Enrichment of Polychlorinated Biphenyls from Aqueous Solutions Using Fe3O4 Grafted Multiwalled Carbon Nanotubes with Poly Dimethyl Diallyl Ammonium Chloride
Abstract
:1. Introduction
2. Results and Discussion
2.1. Characterization of Sorbents
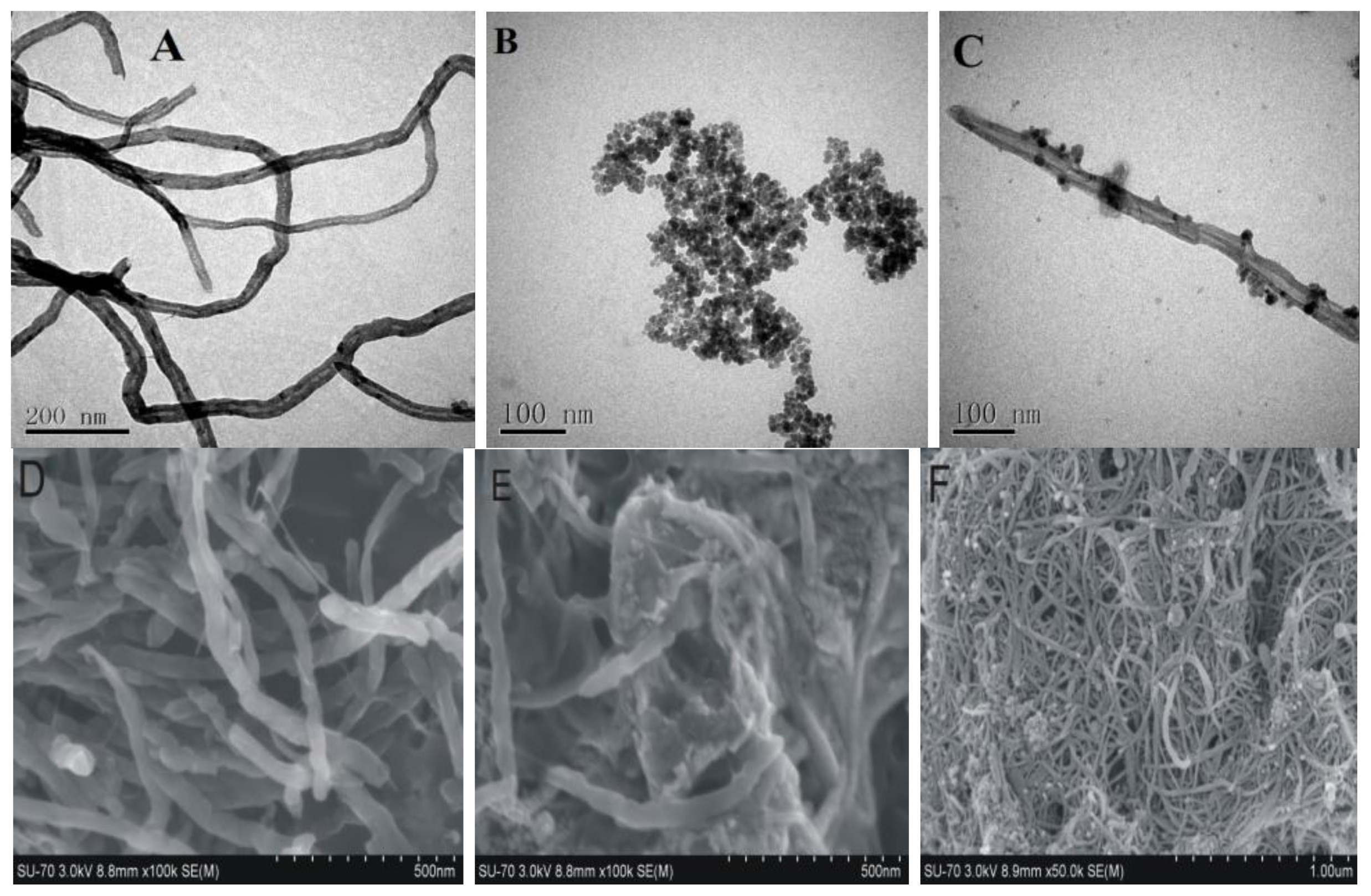
2.1.1. Particle Size and Morphology
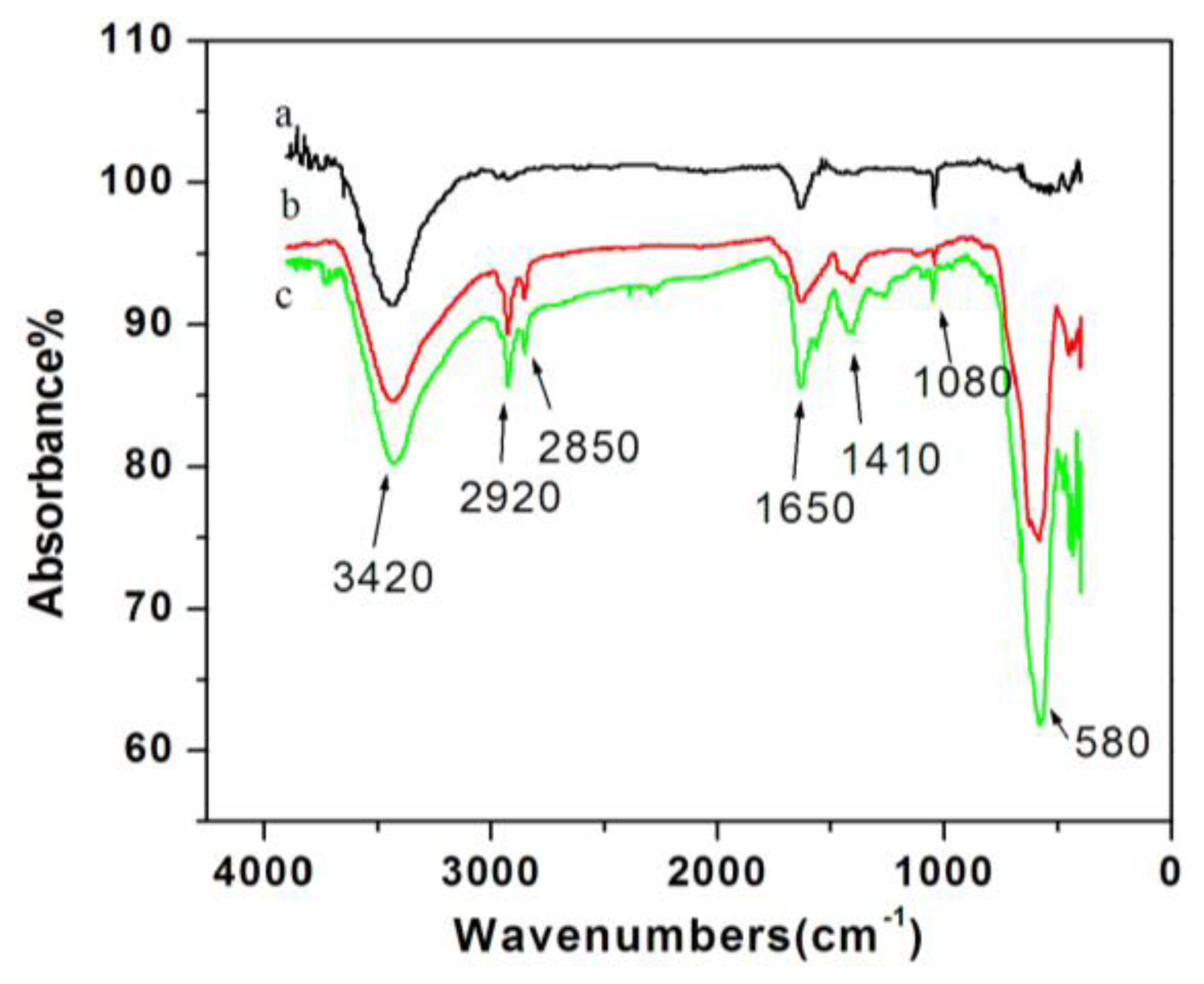
2.1.2. IR Spectra
2.1.3. Magnetic Properties
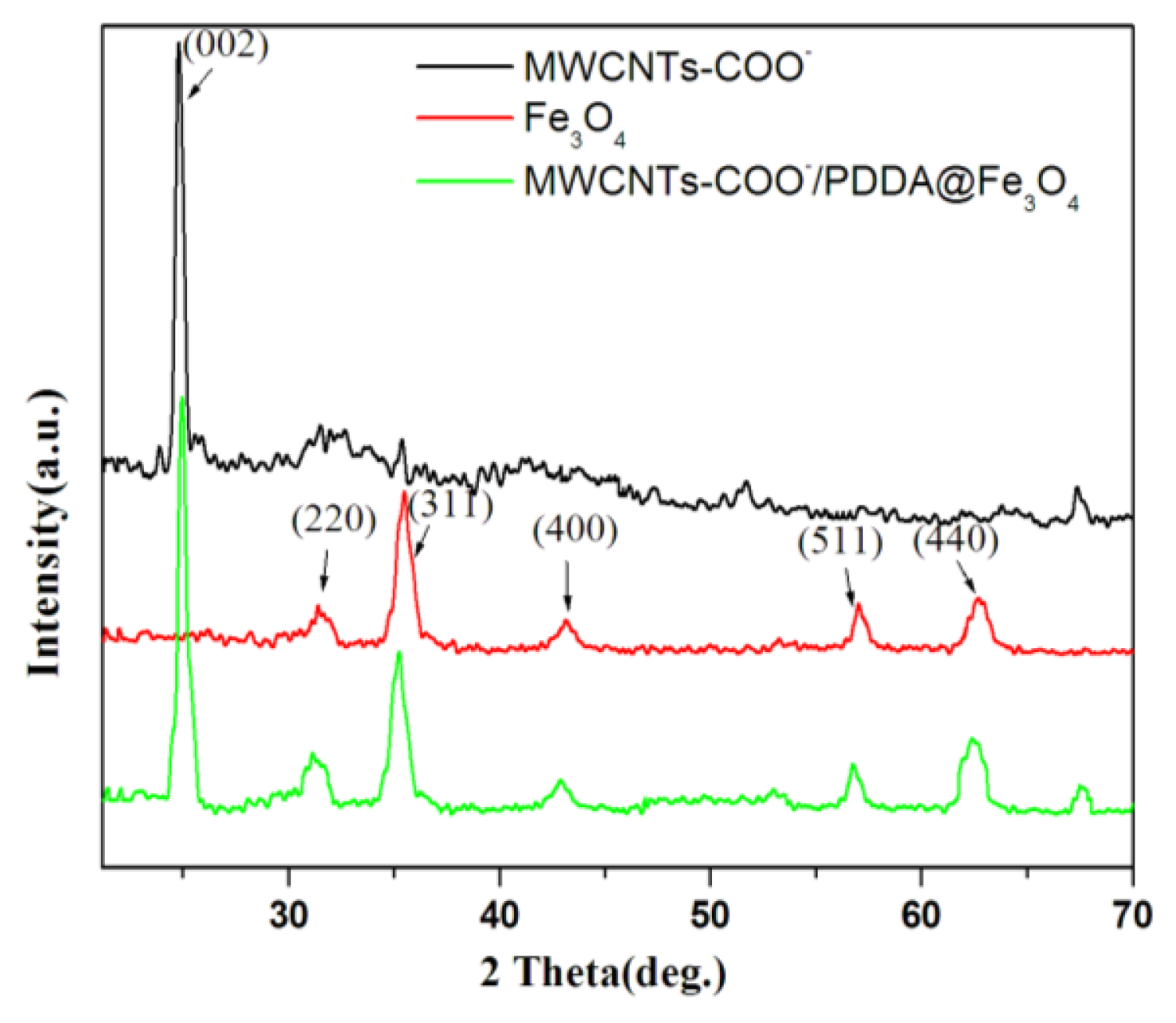
2.1.4. XRD
2.2. Optimization of Extraction Conditions
2.2.1. Magnetic Separation and Adsorption Equilibrium Time
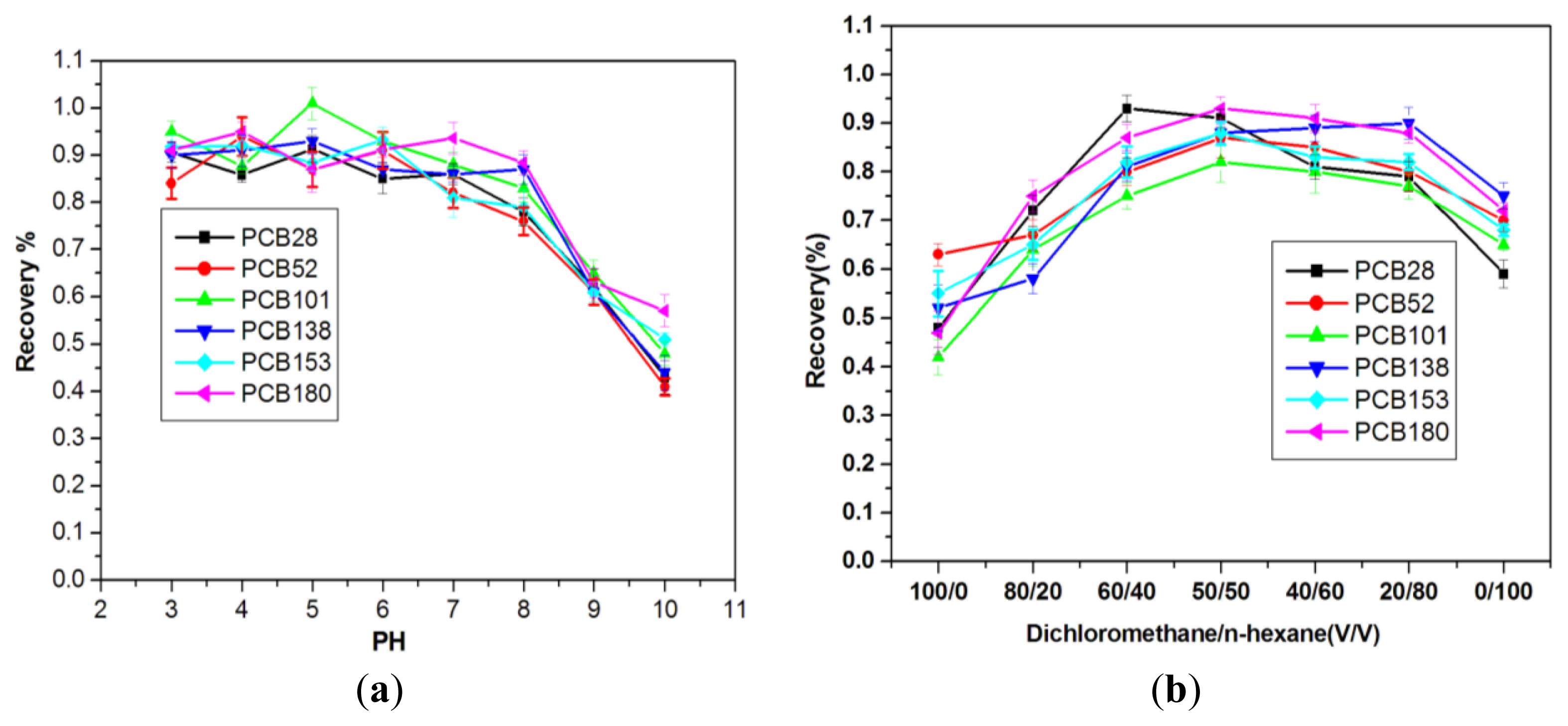
2.2.2. Effect of the Solution pH
2.2.3. Effect of Elution Solvent
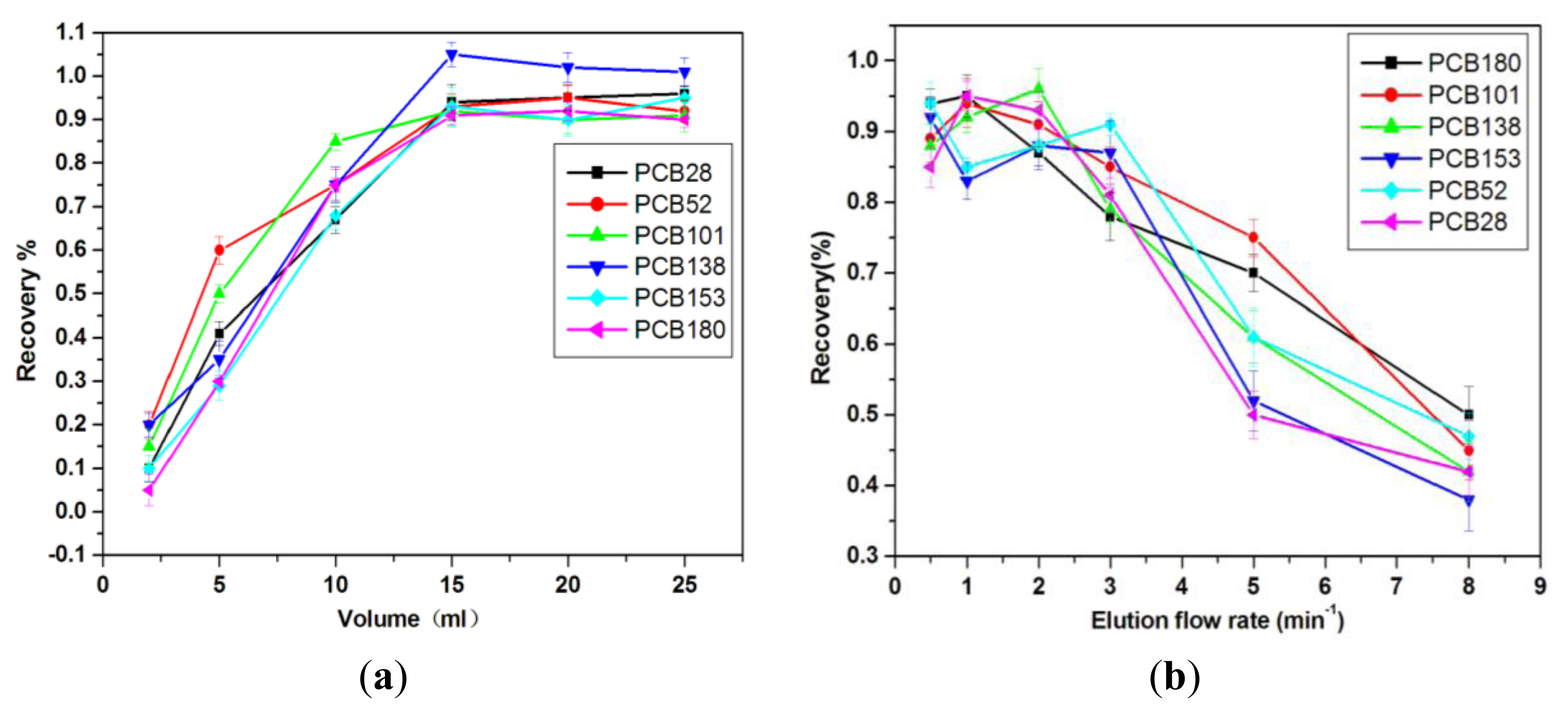
2.2.4. Volume of Elution Solvent
2.2.5. Effect of Desorption Flow Rate
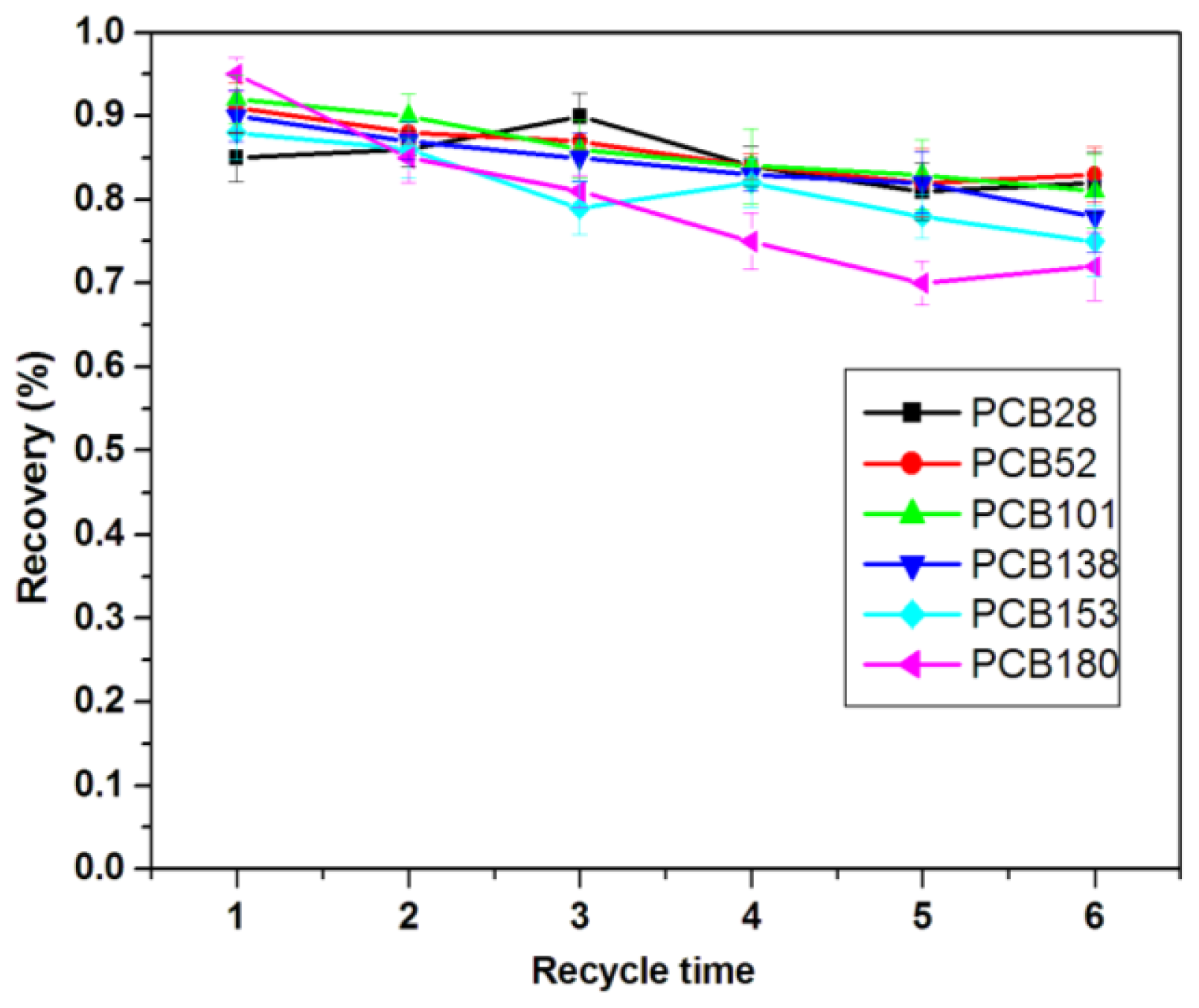
2.3. Reusability of the Sorbents
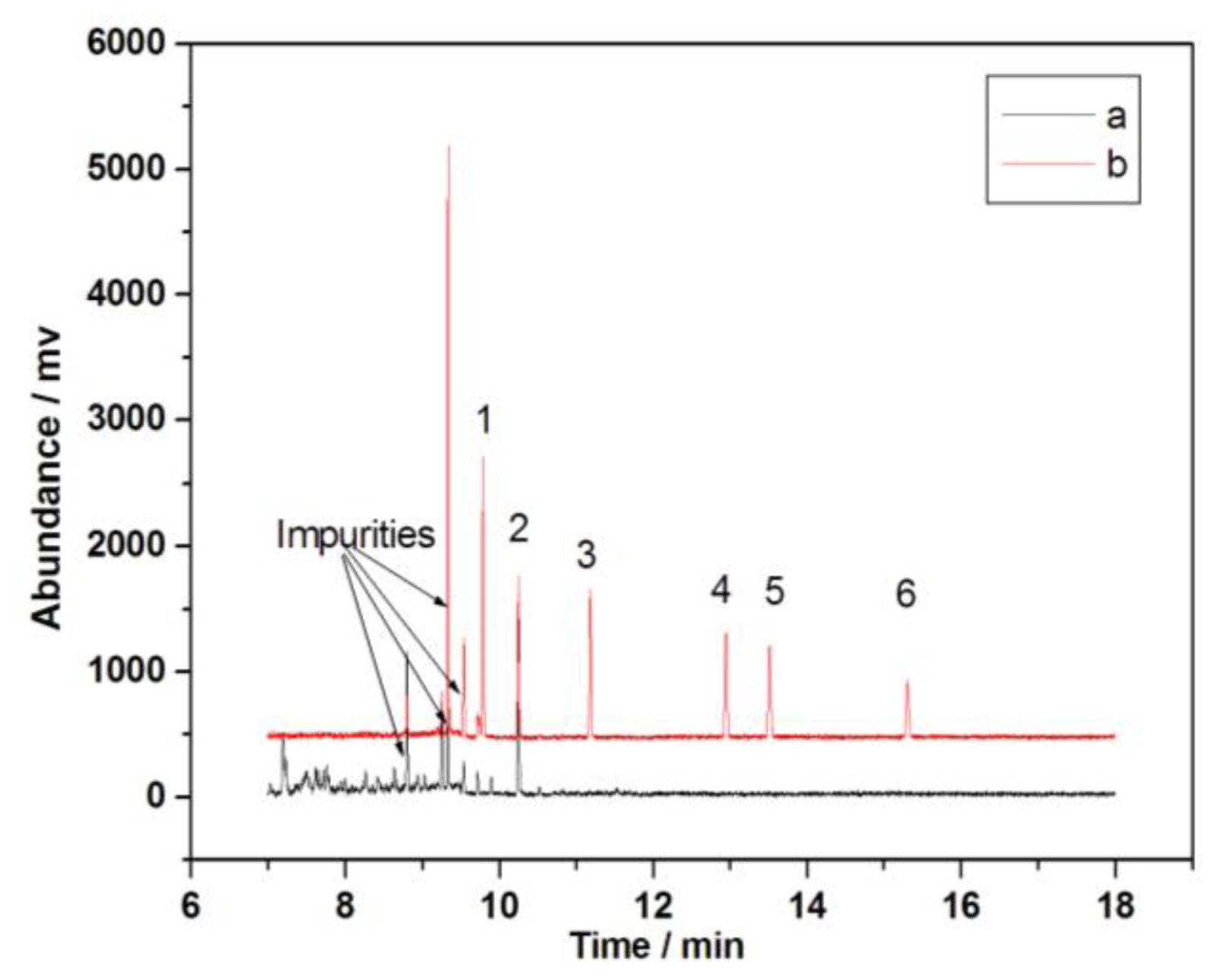
2.4. Method Validation
2.5. Determination of PCBs in Environmental Water Samples
2.6. Comparison of MWCNTs-COO-/PDDA@Fe3O4-SPE with Other Extraction Methods of PCBs
3. Experimental Section
3.1. Chemicals and Materials
3.2. Preparation of Fe3O4 Nanospheres
3.3. Synthesis of MWCNTs-COO−/PDDA@Fe3O4
3.4. Material Characterization
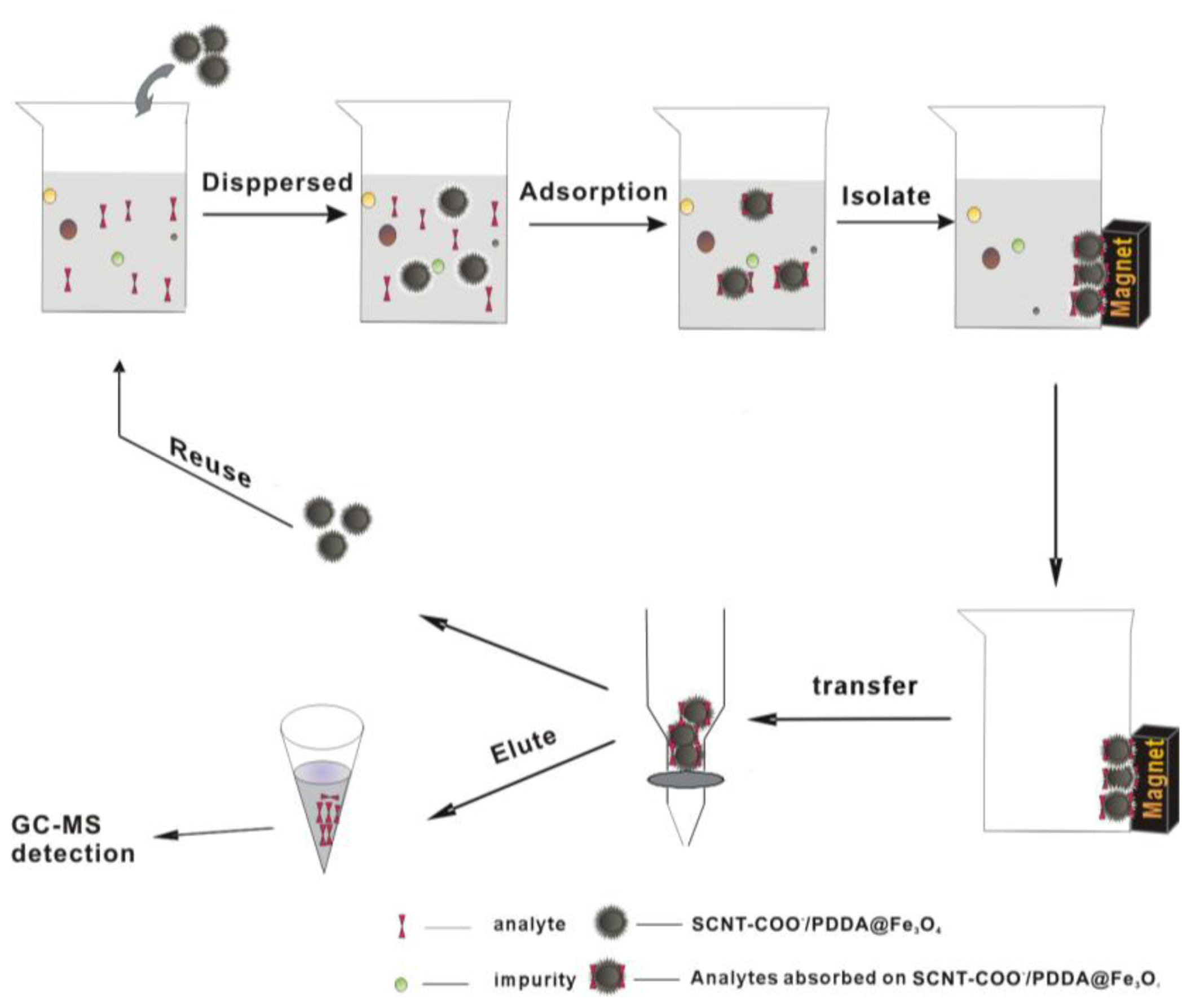
3.5. Magnetic SPE Procedure
3.6. GC-MS Analysis
3.7. Real Sample Preparation
4. Conclusions
Acknowledgments
References
- Liu, P.; Zhang, D.J.; Zhan, J.H. Investigation on the inclusions of PCB 52 with cyclodextrins by performing DFT calculations and molecular dynamics simulations. J. Phys. Chem. A 2010, 114, 13122–13128. [Google Scholar]
- Zhang, Z.L.; Stewart, M.R. Optimized determination of polybrominated diphenyl ethers and polychlorinated biphenyls in sheep serum by solid-phase extraction-gas chromatography-mass spectrometry. Talanta 2011, 84, 487–493. [Google Scholar]
- Li, M.T.; Meng, G.W.; Huang, Q.; Yin, Z.J.; Wu, M.Z.; Zhang, Z.; Kong, M.G. Prototype of a porous ZnO SPV-based sensor for PCB detection at room temperature under visible light illumination. Langmuir 2010, 26, 13703–13706. [Google Scholar]
- Maria, M.S.; Veronica, G. Detection and quantitative analysis of organochlorine compounds (PCBs and DDTs) in deep sea fish liver from Mediterranean Sea Perrone. Environ. Sci. Pollut. Res 2010, 17, 968–976. [Google Scholar]
- Zhou, Q.; Yang, Y.; Ni, J.; Li, Z.C.; Zhang, Z.J. Rapid detection of 2, 3, 3′ 4, 4′-pentachlorinated biphenyls by silver nanorods-enhanced Raman spectroscopy. Phys. E Low-Dimens. Syst. Nanostruct 2010, 42, 1717–1720. [Google Scholar]
- Wei, Y.; Kong, L.T.; Yang, R.; Wang, L.; Liu, J.H.; Huang, X.J. Electrochemical impedance determination of polychlorinated biphenyl using a pyrenecyclodextrin-decorated single-walled carbon nanotube hybrid. Chem. Commun 2011, 47, 5340–5342. [Google Scholar]
- Long, Q.R.; Yang, R.T. Carbon nanotubes as superior sorbent for dioxin removal. J. Am. Chem. Soc 2001, 123, 2058–2059. [Google Scholar]
- Gotovac, S.; Hattori, Y.; Noguchi, D.; Miyamoto, J.; Kanamaru, M.; Utsumi, S.; Kanoh, H.; Kanako, K. Phenanthrene adsorption from solution on single wall carbon nanotubes. J. Phys. Chem. B 2006, 110, 16219–16224. [Google Scholar]
- Yang, K.; Xing, B.S. Adsorption of organic compounds by carbon nanomaterials in aqueous phase: Polanyi theory and its application. Chem. Rev 2010, 110, 5989–6008. [Google Scholar]
- Zhou, Y.L.; Li, Y.Z. Studies of the interaction between poly(diallyldimethyl ammonium chloride) and DNA by spectroscopic methods. Colloids Surf. A 2004, 233, 129–135. [Google Scholar]
- Li, N.; Zhao, H.W.; Yuan, R.; Peng, K.F.; Chai, Y.Q. An amperometric immunosensor with a DNA polyion complex membrane/gold nanoparticles-backbone for antibody immobilisation. Electrochim. Acta 2008, 54, 235–241. [Google Scholar]
- Pascall, M.A.; Zabik, M.E.; Zabik, M.J.; Hernandez, R.J. Uptake of polychlorinated biphenyls (PCBs) from an aqueous medium by polyethylene, polyvinyl chloride, and polystyrene films. J. Agric. Food Chem 2005, 53, 164–169. [Google Scholar]
- Shao, D.D.; Hu, J.; Wang, X.K.; Nagatsu, M. Plasma induced grafting multiwall carbon nanotubes with chitosan for 4,4′-dichlorobiphenyl removal from aqueous solution. Chem. Eng. J 2011, 170, 498–504. [Google Scholar]
- Shao, D.D.; Hu, J.; Jiang, Z.Q.; Wang, X.K. Removal of 4,4′-dichlorinated biphenyl from aqueous solution using methyl methacrylate grafted multiwalled carbon nanotubes. Chemosphere 2011, 82, 751–758. [Google Scholar]
- Shao, D.D.; Sheng, G.D.; Chen, C.L.; Wang, X.K.; Masaaki, N. Removal of polychlorinated biphenyls from aqueous solutions using β-cyclodextrin grafted multiwalled carbon nanotubes. Chemosphere 2010, 79, 679–685. [Google Scholar]
- Yang, F.X.; Jin, S.W.; Meng, D.Y.; Xu, Y. Solid phase extraction with pyrenebutyric acid-bonded silica for analysis of polychlorinated biphenyls in sewage water by gas chromatography-mass spectrometry. Chemosphere 2010, 81, 1000–1005. [Google Scholar]
- Liu, L.; Cheng, J.; Matsadiq, G.; Li, J.K. Novel polymer monolith microextraction using a poly-(methyl methacrylate-co-ethylene dimethacrylate) monolith and its application to the determination of polychlorinated biphenyls in water samples. Chemosphere 2011, 83, 1307–1312. [Google Scholar]
- Dmitrovic, J.; Chan, S.C.; Chan, S.H.Y. Analysis of pesticides and PCB congeners in serum by GC/MS with SPE sample cleanup. Toxicol. Lett 2002, 134, 253–258. [Google Scholar]
- Čonka, K.; Drobná, B.; Conká, B.; Petrík, J. Simple solid-phase extraction method for determination of polychlorinated biphenyls and selected organochlorine pesticides in human serum. J. Chromatogr. A 2005, 1084, 33–38. [Google Scholar]
- Cheng, Y.X.; Liu, Y.J.; Huang, J.J.; Li, K.; Xian, Y.Z.; Zhang, W.; Jin, L.T. Amperometric tyrosinase biosensor based on Fe3O4 nanoparticles-coated carbon nanotubes nanocomposite for rapid detection of coliforms. Electrochim. Acta 2009, 54, 2588–2594. [Google Scholar]
- Lim, S.F.; Zheng, Y.M.; Zou, S.W.; Chen, J.P. Characterization of copper adsorption onto an alginate encapsulated magnetic sorbent by a combined FT-IR, XPS, and mathematical modeling study. Environ. Sci. Technol 2008, 42, 2551–2556. [Google Scholar]
- Ma, Z.Y.; Guan, Y.P.; Liu, H.Z. Synthesis and characterization of micron-sized monodisperse superparamagnetic polymer particles with amino groups. J. Polym. Sci. Part A Polym. Chem 2005, 43, 3433–3439. [Google Scholar]
- Lu, C.Y.; Chung, Y.L.; Chang, K.F. Adsorption of trihalomethanes from water with carbon nanotubes. Water Res 2005, 39, 1183–1189. [Google Scholar]
- Peng, X.J.; Li, Y.H.; Luan, Z.K.; Di, Z.C.; Wang, H.Y.; Tian, B.H.; Jia, Z.P. Adsorption of 1,2- dichlorobenzene from water to carbon nanotubes. Chem. Phys. Lett 2003, 376, 154–158. [Google Scholar]
- Zhou, Y.L.; Li, Y.Z. Studies of the interaction between poly(diallyldimethyl ammonium chloride) and DNA by spectroscopic methods. Colloids Surf. A 2004, 233, 129–135. [Google Scholar]



| Analytes | Calibration equations | Linear correlation coefficients (r2) | LOQ (ng·mL−1) | LOD (ng·mL−1) | RSD/% | |
|---|---|---|---|---|---|---|
| Low | High | |||||
| PCB28 | Y = 110.715x + 24.516 | 0.9988 | 0.10 | 0.031 | 8.5 | 5.2 |
| PCB52 | Y = 70.586x + 16.165 | 0.9998 | 0.17 | 0.052 | 7.8 | 3.5 |
| PCB101 | Y = 25.675x + 18.382 | 0.9992 | 0.23 | 0.069 | 8.3 | 4.2 |
| PCB138 | Y = 36.707x + 22.908 | 0.9993 | 0.19 | 0.058 | 9.4 | 6.7 |
| PCB153 | Y = 31.704x + 13.181 | 0.9994 | 0.14 | 0.042 | 5.8 | 3.8 |
| PCB180 | Y = 20.413x − 15.181 | 0.9985 | 0.16 | 0.047 | 7.2 | 4.1 |
| Analytes | Tap water | School water | River water | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Real | Added | RR 1 | Real | Added | RR 1 | Real | Added | RR 1 | |
| (ng·mL−1) | (ng·mL−1) | (%) | (ng·mL−1) | (ng·mL−1) | (%) | (ng·mL−1) | (ng·mL−1) | (%) | |
| PCB28 | ND 2 | 10 | 73.3% | 2.7 | 10 | 71.2% | 1.1 | 10 | 71.7% |
| 100 | 90.2% | 100 | 88.2% | 100 | 85.8% | ||||
| PCB52 | ND | 10 | 79.8% | 5.3 | 10 | 75.3% | 0.8 | 10 | 78.3% |
| 100 | 94.8% | 100 | 92.8% | 100 | 93.4% | ||||
| PCB101 | ND | 10 | 87.1% | ND | 10 | 77.9% | ND | 10 | 79.5% |
| 100 | 98.9% | 100 | 91.9% | 100 | 91.6% | ||||
| PCB138 | ND | 10 | 90.2% | ND | 10 | 89.2% | ND | 10 | 88.7% |
| 100 | 95.7% | 100 | 93.5% | 100 | 96.3% | ||||
| PCB153 | ND | 10 | 82.5% | ND | 10 | 81.6% | ND | 10 | 80.3% |
| 100 | 93.2% | 100 | 96.7% | 100 | 91.8% | ||||
| PCB180 | ND | 10 | 78.1% | ND | 10 | 79.1% | ND | 10 | 81.1% |
| 100 | 95.7% | 100 | 95.2% | 100 | 95.7% | ||||
| Methods | LODs (ng·mL−1) | RR (%) | Time of pretreatment (h) |
|---|---|---|---|
| MWCNTs-SPE | 0.028–0.051 | 68.2–76.8 | 6.4 |
| MWCNTs-COO−/PDDA-SPE | 0.034–0.063 | 78.4–98.6 | 6.4 |
| MWCNTs-COO−/PDDA@Fe3O4-SPE | 0.027–0.059 | 78.1–98.3 | 2.4 |
| C18-NH2-SPE | 0.007–0.023 | 75.9–105.4 | 6.0 |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zeng, S.; Cao, Y.; Sang, W.; Li, T.; Gan, N.; Zheng, L. Enrichment of Polychlorinated Biphenyls from Aqueous Solutions Using Fe3O4 Grafted Multiwalled Carbon Nanotubes with Poly Dimethyl Diallyl Ammonium Chloride. Int. J. Mol. Sci. 2012, 13, 6382-6398. https://doi.org/10.3390/ijms13056382
Zeng S, Cao Y, Sang W, Li T, Gan N, Zheng L. Enrichment of Polychlorinated Biphenyls from Aqueous Solutions Using Fe3O4 Grafted Multiwalled Carbon Nanotubes with Poly Dimethyl Diallyl Ammonium Chloride. International Journal of Molecular Sciences. 2012; 13(5):6382-6398. https://doi.org/10.3390/ijms13056382
Chicago/Turabian StyleZeng, Shaolin, Yuting Cao, Weiguo Sang, Tianhua Li, Ning Gan, and Lei Zheng. 2012. "Enrichment of Polychlorinated Biphenyls from Aqueous Solutions Using Fe3O4 Grafted Multiwalled Carbon Nanotubes with Poly Dimethyl Diallyl Ammonium Chloride" International Journal of Molecular Sciences 13, no. 5: 6382-6398. https://doi.org/10.3390/ijms13056382




