Poisson Parameters of Antimicrobial Activity: A Quantitative Structure-Activity Approach
Abstract
:1. Introduction
2. Results
2.1. Probability Distribution Analysis
2.2. QSAR Models
2.2.1. Based on DRAGON Descriptors
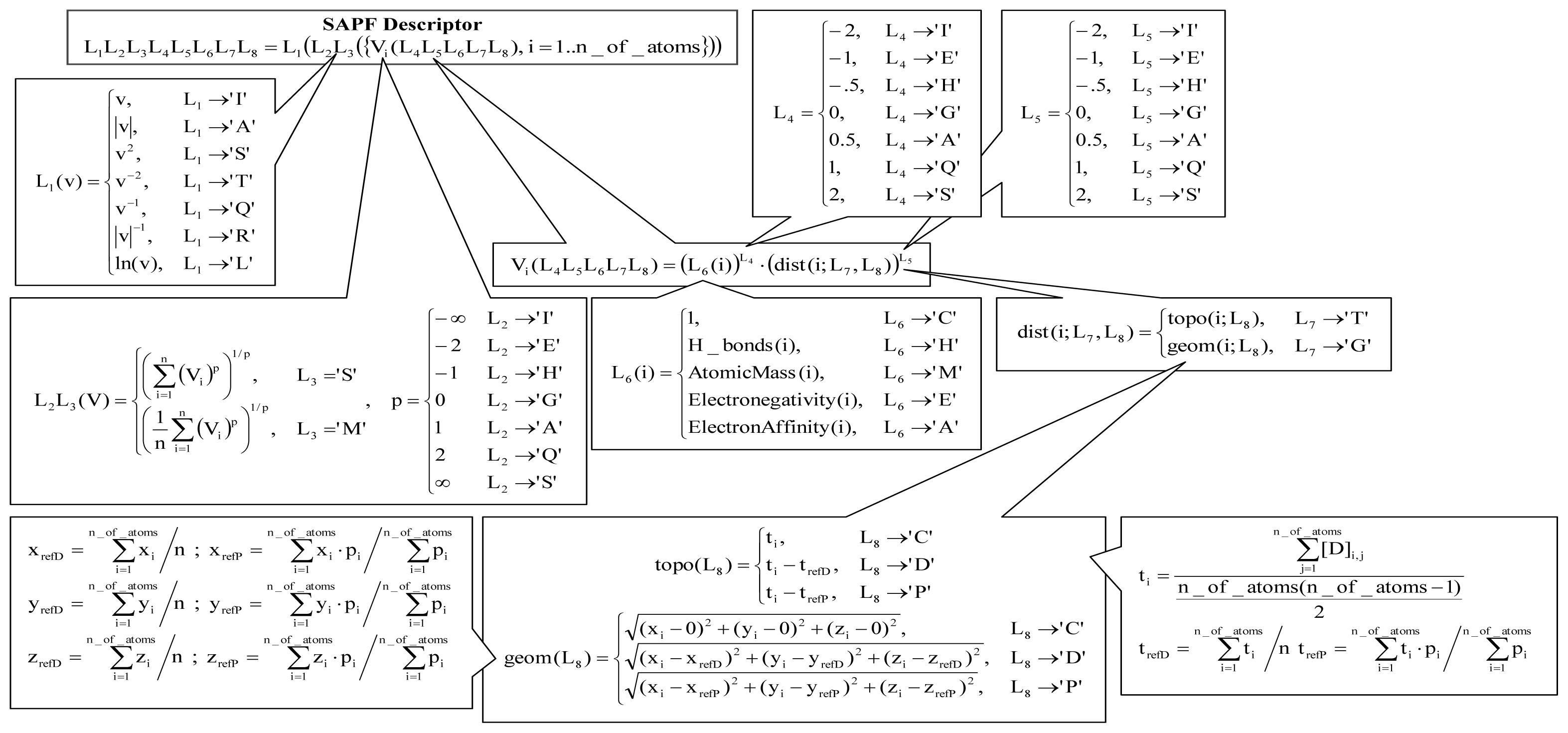
2.2.2. Based on SAPF Descriptors
2.2.3. Models Comparison
3. Discussion
- Compounds series:
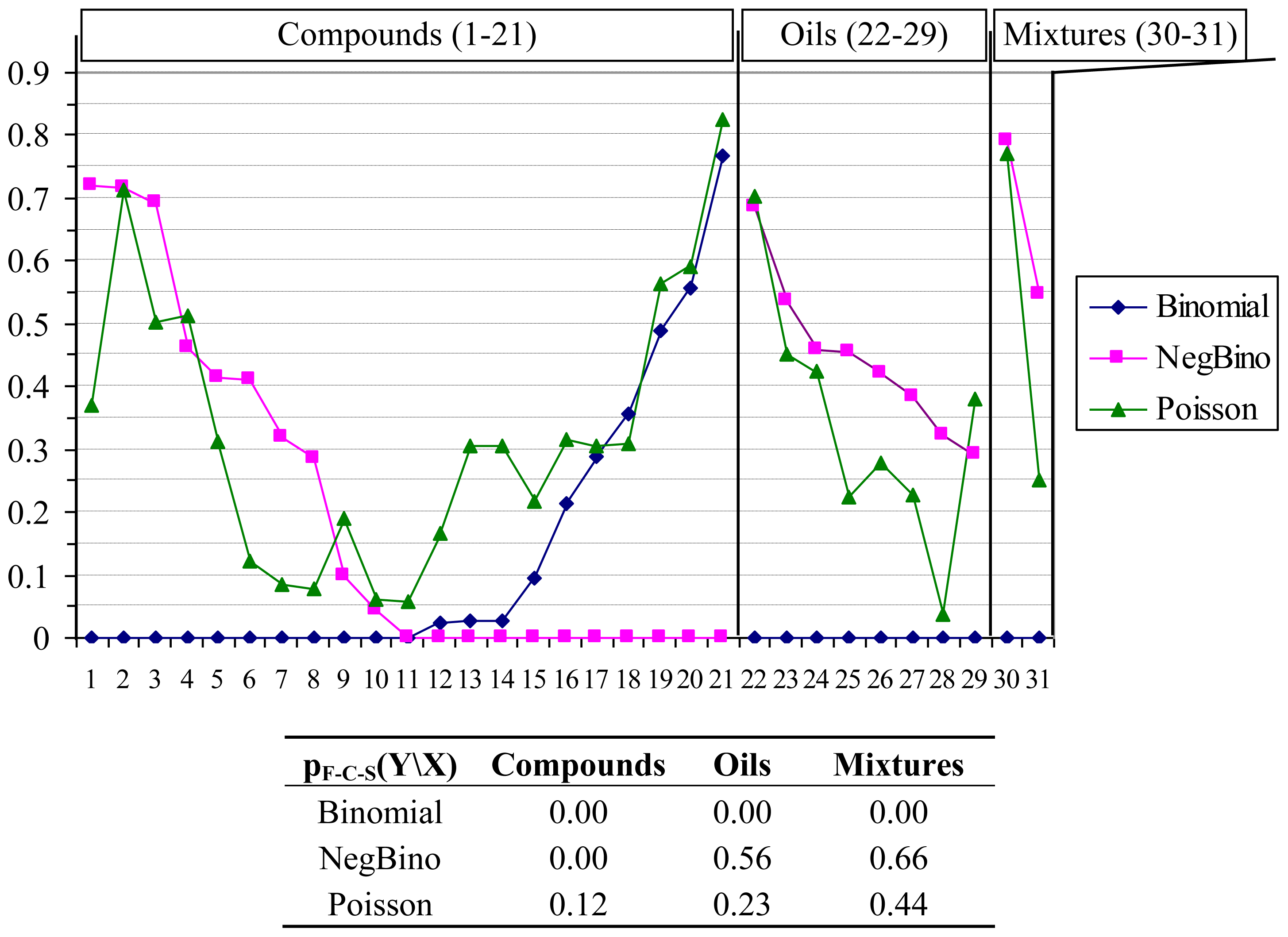
- ○ Without any exception, the antimicrobial effects of all investigated compounds proved to follow Poisson distribution. Moreover, the hypothesis that any compound has a Poisson distribution of antimicrobial activity on bacteria population could not be rejected by F-C-S statistics (F-C-S statistics = 28.79, p = 0.12, Figure 1). Starting with this result, the Poisson λ parameter has been obtained to reflect what happen in the population, this parameter being an estimate for both central tendency and variability of antibacterial effects. The analysis of the obtained Poisson parameters showed to follow more likely a log-normal distribution and a logarithm transformation was applied on these values before quantitative structure-activity relationship search. This transformation was applied to avoid the presence of outliers and to assure the normality assumption needed for linear regression analysis [35,36].
- ○ Negative binomial distribution was rejected by 55% of compounds while Binomial distribution was rejected in 70% of cases. Negative binomial distribution, also known as the Pascal distribution or Pólya distribution, is a twin of Poisson distribution [37,38] widely used in analysis of count data [39,40]. The negative binomial distribution could be obtained by superposition of a continuous distribution over Poisson distribution (Fisher showed the convolution between Chi-Square and Poisson distribution [41]). Other authors showed that the negative binomial distribution might derive from a convolution between the Gamma distribution (Chi-Square distribution is a particular case of Gamma distribution) and Poisson distribution [42,43]. Whenever the separation of factors is possible, it is also possible to separate the convolutions of distributions [44], and this separation give the possibility to analyze separately the factors. The results presented by Jäntschi et al. [44] sustained and/or are sustained by convolution of Poisson distribution with a continuous distribution in regards of both factors (bacteria and chemical compounds) in the expression of antimicrobial activity. The results showed that antimicrobial activity follow a negative binomial distribution under the influence of both factors (bacteria and chemical compound) and Poison distribution under the influence of the bacteria factor [44]. Furthermore, the negative binomial distribution might be obtained by convolution of log-normal with Gamma distribution; although a high number of observations are needed (n > 250) in order to statistically assure the difference between Log-normal and Gamma distributions [45].
- Oils and mixture series:
- ○ Negative Binomial distribution cannot be rejected for oils. Moreover, Negative Binomial distribution for oils had a higher likelihood than Poisson distribution (pF-C-S for Negative Binomial: 0.56; pF-C-S for Poisson: 0.23) while the Binomial distribution was rejected.
- ○ Negative Binomial distribution cannot be rejected for mixtures either. Moreover, Negative Binomial distribution for mixtures had also higher likelihood than Poisson distribution (pF-C-S for Negative Binomial = 0.66; pF-C-S for Poisson = 0.44) while the Binomial distribution was rejected.
- ○ The above-presented facts suggest that in the case of oils and mixtures, the factors of the antibacterial activity are not completely separated when oil/mixture name are taken as factor; this appears to be because the Negative Binomial distribution often occurs when a convolution/superposition of Poisson distributions characterize the observed data [46].
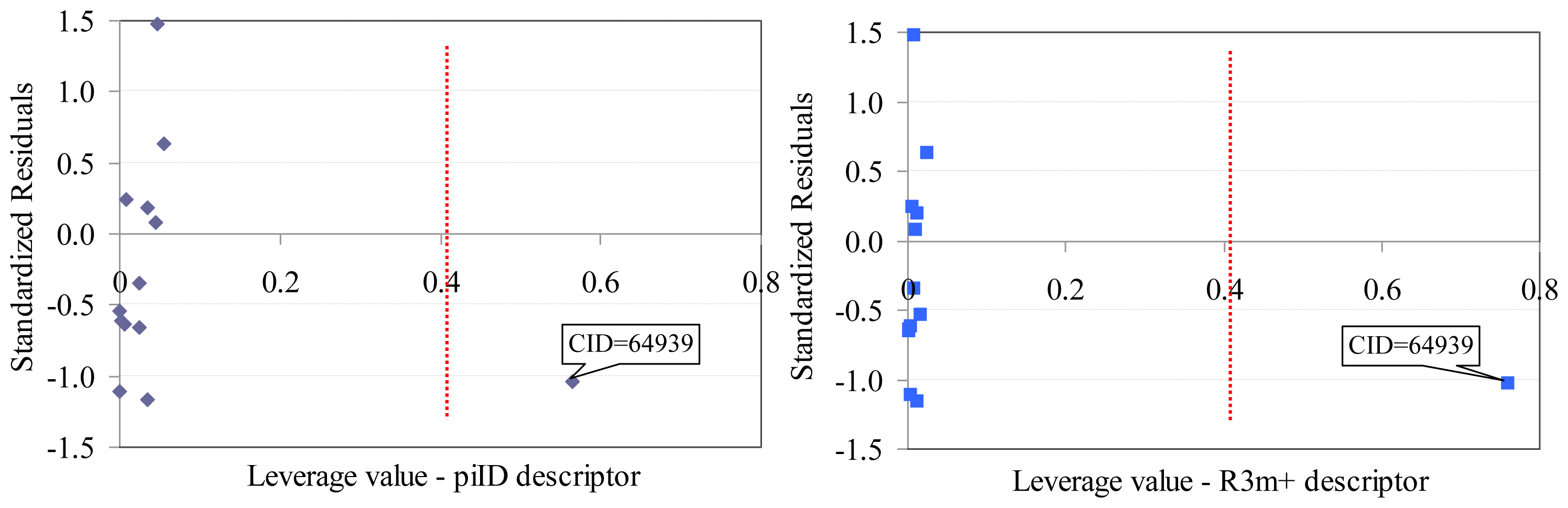
- One compound proved to be influential in the model (CID = 64939, Figure 2). This compound obtained the value of leverage for both Dragon descriptors higher than the accepted threshold (0.41). This compound, which belongs to the training set, was withdrawn, and a model based on 12 compounds in training set was obtained, Equation(1).
- Two descriptors were able to describe the linear relation between overall antimicrobial activities of investigated compounds. One descriptor belongs to the walk and path counts and relates the conventional bond order ID number while the second descriptor relates the maximal autocorrelation of lag 3 divided by mass (R3m+). According with associated coefficients, the R3m+ had a higher contribution in the model compared with piID descriptor, but its contribution is to the significance level threshold (5.8% compared to imposed 5% significance level).
- QSAR-Dragon model proved to be statistically significant (F = 39, p = 3.62 × 10−5). A low value of root mean square error was obtained in leave-one-out analysis (0.1276). The contribution of R3m+ descriptor to the model is questionable since the significance associated to its coefficient is very close to 0.05 but since it has a real contribution in the r2 value its significance of 5.8% was accepted. Moreover, the R3m+ proved not significantly correlate with Poisson parameter (r = −0.2410).
- Multicollianearity is not present in the model since the tolerance value 0.1 < T < 1 and the variance inflation factors (VIF) < 10 even if a significant correlation coefficient was obtained between Dragon descriptors.
- The model proved its abilities in estimation (R2TR = 0.897) as well as in prediction (internal validity of the model in leave-one-out analysis, R2loo = 0.845 and external validation in test set R2TS = 0.652) with a difference in the goodness-of-fit from 0.052 (training vs. interval validation - leave one out analysis) to 0.245 (training vs. external validation-test set). However, the difference of 0.245 proved not statistically significant (p > 0.05).
- Unfortunately, external abilities in prediction were away from the expected abilities. The trend is significant far from the expected line-Figure 3.
- The abilities in estimation (training set) proved not statistically significant from the abilities in prediction (test set) since a probability of 0.3598 was obtained in comparison.
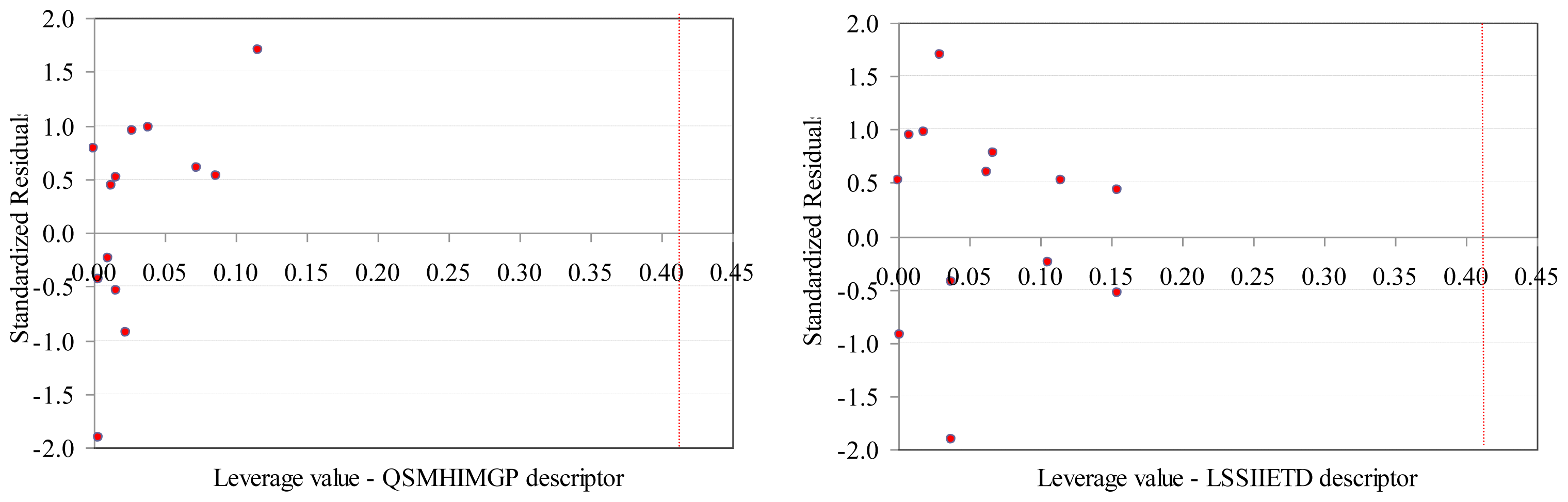
- The values of SAPF descriptors associated to compounds proved that no compound had significant influence on the model (all leverage values where lower than threshold −0.41, Figure 4).
- SAPF model proved statistically significant (F = 24, p = 1.48 × 10−4). The contribution of both descriptors to the model proved statistically significant (p-values associated to coefficients <0.05).
- According to descriptors from Equation(2), the global model of antibacterial activity is related to both molecular geometry and topology: one descriptor identified a relation between the geometry of compounds and the overall antimicrobial activity while the second descriptor identified a relation with compounds’ topology. Moreover, the atomic mass and electronegativity proved to be related to the overall antimicrobial activity by the same split ratio in the expression of the model descriptors.
- Multicollianearity was not identified in the QSAR-SAPF model, even if a statistically significant correlation coefficient between descriptors exists (the tolerance values were higher than 0.1 and smaller than 1 and the variance inflation factors (VIF) had values smaller than 10).
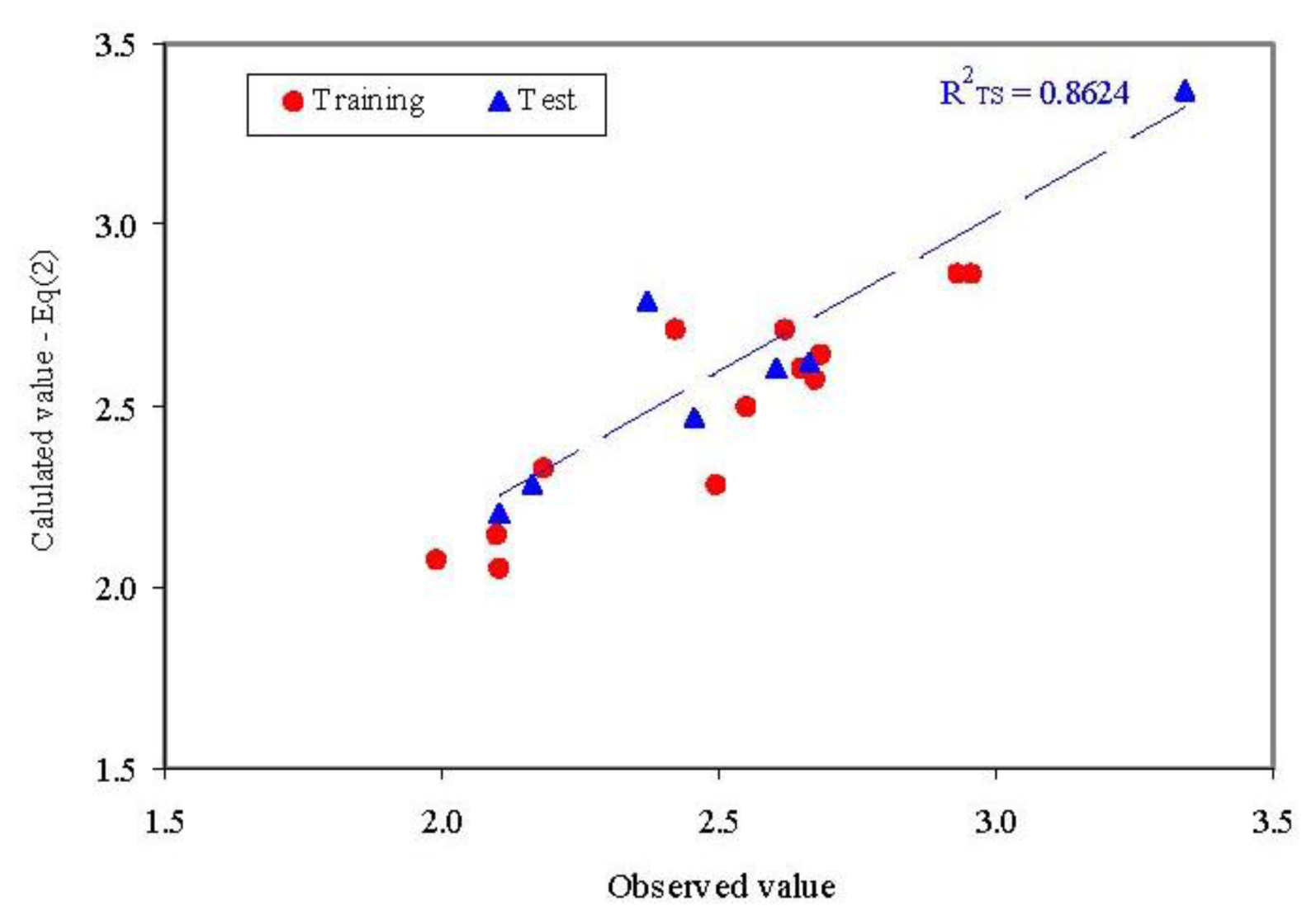
- The model proved its abilities in estimation (R2TR = 0.829) as well as in prediction (internal validity of the model in leave-one-out analysis, R2loo = 0.700 and external validation in test set R2TS = 0.862) with a difference in the goodness-of-fit from −0.034 (training vs. external validation - test set) to 0.129 (training vs. interval validation-leave one out analysis). Moreover, none of these differences were statistically significant (p > 0.05).
- External abilities in prediction proved to be close to expected abilities for QSAR-SAPF model (Figure 5).
- Dragon model has slightly better abilities in estimation compared to SAPF model, but these abilities proved not statistically significant. The determination coefficient obtained both in training set and in leave-one-out analysis was higher compared to SAPF model with 0.068 and respectively 0.145. Moreover, the abilities of prediction seem to be better for SAPF model compared to Dragon model (a difference of 0.211, not statistically significant p < 0.05). This observation is also sustained by the lowest value of residuals in training set for Dragon model and in two compounds from training set and all compounds from test set for SAPF model (Table 2).
- The SAPF model systematically obtained smallest values of parameters presented in Table 3: best explaining the variability in the observation; smallest typical errors; smallest standard error of prediction as well as smallest relative error of prediction. The highest difference is observed with regards to standard error of prediction that is almost 4 times higher for Dragon model compared to SAPF model.
- The analysis of predictive power of the models demonstrated that SAPF model had significantly higher power of prediction (Table 3). According to the obtained results, the Q2 values for Dragon model are smaller than 0.6, being considered unacceptable while all Q2 values for SAPF model are higher than 0.77. These results show that the Dragon model can be rejected from a statistical point of view, taking also into consideration that the relative error of prediction is almost 2 times higher compared to SAPF model.
- Furthermore, the mean of residuals for training, external and external + test set proved not statistically different by zero when the SAPF model was analyzed. The Fisher’s predictive power identified statistically difference by zero of the residuals obtained by Dragon model in both training and test sets (9 compounds) (p < 0.05, Table 3).
- The model with a higher concordance between observed and estimated/predicted could be considered the best model. The analysis of concordance correlation coefficient revealed a substantial strength of agreement for training set but a very poor agreement in test set for Dragon model. A moderate strength of agreement was obtained by SAPF model in both training and test sets (Table 3).
- Steiger’s test was not able to identify any statistically significant differences between Dragon and SAPF model regarding goodness-of-fit neither in training set nor in external set.
4. Experimental Section
4.1. Compounds, Oils and Mixtures
4.2. Distribution Analysis
4.3. Molecular Descriptors Calculation
4.4. Identification and Characterization of Linear Regression Models
5. Conclusions
Supplementary Information
ijms-13-05207-s001.pdfAcknowledgments
References
- Sengul, M.; Ercisli, S.; Yildiz, H.; Gungor, N.; Kavaz, A.; Cetin, B. Antioxidant, antimicrobial activity and total phenolic content within the aerial parts of Artemisia absinthum, Artemisia santonicum and Saponaria officinalis. Iran. J. Pharm. Res 2011, 10, 49–55. [Google Scholar]
- Martini, M.G.; Bizzo, H.R.; Moreira, D.D.; Neufeld, P.M.; Miranda, S.N.; Alviano, C.S.; Alviano, D.S.; Leitao, S.G. Chemical composition and antimicrobial activities of the essential oils from Ocimum selloi and hesperozygis myrtoides. Nat. Prod. Commun 2011, 6, 1027–1030. [Google Scholar]
- Serrano, C.; Matos, O.; Teixeira, B.; Ramos, C.; Neng, N.; Nogueira, J.; Nunes, M.L.; Marques, A. Antioxidant and antimicrobial activity of Satureja montana L. extracts. J. Sci. Food Agric 2011, 91, 1554–1560. [Google Scholar]
- Mothana, R.A.; Alsaid, M.S.; Al-Musayeib, N.M. Phytochemical analysis and in vitro antimicrobial and free-radical-scavenging activities of the essential oils from Euryops arabicus and Laggera decurrens. Molecules 2011, 16, 5149–5158. [Google Scholar]
- Quintans, L.; da Rocha, R.F.; Caregnato, F.F.; Moreira, J.C.F.; da Silva, F.A.; Araujo, A.A.D.; dos Santos, J.P.A.; Melo, M.S.; de Sousa, D.P.; Bonjardim, L.R.; Gelain, D.P. Antinociceptive action and redox properties of citronellal, an essential oil present in lemongrass. J. Med. Food 2011, 14, 630–639. [Google Scholar]
- Ito, K.; Ito, M. Sedative effects of vapor inhalation of the essential oil of Microtoena patchoulii and its related compounds. J. Nat. Med 2011, 65, 336–343. [Google Scholar]
- Garozzo, A.; Timpanaro, R.; Stivala, A.; Bisignano, G.; Castro, A. Activity of Melaleuca alternifolia (tea tree) oil on influenza virus A/PR/8: Study on the mechanism of action. Antivir. Res 2011, 89, 83–88. [Google Scholar]
- Pauli, A. Anticandidal low molecular compounds from higher plants with special reference to compounds from essential oils. Med. Res. Rev 2011, 26, 223–268. [Google Scholar]
- Jaffri, J.M.; Mohamed, S.; Ahmad, I.N.; Mustapha, N.M.; Manap, Y.A.; Rohimi, N. Effects of catechin-rich oil palm leaf extract on normal and hypertensive rats’ kidney and liver. Food Chem 2011, 128, 433–441. [Google Scholar]
- Yu, F.; Gao, J.; Zeng, Y.; Liu, C.X. Effects of adlay seed oil on blood lipids and antioxidant capacity in hyperlipidemic rats. J. Sci. Food Agric 2011, 91, 1843–1848. [Google Scholar]
- Zhang, Y.B.; Guo, J.; Dong, H.Y.; Zhao, X.M.; Zhou, L.; Li, X.Y.; Liu, J.C.; Niu, Y.C. Hydroxysafflor yellow a protects against chronic carbon tetrachloride-induced liver fibrosis. Eur. J. Pharmacol 2011, 660, 438–444. [Google Scholar]
- Yordi, E.G.; Molina Pérez, E.; Joao Matos, M.; Uriarte Villares, E. Structural alerts for predicting clastogenic activity of pro-oxidant flavonoid compounds: Quantitative structure-activity relationship study. J. Biomol. Screen 2012, 17, 216–224. [Google Scholar]
- Rishton, G.M. Natural products as a robust source of new drugs and drug leads: Past successes and present day issues. Am. J. Cardiol 2008, 101, 43D–49D. [Google Scholar]
- Dunn, W.J., III. Quantitative structure-activity relationships (QSAR). Chemom. Intell. Lab 1989, 6, 181–190. [Google Scholar]
- Khan, F.; Yadav, D.K.; Maurya, A.; Srivastava, S.K. Modern methods & web resources in drug design & discovery. Lett. Drug Des. Discov 2011, 8, 469–490. [Google Scholar]
- Vedani, A.; Dobler, M.; Spreafico, M.; Peristera, O.; Smiesko, M. VirtualToxLab—in silico prediction of the toxic potential of drugs and environmental chemicals: Evaluation status and internet access protocol. Altex 2007, 24, 153–161. [Google Scholar]
- Castro, E.A. QSPR-QSAR Studies on Desired Properties for Drug Design; Research Signpost: Kerala, India, 2010. [Google Scholar]
- Gasteiger, J.; Engel, T. Chemoinformatics: A Textbook, 1st ed; Wiley-VCH: Weinheim, Germany, 2003. [Google Scholar]
- Alvarez, J.; Shoichet, B. Virtual Screening in Drug Discovery, 1st ed; CRC Press: Boca Raton, FL, USA, 2005. [Google Scholar]
- Schuster, D.; Wolber, G. Identification of bioactive natural products by pharmacophore-based virtual screening. Curr. Pharm. Des 2010, 16, 1666–1681. [Google Scholar]
- Bartalis, J.; Halaweish, F.T. In vitro and QSAR studies of cucurbitacins on HepG2 and HSC-T6 liver cell lines. Bioorg. Med. Chem 2011, 19, 2757–2766. [Google Scholar]
- Bolboacă, S.D.; Pică, E.M.; Cimpoiu, C.V.; Jäntschi, L. Statistical assessment of solvent mixture models used for separation of biological active compounds. Molecules 2008, 13, 1617–1639. [Google Scholar]
- González-Díaz, H.; Torres-Gomez, L.A.; Guevara, Y.; Almeida, M.S.; Molina, R.; Castanedo, N.; Castañedo, N.; Santana, L.; Uriarte, E. Markovian chemicals “in silico” design (MARCH-INSIDE), a promising approach for computer-aided molecular design III: 2.5D indices for the discovery of antibacterials. J. Mol. Model 2005, 11, 116–123. [Google Scholar]
- Gonzalez-Diaz, H.; Prado-Prado, F.; Ubeira, F.M. Predicting antimicrobial drugs and targets with the MARCH-INSIDE approach. Curr. Top. Med. Chem 2008, 8, 1676–90. [Google Scholar]
- Molina, E.; Díaz, H.G.; González, M.P.; Rodríguez, E.; Uriarte, E. Designing antibacterial compounds through a topological substructural approach. J. Chem. Inf. Comput. Sci 2004, 44, 515–521. [Google Scholar]
- González-Díaz, H.; Romaris, F.; Duardo-Sanchez, A.; Pérez-Montoto, L.G.; Prado-Prado, F.; Patlewicz, G.; Ubeira, F.M. Predicting drugs and proteins in parasite infections with topological indices of complex networks: Theoretical backgrounds, applications and legal issues. Curr. Pharm. Des 2010, 16, 2737–2764. [Google Scholar]
- Prado-Prado, F.J.; Gonzalez-Diaz, H.; Santana, L.; Uriarte, E. Unified QSAR approach to antimicrobials. Part 2: Predicting activity against more than 90 different species in order to halt antibacterial resistance. Bioorg. Med. Chem 2007, 15, 897–902. [Google Scholar]
- Prado-Prado, F.J.; Uriarte, E.; Borges, F.; González-Díaz, H. Multi-target spectral moments for QSAR and complex networks study of antibacterial drugs. Eur. J. Med. Chem 2009, 44, 4516–4521. [Google Scholar]
- Gonzalez-Diaz, H.; Prado-Prado, F.J. Unified QSAR and network-based computational chemistry approach to antimicrobials, part 1: Multispecies activity models for antifungals. J. Comput. Chem 2008, 29, 656–667. [Google Scholar]
- Prado-Prado, F.J.; Ubeira, F.M.; Borges, F.; Gonzalez-Diaz, H. Unified QSAR & network-based computational chemistry approach to antimicrobials. II. Multiple distance and triadic census analysis of antiparasitic drugs complex networks. J. Comput. Chem 2010, 31, 164–173. [Google Scholar]
- Prado-Prado, F.J.; Martinez de la Vega, O.; Uriarte, E.; Ubeira, F.M.; Chou, K.C.; Gonzalez-Diaz, H. Unified QSAR approach to antimicrobials. 4. Multi-target QSAR modeling and comparative multi-distance study of the giant components of antiviral drug-drug complex networks. Bioorg. Med. Chem 2009, 17, 569–575. [Google Scholar]
- Gonzalez-Diaz, H.; Prado-Prado, F.; Sobarzo-Sanchez, E.; Haddad, M.; Maurel Chevalley, S.; Valentin, A.; Quetin-Leclercq, J.; Dea-Ayuela, M.A.; Teresa Gomez-Muños, M.; Munteanu, C.R. NL MIND-BEST: A web server for ligands and proteins discovery-theoretic-experimental study of proteins of Giardia lamblia and new compounds active against Plasmodium falciparum. J. Theor. Biol 2011, 276, 229–249. [Google Scholar]
- Jirovetz, L.; Eller, G.; Buchbauer, G.; Schmidt, E.; Denkova, Z.; Stoyanova, A.S.; Nikolova, R.; Geissler, M. Chemical composition, antimicrobial activities and odor descriptions of some essential oils with characteristic. Recent Res. Dev. Agron. Hortic 2006, 2, 1–12. [Google Scholar]
- Fisher, R.A. On an absolute criterion for fitting frequency curves. Messenger Math 1912, 41, 155–160. [Google Scholar]
- Sacks, J.; Ylvisaker, D. Designs for regression problems with correlated errors III. Ann. Math. Stat 1970, 41, 2057–2074. [Google Scholar]
- Jarque, C.M.; Bera, A.K. A test for normality of observations and regression residuals. Int. Stat. Rev 1987, 55, 163–172. [Google Scholar]
- LeRoy, J.S. Negative Binomial and Poisson Distributions Compared. Proceedings of the Casualty Actuarial Society; Casualty Actuarial Society: Arlington, VA, USA, 1960; XLVII, pp. 20–24. Available online: http://www.casact.org/pubs/proceed/proceed60/60020.pdf accessed on 6 August 2011.
- Furman, E. On the convolution of the negative binomial random variables. Stat. Probab. Lett 2007, 77, 169–172. [Google Scholar]
- Jones, A. Health Econometrics. In Handbook of Health Economics; Culyer, A., Newhouse, J., Eds.; Elsevier: Amsterdam, The Netherland, 2000. [Google Scholar]
- Cameron, A.C.; Trivedi, P.K. Regression Analysis of Count Data; Cambridge University Press: London, UK, 1998. [Google Scholar]
- Fisher, R.A. A theoretical distribution for the apparent abundance of different species. J. Anim. Ecol. 1943, 12, 54–58. [Google Scholar]
- Shaked, M. A family of concepts of dependence for bivariate distributions. J. Am. Stat. Assoc 1977, 72, 642–650. [Google Scholar]
- Marshall, A.W.; Olkin, I. Multivariate distributions generated from mixtures of convolution and product families, lecture notes-monograph series. Top. Stat. Depend 1990, 16, 371–393. [Google Scholar]
- Jäntschi, L.; Bolboacă, S.D.; Bălan, M.C.; Sestraş, R.E. Distribution fitting 13. Analysis of independent, multiplicative effect of factors. Application to effect of essential oils extracts from plant species on bacterial species. Application to factors of antibacterial activity of plant species. Bull. Univ. Agric. Sci. Vet. Med. Cluj-Napoca. Anim. Sci. Biotechnol 2011, 68, 323–331. [Google Scholar]
- Kundu, D.; Manglick, A. Discriminating between the log-normal and gamma distributions. Available online: http://home.iitk.ac.in/~kundu/paper93.pdf accessed on 1 August 2011.
- Bolboacă, S.D.; Jäntschi, L. Modelling the property of compounds from structure: Statistical methods for models validation. Environ. Chem. Lett 2008, 6, 175–181. [Google Scholar]
- Kolmogorov, A. Confidence limits for an unknown distribution function. Ann. Math. Stat 1941, 12, 461–463. [Google Scholar]
- Anderson, T.W.; Darling, D.A. Asymptotic theory of certain “goodness-of-fit” criteria based on stochastic processes. Ann. Math. Stat 1952, 23, 193–212. [Google Scholar]
- Fisher, R.A. Combining independent tests of significance. Am. Stat 1948, 2, 30. [Google Scholar]
- Hobza, P.; Kabeláč, M.; Šponer, J.; Mejzlík, P.; Vondrášek, J. Performance of empirical potentials (AMBER, CFF95, CVFF, CHARMM, OPLS, POLTEV), semiempirical quantum chemical methods (AM1, MNDO/M, PM3), and Ab initio Hartree-Fock method for interaction of DNA bases: Comparison with nonempirical beyond Hartree-Fock results. J. Comput. Chem 1997, 18, 1136–1150. [Google Scholar]
- HyperChem, version 8.0; Hypercube Inc: Gainesville, FL, USA, 2007.
- Jäntschi, L. Computer assisted geometry optimization for in silico modeling. Appl. Med. Inform 2011, 29, 11–18. [Google Scholar]
- Jäntschi, L. Genetic Algorithms and Their Applications (in Romanian). Ph.D. Dissertation, University of Agricultural Sciences and Veterinary Medicine, Cluj-Napoca, Romania, 2010. [Google Scholar]
- Jäntschi, L.; Bolboacă, S.D.; Sestraş, R.E. Quantum Mechanics Study on a Series of Steroids Relating Separation with Structure. Proceedings of 17th International Symposium on Separation Sciences: Book of Abstracts, Cluj-Napoca, Romania, September 5–9, 2011; Casa Cărţii de Ştiinţă: Cluj-Napoca, Romania, 2011; p. 59. [Google Scholar]
- DRAGON, version 5.5; Talete srl: Milano, Italy, 2007.
- Pauling, L. The nature of the chemical bond. IV. The energy of single bonds and the relative electronegativity of atoms. J. Am. Chem. Soc 1932, 54, 3570–3582. [Google Scholar]
- Jäntschi, L.; Bolboacă, S.D. Distribution Fitting 2. Pearson-Fisher, Kolmogorov-Smirnov, Anderson-Darling, Wilks-Shapiro, Kramer-von-Misses and Jarque-Bera statistics. Bull. Univ. Agric. Sci. Vet. Med. Cluj-Napoca. Hortic 2009, 66, 691–697. [Google Scholar]
- Grubbs, F. Procedures for detecting outlying observations in samples. Technometrics 1969, 11, 1–21. [Google Scholar]
- Chatterjee, S.; Hadi, A.S. Influential observations, high leverage points, and outliers in linear regression (with discussion). Stat. Sci 1986, 1, 379–416. [Google Scholar]
- Eriksson, L.; Jaworska, J.; Worth, A.P.; Cronin, M.T.D.; McDowell, R.M.; Gramatica, P. Methods for reliability and uncertainty assessment and for applicability evaluations of classification and regression-based QSARs. Environ. Health Perspect 2003, 111, 1361–1375. [Google Scholar]
- Chirico, N.; Gramatica, P. Real external predictivity of QSAR models: How to evaluate it? Comparison of different validation criteria and proposal of using the concordance correlation coefficient. J. Chem. Inf. Model 2011, 51, 2320–2335. [Google Scholar]
- McBride, G.B. A Proposal for Strength-of-Agreement Criteria for Lin’S Concordance Correlation Coefficient. NIWA Client Report: HAM2005-062; National Institute of Water & Atmospheric Research: Hamilton, New Zeeland, May 2005. Available online: http://www.medcalc.org/download/pdf/McBride2005.pdf accessed on 14 March 2012.
- Shi, L.M.; Fang, H.; Tong, W.; Wu, J.; Perkins, R.; Blair, R.M.; Branham, W.S.; Dial, S.L.; Moland, C.L.; Sheehan, D.M. QSAR models using a large diverse set of estrogens. J. Chem. Inf. Comput. Sci 2001, 41, 186–195. [Google Scholar]
- Schüürmann, G.; Ebert, R.U.; Chen, J.; Wang, B.; Kühne, R. External validation and prediction employing the predictive squared correlation coefficient test set activity mean vs. training set activity mean. J. Chem. Inf. Model 2008, 48, 2140–2145. [Google Scholar]
- Consonni, V.; Ballabio, D.; Todeschini, R. Comments on the definition of the Q2 parameter for QSAR validation. J. Chem. Inf. Model 2009, 49, 1669–1678. [Google Scholar]
- Golbraikh, A.; Tropsha, A. Beware of q2! J. Mol. Gr. Mod 2002, 20, 269–276. [Google Scholar]
- Fisher, R.A. The goodness of fit of regression formulae, and the distribution of regression coefficients. J. Royal Stat. Soc 1922, 85, 597–612. [Google Scholar]
- Steiger, J.H. Tests for comparing elements of a correlation matrix. Psychol. Bull 1980, 87, 245–251. [Google Scholar]

| λ | Mode | Mean | Var | StDev | Skew | EKurt | Median | |
|---|---|---|---|---|---|---|---|---|
| Compound (CID) | ||||||||
| Citral (638011) | 14.125 | 14 | 14.125 | 14.125 | 3.758 | 0.266 | 0.071 | 13.457 |
| Geraniol (637566) | 13.750 | 13 | 13.750 | 13.750 | 3.708 | 0.270 | 0.073 | 13.082 |
| Geranyl formate (5282109) | 8.875 | 8 | 8.875 | 8.875 | 2.979 | 0.336 | 0.113 | 8.207 |
| Geranyl acetate (1549026) | 8.200 | 8 | 8.200 | 8.200 | 2.864 | 0.349 | 0.122 | 7.531 |
| Geranyl butyrate (5355856) | 8.714 | 8 | 8.714 | 8.714 | 2.952 | 0.339 | 0.115 | 8.046 |
| Geranyl tiglate (5367785) | 11.625 | 11 | 11.625 | 11.625 | 3.410 | 0.293 | 0.086 | 10.957 |
| Neral (643779) | 13.500 | 13 | 13.500 | 13.500 | 3.674 | 0.272 | 0.074 | 12.932 |
| Nerol (643820) | 11.250 | 11 | 11.250 | 11.250 | 3.354 | 0.298 | 0.089 | 10.582 |
| Nerol acetate (1549025) | 7.333 | 7 | 7.333 | 7.333 | 2.708 | 0.369 | 0.136 | 6.664 |
| Neryl butyrate (5352162) | 10.714 | 10 | 10.714 | 10.714 | 3.273 | 0.306 | 0.093 | 10.046 |
| Neryl propanoate (5365982) | 10.714 | 10 | 10.714 | 10.714 | 3.273 | 0.306 | 0.093 | 10.046 |
| Citronellal (7794) | 14.600 | 14 | 14.600 | 14.600 | 3.821 | 0.262 | 0.068 | 13.932 |
| Citronellyl formate (7778) | 12.143 | 12 | 12.143 | 12.143 | 3.485 | 0.287 | 0.082 | 11.475 |
| Citronellyl acetate (9017) | 7.286 | 7 | 7.286 | 7.286 | 2.699 | 0.370 | 0.137 | 6.617 |
| Citronellyl butyrate (8835) | 8.167 | 8 | 8.167 | 8.167 | 2.858 | 0.350 | 0.122 | 7.498 |
| Citronellyl isobutyrate (60985) | 8.200 | 8 | 8.200 | 8.200 | 2.864 | 0.349 | 0.122 | 7.531 |
| Citronellyl propionate (8834) | 14.333 | 14 | 14.333 | 14.333 | 3.786 | 0.264 | 0.070 | 13.665 |
| Hydroxycitronellal (7888) | 18.750 | 18 | 18.750 | 18.750 | 4.330 | 0.231 | 0.053 | 18.083 |
| Rose oxide (27866) | 12.800 | 12 | 12.800 | 12.800 | 3.578 | 0.280 | 0.078 | 12.132 |
| Eugenol (3314) | 28.250 | 28 | 28.250 | 28.250 | 5.315 | 0.188 | 0.035 | 27.583 |
| Sulfametrole (64939) | 19.200 | 19 | 19.200 | 19.200 | 4.382 | 0.228 | 0.052 | 18.533 |
| Oil | ||||||||
| Citronella | 9.750 | 9 | 9.750 | 9.750 | 3.122 | 0.320 | 0.103 | 9.082 |
| Geranium Africa | 13.250 | 13 | 13.250 | 13.250 | 3.640 | 0.275 | 0.075 | 12.582 |
| Geranium Bourbon | 12.500 | 12 | 12.500 | 12.500 | 3.536 | 0.283 | 0.080 | 11.832 |
| Geranium China | 13.625 | 13 | 13.625 | 13.625 | 3.691 | 0.271 | 0.073 | 12.957 |
| Helichrysum | 10.667 | 10 | 10.667 | 10.667 | 3.266 | 0.306 | 0.094 | 9.999 |
| Palmarosa | 11.625 | 11 | 11.625 | 11.625 | 3.410 | 0.293 | 0.086 | 10.957 |
| Rose | 12.750 | 12 | 12.750 | 12.750 | 3.571 | 0.280 | 0.078 | 12.082 |
| Verbena | 16.500 | 16 | 16.500 | 16.500 | 4.062 | 0.246 | 0.061 | 15.833 |
| Mixture | ||||||||
| Tetracycline hydrochloride | 15.143 | 15 | 15.143 | 15.143 | 3.891 | 0.257 | 0.066 | 14.476 |
| Ciproxin | 26.000 | 26 | 26.000 | 26.000 | 5.099 | 0.196 | 0.038 | 25.333 |
| Set | CID | Y | ŶDragon | ResDragon | ŶSAPF | ResSAPF |
|---|---|---|---|---|---|---|
| Training | 1549025 | 1.9924 | 2.0070 | −0.0146 | 2.0761 | −0.0836 |
| Training | 8835 | 2.1001 | 2.0564 | 0.0437 | 2.1461 | −0.0460 |
| Training | 60985 | 2.1041 | 2.0768 | 0.0273 | 2.0553 | 0.0488 |
| Training | 5282109 | 2.1832 | 2.2596 | −0.0764 | 2.3267 | −0.1435 |
| Training | 643820 | 2.4204 | 2.6106 | −0.1902 | 2.7127 | −0.2923 |
| Training | 7778 | 2.4968 | 2.4132 | 0.0835 | 2.2816 | 0.2151 |
| Training | 27866 | 2.5494 | 2.5905 | −0.0411 | 2.4957 | 0.0538 |
| Training | 637566 | 2.6210 | 2.6106 | 0.0104 | 2.7127 | −0.0917 |
| Training | 638011 | 2.6479 | 2.7061 | −0.0582 | 2.6042 | 0.0437 |
| Training | 8842 | 2.6741 | 2.6435 | 0.0307 | 2.5713 | 0.1029 |
| Training | 7794 | 2.6810 | 2.6929 | −0.0118 | 2.6430 | 0.0380 |
| Training | 7888 | 2.9312 | 2.7346 | 0.1966 | 2.8638 | 0.0674 |
| Training | 64939 | 2.9549 | 2.8674 | 0.0875 | ||
| Test | 1549026 | 2.1041 | 2.0070 | 0.0971 | 2.2012 | −0.0971 |
| Test | 5355856 | 2.1650 | 1.9271 | 0.2379 | 2.2830 | −0.1180 |
| Test | 5352162 | 2.3716 | 1.9271 | 0.4445 | 2.7847 | −0.4132 |
| Test | 5367785 | 2.4532 | 1.8661 | 0.5870 | 2.4642 | −0.0111 |
| Test | 643779 | 2.6027 | 2.7061 | −0.1034 | 2.6006 | 0.0021 |
| Test | 8834 | 2.6626 | 2.4108 | 0.2518 | 2.6207 | 0.0418 |
| Test | 3314 | 3.3411 | 2.7843 | 0.5568 | 3.3685 | −0.0274 |
| External | 9017 | 1.9859 | 2.1432 | −0.1572 | 2.0053 | −0.0194 |
| External | 5365982 | 2.3716 | 2.2688 | 0.1028 | 2.2889 | 0.0827 |
| Parameter (Abbreviation) | Dragon–Equation(1)–n = 21 | SAPF–Equation(2)–n = 22 | ||||
|---|---|---|---|---|---|---|
| Root-mean-square error (RMSE) | 0.2314 | 0.1357 | ||||
| Mean absolute error (MAE) | 0.1582 | 0.0967 | ||||
| Mean Absolute Percentage Error (MAPE) | 0.0628 | 0.0403 | ||||
| Standard error of prediction (SEP) | 0.2371 | 0.0628 | ||||
| Relative error of prediction (REP%) | 9.2964 | 5.4523 | ||||
| Predictive Power of the Model | ||||||
| Q2F1 | 0.2121 * | 0.8436 * | ||||
| Q2F2 | 0.2041 * | 0.8421 * | ||||
| Q2F3 | n.a. | 0.7742 * | ||||
| ρc-TR | 0.9457 a | 0.9063 c | ||||
| ρc-TS | 0.4885 b | 0.9219 d | ||||
| Fisher’s Predictive Power | TS | EX e | TS + EX f | TS | EX | TS + EX |
| n | 7 | 2 | 9 | 7 | 2 | 9 |
| t-value | 3.1148 | −0.2095 | 2.5071 | −1.5344 | 0.6198 | −1.2830 |
| p-value | 0.0104 | 0.4343 | 0.0230 | 0.0879 | 0.3234 | 0.1234 |
| SA | EF | EC | PV | PA | Ss | KP | CA | n | ||
|---|---|---|---|---|---|---|---|---|---|---|
| Compound (CID) | ||||||||||
| 1 | Citral (638011) | 15 | 23 | 11 | 9 | 10 | 8 | 9 | 28 | 8 |
| 2 | Geraniol (637566) | 15 | 12 | 15 | 12 | 11 | 10 | 10 | 25 | 8 |
| 3 | Geranyl formate (5282109) | 10 | 9 | 7 | 8 | 8 | 7 | 7 | 15 | 8 |
| 4 | Geranyl acetate (1549026) | 10 | 8 | 7 | NIO | NIO | 7 | NIO | 9 | 5 |
| 5 | Geranyl butyrate (5355856) | 10 | 11 | 7 | NIO | 9 | 7 | 7 | 10 | 7 |
| 6 | Geranyl tiglate (5367785) | 17 | 10 | 11 | 9 | 8 | 8 | 15 | 15 | 8 |
| 7 | Neral (643779) | 15 | 20 | 10 | 6 | 12 | 10 | 10 | 25 | 8 |
| 8 | Nerol (643820) | 11 | 8 | 10 | 10 | 10 | 7 | 7 | 27 | 8 |
| 9 | Nerol acetate (1549025) | 8 | NIO | 7 | 7 | 7 | 8 | 7 | NIO | 6 |
| 10 | Neryl butyrate (5352162) | 25 | 8 | 8 | 8 | NIO | 8 | 8 | 10 | 7 |
| 11 | Neryl propanoate (5365982) | 17 | 10 | NIO | 7 | 8 | 9 | 10 | 14 | 7 |
| 12 | Citronellal (7794) | 25 | 18 | NIO | 9 | NIO | 7 | 14 | NIO | 5 |
| 13 | Citronellyl formate (7778) | 18 | 20 | 10 | 8 | 9 | 7 | NIO | 13 | 7 |
| 14 | Citronellyl acetate (9017) | 10 | 6 | NIO | 6 | 7 | 6 | 7 | 9 | 7 |
| 15 | Citronellyl butyrate (8835) | 8 | 8 | NIO | NIO | 8 | 7 | 8 | 10 | 6 |
| 16 | Citronellyl isobutyrate (60985) | 8 | 10 | 9 | 7 | NIO | NIO | 7 | NIO | 5 |
| 17 | Citronellyl propionate (8834) | 15 | 20 | NIO | NIO | 10 | 15 | 11 | 15 | 6 |
| 18 | Hydroxycitronellal (7888) | 20 | 20 | 23 | 16 | 17 | 15 | 14 | 25 | 8 |
| 19 | Rose oxide (27866) | 8 | 10 | NIO | 11 | 7 | NIO | NIO | 28 | 5 |
| 20 | Eugenol (3314) | 30 | 30 | 28 | 28 | 25 | 25 | 28 | 32 | 8 |
| 21 | Sulfametrole (64939) | 27 | 27 | 11 | 23 | NIO | 8 | NIO | NIO | 5 |
| 32 | Citronellol (8842) | 25 | 18 | NIO | 8 | NIO | 7 | NIO | NIO | 4 |
| Oil | ||||||||||
| 22 | Citronella | 10 | 10 | 7 | 10 | 7 | 7 | 7 | 20 | 8 |
| 23 | Geranium Africa | 16 | 12 | 10 | 10 | 10 | 9 | 11 | 28 | 8 |
| 24 | Geranium Bourbon | 13 | 12 | 8 | 12 | 10 | 10 | 10 | 25 | 8 |
| 25 | Geranium China | 20 | 13 | 14 | 9 | 9 | 9 | 10 | 25 | 8 |
| 26 | Helichrysum | 20 | 13 | 8 | NIO | 9 | NIO | 7 | 7 | 6 |
| 27 | Palmarosa | 8 | 13 | 12 | 9 | 11 | 10 | 10 | 20 | 8 |
| 28 | Rose | 20 | 15 | 10 | 10 | 8 | 9 | 10 | 20 | 8 |
| 29 | Verbena | 27 | 25 | 10 | 13 | 10 | 12 | 10 | 25 | 8 |
| Mixture | ||||||||||
| 30 | Tetracycline hydrochloride | 15 | 22 | 11 | 13 | 15 | 10 | 20 | NIO | 7 |
| 31 | Ciproxin | 35 | 33 | 22 | 25 | 32 | 10 | 25 | NIO | 7 |
| Parameter (Abbreviation) | Formula [ref] | Remarks |
|---|---|---|
| Root-mean-square error (RMSE) | RMSE > MAE → variation in the errors exist | |
| Mean absolute error (MAE) | ||
| Mean Absolute Percentage Error (MAPE) n | MAPE ~ 0 → perfect fit | |
| Standard error of prediction (SEP) | Lower value indicate a good model | |
| Relative error of prediction (REP%) | Lower value indicate a good model | |
| Concordance analysis (ρc) | [61] | Strength of agreement [62]: >0.99 almost perfect; (0.95; 0.99) substantial; (0.90; 0.95) moderate; <0.90 poor |
| Predictive Power of the Model Prediction is considered accurate if the predictive power of the model is > 0.6 [66] | [63] | Prediction power relative to mean value of observable in training set |
| [64] | Prediction power relative to mean value of observable in test set | |
| [65] | Overall prediction weighted by test set sample size relative to observable weighted by mean of observed value in training set weighted by sample size in training set | |
| Predictive Power: Fisher’s approach | [67] | Evaluate if the mean of residual is statistically different by the expected value (0) |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Sestraş, R.E.; Jäntschi, L.; Bolboacă, S.D. Poisson Parameters of Antimicrobial Activity: A Quantitative Structure-Activity Approach. Int. J. Mol. Sci. 2012, 13, 5207-5229. https://doi.org/10.3390/ijms13045207
Sestraş RE, Jäntschi L, Bolboacă SD. Poisson Parameters of Antimicrobial Activity: A Quantitative Structure-Activity Approach. International Journal of Molecular Sciences. 2012; 13(4):5207-5229. https://doi.org/10.3390/ijms13045207
Chicago/Turabian StyleSestraş, Radu E., Lorentz Jäntschi, and Sorana D. Bolboacă. 2012. "Poisson Parameters of Antimicrobial Activity: A Quantitative Structure-Activity Approach" International Journal of Molecular Sciences 13, no. 4: 5207-5229. https://doi.org/10.3390/ijms13045207