Inhibitory Action of Antidepressants on Mouse Betaine/GABA Transporter (BGT1) Heterologously Expressed in Cell Cultures
Abstract
:1. Introduction
2. Results
2.1. Establishment of the GAT-Expressing Cell Lines
2.2. Comparison of the Inhibitory Potency of Antidepressants
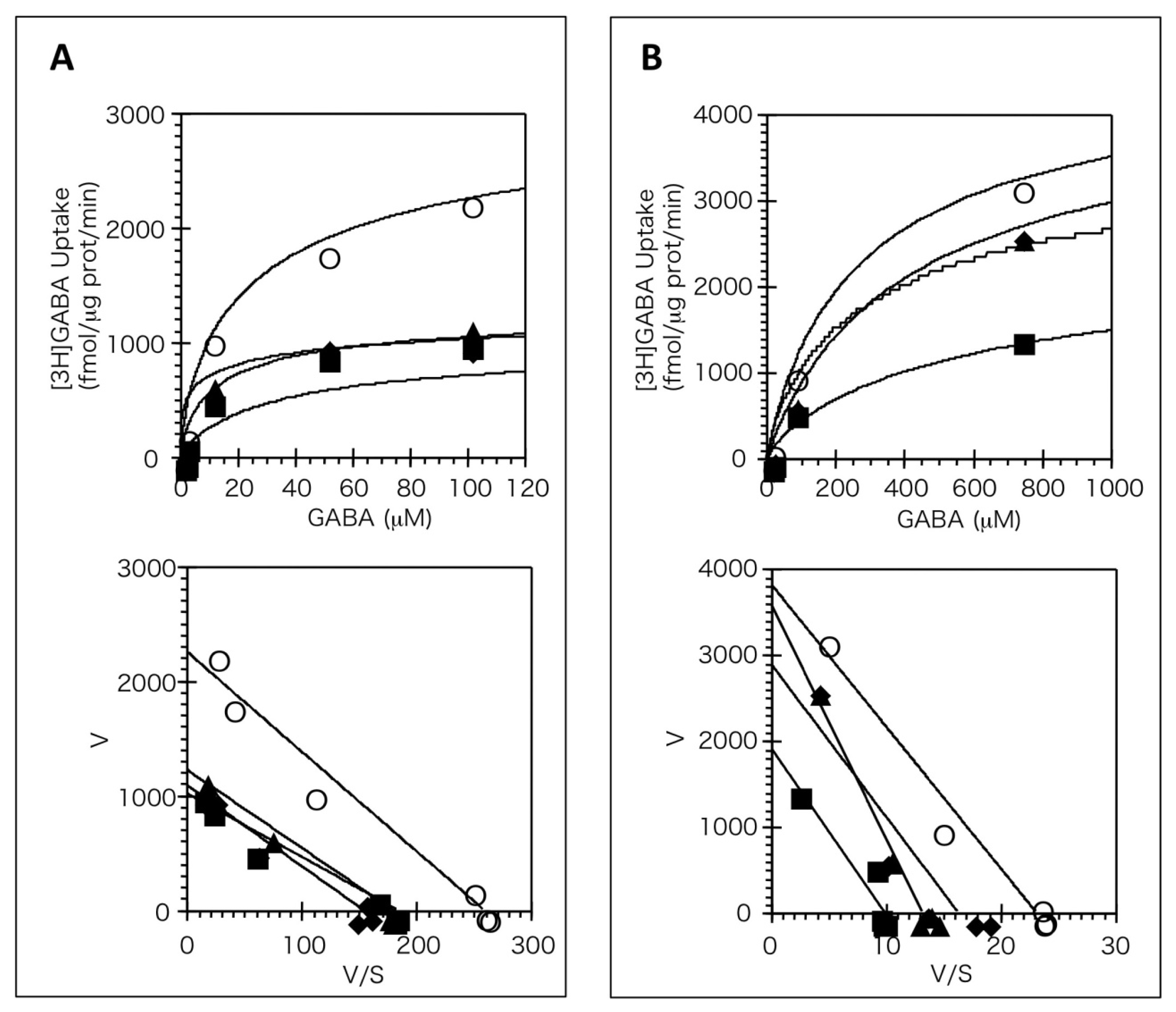
2.3. Kinetic Analyses of the Effects of Antidepressants on BGT1
3. Discussion
4. Experimental Section
4.1. Materials
4.2. cDNA Constructs
4.3. Cell Culture and Expression
4.4. Uptake Assay
4.5. Statistical Analysis
5. Conclusions
Supplementary Information
ijms-13-02578-s001.pdfAcknowledgments
References
- Iversen, L.L. Role of transmitter uptake mechanisms in synaptic neurotransmission. Br. J. Pharmacol 1971, 41, 571–591. [Google Scholar]
- Yamauchi, A.; Uchida, S.; Kwon, H.M.; Preston, A.S.; Robey, R.B.; Garcia-Perez, A.; Burg, M.B.; Handler, J.S. Cloning of a Na+- and Cl−-dependent betaine transporter that is regulated by hypertonicity. J. Biol. Chem 1992, 267, 649–652. [Google Scholar]
- Borden, L.A.; Smith, K.E.; Gustafson, E.L.; Branchek, T.A.; Weinshank, R.L. Cloning and expression of a betaine/GABA transporter from human brain. J. Neurochem 1995, 64, 977–984. [Google Scholar]
- Borden, L.A. GABA transporter heterogeneity: Pharmacology and cellular localization. Neurochem. Int 1996, 29, 335–356. [Google Scholar]
- Amara, S.G.; Kuhar, M.J. Neurotransmitter transporters: Recent progress. Ann. Rev. Neurosci 1993, 16, 73–93. [Google Scholar]
- Dalby, N.O. Inhibition of γ-aminobutyric acid uptake: Anatomy, physiology and effects against epileptic seizures. Eur. J. Pharmacol 2003, 479, 127–137. [Google Scholar]
- Madsen, K.K.; White, H.S.; Schousboe, A. Neuronal and non-neuronal GABA transporters as targets for antiepileptic drugs. Pharmacol. Ther 2010, 125, 394–401. [Google Scholar]
- Liu, Q.-R.; Lopez-Corcuera, B.; Mandiyan, S.; Nelson, H.; Nelson, N. Molecular characterization of four pharmacologically distinct γ-aminobutyric acid transporters in mouse brain. J. Biol. Chem 1993, 268, 2106–2112. [Google Scholar]
- Schousboe, A.; Larsson, O.M.; Sarup, A.; White, H.S. Role of the betaine/GABA transporter (BGT-1/GAT2) for the control of epilepsy. Eur. J. Pharmacol 2004, 500, 281–287. [Google Scholar]
- Langer, Z.S.; Schoemaker, H. Effects of antidepressants on monoamine transporters. Prog. Neuropsychopharmacol. Biol. Psychiat 1988, 12, 193–216. [Google Scholar]
- Todd, A.J. Neuronal circuitry for pain processing in the dorsal horn. Nat. Rev. Neurosci 2010, 11, 823–836. [Google Scholar]
- Mico, J.A.; Ardid, D.; Berrocoso, E.; Eschalier, A. Antidepressants and pain. Trends Pharmacol. Sci 2006, 27, 348–354. [Google Scholar]
- Nakashita, M.; Sasaki, K.; Sakai, N.; Saito, N. Effects of tricyclic and tetracyclic antidepressants on the three types of GABA transporter. Neurosci. Res 1997, 29, 87–91. [Google Scholar]
- Yamashita, A.; Singh, S.K.; Kawate, T.; Jin, Y.; Gouaux, E. Crystal structure of a bacterial homologue of Na+/Cl− -dependent neurotransmitter transporters. Nature 2005, 437, 215–223. [Google Scholar]
- Singh, S.K.; Yamashita, A.; Gouaux, E. Antidepressant binding site in a bacterial homolog of neurotransmitter transporters. Nature 2007, 448, 952–956. [Google Scholar]
- Zhou, Z.; Zhen, J.; Karpowich, N.K.; Goetz, R.M.; Law, C.J.; Reith, M.E.A.; Wang, D.N. LeuT-desipramine structure reveals how antidepressants block neurotransmitter reuptake. Science 2007, 317, 1390–1393. [Google Scholar]
- Nyola, A.; Karpowich, N.K.; Zhen, J.; Marden, J.; Reith, M.E.; Wang, D.-N. Substrate and drug binding sites in LeuT. Curr. Opin. Struct. Biol 2010, 20, 415–422. [Google Scholar]
- Skovstrup, S.; Taboureau, O.; Brauner-Osborne, H.; Jorgensen, F.S. Homology modeling of the GABA transporter and analysis of tiagabine binding. Chem. Med. Chem 2010, 5, 986–1000. [Google Scholar]
- Kardos, J.; Pallo, A.; Bencsura, A.; Simon, A. Assessing structure, function and druggability of major inhibitory neurotransmitter γ-aminobutyrate symporter subtypes. Curr. Med. Chem 2010, 17, 2203–2213. [Google Scholar]
- Cherubino, F.; Miszner, A.; Renna, M.D.; Sangaletti, R.; Giovannardi, S.; Bossi, E. GABA transporter lysine 448: A key residue for tricyclic antidepressants interaction. Cell. Mol. Life Sci 2009, 66, 3797–3808. [Google Scholar]
- Kvist, T.; Christiansen, B.; Jensen, A.A.; Brauner-Osborne, H. The four human γ-aminobutyric acid (GABA) transporters: Pharmacological characterization and validation of a highly efficient screening assay. Comb. Chem. High Throughput Screen 2009, 12, 241–249. [Google Scholar]
- Sato, T.; Kitayama, S.; Mitsuhata, C.; Ikeda, T.; Morita, K.; Dohi, T. Selective inhibition of monoamine neurotransmitter transporters by synthetic local anesthetics. Naunyn-Schmiedeberg’s Arch. Pharmacol 2000, 361, 214–220. [Google Scholar]
- Sogawa, C.; Sogawa, N.; Tagawa, J.; Fujino, A.; Ohyama, K.; Asanuma, M.; Funada, M.; Kitayama, S. 5-Methoxy-N,N-diisopropyltryptamine (Foxy), a selective and high affinity inhibitor of serotonin transporter. Toxicol. Lett 2007, 170, 75–82. [Google Scholar]
- Rudnick, G. What is an antidepressant binding site doing in a bacterial transporter? ACS Chem. Biol 2007, 2, 606–609. [Google Scholar]
- Barker, E.L.; Kimmel, H.L.; Blakely, R.D. Chimeric human and rat serotonin transporters reveal domains involved in recognition of transporter ligands. Mol. Pharmacol 1994, 46, 799–807. [Google Scholar]
- Barker, E.L.; Blakely, R.D. Identification of a single amino acid, phenylalanine 586, that is responsible for high affinity interactions of tricyclic antidepressants with the human serotonin transporter. Mol. Pharmacol 1996, 50, 957–965. [Google Scholar]
- Lehre, A.C.; Rowley, N.M.; Zhou, Y.; Holmseth, S.; Guo, C.; Holen, T.; Hua, R.; Laake, P.; Olofsson, A.M.; Poblete-Naredo, I. Deletion of the betaine-GABA transporter (BGT1; slc6a12) gene does not affect seizure thresholds of adult mice. Epilepsy Res 2011, 95, 70–81. [Google Scholar]
- Zhou, Y.; Holmseth, S.; Hua, R.; Lehre, A.C.; Olofsson, A.M.; Poblete-Naredo, I.; Kempson, S.A.; Danbolt, N.C. The betaine-GABA transporter (BGT1, slc6a12) is predominantly expressed in the liver and at lower levels in the kidneys and at the brain surface. Am. J. Physiol. Renal Physiol 2012, 302, F316–F328. [Google Scholar]
- Gregor, P.; Patel, A.; Shimada, S.; Lin, C.L.; Rochelle, J.M.; Kitayama, S.; Seldin, M.F.; Uhl, G.R. Murine serotonin transporter: sequence and localization to chromosome 11. Mammal. Genome 1993, 4, 283–284. [Google Scholar]
- Carninci, P.; Kasukawa, T.; Katayama, S.; Gough, J.; Frith, M.C.; Maeda, N.; Oyama, R.; Ravasi, T.; Lenhard, B.; Wells, C.; et al. The Transcriptional landscape of the mammalian genome. Science 2005, 309, 1559–1563. [Google Scholar]
- Kitayama, S.; Shimada, S.; Xu, H.; Markham, L.; Donovan, M.D.; Uhl, G.R. Dopamine transporter site-directed mutations differentially alter substrate transport and cocaine binding. Proc. Natl. Acad. Sci. USA 1992, 89, 7782–7785. [Google Scholar]
- Sugimura, M.; Kitayama, S.; Morita, K.; Irifune, M.; Takarada, T.; Kawahara, M.; Dohi, T. Effects of volatile and intravenous anesthetics on the uptake of GABA, glutamate and dopamine by their transporters heterologously expressed in COS cells and in rat brain synaptosomes. Toxicol. Lett 2001, 123, 69–76. [Google Scholar]
| Drugs | IC50 (μM) | ||||
|---|---|---|---|---|---|
| mBGT1 | mGAT1 | mGAT2 | mGAT3 | rSERT | |
| Amitriptyline | 33.0 ± 9.9 * | 100.6 ± 11.7 | 45.8 | 61.5 | 0.14 |
| Amoxapine | 78.3 ± 9.5 * | 287.9 ± 80.5 | 134.6 | 195.4 | 1.62 |
| Clomipramine | 27.2 ± 6.1 | 93.3 ± 39.8 | 41.8 | 40.2 | 0.05 |
| Desipramine | 75.3 ± 6.3 * | 268.4 ± 36.7 | 122.5 | 80.3 | 1.23 |
| Doxepine | 59.9 ± 11.2 ** | 222.1 ± 14.7 | 129.2 | 115.5 | 0.58 |
| Imipramine | 44.5 ± 2.2 * | 222.5 ± 58.5 | 105.9 | 108.1 | 0.75 |
| Maprotilline | 61.3 ± 2.1 ** | 123.5 ± 8.4 | 98.0 | 106.1 | 27.5 |
| Mianserine | 57.4 ± 3.9 ** | 221.6 ± 9.9 | 131.1 | 91.0 | 7.63 |
| Nortriptyline | 47.0 ± 2.1 * | 135.7 ± 21.9 | 87.1 | 74.1 | 0.63 |
| Protriptyline | 43.6 ± 10.2 ** | 166.8 ± 22.1 | 77.2 | 113.0 | 0.91 |
| Trimipramine | 21.8 ± 3.8 * | 181.4 ± 72.9 | 85.6 | 64.6 | 5.89 |
| Transporter | Drugs | Km (μM) | Vmax (ratio to control #) |
|---|---|---|---|
| mGAT1 | Control | 13.04 ± 1.87 | 1.00 |
| Maprotiline | 9.38 ± 2.44 | 0.48 ± 0.04 * | |
| Mianserine | 9.93 ± 2.67 | 0.47 ± 0.04 * | |
| Trimipramine | 9.33 ± 2.39 | 0.37 ± 0.05 * | |
| mBGT1 | Control | 202.2 ± 34.8 | 1.00 |
| Maprotiline | 242.7 ± 56.2 | 0.47 ± 0.06 * | |
| Mianserine | 305.2 ± 56.4 * | 0.90 ± 0.03 | |
| Trimipramine | 254.9 ± 34.4 | 0.86 ± 0.06 | |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Gerile; Sogawa, C.; Ohyama, K.; Masuko, T.; Kusama, T.; Morita, K.; Sogawa, N.; Kitayama, S. Inhibitory Action of Antidepressants on Mouse Betaine/GABA Transporter (BGT1) Heterologously Expressed in Cell Cultures. Int. J. Mol. Sci. 2012, 13, 2578-2589. https://doi.org/10.3390/ijms13032578
Gerile, Sogawa C, Ohyama K, Masuko T, Kusama T, Morita K, Sogawa N, Kitayama S. Inhibitory Action of Antidepressants on Mouse Betaine/GABA Transporter (BGT1) Heterologously Expressed in Cell Cultures. International Journal of Molecular Sciences. 2012; 13(3):2578-2589. https://doi.org/10.3390/ijms13032578
Chicago/Turabian StyleGerile, Chiharu Sogawa, Kazumi Ohyama, Takashi Masuko, Tadashi Kusama, Katsuya Morita, Norio Sogawa, and Shigeo Kitayama. 2012. "Inhibitory Action of Antidepressants on Mouse Betaine/GABA Transporter (BGT1) Heterologously Expressed in Cell Cultures" International Journal of Molecular Sciences 13, no. 3: 2578-2589. https://doi.org/10.3390/ijms13032578





