Characterization of CdTe Films Deposited at Various Bath Temperatures and Concentrations Using Electrophoretic Deposition
Abstract
:1. Introduction
2. Experiment
2.1. Sample Preparation
2.2. Characterization of Films
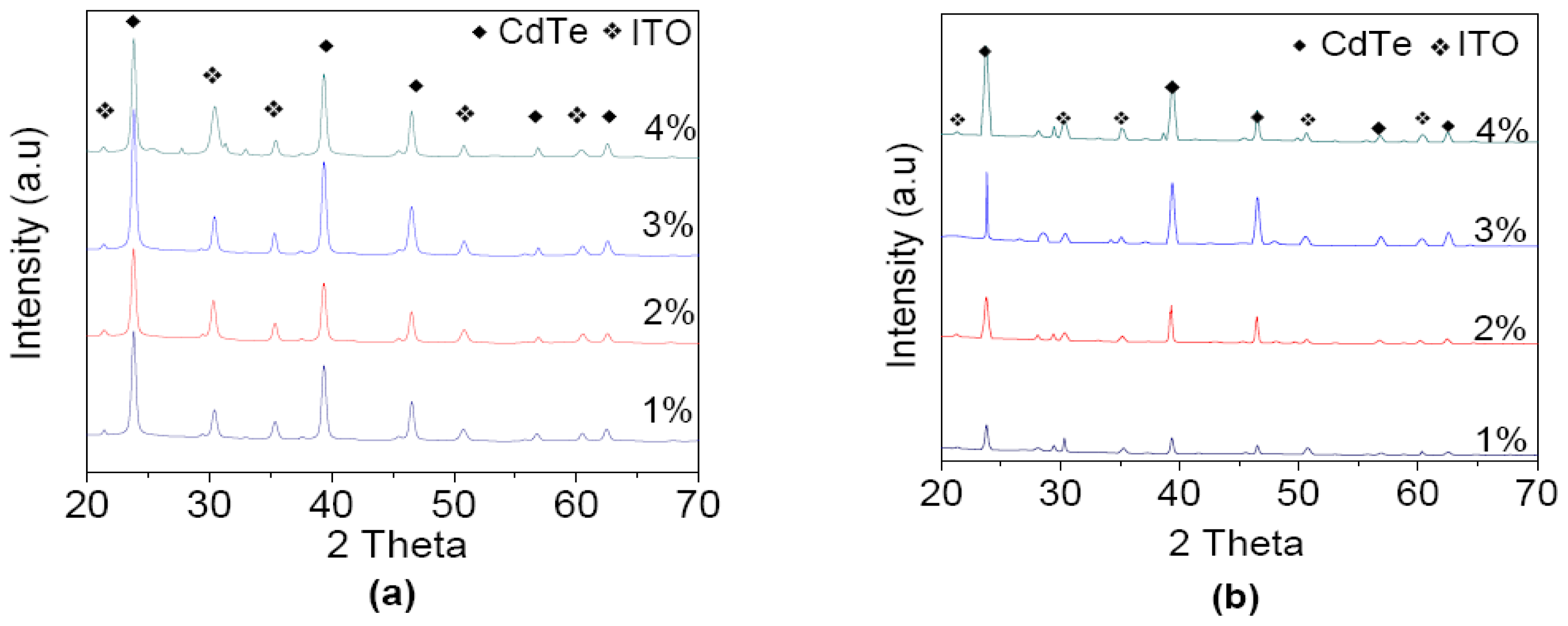
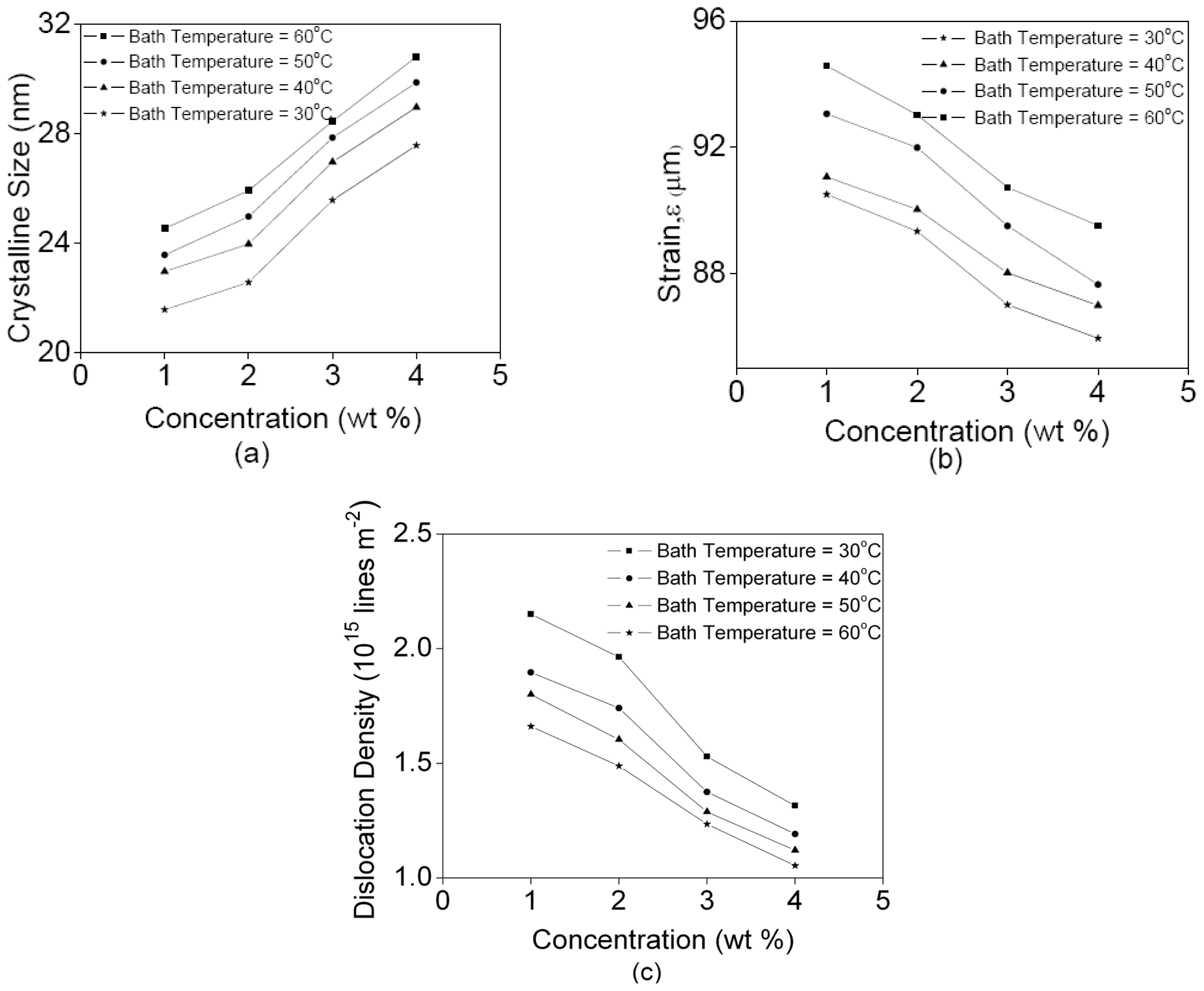
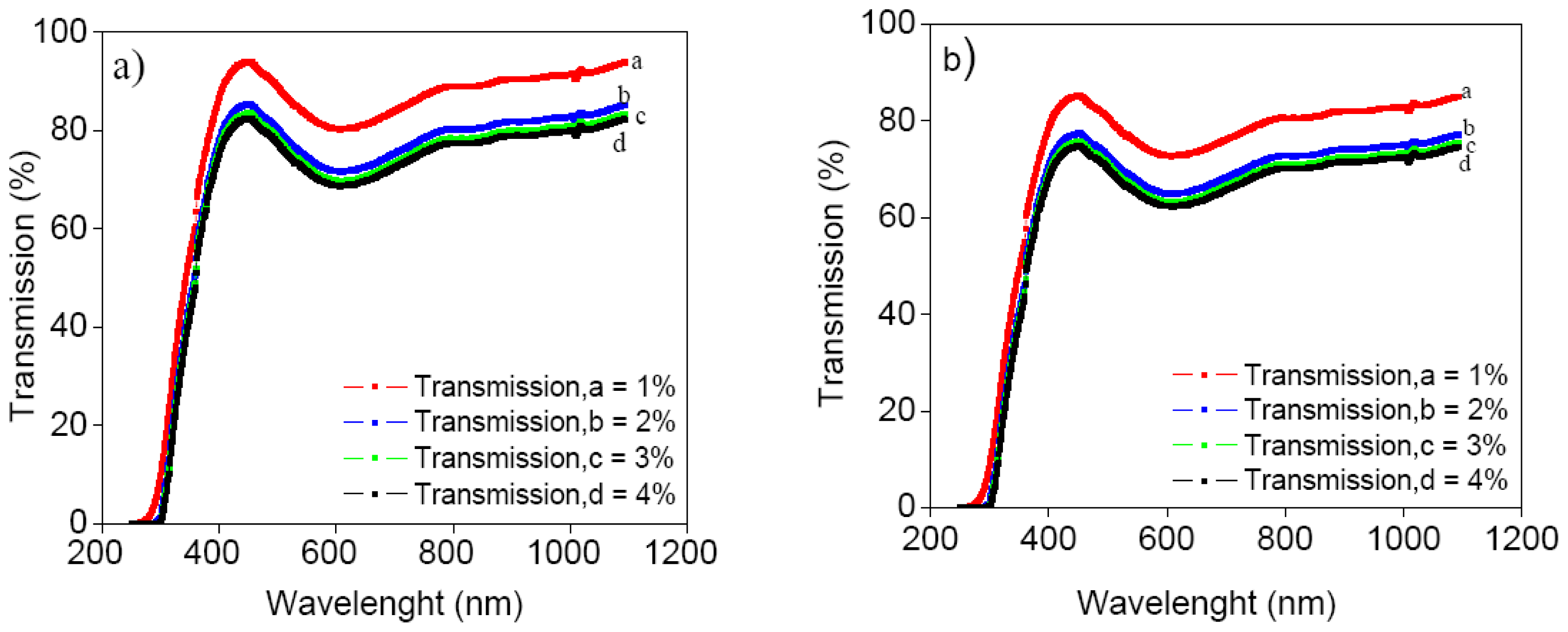
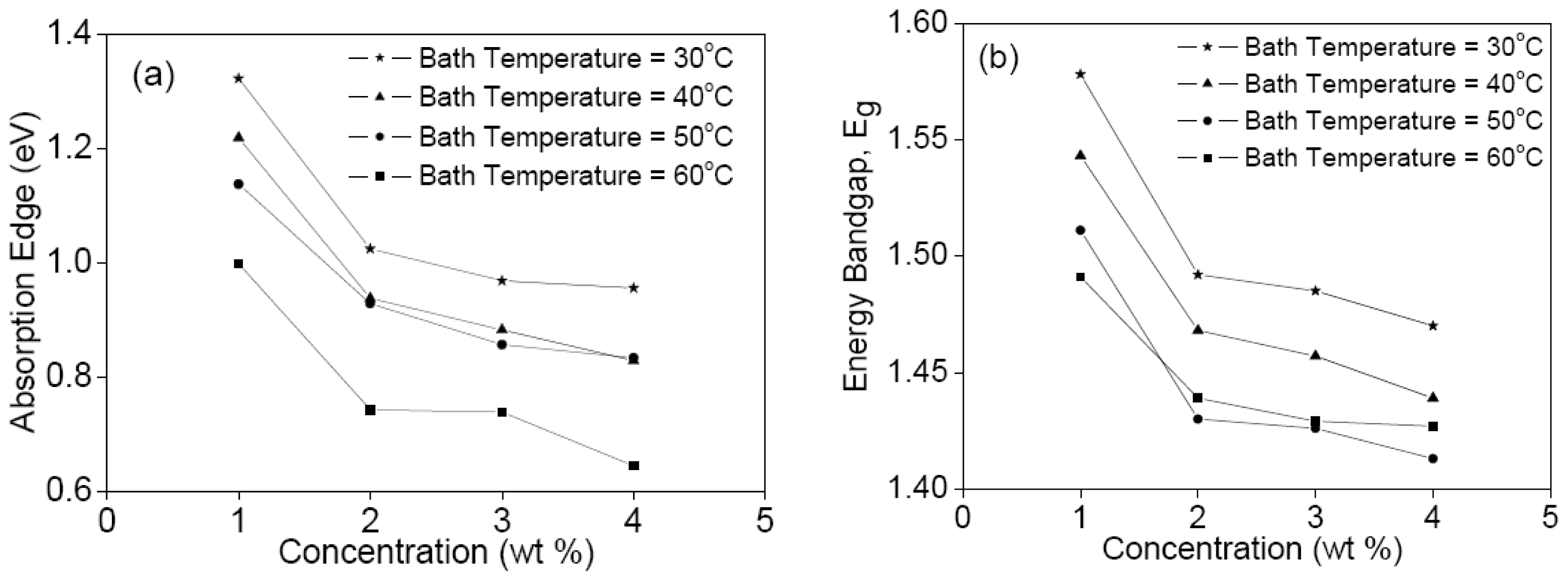
3. Results and Discussion
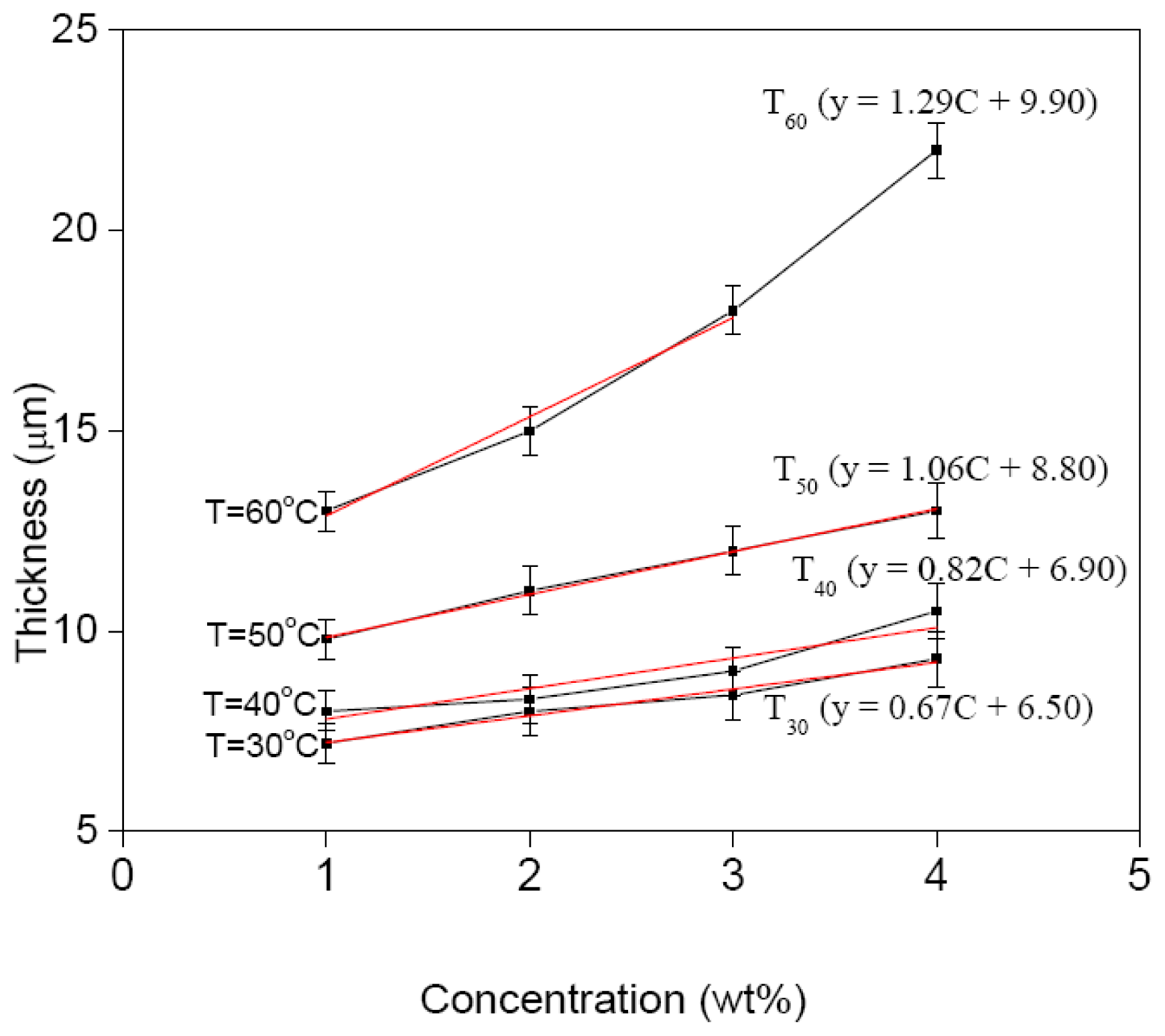
4. Numerical Relationship of Film Thickness
5. Conclusions
Acknowledgments
References
- Boccaccini, A.R.; Cho, J.; Roether, J.A.; Thomas, B.J.C.; Minary, E.J.; Shaffer, M.S.P. Electrophoretic deposition of carbon nanotubes. Carbon 2006, 44, 3149–3160. [Google Scholar]
- Ismail, B.B.; Deraman, K.B.; Woon, H.Y. X-ray diffraction study of evaporated cadmium telluride thin films. J. Fizik UTM 2009, 4, 26–34. [Google Scholar]
- Ogata, N.; van Tassel, J.; Randall, C.A. Electrode formation by electrophoretic deposition of nanopowder. Mater. Lett 2001, 49, 7–14. [Google Scholar]
- Hasan, S.A.; Kavich, D.W.; Mahajan, S.V.; Dickson, J.H. Electrophoretic deposition of CdSe nanocrystal films onto dielectric polymer thin films. Thin Solid Films 2009, 517, 2665–2669. [Google Scholar]
- Salant, A.; Shalom, M.; Hod, I.; Faust, A.; Zaban, A.; Banin, U. Quantum dot sensitized solar cells with improved efficiency prepared using electrophoretic deposition. ACS Nano 2010, 4, 5962–5968. [Google Scholar]
- Razak, M.F.S.A.; Zainal, Z. Electrophoretic deposition and characterization of copper selenide thin films. Malays. J. Anal. Sci 2007, 11, 324–330. [Google Scholar]
- Savadogo, O. Chemically and electrochemically deposited materials. Solar Energy Mater. Solar Cells 1998, 52, 361–388. [Google Scholar]
- Corni, I.; Ryan, M.P.; Boccaccini, A.R. Electrophoretic deposition: From traditional ceramic to nanotechnology. J. Eur. Ceram. Soc 2008, 28, 1353–1367. [Google Scholar]
- Sarkar, P.; Nicholson, P.S. Electrophoretic Deposition (EPD): Mechanism, kinetic and application to ceramic. J. Am. Ceram. Soc 1996, 79, 1987–2002. [Google Scholar]
- Mathew, X.; Sebasian, P.J. Optical properties of electrodeposited CdTe thin films. Solar Energy Mater. Solar Cells 1999, 59, 85–98. [Google Scholar]
- Du, C.; Heldbrant, D.; Pan, N. Preparation and preliminary property study of carbon nanotubes films by electrophoretic deposition. Mater. Lett 2002, 57, 434–438. [Google Scholar]
- Khairnar, U.P.; Bhavsar, D.S.; Vaidya, R.U.; Bhavsar, G.P. Optical properties of thermally evaporated cadmium telluride thin films. Mater. Chem. Phys 2003, 80, 421–427. [Google Scholar]
- Chaudhuri, S.; Das, S.K.; Pal, A.K. Optical transitions in CdTe thin films produced by hot-wall vacuum evaporation. Thin Solid Films 1987, 147, 9–16. [Google Scholar]
- Myers, T.H.; Edwards, S.W.; Schetzina, J.F. Optical properties of CdTe films. J. Appl. Phys 1981, 52, 4231–4237. [Google Scholar]
- Matsune, K.; Oda, H.; Toyoma, T.; Okamoto, H.; Kudriavysevend, Y.; Asomoza, R. 15% efficiency CdS/CdTe thin film solar cells using CdS layers doped with metal organic compounds. Solar Energy Mater. Solar Cells 2006, 90, 3108–3114. [Google Scholar]
- Lalitha, S.; Sathyamoorthy, R.; Senthilarasu, S.; Subbarayan, A.; Natarajan, K. Characterization of CdTe thin film depence of structural and optical properties on temperature and thickness. Solar Energy Mater. Solar Cells 2004, 82, 187–199. [Google Scholar]
- Sathyamoorthy, R.; Narayandass; Sa, K.; Mangalaraj, D. Effect of substrate temperature on the structure and optical properties of CdTe thin films. Solar Energy Mater. Solar Cells 2003, 76, 339–346. [Google Scholar]
- Chopra, K.L. Thin Film Phenomena; McGraw-Hill: New York, NY, USA, 1970. [Google Scholar]
- Anuar, K.; Ho, S.M.; Tan, W.T.; Atan, S.; Kuang, Z.; Haron, M.J.; Saravanan, N. Effect of bath temperature on the electrodeposition of Cu4SnS4 thin films. J. Appl. Sci. Res 2008, 4, 1701–1707. [Google Scholar]
- Rizwan, Z.; Norizam, A.Z.M.; Sabri, M.G.M.; Noroozi, M. Characterization of nanostructured CdS film deposited at low growth rate using CBD technique. Chalcogenide Lett 2010, 7, 423–429. [Google Scholar]
- Murali, K.R.; Radhakrishna, I.; Nagaraja Rao, K.; Venkatesan, V.K. Properties of CdTe films deposited by electron beam evaporationn. Surface Coat. Technol 1990, 41, 211–219. [Google Scholar]
- Eibed, M.R.; Ahmed, M.F.; Ramadan, A.A.; Abdel-Hady, K. Thickness and deposition rate dependence of structural characteristics of evaporated CdTe films. Egypt J. Solids 2005, 28, 231–240. [Google Scholar]

© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Daud, M.N.M.; Zakaria, A.; Jafari, A.; Ghazali, M.S.M.; Abdullah, W.R.W.; Zainal, Z. Characterization of CdTe Films Deposited at Various Bath Temperatures and Concentrations Using Electrophoretic Deposition. Int. J. Mol. Sci. 2012, 13, 5706-5714. https://doi.org/10.3390/ijms13055706
Daud MNM, Zakaria A, Jafari A, Ghazali MSM, Abdullah WRW, Zainal Z. Characterization of CdTe Films Deposited at Various Bath Temperatures and Concentrations Using Electrophoretic Deposition. International Journal of Molecular Sciences. 2012; 13(5):5706-5714. https://doi.org/10.3390/ijms13055706
Chicago/Turabian StyleDaud, Mohd Norizam Md, Azmi Zakaria, Atefeh Jafari, Mohd Sabri Mohd Ghazali, Wan Rafizah Wan Abdullah, and Zulkarnain Zainal. 2012. "Characterization of CdTe Films Deposited at Various Bath Temperatures and Concentrations Using Electrophoretic Deposition" International Journal of Molecular Sciences 13, no. 5: 5706-5714. https://doi.org/10.3390/ijms13055706



