; Nadaradjane, A.; Neumann, D.; Ismaïli, L.; Refouvelet, B.; Pellequer, Y.; Lamprecht, A. 5-amino salicylic acid bound nanoparticles for the therapy of inflammatory bowel disease. J. Control. Release 2007, 123, 211–218. [Google Scholar]
- Lamprecht, A.; Stallmach, A.; Kawashima, Y.; Lehr, C.M. Carrier systems for the treatment of inflammatory bowel disease. Drugs Future 2002, 27, 961–971. [Google Scholar]
- Friend, D.R. New oral delivery systems for treatment of inflammatory bowel disease. Adv. Drug Deliv. Rev 2005, 57, 247–265. [Google Scholar]
- Lamprecht, A. Multiparticulate systems in the treatment of inflammatory bowel disease. Curr. Drug Targets Inflamm. Allergy 2003, 2, 137–144. [Google Scholar]
- Lamprecht, A.; Yamamoto, H.; Takeuchi, H.; Kawashima, Y. Nanoparticles enhance therapeutic efficiency by selectively increased local drug dose in experimental colitis in rats. J. Pharmacol. Exp. Ther 2005, 315, 196–202. [Google Scholar]
- Lamprecht, A.; Schäfer, U.; Lehr, C.M. Size-dependent bioadhesion of micro-and nanoparticulate carriers to the inflamed colonic mucosa. Pharm. Res 2001, 18, 788–793. [Google Scholar]
- Lamprecht, A.; Ubrich, N.; Yamamoto, H.; Schäfer, U.; Takeuchi, H.; Maincent, P.; Kawashima, Y.; Lehr, C.M. Biodegradable nanoparticles for targeted drug delivery in treatment of inflammatory bowel disease. J. Pharmacol. Exp. Ther 2001, 299, 775–781. [Google Scholar]
- Moulari, B.; Pertuit, D.; Pellequer, Y.; Lamprecht, A. The targeting of surface modified silica nanoparticles to inflamed tissue in experimental colitis. Biomaterials 2008, 29, 4554–4560. [Google Scholar]
- Kshirsagar, S.J.; Bhalekar, M.R.; Patel, J.N.; Mohapatra, S.K.; Shewale, N.S. Preparation and characterization of nanocapsules for colon-targeted drug delivery system. Pharm. Dev. Technol 2011. [Google Scholar] [CrossRef]
- Nakase, H.; Okazaki, K.; Tabata, Y.; Uose, S.; Ohana, M.; Uchida, K.; Matsushima, Y.; Kawanami, C.; Oshima, C.; Ikada, Y.; Chiba, T. Development of an oral drug delivery system targeting immune-regulating cells in experimental inflammatory bowel disease: A new therapeutic strategy. J. Pharmacol. Exp. Ther 2000, 292, 15–21. [Google Scholar]
- Kalani, M.; Yunus, R. Application of supercritical antisolvent method in drug encapsulation: A review. Int. J. Nanomedicine 2011, 6, 1429–1442. [Google Scholar]
- Mishima, K. Biodegradable particle formation for drug and gene delivery using supercritical fluid and dense gas. Adv. Drug Deliv. Rev 2008, 60, 411–432. [Google Scholar]
- Zu, Y.; Wang, D.; Zhao, X.; Jiang, R.; Zhang, Q.; Zhao, D.; Li, Y.; Zu, B.; Sun, Z. A novel preparation method for camptothecin (CPT) loaded folic acid conjugated dextran tumor-targeted nanoparticles. Int. J. Mol. Sci 2011, 12, 4237–4249. [Google Scholar]
- Zhao, X.; Zu, Y.; Jiang, R.; Wang, Y.; Li, Y.; Li, Q.; Zhao, D.; Zu, B.; Zhang, B.; Sun, Z.; et al. Preparation and physicochemical properties of 10-hydroxycamptothecin (HCPT) nanoparticles by supercritical antisolvent (SAS) process. Int. J. Mol. Sci 2011, 12, 2678–26591. [Google Scholar]
- Kang, Y.; Yin, G.; Ouyang, P.; Huang, Z.; Yao, Y.; Liao, X.; Chen, A.; Pu, X. Preparation of PLLA/PLGA microparticles using solution enhanced dispersion by supercritical fluids (SEDS). J. Colloid Interface Sci 2008, 322, 87–94. [Google Scholar]
- Yeo, S.D.; Kim, M.S.; Lee, J.C. Recrystallization of sulfathiazole and chlorpropamide using the supercritical fluid antisolvent process. J. Supercrit. Fluids 2003, 25, 143–154. [Google Scholar]
- Yeo, S.D.; Lee, J.C. Crystallization of sulfamethizole using the supercritical and liquid antisolvent processes. J. Supercrit. Fluids 2004, 30, 315–323. [Google Scholar]
- Lee, B.M.; Jeong, J.S.; Lee, Y.H.; Lee, B.C.; Kim, H.S.; Kim, H.; Lee, Y.W. Supercritical antisolvent micronization of cyclotrimethylenetrinitramin: Influence of the organic solvent. Ind. Eng. Chem. Res 2009, 48, 11162–11167. [Google Scholar]
- Jin, H.Y.; Xia, F.; Zhao, Y.P. Preparation of hydroxypropyl methyl cellulose phthalate nanoparticles with mixed solvent using supercritical antisolvent process and its application in co-precipitation of insulin. Adv. Power Technol 2012, 23, 157–163. [Google Scholar]
- He, W.Z.; Suo, Q.L.; Hong, H.L.; Li, G.M.; Zhao, X.H.; Li, C.P.; Shan, A. Supercritical antisolvent micronization of natural carotene by the SEDS process through prefilming atomization. Ind. Eng. Chem. Res 2006, 45, 2108–2115. [Google Scholar]
- Hu, D.D.; Lin, C.C.; Liu, L.; Li, S.; Zhao, Y.P. Preparation, characterization, and in vitro release investigation of lutein/zein nanoparticles via solution enhanced dispersion by supercritical fluids. J. Food Eng 2011, 109, 545–552. [Google Scholar]
- Patomchaiviwat, V.; Paeratakul, O.; Kulvanich, P. Formation of inhalable rifampicin-poly (l-lactide) microparticles by supercritical anti-solvent process. AAPS PharmSciTech 2008, 9, 1119–1129. [Google Scholar]
- Bleich, J.; Kleinebudde, P.; Müller, B.W. Influence of gas density and pressure on microparticles produced with the ASES process. Int. J. Pharm 1994, 106, 77–84. [Google Scholar]
- Xia, F.; Hu, D.D.; Jin, H.Y.; Zhao, Y.P.; Liang, J.M. Preparation of lutein proliposomes by supercritical anti-solvent technique. Food Hydrocoll 2012, 26, 456–463. [Google Scholar]
- Reverchon, E.; Antonacci, A. Polymer microparticles production by supercritical assisted atomization. J. Supercrit. Fluids 2007, 39, 444–452. [Google Scholar]
- Reverchon, E.; Macro, I.D.; Porta, G.D. Tailoring of nano- and micro-particles of some superconductor precursors by supercritical antisolvent precipitation. J. Supercrit. Fluids 2002, 23, 81–87. [Google Scholar]
- Franceschi, E.; de Cesaro, A.M.; Feiten, M.; Ferreira, S.R.S.; Dariva, C.; Kunita, M.H.; Rubira, A.F.; Muniz, E.C.; Corazza, M.L.; Oliveira, J.V. Precipitation of β-carotene and PHBV and co-precipitation from SEDS technique using supercritical CO2. J. Supercrit. Fluids 2008, 47, 259–269. [Google Scholar]
- Wang, Y.; Wang, Y.; Yang, J.; Pfeffer, R.; Dave, R.; Michniak, B. The application of a supercritical antisolvent process for sustained drug delivery. Power Technol 2006, 164, 94–102. [Google Scholar]
- Franceschi, E.; de Cesaro, A.M.; Ferreira, S.R.S. Precipitation of β-carotene microparticles from SEDS technique using supercritical CO2. J. Food Eng 2009, 95, 656–663. [Google Scholar]
- Reverchon, E.; Della Porta, G.; Falivene, M.G. Process parameters and morphology in amoxicillin micro and submicro particles generation by supercritical antisolvent precipitation. J. Supercrit. Fluids 2000, 17, 239–248. [Google Scholar]
- Bharate, S.S.; Bharate, S.B.; Bajaj, A.N. Interactions and incompatibilities of pharmaceutical excipients with active pharmaceutical ingredients: A comprehensive review. J. Excip. Food Chem 2010, 1, 3–26. [Google Scholar]
- Pignatello, R.; Ferro, M.; de Guidi, G.; Salemi, G.; Vandelli, M.A.; Guccione, S.; Geppi, M.; Forte, C.; Puglisi, G. Preparation, characterisation and photosensitivity studies of solid dispersions of diflunisal and Eudragit RS100 and RL100. Int. J. Pharm 2001, 7, 27–42. [Google Scholar]
- Maghsoodi, M.; Sadeghpoor, F. Preparation and evaluation of solid dispersions of piroxicam and Eudragit S100 by spherical crystallization technique. Drug Dev. Ind. Pharm 2010, 36, 917–925. [Google Scholar]
- Mladenovska, K.; Cruaud, O.; Richomme, P.; Belamie, E.; Raicki, R.S.; Venier-Julienne, M.C.; Popovski, E.; Benoit, J.P.; Goracinova, K. 5-ASA loaded chitosan-Ca-alginate microparticles: Preparation and physicochemical characterization. Int. J. Pharm 2007, 345, 59–69. [Google Scholar]





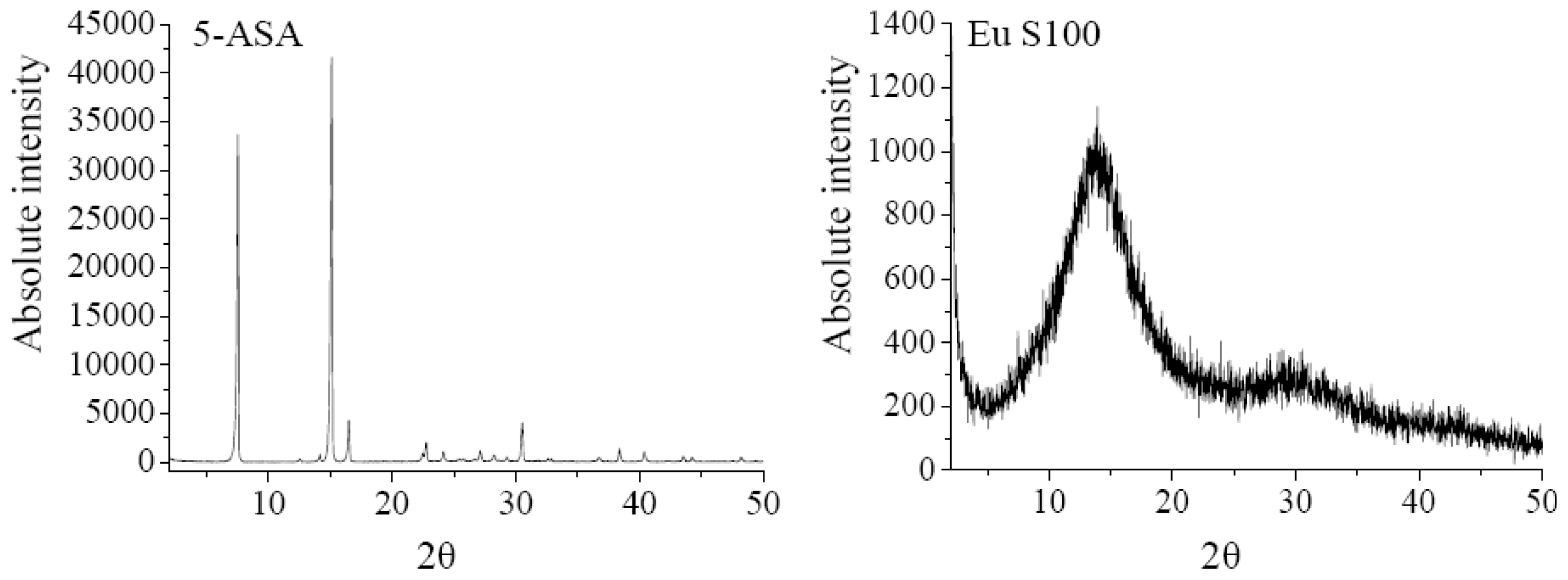


| Pressure MPa | Temperature °C | 5-ASA concentration mg/mL | Solution flow rate mL/min | 5-ASA loading % a | Entrapment efficiency % a | Mean particle size nm a | Span a |
|---|---|---|---|---|---|---|---|
| 8 | 35 | 1.0 | 1.0 | 12.78 ± 0.28 | 18.90 ± 1.20 | 395 ± 89 | 1.98 ± 0.25 |
| 10 | 35 | 1.0 | 1.0 | 13.06 ± 0.86 | 37.26 ± 0.98 | 192 ± 30 | 1.05 ± 0.12 |
| 12 | 35 | 1.0 | 1.0 | 7.21 ± 0.26 | 47.93 ± 1.36 | 159 ± 22 | 0.81 ± 0.05 |
| 15 | 35 | 1.0 | 1.0 | 3.35 ± 0.21 | 51.12 ± 1.85 | 147 ± 16 | 0.62 ± 0.05 |
| 12 | 40 | 1.0 | 1.0 | 6.20 ± 0.08 | 46.63 ± 0.48 | 169 ± 26 | 1.14 ± 0.93 |
| 12 | 45 | 1.0 | 1.0 | 4.16 ± 0.35 | 30.36 ± 0.35 | aggregated | - |
| 12 | 35 | 0.5 | 1.0 | 3.49 ± 0.08 | 60.51 ± 0.21 | 137 ± 20 | 0.67 ± 0.04 |
| 12 | 35 | 1.3 | 1.0. | 9.87 ± 0.35 | 36.45 ± 1.24 | 257 ± 40 | 1.83 ± 0.20 |
| 12 | 35 | 1.0 | 0.5 | 21.91 ± 0.56 | 39.48 ± 1.54 | 137 ± 16 | 0.78 ± 0.05 |
| 12 | 35 | 1.0 | 1.5 | 5.99 ± 0.32 | 3.21 ± 0.38 | 227 ± 48 | 1.57 ± 0.15 |
| Eu S100 | 5-ASA | Physical mixture | Nanoparticles | ||||
|---|---|---|---|---|---|---|---|
| Wavenumber (cm−1) | Functional group | Wavenumber (cm−1) | Functional group | Wavenumber (cm−1) | Functional group | Wavenumber (cm−1) | Functional group |
| 3438 | O–H | 3445 | N–H | 3438 | O–H | 3438 | O–H |
| 1705 | C=O | 3445 | O–H | 1705 | C=O | 1705 | C=O |
| 1730 | C=O | 1650 | C=0 | 1730 | C=O | 1730 | C=O |
| 1620 | N–H | 1650 | C=0 | 1650 | C=0 | ||
| 1356 | C–N | 1356 | C–N | 1356 | C–N | ||
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hu, D.; Liu, L.; Chen, W.; Li, S.; Zhao, Y. A Novel Preparation Method for 5-Aminosalicylic Acid Loaded Eudragit S100 Nanoparticles. Int. J. Mol. Sci. 2012, 13, 6454-6468. https://doi.org/10.3390/ijms13056454
Hu D, Liu L, Chen W, Li S, Zhao Y. A Novel Preparation Method for 5-Aminosalicylic Acid Loaded Eudragit S100 Nanoparticles. International Journal of Molecular Sciences. 2012; 13(5):6454-6468. https://doi.org/10.3390/ijms13056454
Chicago/Turabian StyleHu, Daode, Liang Liu, Wenjuan Chen, Sining Li, and Yaping Zhao. 2012. "A Novel Preparation Method for 5-Aminosalicylic Acid Loaded Eudragit S100 Nanoparticles" International Journal of Molecular Sciences 13, no. 5: 6454-6468. https://doi.org/10.3390/ijms13056454




