Biological Properties of Acidic Cosmetic Water from Seawater
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents and Materials
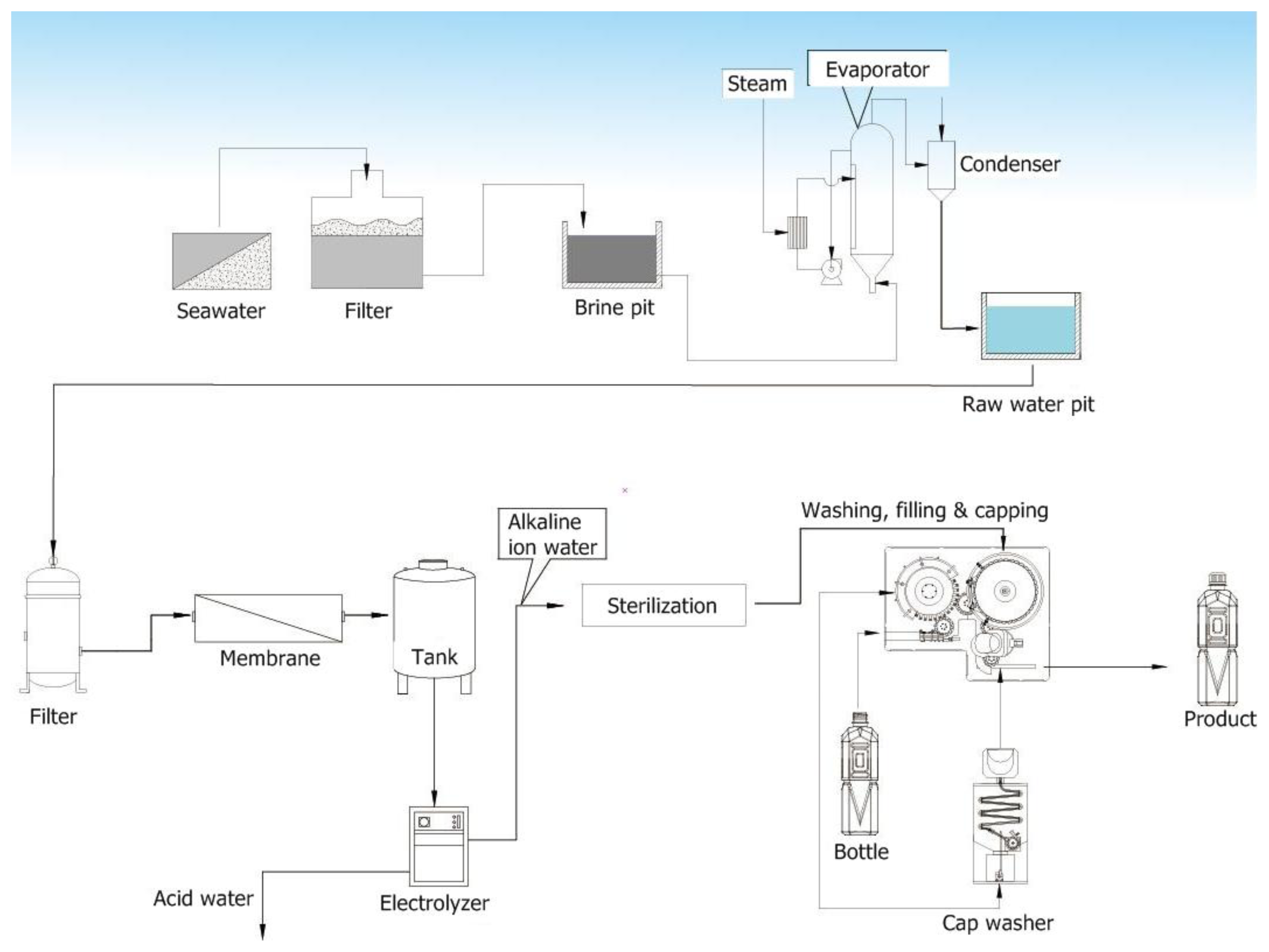
2.2. Preparation of ACW from Seawater
2.3. Determination of Antimicrobial Activity
2.4. Cell Cultures
2.5. Cytotoxicity Assay—Methylthiazoleterazolium (MTT) Assay
2.6. The Inflammation Evaluation
2.7. The Measurement of De-Granulation
2.8. Determination of Anti-Oxidative Properties of ACW in Vitro
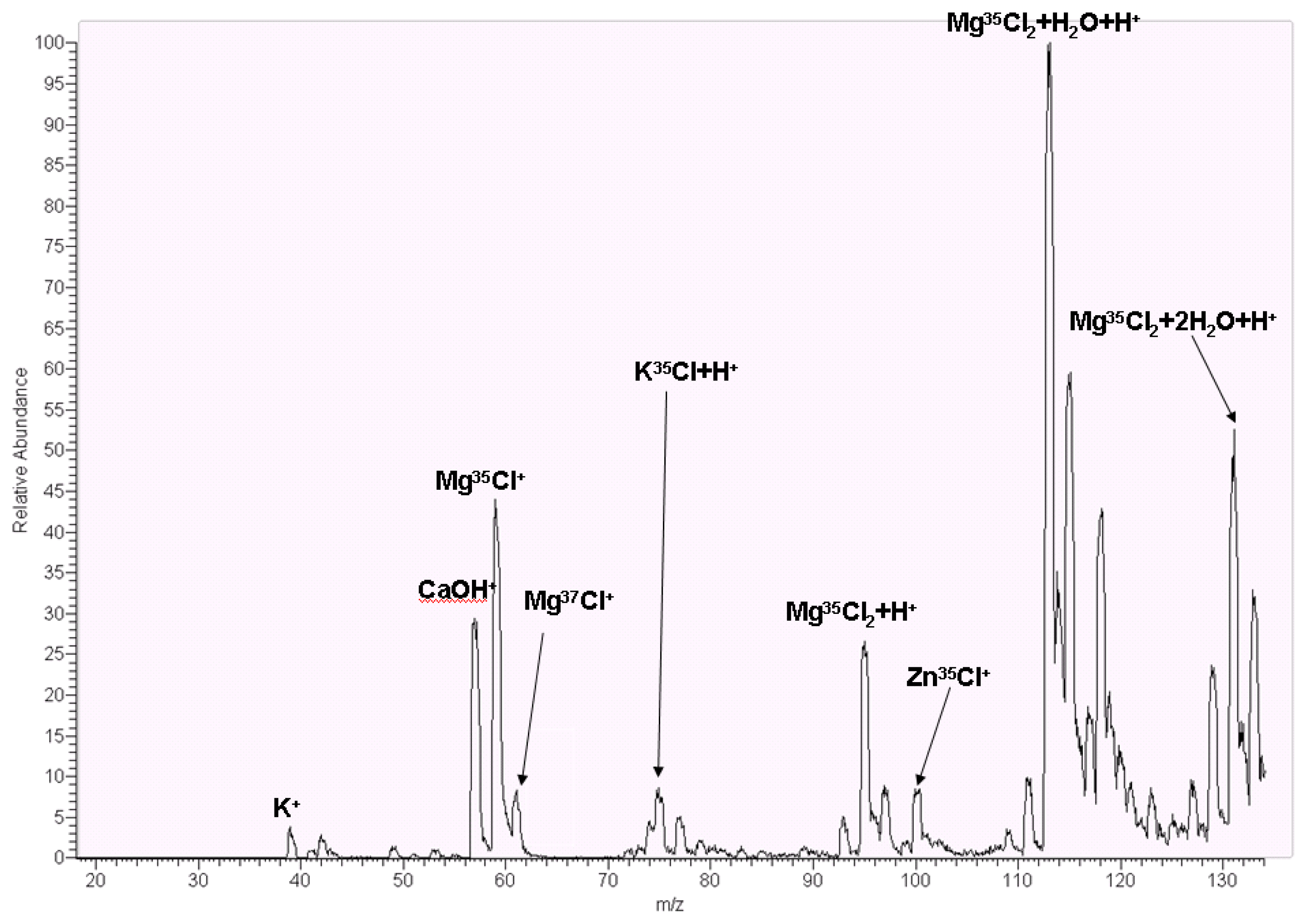
2.9. Mass Spectrometry Analysis
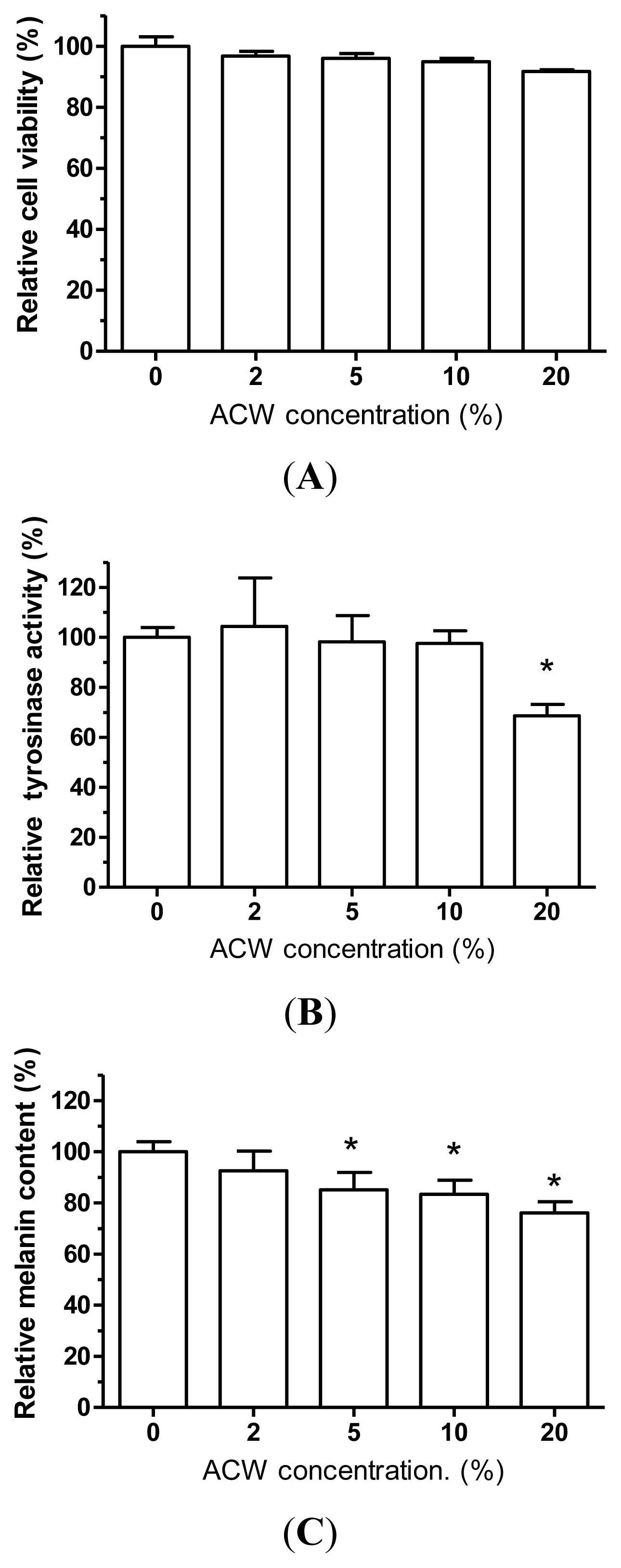
2.10. Cellular Tyrosinase Activity Assay and Melanin Quantification Examination
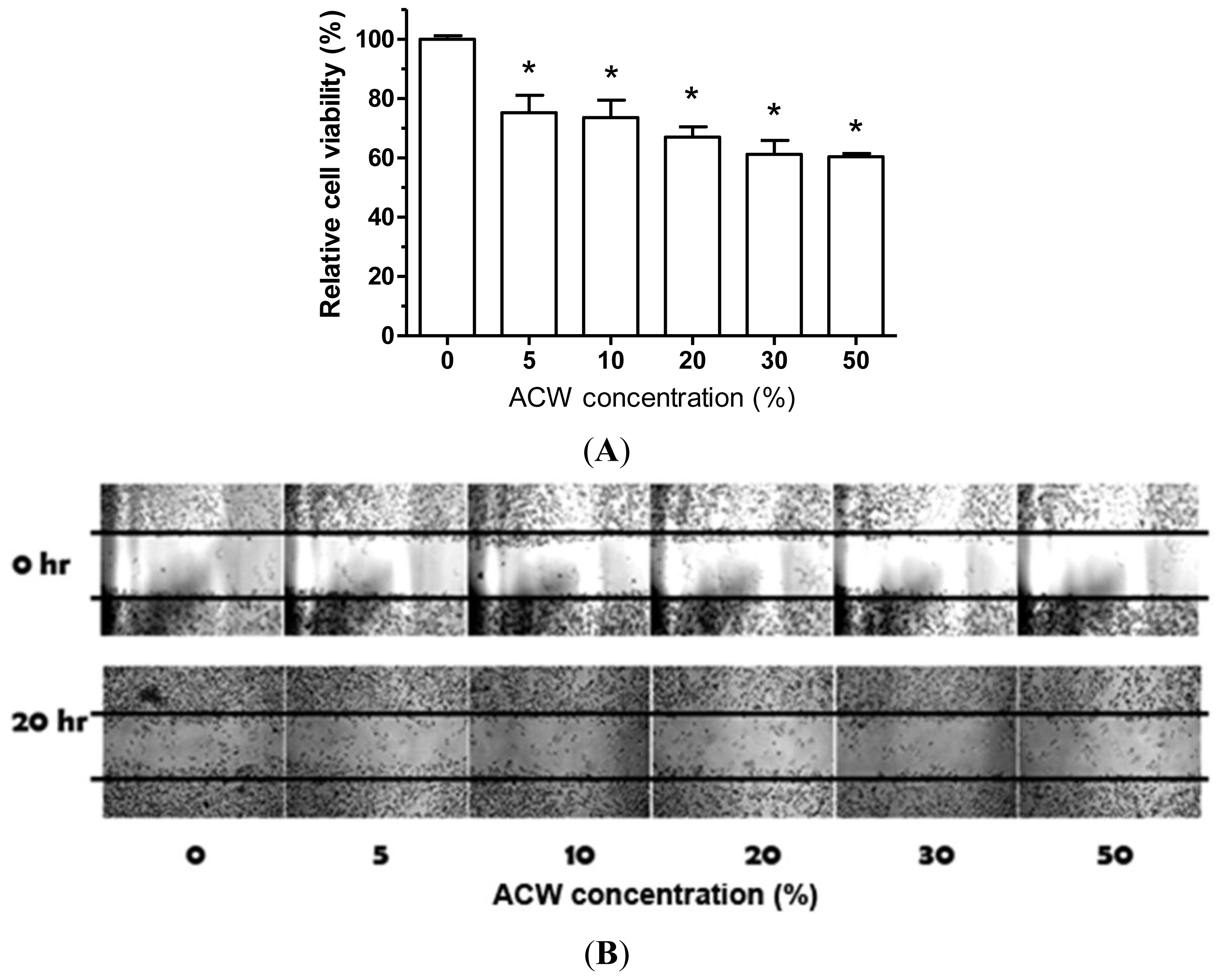
2.11. The Assessment of Cell Migration
2.12. Statistical Analysis
3. Results and Discussion
3.1. Antimicrobial Activity against E. coli and S. aureus
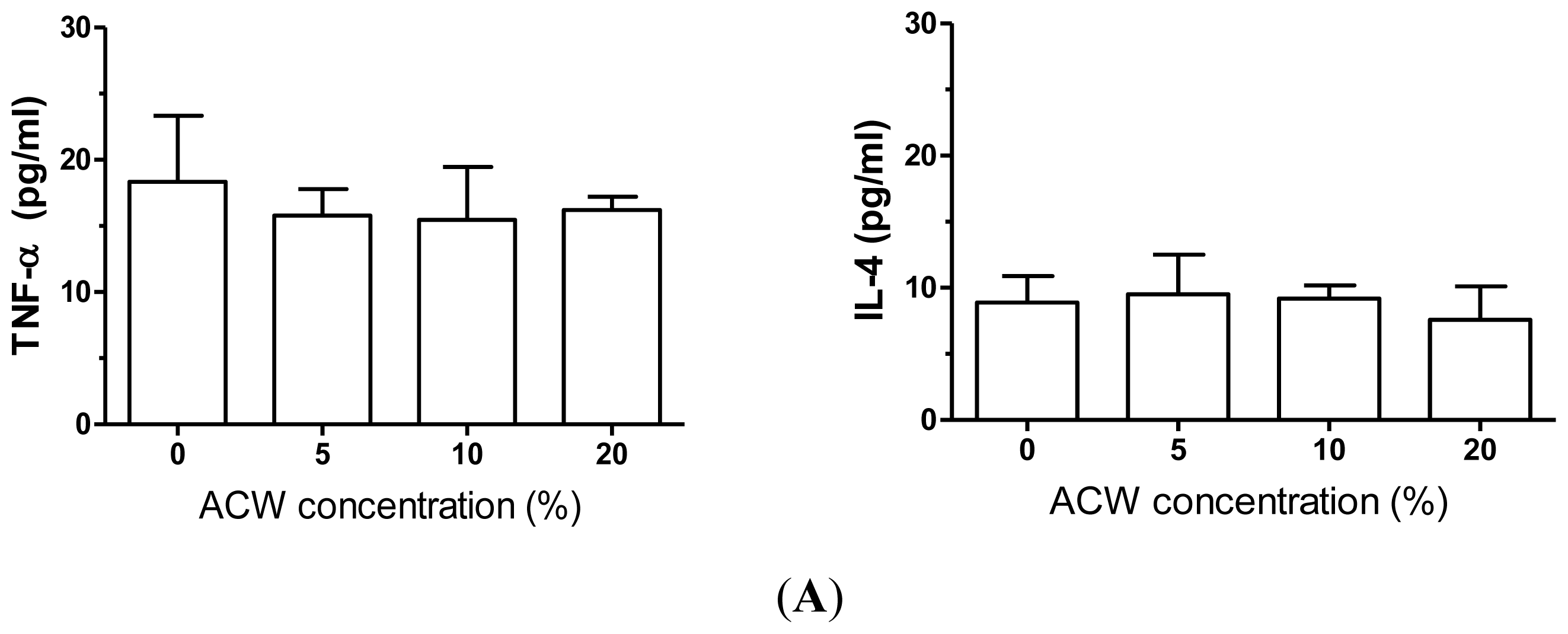
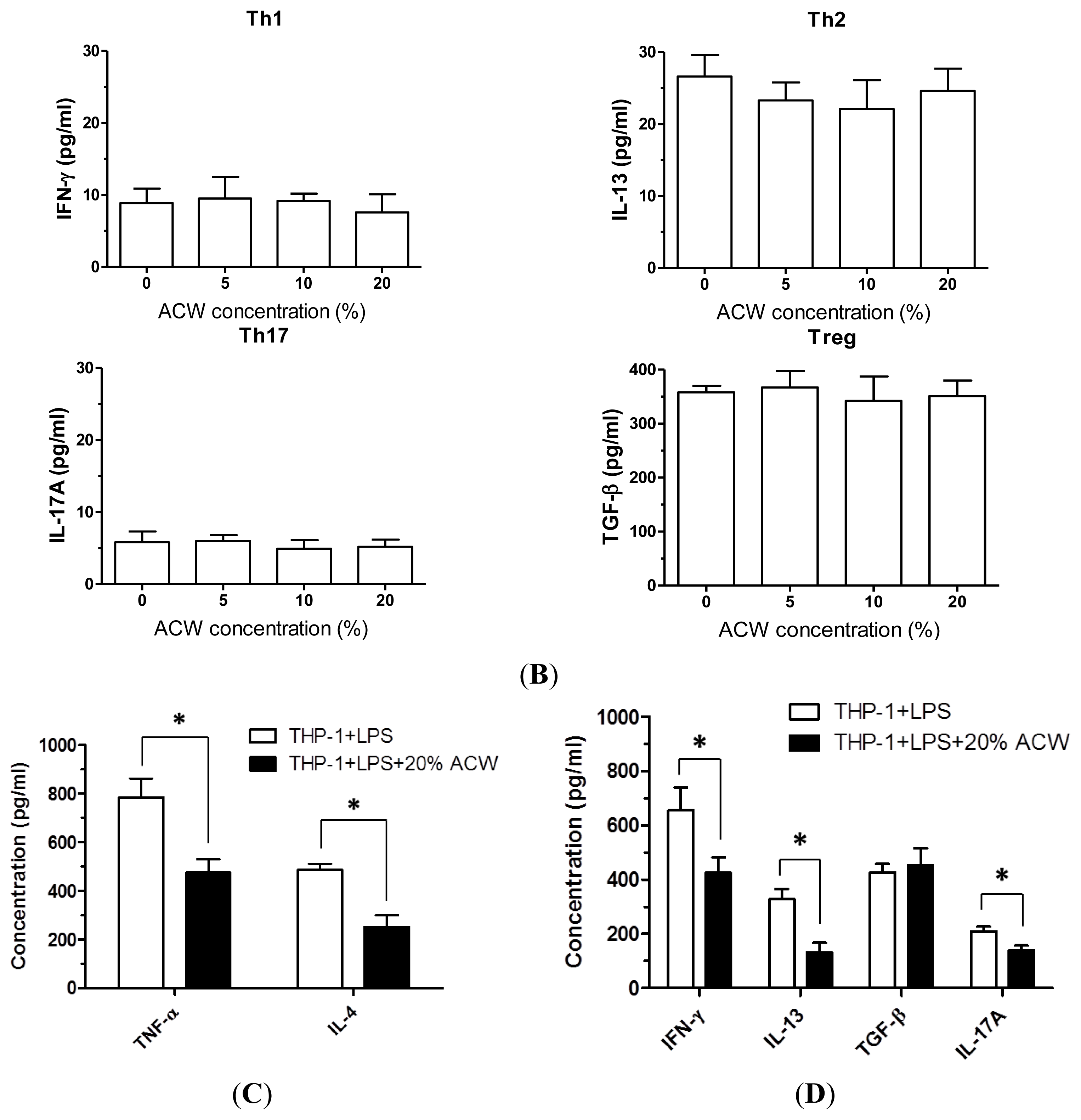
3.2. Anti-Inflammation Measurements of ACW
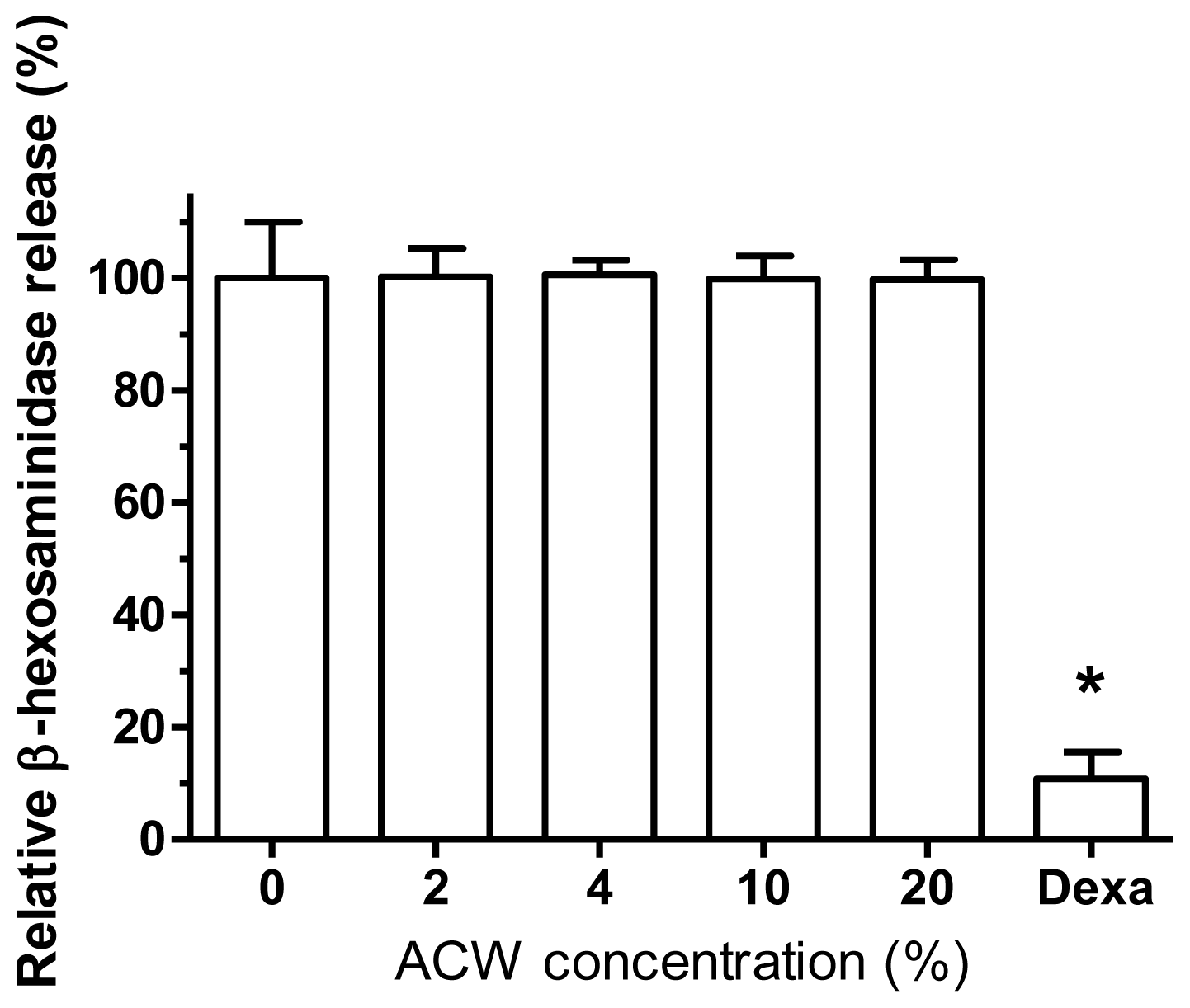
3.3. Allergy-Free Test on ACW
3.4. Antioxidant Capacity of ACW
3.5. Mass Spectrometry Analysis
3.6. Skin-Whitening Examination on ACW
3.7. ACW Attenuated A375.S2 Melanoma Proliferation and Migration
4. Conclusion
Acknowledgements
References
- Shimizu, Y.; Furusawa, T. Killing action of virus, bacteria and fungus by oxidative potential water induced by electrolysis. J. Dent. Med 1992, 36, 1055–1060. [Google Scholar]
- Mori, Y.; Komatsu, S.; Hata, Y. Toxicity of electrolyzed strong acid aqueous solution-subacute toxicity test and effect on oral tissue in rats. Odontology 1997, 84, 619–626. [Google Scholar]
- Kohno, S.; Kawata, T.; Kaku, M.; Fuita, T.; Tsutsui, K.; Ohtani, J.; Tenjo, K.; Motokawa, M.; Tohma, Y.; Shigekawa, M.; et al. Bactericidal effects of acidic electrolyzed water on the dental unit waterline. Jpn. J. Infect. Dis 2004, 57, 52–54. [Google Scholar]
- Shimmura, S.; Matsumoto, K.; Yaguchi, H.; Okuda, T.; Miyajima, S.; Negi, A.; Shimazaki, J.; Tsubot, K. Acidic electrolysed water in the disinfection of the ocular surface. Exp. Eye Res 2000, 70, 1–6. [Google Scholar]
- Harrington, L.E.; Hatton, R.D.; Mangan, P.R.; Turner, H.; Murphy, T.L.; Murphy, K.M.; Weaver, C.T. Interleukin 17-producing CD4+ effector T cells develop via a lineage distinct from the T helper type 1 and 2 lineages. Nat. Immunol 2005, 6, 1123–1132. [Google Scholar]
- Mantovani, A.; Sica, A.; Sozzani, S.; Allavena, P.; Vecchi, A.; Locati, M. The chemokine system in diverse forms of macrophage activation and polarization. Trends Immunol 2004, 25, 677–686. [Google Scholar]
- Chen, B.H.; Wu, P.Y.; Chen, K.M.; Fu, T.F.; Wang, H.M.; Chen, C.Y. Antiallergic potential on RBL-2H3 cells of some phenolic constituents of Zingiber officinale (Ginger). J. Nat. Prod 2009, 72, 950–953. [Google Scholar]
- Wang, H.M.; Pan, J.L.; Chen, C.Y.; Chiu, C.C.; Yang, M.H.; Chang, H.W.; Chang, J.S. Identification of anti-lung cancer extract from Chlorella vulgaris C-C by antioxidant property using supercritical carbon dioxide extraction. Process Biochem 2010, 45, 1865–1872. [Google Scholar]
- Wang, H.M.; Chou, Y.T.; Hong, Z.L.; Chen, H.A.; Chang, Y.C.; Yang, W.L.; Chang, H.C.; Mai, C.T.; Chen, C.Y. Bioconstituents from stems of Synsepalum dulcificum Daniell (Sapotaceae) inhibit human melanoma proliferation, reduce mushroom tyrosinase activity and have antioxidant properties. J. Taiwan Inst. Chem. E 2011, 42, 204–211. [Google Scholar]
- Wang, H.M.; Chen, C.Y.; Chen, C.Y.; Ho, M.L.; Chou, Y.T.; Chang, H.C.; Lee, C.H.; Wang, C.Z.; Chu, I.M. (−)-N-Formylanonaine from Michelia alba as a human tyrosinase inhibitor and antioxidant. Bioorg. Med. Chem 2010, 18, 5241–5247. [Google Scholar]
- Wang, H.M.; Chen, C.Y.; Wen, Z.H. Identifying melanogenesis inhibitors from Cinnamomum subavenium with in vitro and in vivo screening systems by targeting the human tyrosinase. Exp. Dermatol 2011, 20, 242–248. [Google Scholar]
- Baldea, I.; Mocan, T.; Cosgarea, R. The role of ultraviolet radiation and tyrosine stimulated melanogenesis in the induction of oxidative stress alterations in fair skin melanocytes. Exp. Oncol 2009, 31, 200–208. [Google Scholar]
- Wang, H.M.; Chiu, C.C.; Wu, P.F.; Chen, C.Y. Subamolide E from Cinnamomum subavenium induces sub-G1 cell-cycle arrest and caspase-dependent apoptosis and reduces the migration ability of human melanoma cells. J. Agric. Food Chem 2011, 59, 8187–8192. [Google Scholar]
- Tran, H.; Chen, K.; Shumack, S. Epidemiology and aetiology of basal cell carcinoma. Br. J. Dermatol 2003, 149(Suppl 66), 50–52. [Google Scholar]
- White, N.; Knight, G.E.; Butler, P.E.; Burnstock, G. An in vivo model of melanoma: Treatment with ATP. Purinergic Signal 2009, 5, 327–333. [Google Scholar]
- Tanaka, R.; Koyanagi, K.; Narita, N.; Kuo, C.; Hoon, D.S. Prognostic molecular biomarkers for cutaneous malignant melanoma. J. Surg. Oncol 2011, 104, 438–446. [Google Scholar]
- Yoshioka, S.; Hamada, A.; Cui, T.; Yokota, J.; Yamamoto, S.; Kusunose, M.; Miyamura, M.; Kyotani, S.; Kaneda, R.; Tsutsui, Y.; Odani, K.; Odani, I.; Nishioka, Y. Pharmacological activity of deep-sea water: Examination of hyperlipemia prevention and medical treatment effect. Biol. Pharm. Bull 2003, 26, 1552–1559. [Google Scholar]
- Lee, J.B.; Hayashi, K.; Hirata, M.; Kuroda, E.; Suzuki, E.; Kubo, Y.; Hayashi, T. Antiviral sulfated polysaccharide from Navicula directa, a diatom collected from deep-sea water in Toyama Bay. Biol. Pharm. Bull 2006, 29, 2135–2139. [Google Scholar]
- Hwang, H.S.; Kim, H.A.; Lee, S.H.; Yun, J.W. Anti-obesity and antidiabetic effects of deep sea water on ob/ob mice. Mar. Biotechnol. (N.Y.) 2009, 11, 531–539. [Google Scholar]
- Katsuda, S.; Yasukawa, T.; Nakagawa, K.; Miyake, M.; Yamasaki, M.; Katahira, K.; Mohri, M.; Shimizu, T.; Hazama, A. Deep-sea water improves cardiovascular hemodynamics in Kurosawa and Kusanagi-Hypercholesterolemic (KHC) rabbits. Biol. Pharm. Bull 2008, 31, 38–44. [Google Scholar]
- Boyum, A.; Lovhaug, D.; Tresland, L.; Nordlie, E.M. Separation of leucocytes: Improved cell purity by fine adjustments of gradient medium density and osmolality. Scand. J. Immunol 1991, 34, 697–712. [Google Scholar]
- Chen, C.Y.; Cheng, K.C.; Chang, A.Y.; Lin, Y.T.; Hseu, Y.C.; Wang, H.M. 10-Shogaol, an antioxidant from Zingiber officinale for skin cell proliferation and migration enhancer. Int. J. Mol. Sci 2012, 13, 1762–1777. [Google Scholar]
- Iveković, D.; Milardović, S.; Roboz, M.; Grabarić, B.S. Evaluation of the antioxidant activity by flow injection analysis method with electrochemically generated ABTS radical cation. Analyst 2005, 130, 708–714. [Google Scholar]
- Oyaizu, M. Studies on products of the browning reaction. Antioxidative activities of browning reaction products prepared from glucosamine. Jpn. J. Nutr 1986, 44, 307–315. [Google Scholar]
- Chiu, C.C.; Liu, P.L.; Huang, K.J.; Wang, H.M.; Chang, K.F.; Chou, C.K.; Chang, F.R.; Chong, I.W.; Fang, K.; Chen, J.S.; et al. Goniothalamin inhibits growth of human lung cancer cells through DNA damage, apoptosis, and reduced migration ability. J. Agric. Food Chem 2011, 59, 4288–4293. [Google Scholar]
- Geback, T.; Schulz, M.M.; Koumoutsakos, P.; Detmar, M. TScratch: A novel and simple software tool for automated analysis of monolayer wound healing assays. Biotechniques 2009, 46, 265–274. [Google Scholar]
- Yamada, P.; Isoda, H.; Han, J.K.; Talorete, T.P.N.; Yamaguchi, T.; Abe, Y. Inhibitory effect of fulvic acid extracted from Canadian sphagnum peat on chemical mediator release by RBL-2H3 and KU812 cells. Biosci. Biotechnol. Biochem 2007, 71, 1294–1305. [Google Scholar]
- Luo, C.; Wu, X.G. Lycopene enhances antioxidant enzyme activities and immunity function in N-methyl-N′-nitro-N-nitrosoguanidine-induced gastric cancer rats. Int. J. Mol. Sci 2011, 12, 3340–3351. [Google Scholar]
- Greiner, J.; Diezel, W. Inflammation-inhibiting effect of magnesium ions in contact eczema reactions (in German). Hautarzt 1990, 41, 602–605. [Google Scholar]
- Chen, B.W.; Wang, H.H.; Liu, J.X.; Liu, X.G. Zinc sulfate solution enema decreases inflammation in experimental colitis in rats. J. Gastroenterol. Hepatol 1999, 14, 1088–1092. [Google Scholar]
- Kwon, B.S.; Haq, A.K.; Pomerantz, S.H.; Halaban, R. Isolation and sequence of a cDNA clone for human tyrosinase that maps at the mouse c-albino locus. Proc. Natl. Acad. Sci. USA 1987, 84, 7473–7477. [Google Scholar]
- Muller, G.; Ruppert, S.; Schmid, E.; Schutz, G. Functional analysis of alternatively spliced tyrosinase gene transcripts. EMBO J 1988, 7, 2723–2730. [Google Scholar]
- Jacobsohn, G.M.; Jacobsohn, M.K. Incorporation and binding of estrogens into melanin: Comparison of mushroom and mammalian tyrosinases. Biochim. Biophys. Acta 1992, 1116, 173–182. [Google Scholar]
- Belkhiri, A.; Richards, C.; Whaley, M.; McQueen, S.A.; Orr, F.W. Increased expression of activated matrix metalloproteinase-2 by human endothelial cells after sublethal H2O2 exposure. Lab. Invest 1997, 77, 533–539. [Google Scholar]
- Reth, M. Hydrogen peroxide as second messenger in lymphocyte activation. Nat. Immunol 2002, 3, 1129–1134. [Google Scholar]
- Huo, Y.; Qiu, W.Y.; Pan, Q.; Yao, Y.F.; Xing, K.; Lou, M.F. Reactive oxygen species (ROS) are essential mediators in epidermal growth factor (EGF)-stimulated corneal epithelial cell proliferation, adhesion, migration, and wound healing. Exp. Eye Res 2009, 89, 876–886. [Google Scholar]
- Chiu, C.C.; Chou, H.L.; Wu, P.F.; Wang, H.M.; Chen, C.Y. Bio-functional constituents from the stems of Liriodendron tulipifera. Molecules 2012, 17, 4357–4372. [Google Scholar]
- Ishikawa, K.; Takenaga, K.; Akimoto, M.; Koshikawa, N.; Yamaguchi, A.; Imanishi, H.; Nakada, K.; Honma, Y.; Hayashi, J.I. ROS-generating mitochondrial DNA mutations can regulate tumor cell metastasis. Science 2008, 320, 661–664. [Google Scholar]
- Wu, W.S. The signaling mechanism of ROS in tumor progression. Cancer Metastasis Rev 2006, 25, 695–705. [Google Scholar]
- Melo-Silveira, R.F.; Fidelis, G.P.; Costa, M.S.S.P.; Telles, C.B.S.; Dantas-Santos, N.; Elias, S.D.O.; Ribeiro, V.B.; Barth, A.L.; Macedo, A.J.; Leite, E.L.; Rocha, H.A.O. In vitro antioxidant, anticoagulant and antimicrobial activity and in inhibition of cancer cell proliferation by xylan extracted from corn cobs. Int. J. Mol. Sci 2011, 13, 409–426. [Google Scholar]
| ACW | Colony Count (×106 CFU/mL) (% Bacterial Growth) | ||||||
|---|---|---|---|---|---|---|---|
| Control | Exposure Time (s) | ||||||
| 5 | 30 | 60 | 180 | 300 | 900 | ||
| S. aureus (29,213) | 272 | 30 (11%) | 14 (5%) | 18 (7%) | 43 (16%) | 50 (18%) | 23 (8%) |
| E. coli (35,218) | 243 | 159 (65%) | 180 (74%) | 212 (87%) | 191 (79%) | 150 (62%) | 117 (48%) |
| ACW | Superoxide Anion Scavenging (%) | DPPH· Scavenging (%) | ABTS+ Scavenging (%) | Reducing Power c (OD700) | FRAP Assay (FeSO4 mg/mL) | Metal Chelating Activity (%) |
|---|---|---|---|---|---|---|
| Gallic acid a | 100 ± 0.00 | 100 ± 0.00 | 100 ± 0.00 | 0.284 ± 0.04 | 10.31 ± 0.23 | – |
| EDTA b | – | – | – | – | – | 100 ± 0.00 |
| 2% | 93.97 ± 0.25 | nd | nd | 0.044 ± 0.05 | nd | nd |
| 5% | 95.88 ± 0.39 | nd | nd | 0.044 ± 0.08 | nd | nd |
| 10% | 96.47 ± 0.49 | <10.0 | nd | 0.045 ± 0.01 | <1.0 | <10.0 |
| 20% | 96.86 ± 0.10 | <10.0 | nd | 0.043 ± 0.05 | <1.0 | <10.0 |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Liao, W.-T.; Huang, T.-S.; Chiu, C.-C.; Pan, J.-L.; Liang, S.-S.; Chen, B.-H.; Chen, S.-H.; Liu, P.-L.; Wang, H.-C.; Wen, Z.-H.; et al. Biological Properties of Acidic Cosmetic Water from Seawater. Int. J. Mol. Sci. 2012, 13, 5952-5971. https://doi.org/10.3390/ijms13055952
Liao W-T, Huang T-S, Chiu C-C, Pan J-L, Liang S-S, Chen B-H, Chen S-H, Liu P-L, Wang H-C, Wen Z-H, et al. Biological Properties of Acidic Cosmetic Water from Seawater. International Journal of Molecular Sciences. 2012; 13(5):5952-5971. https://doi.org/10.3390/ijms13055952
Chicago/Turabian StyleLiao, Wei-Ting, Tsi-Shu Huang, Chien-Chih Chiu, Jian-Liang Pan, Shih-Shin Liang, Bing-Hung Chen, Shi-Hui Chen, Po-Len Liu, Hui-Chun Wang, Zhi-Hong Wen, and et al. 2012. "Biological Properties of Acidic Cosmetic Water from Seawater" International Journal of Molecular Sciences 13, no. 5: 5952-5971. https://doi.org/10.3390/ijms13055952





