1. Introduction
Environmental stresses constitute the main factor depressing wheat production across the world, with wheat yield particularly suppressed by untimely temperature extremes and water deficit. This situation is emphasized by the 25 and 18% reduction in durum yields experienced across Southern Europe in 2003 and 2005, respectively, attributable to the high temperatures and water shortages experienced by crops in the field in this region in these years, compared with 2004 [
1]. Depending on the timing, duration and intensity of temperature extremes/drought, wheat yield maybe depressed by 10% to 90% [
2].
Climate change models project that summer rainfall will decrease substantially (in some areas up to 70%) across Southern and Central Europe [
3] and many—like that of Döll and Flörke [
4]—predict that summer droughts experienced during grain filling like those experienced in 2003 and 2005 will constitute a growing influence limiting European wheat production, especially at Southern latitudes and in parts of Eastern Europe. Temperatures during grain-filling already regularly exceed 30 °C, and the Intergovernmental Panel on Climate Change [
5] predicts the Mediterranean region as a whole will be likely adversely affected. Given current pressures on food security it is vital that breeders strive to maintain wheat yields by improving crop adaptability, stability and sustainability in a bid to combat a multitude of environmental challenges, not least water shortage. In this context, a better understanding of the genetic control of yield and the main traits that underlie the adaptive response of crops across a broad range of water availabilities is essential for more effective and targeted breeding activity [
6].
The development of molecular marker technologies to identify a particular chromosomal location of genes regulating specific traits has revolutionized our understanding of the genetic control of these traits. Quantitative trait loci (QTL) analysis and more recently association mapping (AM), exploiting linkage disequilibrium [
7], have become valuable tools in identifying the genetic control of yield and agronomically-relevant traits across a range of environments [
8–
11]. By performance comparison of the same genotypes across environments it is possible to identify those environments in which particular QTLs are expressed. Association analysis based on elite lines and breeding material provides a particularly useful tool to detect loci for traits with low heritability such as yield and its components [
12,
13]. However, other traits associated with wheat adaptability to soil water deficit (e.g., phenological and physiological traits, plant height, harvest index), with higher heritability than grain yield, can be also employed to improve yield in drought-stressed environments [
10,
14–
17].
The choice of germplasm is a key factor determining the resolution of AM. In order to detect more alleles, the germplasm selected should theoretically include all the genetic variation of a specific species because diverse germplasm includes more extensive recombination during its history and allows a high level of resolution [
18]. A species for which a core collection has been established would constitute the ideal material for AM [
19]. In this study we used a core collection of 96 winter wheat accessions sampled from 21 countries across five continents as the AM population. The objectives of this research were: (a) to undertake genetic analysis of the AM population; and (b) to identify chromosome regions that are highly correlated with yield and agronomic traits under contrasting environments (near optimal
versus rainfed and moderate-to-severe soil water deficit).
2. Results
2.1. Phenotypic Evaluation
The analysis of variance for each treatment revealed highly significant differences (
P < 0.01) in all 24 studied traits between the 96 genotypes (data not shown). The mean value across three years in each treatment is presented in
Table 1. Significant differences (
P < 0.05) between irrigated (IP) and drought (DP) were found for all traits except stem height (SH), peduncle length (PL), peduncle extrusion (PE), spike density (SD), spike index (SI) and flag leaf chlorophyll content at flowering date (CH1). The average reduction due to drought (sheltered plots) ranged from 1.4% for SI to 64.5% for grain yield (GY) (
Table 1). GY along with above-ground biomass per plant (BPP) (62.3% reduction) and kernel number per square meter (KN) (60.3%) were the traits most affected by drought stress. In addition, drought stress resulted in a significant increase in sterile spikelets per spike (SSS) (62%), and caused heading and flowering to be accelerated by 4–5 days across all genotypes. In most cases the values of the traits in the rainfed plots (RP) were intermediate between those of the IP and DP. The estimated variance of components of random effects which demonstrate the importance of accessions, environment and their interactions on each of 24 agronomic traits is presented in
Electronic Supplementary Table 1.
2.2. Heritability of the Traits
The estimates of overall heritability (combined across all treatments) and within each treatment are presented in
Table 2.
The overall heritability was high (>0.75) for days to heading (DTH), days to flowering (DTF), SH, PL, PE, spike length (SL), SD, fertile spikelets per spike (FSS), SSS, kernels per spike (KS), 1000 kernel weight (TKW), harvest index (HI), production per spike (PPS), flag leaf area (LA) and flag leaf width (LW), moderately high (0.50–0.75) for kernels per spikelets (KSL), SI, CH1, flag leaf chlorophyll content at grain filling (CH2) and GY and low for KN (0.48), BPP (0.44) and days between heading and flowering (HF) (0.37). The heritability in IP ranged from 0.46 (HF) to 0.93 (SL) and in DP from 0.26 (HF) to 0.89 (SD). In general, heritability values were slightly lower under drought than irrigated condition. However, the heritability estimate for GY under drought stress was rather lower than the same value obtained for irrigated plots (0.47 vs. 0.74, respectively). This is also true for BPP (0.39 vs. 0.64) and KN (0.38 vs. 0.61). Among main yield components (PT, KS and TKW) the highest heritability in DP was KS (0.78) while in IP was TKW (0.87).
2.3. Genetic Correlations
The genetic correlations between GY and other studied traits, including drought susceptibility index (DSI) and stress tolerance index (TOL), across all treatments and within each treatment are presented in
Table 3.
Across all treatments significant positive correlations ranged from 0.21 (KS) to 0.94 (TOL) and significant negative correlations ranged from −0.22 (HF) to −0.42 (SSS). In plats subject to drought, GY was highly associated with BPP (rg = 0.86), KN (rg = 0.81) and TOL (rg = 0.72). Higher grain yield in DP was moderately associated (0.30 < |rg| < 0.58) with KS, SH, CH2, PT, FSS, CH1, HI, PPS and TKW and, to lesser extent with lower SSS (rg = −0.30) and shorter DTH (rg = −0.28), DTF (rg = −0.28) and HF (rg = −0.25). Several traits such as FSS, KS and LA appeared to be significant for GY in DP but not in RP and/or IP. In contrast, DTH, DTF, PL and PE were significant for GY in RP and/or IP treatments, but not under drought. In IP and RP treatment the highest negative correlation was with SSS (rg = −0.42 and −0.41, respectively) while the highest positive correlation was with TOL (rg = 0.97 and 0.95, respectively). Note that for TOL positive correlations it should be interpreted as negative, because a higher TOL value indicates greater susceptibility, not a higher level of tolerance.
2.4. Model-Based Groups within AM Population and Linkage Disequilibrium
The genetic relationships among the accessions were investigated using a model-based Bayesian clustering method. A higher number (>4) of Bayesian clustering-based subgroups improved the overall fit to the model but led to a general decrease in the number of accessions assigned to a specific subgroup with high membership probability (>0.80), suggesting excessive partitioning of the diversity structure. The accessions included in each of the four subgroups (A–D) together with their corresponding membership probability estimates are reported in
Electronic Supplementary Table 2.
The effect of population structure on phenotypic traits was investigated by means of regression analysis (
Table 4).
Using the mean values across treatments, the greatest effect was observed for stem related traits SH, PL and PE (R2 = 57.8%, 53.3 % and 42.9%, respectively); a modest influence of population structure was detected for sterile spikelets per spike (SSS), CH1, CH2, HI and BPP, with R2 values ranging from 12.6% to 25.6%. Interestingly, the effect of population structure on GY and its components (PT, KN, TKW), including earliness (DTH and DTH), was low (R2 ranged from 1.2% to 6.4%). Similar results were also obtained when considering data of each treatment (data not presented).
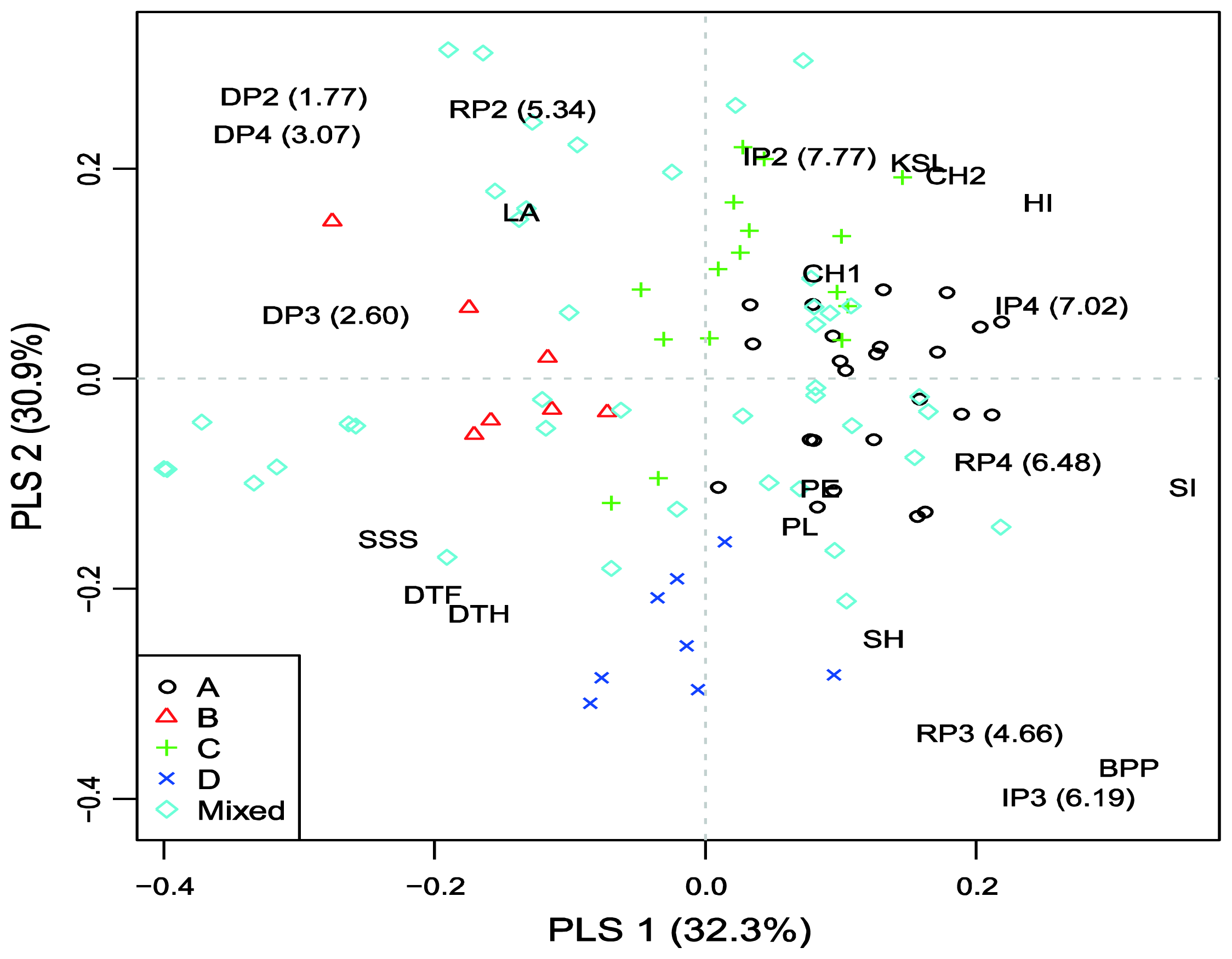
To explore model-based group × environment (treatment/year combination) interactions a partial least squares (PLS) regression approach was adopted using agronomic traits as explanatory variables. Only traits significantly affected (
P < 0.05) by population structure were included in the analysis (see
Table 4). The results of PLS regression for grain yield (denoted according to sub-populations assignments, environments and agronomic traits affecting interactions across the 96 accessions) is presented in
Figure 1.
Most accessions from sub-populations A and C are characterized by high chlorophyll content, KSL and HI, as well as low DSI and shorter vegetation, and had positive interaction with more favorable environments (IP2 and IP4). Accessions from sub-population B had low stature and large leaf area and showed positive interactions with drought stress environments. Accessions from sub-population D were late and tall and showed positive interactions in medium yielding environments (RP3 and IP3). Generally, the results from PLS regression for exploring specific interactions for grain yield in the trial indicated similar responses of accessions within model-based groups to environmental factors.
The extent of genome-wide LD among the entire set of accessions was evaluated through pairwise comparisons among 46 SSR loci yielding 1035 estimates. Among them, 178 (17.2%) showed significant association at a comparison-wise 0.01 level. The pairwise r2 estimates among 46 SSR loci ranged from 0 to 0.606, with a mean of 0.020 and a median of 0.015 (data not shown).
2.5. Marker-Trait Association
Using subpopulation assignments as covariates, a total of 517 significant marker–trait associations (MTAs) for 24 agronomic traits were detected in IP, RP and DP treatments at 41 loci. Fifty-eight percent of the associations were significant at the 0.05 level, 30% at the level 0.01 and 12% at the level 0.001. In the following special emphasis is given to highly significant results (with
P < 0.001); findings with lower significance are not reported. A total of 64 highly significant MTAs were identified in IP (24), RP (16) and DP (24) with 11 different SSR markers, and
R2 ranged from 11.4 to 42.2% (
Table 5). Thirty seven associations survived a Bonferroni correction for multiple testing (
Table 5). Combined across all treatments 28 significant (
P < 0.001) MTAs were identified with nine markers (
R2 ranged from 13.4 to 41.4%) while 15 associations survived Bonferroni correction (
Table 5).
DTF was involved in the highest number of MTAs within three treatments (9), followed by PT (7) and DTH, SL and FSS (all 6), while the fewest MTAs were associated with KN, GY and CH2 (all 2). No associated marker was found for HF, SH, PL, PE, SD, SI, TKW, BPP and CH1 within treatments. Of a total of 11 markers which showed highly significant effect on traits, eight (gwm99, gwm130, gwm369, gwm389, gwm458, gwm 540, psp3050 and psp3088) were associated with only one trait and therefore can be called trait-specific MTAs; the other three (gwm257, gwm484 and psp3200) were associated with more than one trait and can be referred as multi-trait MTAs. Marker gwm484 was found to be associated with 10 traits in three treatments and both drought indices (explained up to 42.2% of the phenotypic variation), while psp3200 was observed significant associated with nine traits in three treatments (explained up to 31.8% of the phenotypic variation). Marker gwm257 was associated with DTH, DTF and TOL (explained up to 16.3% of the phenotypic variation).
3. Discussion
We report a genetic analysis and a SSR-based association study of a breeder’s core collection of 96 winter wheat accessions of diverse origin, evaluated for a set of 24 agronomic traits and two drought indices over three years and three treatments (nine environments in toto). The 96 accessions were chosen from a wider collection of wheat genotypes from the, Novi Sad, Serbia with the aim to accumulate as much variation as possible within traits of importance for wheat breeding programs. Treatments allowed us to measure yield and other traits across a range of water availabilities; from 54.3 mm (drought plots in 2002–2003 season) to 418.2 mm (irrigated plots in 2003–2004 season). A rain-out shelter was effective in providing a drought treatment in each season; average yield reductions in comparison with the irrigated treatments were 77.2%, 58.0%, and 56.3% in seasons one to three, respectively.
Considerable phenotypic variation in GY [coefficient of variation (CV) = 24.8%)] and other measured traits [CV ranged from 6.2% (CH1) to 82.2 (SSS)] was observed across the 96 accessions. The total number of alleles per SSR locus (7.96) and estimated gene diversity (0.64) previously reported for this core collection [
20,
21] are comparable with that reported in the wheat elite germplasm collections that have been molecularly characterized so far [
11,
22–
24] and confirm the broad genetic base of the 96 accessions comprising the core collection investigated. Higher heritability estimates for GY under IP and RP than DP supported previous evidence that significant genotype by environment interactions exist under drought conditions [
10,
25,
26]. Low heritability for BPP and more predictable genotype by environment interaction and little effect of the environment on SH, HI and PPS were also suggested in earlier studies [
10,
27,
28].
The percentage of SSR loci pairs in LD observed in our mapping population (17%) was comparable with reports in maize (10%) [
29] and cotton (11–12%) [
30]. However, results from a SSR survey of other small grain cereals, including cultivated barley [
31], elite durum wheat germplasm [
32] and bread wheat collection [
22], showed much high levels of LD (45–82%). This difference could be due, in part, to the small number of used loci (46) in this study which may give limited genome coverage with only 2–3 markers per chromosome. The wide genetic diversity found among the 96 accessions [
20,
21] may have also contributed to lower levels of LD detected.
Among the defined sub-populations, the smallest sub-population (B) is the most easily distinguished (there were no overlapping points with other subgroups) (
Figure 1). The most distinct from B is sub-population D. Phenotypically, D accessions were significantly taller (stem height = 107.7
versus 54.7 cm) and flowered seven days later than B accessions. Sub-population B was dominated by accessions from Western Europe (GBR and FRA) while D centered on accessions of USA origin (
Electronic Supplementary Table 2). It might be surprising that relatively late and short genotypes from sub-population B showed positive interaction with drought conditions (
Figure 1). According to the conceptual model for drought resistance [
2], taller plants are considered to have better yield stability under adverse conditions, while shorter plants are better adapted to irrigated and high-input environments. Also, shortening of the growing season has been a very successful strategy when breeding wheat for variable rainfall conditions [
33]. Possibly an appropriate balance between water use during plant development and water use during grain fill existed in the genotypes from sub-population B. Hence, this sub-population may be of great interest to explore further for drought tolerance. The least differentiated pair was A–C. Sub-population C was the most diverse among sub-populations (spread over three quadrants) and consisted of genotypes from five continents. Sub-population A (mainly consisted of European accessions) exhibited positive interaction with irrigation treatments. This is not unexpected since many wheat-breeding programs in Europe are being carried out under non-limiting conditions.
We used sub-population assignments as covariates in applied AM for identification of genetic markers associated with grain yield and agronomic traits. Many studies in the past have demonstrated that all seven chromosome groups are involved in the genetic control of yield and yield-related traits in bread wheat [
34]. Due to the complex genetic control of grain yield, we found only two markers (gwm99 and gwm484) linked to genomic regions contributing to grain yield. Both were associated with GY only in well watered conditions. This is in keeping with Maccaferri
et al. [
11] who suggested that as the level of moisture stress increases, the power to detect the relevant loci for grain yield via AM decreases. They explained that under such conditions similar grain yield values can be attained by different genotypes through different adaptive strategies and corresponding gene networks, thus undermining the occurrence of significant marker–trait associations.
Markers gwm99 (on chromosome arm 1AL) and gwm484 (on chromosome arm 2DS) explained relatively high portion of the phenotypic variation of GY (14.4 and 22.3%, respectively). Furthermore, gwm484 effect for GY combined across all treatments was also rather high (19.8%). Such a high percentage of explained variations of GY by QTLs are not so common in association studies in cereals. Previous studies have also shown regions on chromosome arms 1AL and 2DS associated with grain yield in bread wheat. Using a meta-QTL analysis (MTQL), Zhang
et al. [
34] located three MTQLs for yield and yield-related traits on chromosome arm 1AL. One of them was positioned at 115 cM away from the terminal of 1AL which is ~11 cM from gwm99 based on the consensus map Ta-SSR-2004 [
35]. In addition, Zhang
et al. [
34] identified four MQTLs for yield and yield-related traits on chromosome arm 2DS, located between 17 and 50 cM. Marker gwm484 is positioned within this interval (41 cM) based on the consensus map Ta-SSR-2004. A significant QTL for grain yield at 35 cM on chromosome arm 2DS was located by Kumar
et al. [
36]. In our study loci gwm99 was associated only with GY in only IP and this can be referred to as trait- and treatment-specific MTA. On the other hand loci gwm484 (2DS) was associated with a number of other agronomic and developmental traits including DTF, PT, SL, FSS, SSS, HI, LA, LW and CH2. Significant genetic correlations were observed between GY and most of these traits suggesting pleiotropy and/or linked loci controling them.
The highly-significant association between allele size and different traits, including DTF, at the locus gwm484 is probably due to the proximity to
Ppd-D1 photoperiod sensitivity gene on 2DS [
37]. This gene is well known as having an influence on wheat yield through the optimization of flowering time [
38]. Recently the existence of at least six haplotypes of
Ppd-D1 has been reported [
39], with apparent adaptive significance, suggesting many more opportunities for fine-tuning genotypes to environmental conditions. It is typical that population structure coincides strongly with origin, adaptation and earliness, as was found in the present study. To minimize the effects that a high variability in phenology could have on GY under varying water regimes Maccaferri
et al. [
11] purposely kept the heading date within a narrow range when assembling a durum wheat mapping population. The correlation of earliness and GY was often significant suggesting that even relatively small range in heading date can significantly influence adaptation to the conditions commonly present in Mediterranean environments.
Besides gwm484 another microsatellite locus, psp3200 on chromosome arm 6DS, was associated with a large number of traits and involved in many MTAs, explained from 12.7% to 33.0% of phenotypic variation. As in the case of gwm484 locus most of detected MTAs for locus psp3200 survived a Bonferroni correction for multiple testing. The genomic region on the chromosome arm 6DS had a considerable effect on PT, SL, FSS, KS, PPS, LA, LW under optimum and water deficit conditions. Although no major flowering time effect has yet been reported on the short arm of chromosome 6D of wheat, the marker psp3200 was found to have highly significant allele associations with DTH and DTF under non-optimum conditions. This possibly new source of heading/flowering time gene(s) on chromosome arm 6DS could provide new opportunities for breeders to adapt their varieties better to the variable rainfed conditions, and under drought conditions early heading/flowering is generally advantageous. Recently, McIntyre
et al. [
10] reported a QTL for increased grain yield on chromosome 6B which co-located with a possible QTL for increased water-soluble carbohydrates (WSC) concentration in stem. A high stem carbohydrate concentration is considered to be a potentially useful trait for improving grain weight and yield in water-limited wheat production environments [
40,
41]. High WSC lines appeared to flower earlier than low WSC lines [
10,
42].
Although breeding for GY is the ultimate way to produce stress tolerant crop plants, due to the low heritability and complexity of grain yield, other traits such as yield components can be employed. Grain yield can be divided into a number of components including PT, KS and TKW. Several genes also control yield components; however, these components are usually less environmentally sensitive and have higher heritability than grain yield [
43]. This is confirmed in our study as PT, KS and TKW had higher heritability than GY in all three treatments as well as across all treatments. However there were no significant MTAs for TKW, while three and two markers were detected to be associated with PT and KS, respectively. Markers gwm484 (2DS) and psp3200 (6DS) were associated with PT in more than one treatment and both were related to several other traits. Markers gwm389 (3BS) was trait-specificand related to PT only in DP, explaining 14.4% of the phenotypic variation. The existence of QTLs for the tiller number was also observed on chromosomes 1A, 1B, 2B, 4B, 5B and 6A [
10,
28].
For KS one trait-specific and one multi-trait MTAs were located on 5BS (gwm540) and 6DS (psp3200), respectively. Both MTAs were detected under non-stress conditions and across all treatments. In the study by Quarrie
et al. [
8] the yield component most strongly associated with yield QTLs clusters was KS. Other studies also indicate a high correlation between GY and KS [
44,
45]. In our study KN (which combines PT and KS) was the trait most highly associated with GY across all treatments. Marker related to increased KN was detected on chromosome arm 2AL (psp3088) for IP and DP treatments as well as across all treatments. In a recent study, QTLs for increased KN were found on chromosomes 1A and 7A both in bread [
10] and durum wheat [
11].
In addition to gwm99 another two trait- and treatment-specific markers were identified on chromosomes 1DL and 7AS. Marker gwm458 (1DL) was related to HI while marker gwm130 was associated with PPS, both in DP, explaining 12.2% and 15.3% of the phenotypic variance, respectively. Both traits were highly correlated with GY in all treatments and can be effective in contributing GY in different conditions. Golobadi
et al. [
28] located MTA for HI detected only under drought stress on chromosome 2B, which explained 26.5% of the phenotypic variance, while the most significant marker for HI under normal conditions was located on 4B chromosome. Specific MTAs for HI in bread wheat were also reported by Neumann
et al. [
13] on 1A, 3A, 7A and 7B chromosomes. Besides the MTA related to PPS (gwm130), chromosome arm 7AS harbored MTA for LW (psp3200) also only under drought stress. The variation in flag leaf width was found to be related with major yield QTL on 7AL chromosome, expressed mainly under stressed conditions [
46]. The differences in width of flag leaves associated with the yield QTL on 7AL were due to variation in numbers of cell files across the leaf,
i.e., variation in cell division during leaf ontogeny. In recent studies QTLs for PPS were reported on several chromosomes including 7A short arm [
10,
13,
28].
Three MTAs were found for drought indices on chromosome arms 2BS (gwm257) and 2DS (gwm484), explaining a relatively high proportion of the observed phenotypic variation (14.9 to 25.3%) for drought tolerance. Not surprisingly, markers gwm257 and gwm484 were also associated with variation in DTH and/or DTF as they are known to map close to photoperiod sensitivity genes
Ppd-B1and
Ppd-D1, respectively [
47]. Genomic region on chromosome arms 2DS also exerted a considerable influence on SL, FSS, SSS, HI, LA, LW, CH2 (under drought stress) and PT, SL, FSS, SSS, HI, LA and GY (under optimum conditions). This finding concurs with that of Kumar
et al. [
36] who explained that a large proportion of phenotypic variation was not only consistent over environments, but was also pleiotropic, and/or coincident with QTLs for other traits. The coincidence of the gwm484 locus on chromosome 2DS with a number of traits across treatments may explain its effect on adaptation to different environmental conditions. The reports of QTLs for water-soluble carbohydrates [
14,
17] osmotic potential, chlorophyll content and flag leaf rolling [
48] in homoelogous Triticae group 2 chromosomes highlight the importance of this chromosome group for physiological responses to drought stress of wheat. In previous QTL mapping studies in biparental populations the QTLs controlling drought tolerance indices have been reported on chromosomes 5B for TOL [
49] and 4A, 4B, 5B, 7A for DSI [
44,
49], explaining between 13 and 41% of phenotypic variation.
Many of the reported MTAs within treatments were observed also in combined analysis across treatments. However, as a result of marker × environment interactions, not all these MTA are expressed in all environments (treatment/year combination). Nevertheless, the (pleiotropic) effect on spike morphology (SL and FSS) appears to be expressed in all environments on chromosome arm 6DS (psp3200), and the overall phenotypic variance for both trait explained by this marker was above 27%. This locus was also included in determination of KS and PPS in eight environments, suggesting a high value target for wheat improvement. The locus probably exerts its effect on PPS by controlling KS, which results from a combination of SL and FSS. With respect to spike morphology, another consistent MTA were related to gwm484 on chromosome arm 2DS and explained about 35 and 42% of SL and FSS phenotypic variance, respectively. Recently, a stable across environment QTL involved in the determination ear morphology/grain yield were detected on chromosome arm 4AL [
50].