Molecular Characterization of Cultivated Bromeliad Accessions with Inter-Simple Sequence Repeat (ISSR) Markers
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Materials
2.2. DNA Extraction
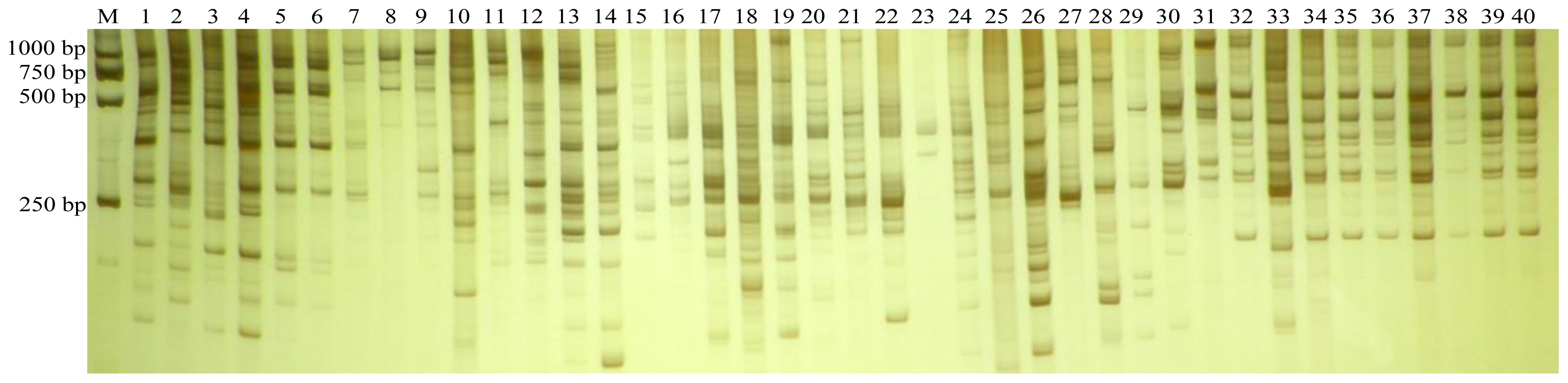
2.3. ISSR Analysis and Band Scoring
2.4. Data Analysis
3. Results
3.1. ISSR Polymorphism
3.2. Cluster Analysis and PCoA
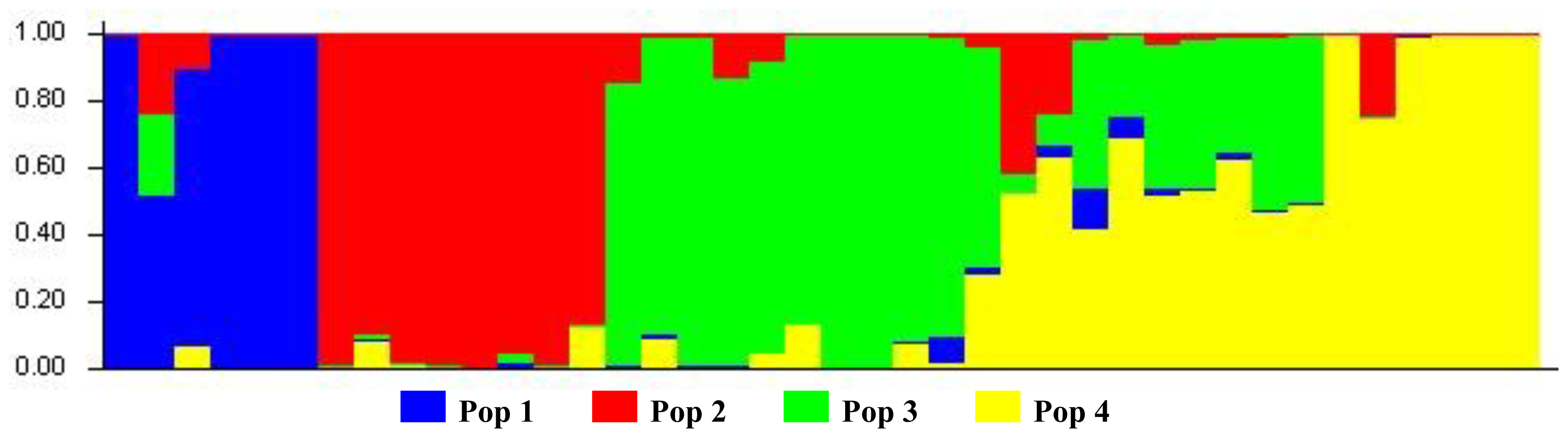
3.3. Bayesian-Modeled Population Structure
4. Discussion
5. Conclusions
Supplementary Material
ijms-13-06040-s001.xlsAcknowledgments
- Conflict of InterestThe authors declare no conflict of interest.
References
- Luther, H.E. An Alphabetical List of Bromeliad Binomials, 9th ed; The Bromeliad Society International, Inc: Orlando, FL, USA, 2004. [Google Scholar]
- Barfuss, M.H.J.; Samuel, R.; Till, W.; Stuessy, T.F. Phylogenetic relationships in subfamily Tillandsioideae (Bromeliaceae) based on DNA sequence data from seven plastid regions. Am. J. Bot 2005, 92, 337–351. [Google Scholar]
- Versieux, L.M.; Wendt, T. Bromeliaceae diversity and conservation in Minas Gerais state, Brazil. Biodivers. Conserv 2007, 16, 2989–3009. [Google Scholar]
- Izquierdo, L.Y.; Pinero, D. High genetic diversity in the only known population of Aechmea tuitensis (Bromeliaceae). Aust. J. Bot 2000, 48, 645–650. [Google Scholar]
- Sass, C.; Specht, C.D. Phylogenetic estimation of the core Bromeliads with an emphasis on the genus Aechmea (Bromeliaceae). Mol. Phylogenet. Evol 2010, 55, 559–571. [Google Scholar]
- Barbará, T.; Lexer, C.; Martinelli, G.; Mayo, S.; Fay, M.F.; Heuertz, M. Within-population spatial genetic structure in four naturally fragmented species of a neotropical inselberg radiation, Alcantarea imperialis, A. geniculata, A glaziouana, and A. regina (Bromeliaceae). Heredity 2008, 101, 285–296. [Google Scholar]
- Barbará, T.; Martinelli, G.; Palma-Silva, C.; Fay, M.F.; Mayo, S.; Lexer, C. Genetic relationships and variation in reproductive strategies in four closely related bromeliads adapted to neotropical “inselbergs”: Alcantarea glaziouana, A. regina, A. geniculata and A. imperialis (Bromeliaceae). Ann. Bot 2009, 103, 65–77. [Google Scholar]
- Gonzalez-Astorga, J.; Cruz-Angon, A.; Flores-Palacios, A.; Vovides, A.P. Diversity and genetic structure of the Mexican endemic epiphyte Tillandsia achyrostachys E. Morr. ex Baker var. achyrostachys (Bromeliaceae). Ann. Bot 2004, 94, 545–551. [Google Scholar]
- Palma-Silva, C.; Lexer, C.; Paggi, G.M.; Barbará, T.; Bered, F.; Bodanese-Zanettini, M.H. Range-wide patterns of nuclear and chloroplast DNA diversity in Vriesea gigantean (Bromeliaceae), a neotropical forest species. Heredity 2009, 103, 503–512. [Google Scholar]
- Paggi, G.M.; Sampaio, J.A.T.; Bruxel, M.; Zanella, C.M.; Göetze, M.; Büttow, M.V.; Palma-Silva, C.; Bered, F. Seed dispersal and population structure in Vriesea gigantea, a bromeliad from the Brazilian Atlantic Rainforest. Bot. J. Linn. Soc 2010, 164, 317–325. [Google Scholar]
- Cavallari, M.M.; Forzza, R.C.; Veasey, E.A.; Zucchi, M.I.; Oliveira, G.C.X. Genetic variation in three endangered species of Encholirium (Bromeliaceae) from Cadeia do Espinhaco, Brazil, detected using RAPD Markers. Biodivers. Conserv 2006, 15, 4357–4373. [Google Scholar]
- Sgorbati, S.; Labra, M.; Grugni, E.; Barcaccia, G.; Galasso, G.; Boni, U.; Mucciarelli, M.; Citterio, S.; Benavides, I.A.; Venero, G.L.; et al. A survey of genetic diversity and reproductive biology of Puya raimondii (Bromeliaceae), the endangered queen of the Andes. Plant Biol 2004, 6, 222–230. [Google Scholar]
- Zhang, F.; Wang, W.; Ge, Y.; Shen, X.; Tian, D.; Liu, J.; Liu, X.; Yu, X.; Zhang, Z. Genetic relatedness among Aechmea species and hybrids inferred from AFLP markers and pedigree data. Sci. Hortic 2012, 139, 39–45. [Google Scholar]
- Zietkiewicz, E.; Rafalski, A.; Labuda, D. Genome fingerprinting by simple sequence repeat (SSR)-anchored polymerase chain reaction amplification. Genomics 1994, 20, 176–183. [Google Scholar]
- Carlier, J.D.; Reis, A.; Duval, M.F.; d’Eeckenbrugge, G.C.; Leitão, J.M. Genetic maps of RAPD, AFLP and ISSR markers in Ananas bracteatus and A. comosus using the pseudo-testcross strategy. Plant Breed 2008, 123, 186–192. [Google Scholar]
- Carlier, J.D.; Sousa, N.H.; Santo, T.E.; d’Eeckenbrugge, G.C.; Leitão, J.M. A genetic map of pineapple (Ananas comosus (L.) Merr.) including SCAR, CAPS, SSR and EST-SSR markers. Mol. Breed 2012, 29, 245–260. [Google Scholar]
- Murray, M.G.; Thompson, W.F. Rapid isolation of high molecular weight plant DNA. Nucleic Acids Res 1980, 8, 4321–4325. [Google Scholar]
- Mortz, E.; Krogh, T.N.; Vorum, H.; Gorg, A. Improved silver staining protocols for high sensitivity protein identification using matrix-assisted desorption/ionization-time of flight analysis. Proteomics 2001, 1, 1359–1363. [Google Scholar]
- Liu, K.; Muse, S.V. PowerMarker: An integrated analysis environment for genetic marker analysis. Bioinformatics 2005, 21, 2128–2129. [Google Scholar]
- Rohlf, F.J. NTSYSpc Numerical Taxonomy and Multivariate Analysis System, version 2.2; Exeter Software: New York, NY, USA, 2005. [Google Scholar]
- Hampl, V.; Pavlicek, A.; Flegr, J. Construction and bootstrap of DNA fingerprinting-based phylogenetic trees with the freeware program FreeTree: Application to trichomonad parasites. Int. J. Syst. Evol. Microbiol 2001, 51, 731–735. [Google Scholar]
- Pritchard, J.; Stephens, M.; Donnelly, P. Inference of population structure using multilocus genotype data. Genetics 2000, 1555, 945–959. [Google Scholar]
- Hubisz, M.J.; Falush, D.; Stephens, M.; Pritchard, J.K. Inferring weak population structure with the assistance of sample group information. Mol. Ecol. Resour 2009, 9, 1322–1332. [Google Scholar]
- Pritchard, J.K.; Falus, D. Documentation for STRUCTURE Software, version 2.3; The University of Chicago Press: Chicago, IL, USA, 2009. [Google Scholar]
- Evanno, G.; Regnaut, S.; Goudet, J. Detecting the number of clusters of individuals using the software STRUCTURE: A simulation study. Mol. Ecol 2005, 14, 2611–2620. [Google Scholar]
- Sikdar, B.; Bhattacharya, M.; Mukherjee, A.; Banerjee, A.; Ghosh, E.; Ghosh, B.; Roy, S.C. Genetic diversity in important members Cucurbitaceae using isozyme, RAPD and ISSR markers. Biol. Plant 2010, 54, 135–140. [Google Scholar]
- Farsani, T.M.; Etemadi, N.; Sayed-Tabatabaei, B.E.; Talebi, M. Assessment of genetic diversity of Bermudagrass (Cynodon dactylon) using ISSR markers. Int. J. Mol. Sci 2012, 13, 383–392. [Google Scholar]
- Campa, A.; Trabanco, N.; Perez-Vega, E.; Rovira, M.; Ferreira, J.J. Genetic relationship between cultivated and wild hazelnuts (Corylus avellana L.) collected in northern Spain. Plant Breed 2011, 130, 360–366. [Google Scholar]
- Gaiero, P.; Mazzella, C.; Agostini, G.; Bertolazzi, S.; Rossato, M. Genetic diversity among endangered Uruguayan populations of Butia Becc. Species based on ISSR. Plant Syst. Evol 2011, 292, 105–116. [Google Scholar]
- Zhang, F.; Ge, Y.; Wang, W.; Shen, X.; Liu, X.; Liu, J.; Tian, D.; Yu, X. Genetic diversity and population structure of cultivated bromeliad accessions assessed by SRAP markers. Sci. Hortic 2012, 141, 1–6. [Google Scholar]
- Bianco, C.L.; Fernández, J.A.; Migliaro, D.; Crinò, P.; Egea-Gilabert, C. Identification of F1 hybrids of artichoke by ISSR markers and morphological analysis. Mol. Breed 2011, 27, 157–170. [Google Scholar]
- Goldman, J.J. The use of ISSR markers to identify Texas bluegrass interspecific hybrids. Plant Breed 2008, 127, 644–646. [Google Scholar]
- Carvalho, A.; Matos, M.; Lima-Brito, J.; Guedes-Pinto, H.; Benito, C. DNA fingerprint of F1 interspecific hybrids from the Triticeae tribe using ISSRs. Euphytica 2005, 143, 93–99. [Google Scholar]
- Poczai, P.; Mátyás, K.K.; Szabó, I.; Varga, I.; Hyvönen, J.; Cernák, I.; Gorji, A.M.; Decsi, K.; Taller, J. Genetic variability of Thermal Nymphaea (Nymphaeaceae) population based on ISSR markers: Implications on relationships, hybridization, and conservation. Plant Mol. Biol. Rep 2011, 29, 906–918. [Google Scholar]
- Fang, D.Q.; Roose, M.L. Identification of closely related cultivars with inter-simple sequence repeat markers. Theor. Appl. Genet 1997, 95, 408–417. [Google Scholar]
- Palai, S.K.; Rout, G.R. Characterization of new variety of Chrysanthemum by using ISSR markers. Hortic. Bras 2011, 29, 613–617. [Google Scholar]
- Sabir, A.; Kafkas, S.; Tangolar, S.; Büyükalaca, S. Genetic relationship of grap cultivar by ISSR (inter-simple sequence repeats) markers. Eur. J. Hort. Sci 2008, 73, 84–88. [Google Scholar]
- Sarthou, C.; Samadi, S.; Boisselier-Dubayle, M.C. Genetic structure of the saxicole Pitcairnia geyskesii (Bromeliaceae) on inselbergs in French Guiana. Am. J. Bot 2001, 88, 861–868. [Google Scholar]
- Boisselier-Dubayle, M.C.; Leblois, R.; Samadi, S.; Lambourdic, J.; Sarthou, C. Genetic structure of the xerophilous bromliad Pitcairnia geyskesii on inselbergs in French Guiana—A test of the forest refuge hypothesis. Ecography 2010, 33, 175–184. [Google Scholar]
- Bromeliad Encyclopedia. Florida Council of Bromeliad Societies. Available online: http://fcbs.org accessed on 20 January 2012.
- Gross, C.L.; Nelson, P.A.; Haddadchi, A.; Fatemi, M. Somatic mutations contribute to genotypic diversity in sterile and fertile populations of the threatened shrub, Grevillea rhizomatosa (Proteaceae). Ann. Bot 2012, 109, 331–342. [Google Scholar]
- Meneghetti, S.; Costacurta, A.; Morreale, G.; Calò, A. Study of intra-varietal genetic variability in grapevine cultivars by PCR-derived molecular markers and correlations with the geographic origins. Mol. Biotechnol 2012, 50, 72–85. [Google Scholar]
- Meneghetti, S.; Poljuha, D.; Frare, E.; Costacurta, A.; Morreale, G.; Bavaresco, L.; Calò, A. Inter- and intra-varietal genetic variability in Malvasia cultivars. Mol. Biotechnol 2012, 50, 189–199. [Google Scholar]



| No. | Sample Code | Name | Botanical Classification |
|---|---|---|---|
| 1 | N3 | “Picolo” | Neoregelia |
| 2 | N22 | “Herb Hill” | N. ampullacea |
| 3 | N107 | “Orange” | Neoregelia |
| 4 | N156 | “Burnsie”s Spiral” | N. carolinae f. tricolor |
| 5 | N4 | “Flandria” | N. carolinae |
| 6 | N4-1 | Sport of “Flandria” | N. carolinae |
| 7 | G1 | “Diana” | Guzmania |
| 8 | G8 | “Chloe” | Guzmania |
| 9 | G26 | “Scarlet Star” | Guzmania |
| 10 | G35 | “Torch” | G. lingulata × G. conifera |
| 11 | G40 | “Clementina” | G. lingulata var. minor |
| 12 | G47 | “Pink” | Guzmania |
| 13 | G54 | “Red” | Guzmania |
| 14 | G63 | “Caitlin” | Guzmania |
| 15 | V5 | “Carly” | Vriesea |
| 16 | V6 | “Barbara” | Vriesea |
| 17 | V7 | “Kallisto” | Vriesea |
| 18 | V8 | “Tiffany” | Vriesea |
| 19 | V9 | “Shannon” | Vriesea |
| 20 | V11 | “Kida” | Vriesea |
| 21 | V12 | “Christiane” | Vriesea |
| 22 | V13 | “Darmo” | Vriesea |
| 23 | V21 | “Annie” | Vriesea |
| 24 | V48 | “Select Red” | Vriesea |
| 25 | A5 | Unknown | Aechmea |
| 26 | A6 | “Fireman Sam” | A. dealbata |
| 27 | A36 | “Bert” | A. orlandiana × A. fosteriana |
| 28 | A38 | “Friederike” | A. chantinii × A. fasciata |
| 29 | A39 | “Marqarita L.” | A. dealbata × A. fasciata |
| 30 | A42 | Selected hybrid | A. distichantha × A. caudata |
| 31 | A50 | Species | A. gamosepala |
| 32 | A51 | “Ilha Grande” | A. gracilis |
| 33 | A54 | “Lucky Stripes” | A. gamosepala |
| 34 | A65 | “Red flamingo” | A. fendler × A. chantinii |
| 35 | S1 | Selected hybrid | A50 × A. recurvata var. recurvata |
| 36 | S2 | Selected hybrid | A50 × A. recurvata var. recurvata |
| 37 | S3 | Selected hybrid | A50 × A. recurvata var. recurvata |
| 38 | S4 | Selected hybrid | A50 × A. recurvata var. recurvata |
| 39 | S5 | Selected hybrid | A50 × A. recurvata var. recurvata |
| 40 | S6 | Selected hybrid | A50 × A. recurvata var. recurvata |
| Primer | Sequence (5′ → 3′) | TB | PB | PPB | PIC |
|---|---|---|---|---|---|
| U808 | (AG)8C | 29 | 23 | 79.31 | 0.31 |
| U809 | (AG)8G | 24 | 19 | 79.17 | 0.26 |
| U810 | (GA)8T | 31 | 26 | 83.87 | 0.32 |
| U812 | (GA)8A | 18 | 17 | 94.44 | 0.34 |
| U815 | (CT)8G | 36 | 29 | 80.56 | 0.26 |
| U834 | (AG)8YT | 23 | 18 | 78.26 | 0.27 |
| U840 | (GA)8YT | 39 | 33 | 84.62 | 0.27 |
| U844 | (CT)8RC | 25 | 21 | 84.00 | 0.35 |
| U848 | (CA)8RG | 26 | 25 | 96.15 | 0.32 |
| U873 | (GACA)4 | 36 | 34 | 94.44 | 0.28 |
| U880 | G(GA)2G(GA)2G(GA)2 | 28 | 19 | 67.86 | 0.37 |
| U892 | TAGATCTGATATCTGAATTCCC | 27 | 23 | 85.19 | 0.28 |
| Average | - | 28.50 | 23.92 | 83.92 | 0.30 |
| Total | - | 342 | 287 | - | - |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhang, F.; Ge, Y.; Wang, W.; Yu, X.; Shen, X.; Liu, J.; Liu, X.; Tian, D.; Shen, F.; Yu, Y. Molecular Characterization of Cultivated Bromeliad Accessions with Inter-Simple Sequence Repeat (ISSR) Markers. Int. J. Mol. Sci. 2012, 13, 6040-6052. https://doi.org/10.3390/ijms13056040
Zhang F, Ge Y, Wang W, Yu X, Shen X, Liu J, Liu X, Tian D, Shen F, Yu Y. Molecular Characterization of Cultivated Bromeliad Accessions with Inter-Simple Sequence Repeat (ISSR) Markers. International Journal of Molecular Sciences. 2012; 13(5):6040-6052. https://doi.org/10.3390/ijms13056040
Chicago/Turabian StyleZhang, Fei, Yaying Ge, Weiyong Wang, Xinying Yu, Xiaolan Shen, Jianxin Liu, Xiaojing Liu, Danqing Tian, Fuquan Shen, and Yongming Yu. 2012. "Molecular Characterization of Cultivated Bromeliad Accessions with Inter-Simple Sequence Repeat (ISSR) Markers" International Journal of Molecular Sciences 13, no. 5: 6040-6052. https://doi.org/10.3390/ijms13056040




