Antioxidant and Hepatoprotective Effects of the Red Ginseng Essential Oil in H2O2-Treated HepG2 Cells and CCl4-Treated Mice
Abstract
:1. Introduction
2. Results and Discussion
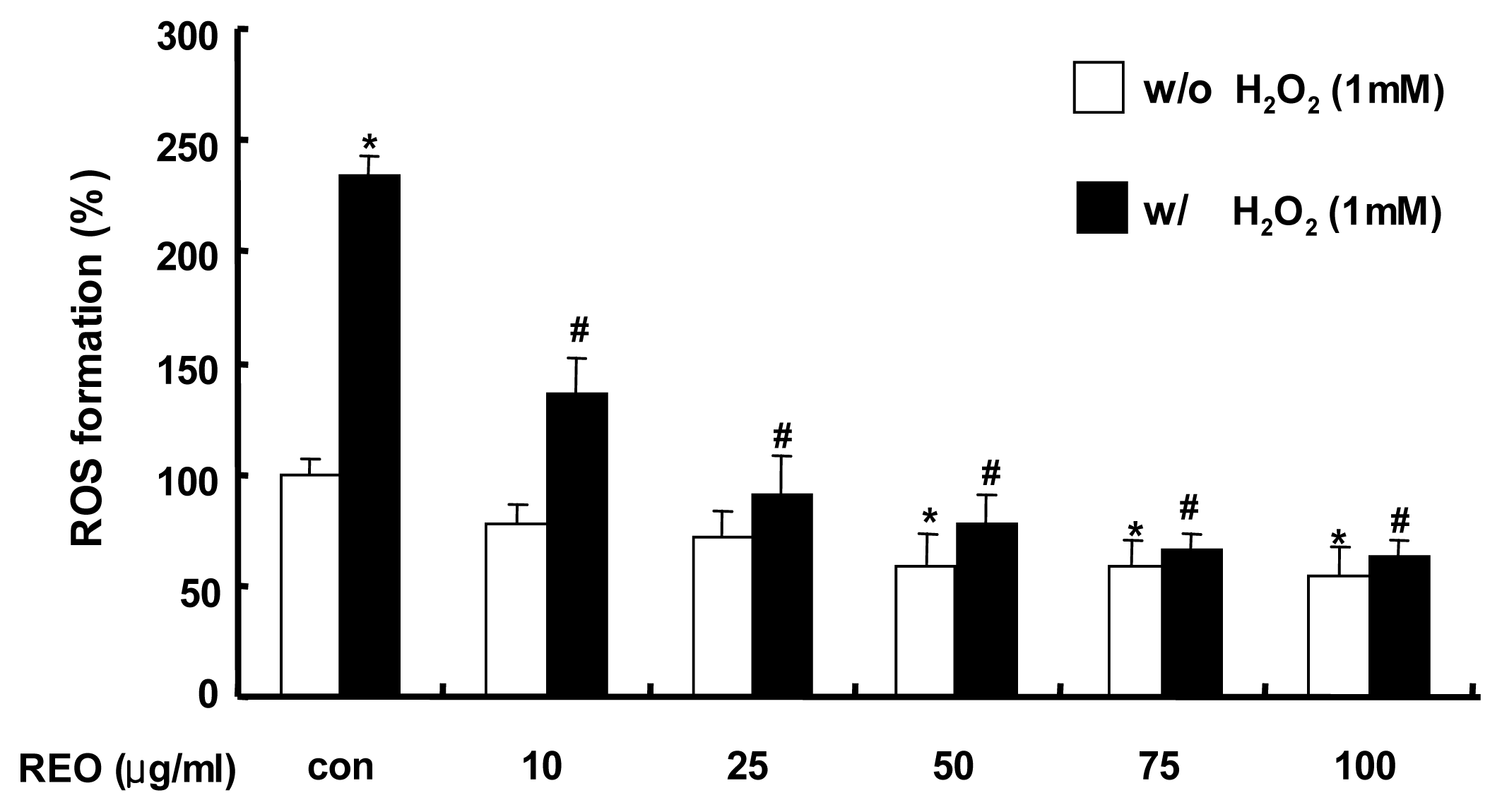
2.1. Inhibition of ROS Production in HepG2 Cells
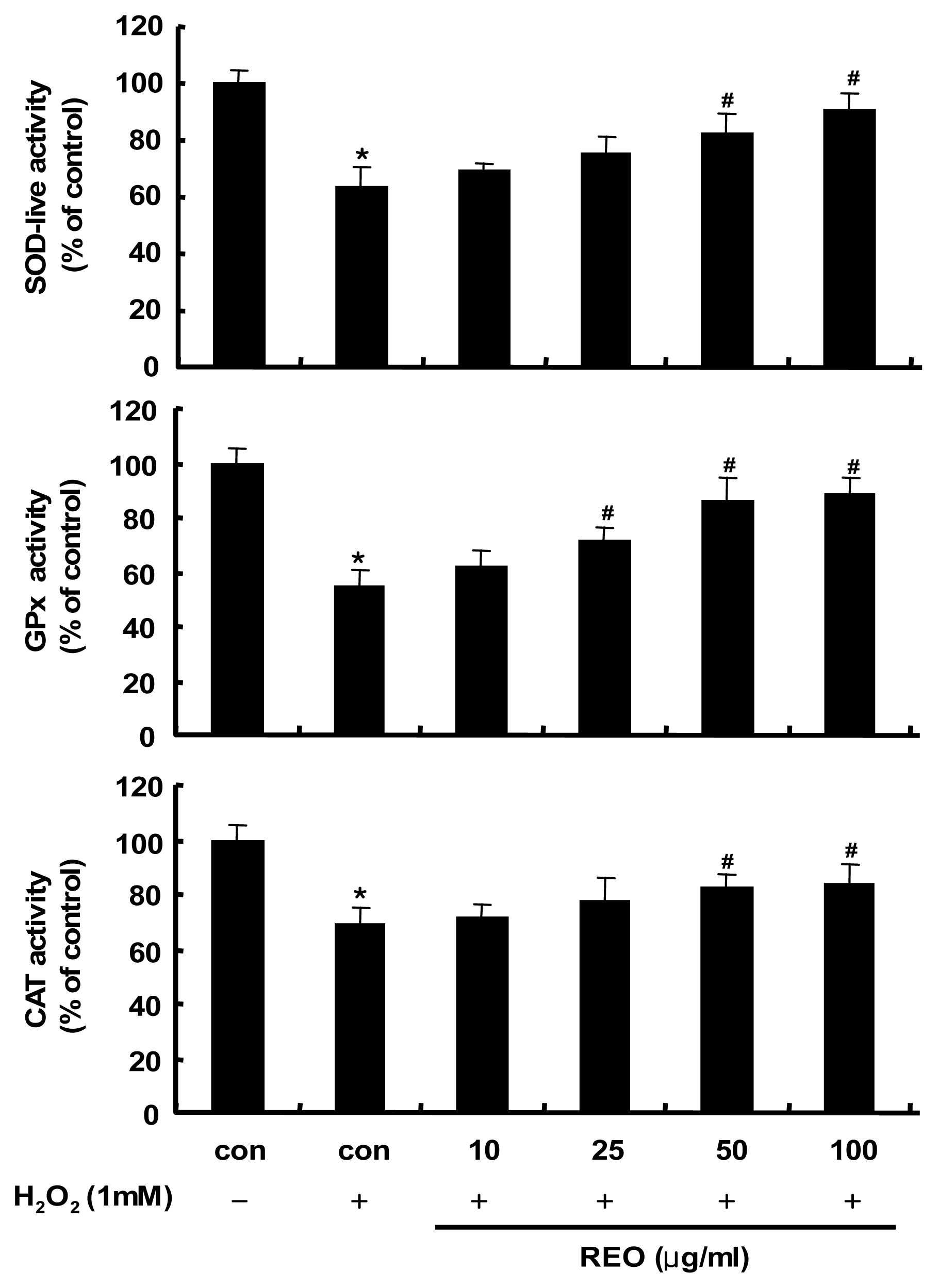
2.2. Induction of Antioxidant Enzyme Activities in HepG2 Cells
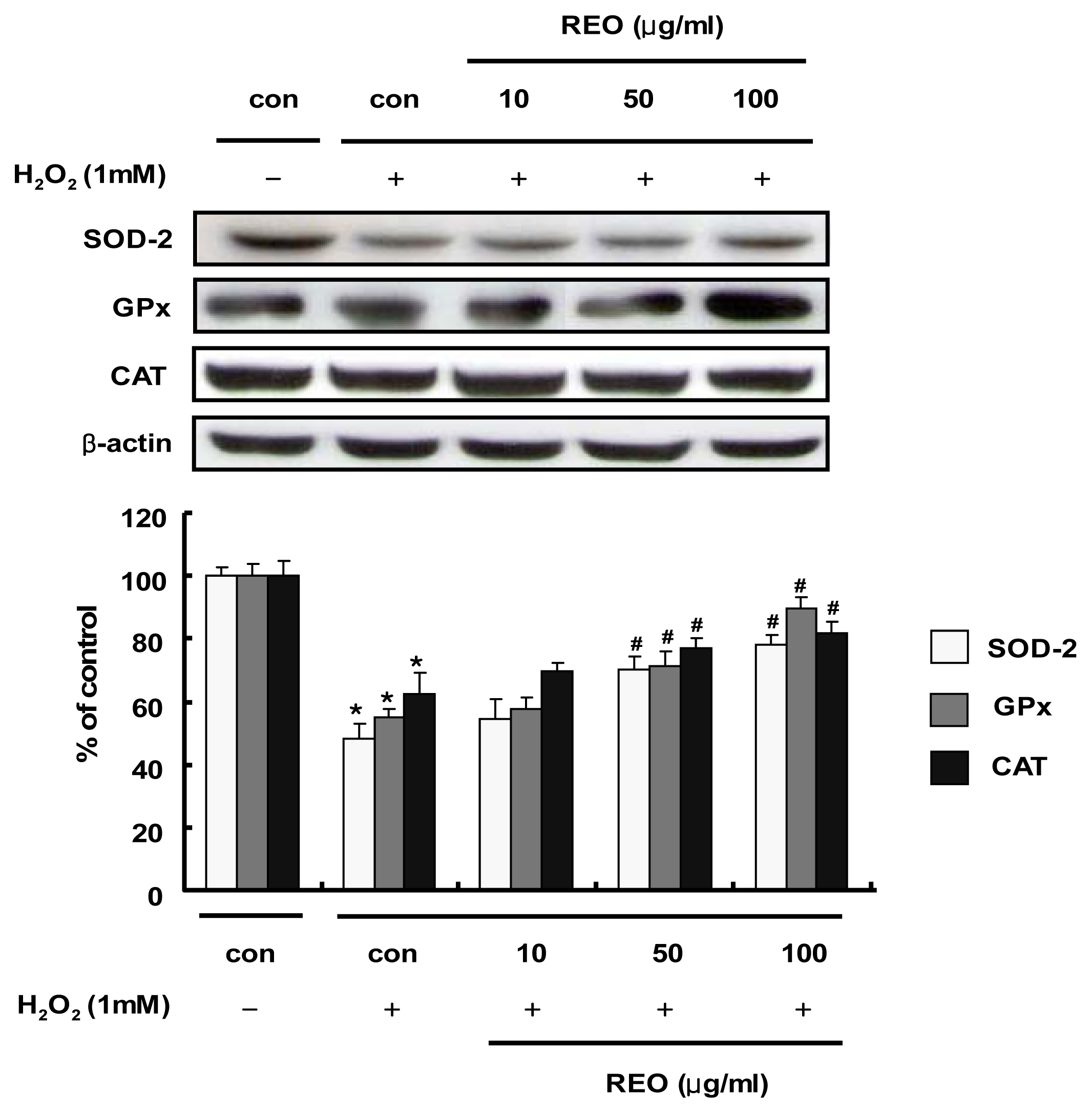
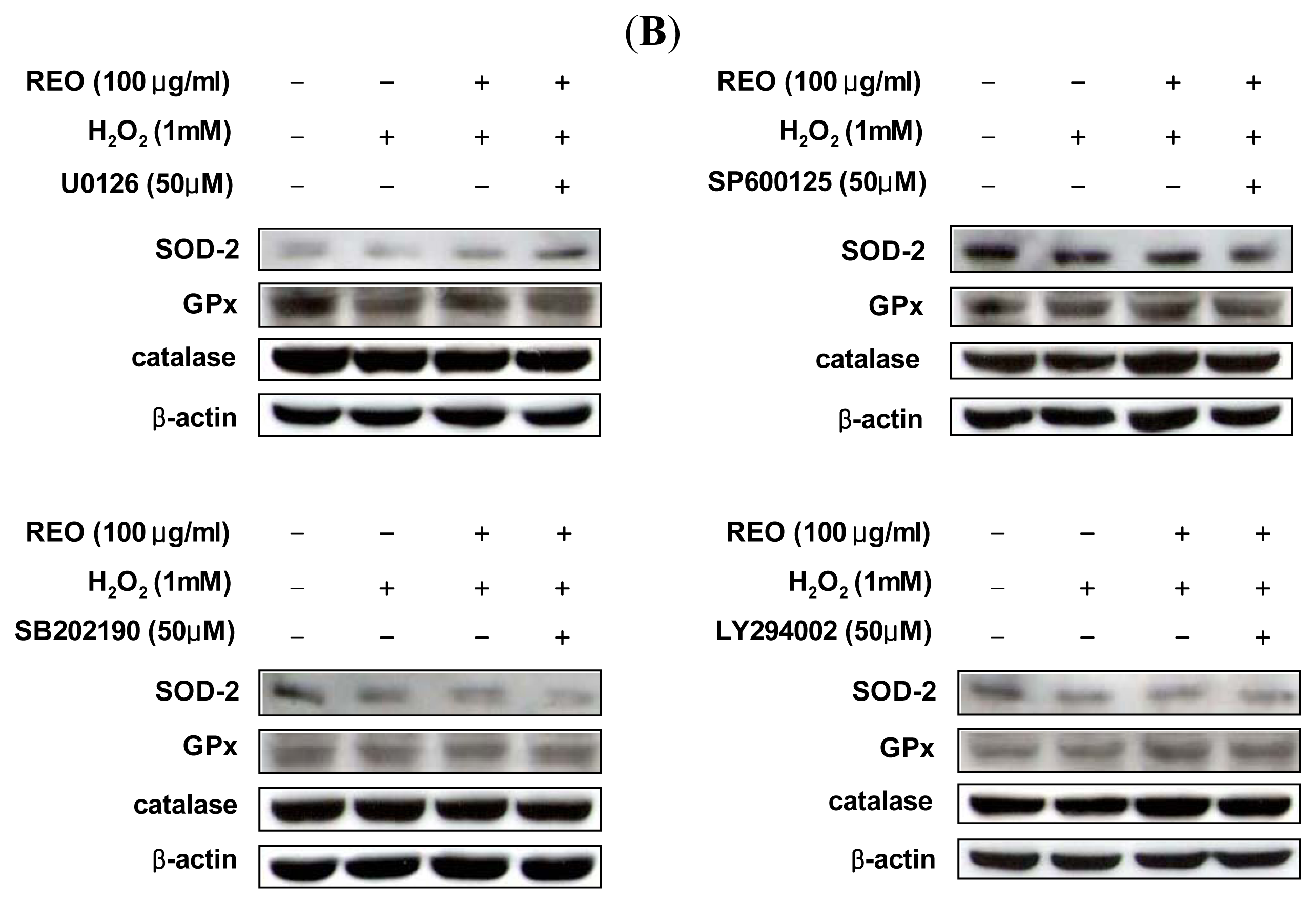
2.3. Induction of Antioxidant Enzyme Expressions in HepG2 Cells
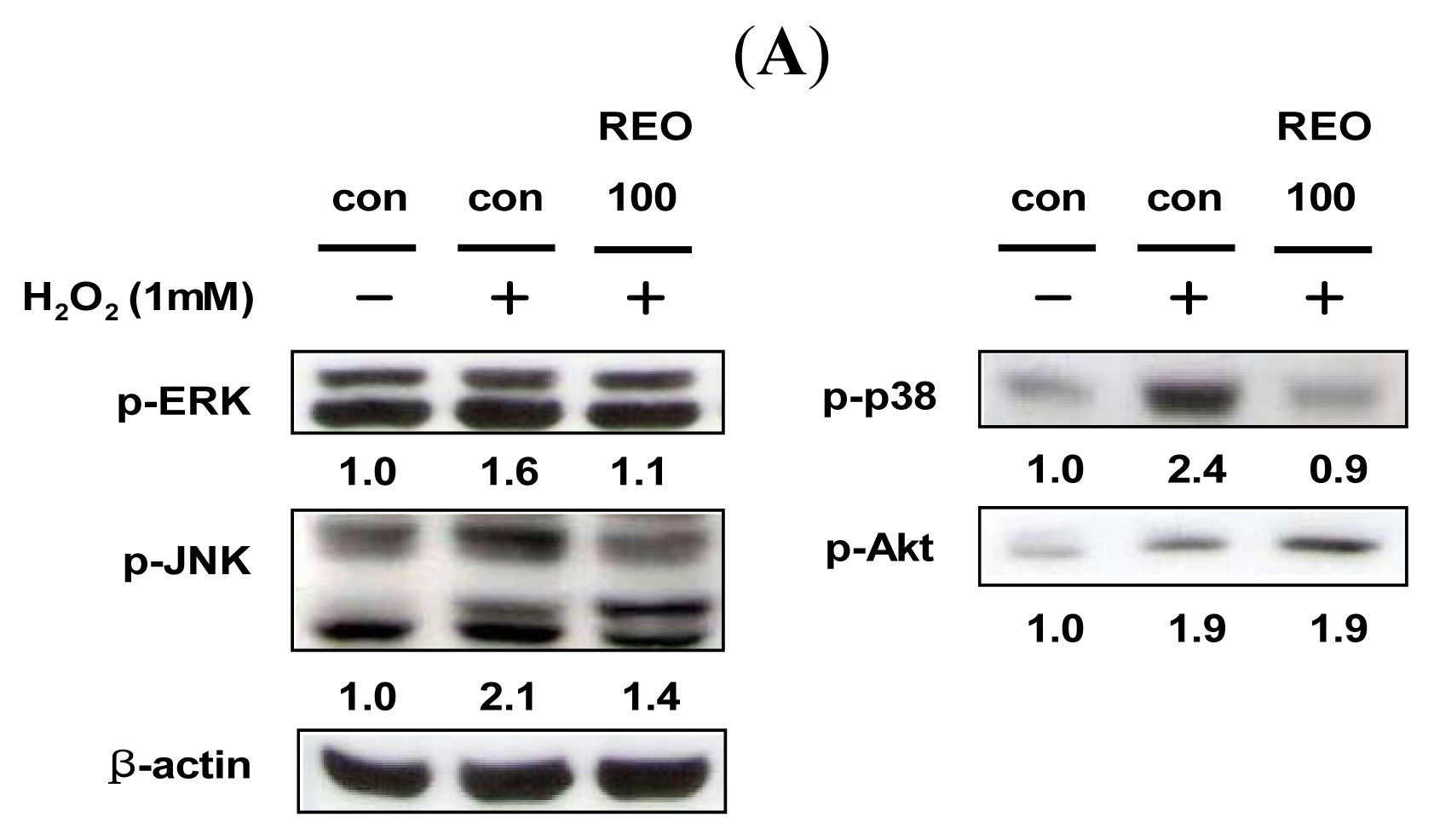
2.4. Effects of REO on Phosphorylations of Upstream Kinases in HepG2 Cells
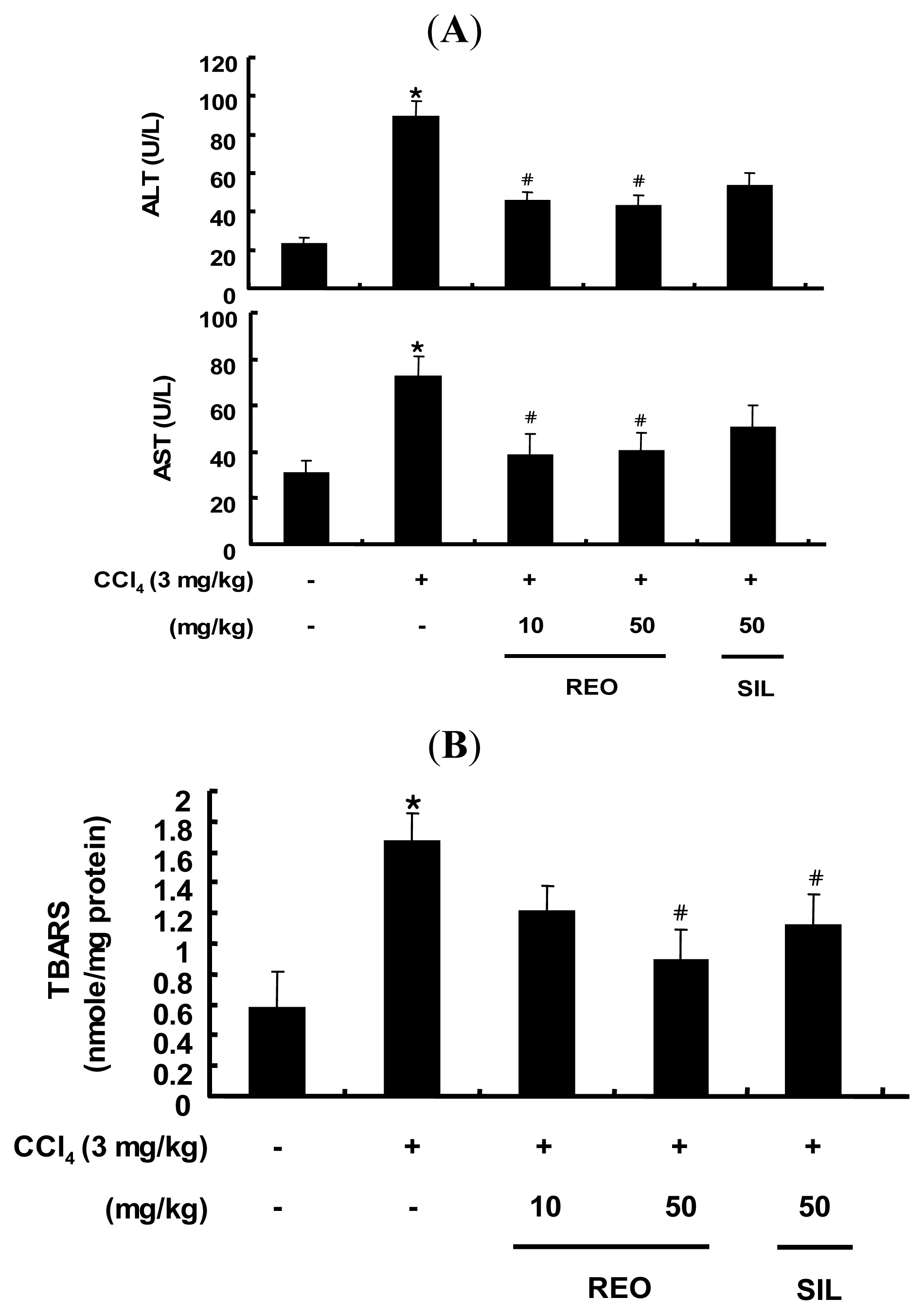
2.5. Effects of REO on Serum ALT and AST Activity in Mice
2.6. Effect of REO on Hepatic TBARS Content in Mice
2.7. Effects of REO on the Activity and Expression of Hepatic Antioxidant Enzymes in Mice
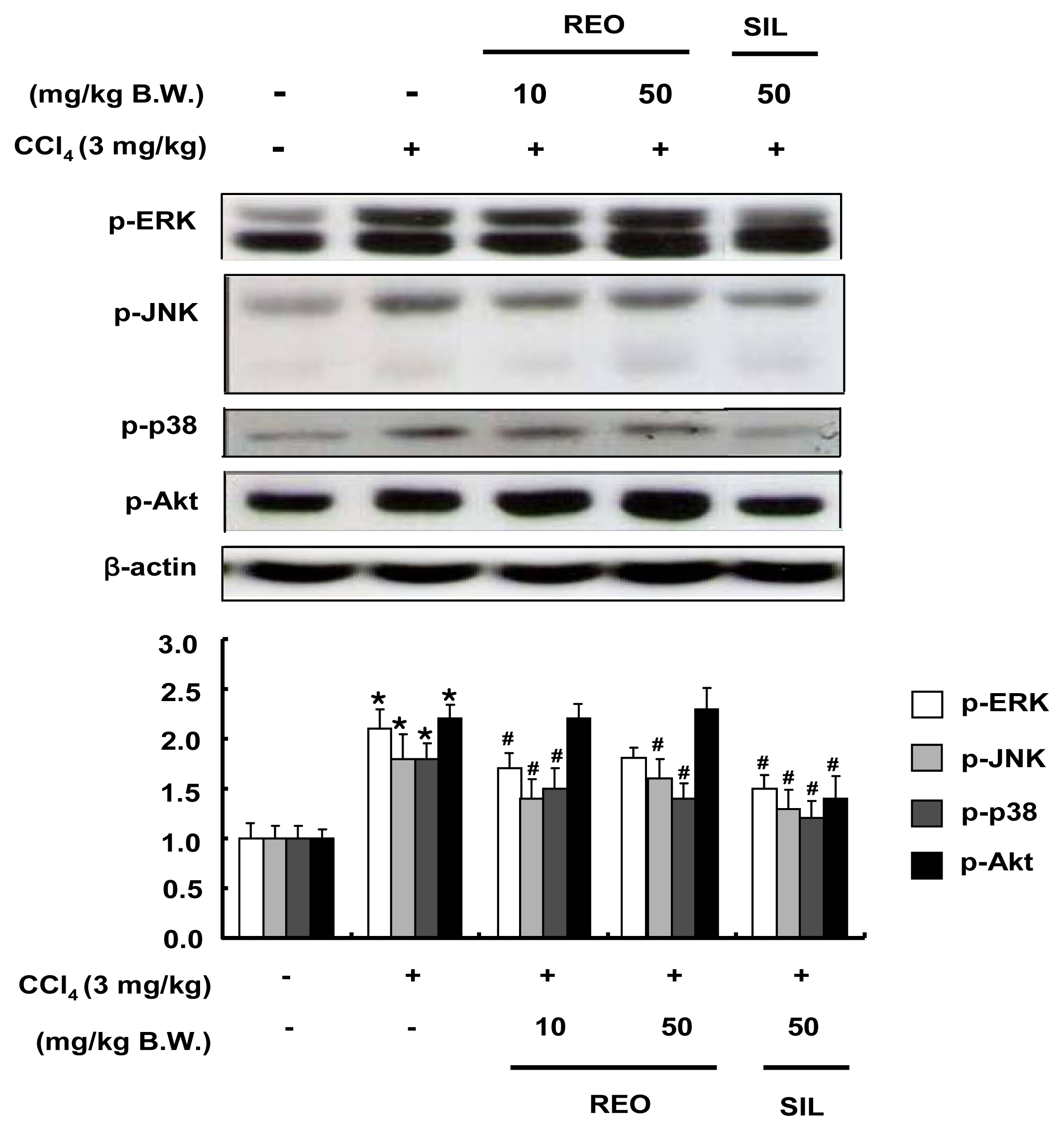
2.8. Effects of REO on Phosphorylations of Upstream Kinases in Mice
3. Experimental Section
3.1. Chemicals and Reagents
3.2. Preparation of REO
3.3. Cell Culture
3.4. Animals
3.5. Assessment of ROS Production in Cells
3.6. Assessment of CAT and GPx Activities in HepG2 Cells and Liver Tissues
3.7. Assessment of SOD-Like Activity in HepG2 Cells and Liver Tissues
3.8. Western Blotting Analysis
3.9. Assessment of Serum Marker Enzymes
3.10. Assessment of Thiobarbituric Acid Reactive Substance (TBARS) in Tissue
3.11. Statistical Analysis
4. Conclusions
Acknowledgments
References
- Aruoma, O.I.; Grootveld, M.; Bahorun, T. Free radicals in biology and medicine: from inflammation to biotechnology. Biofactors 2006, 27, 1–3. [Google Scholar]
- Heyman, S.N.; Rosen, S.; Rosenberger, C. A role for oxidative stress. Contrib. Nephrol 2011, 174, 138–148. [Google Scholar]
- Whaley-Connell, A.; McCullough, P.A.; Sowers, J.R. The role of oxidative stress in the metabolic syndrome. Rev. Cardiovasc. Med 2011, 12, 21–29. [Google Scholar]
- Datta, K.; Sinha, S.; Chattopadhyay, P. Reactive oxygen species in health and disease. Natl. Med. J. India 2000, 13, 304–310. [Google Scholar]
- Winterbourn, C.C. Nutritional antioxidants: their role in disease prevention. N. Z. Med. J 1995, 108, 447–449. [Google Scholar]
- Valko, M.; Rhodes, C.J.; Moncol, J.; Izakovic, M.; Mazur, M. Free radicals, metals and antioxidants in oxidative stress-induced cancer. Chem. Biol. Interact 2006, 160, 1–40. [Google Scholar]
- Bak, M.J.; Jeong, J.H.; Kang, H.S.; Jin, K.S.; Jun, M.; Jeong, W.S. Stimulation of Activity and Expression of Antioxidant Enzymes by Solvent Fractions and Isolated Compound from Cedrela sinensis Leaves in HepG2 Cells. J. Med. Food 2011, 14, 405–412. [Google Scholar]
- El-Mowafy, A.M.; Salem, H.A.; Al-Gayyar, M.M.; El-Mesery, M.E.; El-Azab, M.F. Evaluation of renal protective effects of the green-tea (EGCG) and red grape resveratrol: Role of oxidative stress and inflammatory cytokines. Nat. Prod. Res 2011, 25, 850–856. [Google Scholar]
- Yurt, B.; Celik, I. Hepatoprotective effect and antioxidant role of sun, sulphited-dried apricot (Prunus armeniaca L.) and its kernel against ethanol-induced oxidative stress in rats. Food Chem. Toxicol 2011, 49, 508–513. [Google Scholar]
- Aruoma, O.I.; Sun, B.; Fujii, H.; Neergheen, V.S.; Bahorun, T.; Kang, K.S.; Sung, M.K. Low molecular proanthocyanidin dietary biofactor Oligonol: Its modulation of oxidative stress, bioefficacy, neuroprotection, food application and chemoprevention potentials. Biofactors 2006, 27, 245–265. [Google Scholar]
- Jang, D.J.; Lee, M.S.; Shin, B.C.; Lee, Y.C.; Ernst, E. Red ginseng for treating erectile dysfunction: a systematic review. Br. J. Clin. Pharmacol 2008, 66, 444–450. [Google Scholar]
- Hwang, J.T.; Lee, M.S.; Kim, H.J.; Sung, M.J.; Kim, H.Y.; Kim, M.S.; Kwon, D.Y. Antiobesity effect of ginsenoside Rg3 involves the AMPK and PPAR-gamma signal pathways. Phytother. Res 2009, 23, 262–266. [Google Scholar]
- Ryu, J.K.; Lee, T.; Kim, D.J.; Park, I.S.; Yoon, S.M.; Lee, H.S.; Song, S.U.; Suh, J.K. Free radical-scavenging activity of Korean red ginseng for erectile dysfunction in non-insulin-dependent diabetes mellitus rats. Urology 2005, 65, 611–615. [Google Scholar]
- Kim, J. Protective effects of Asian dietary items on cancers—soy and ginseng. Asian Pac. J. Cancer Prev 2008, 9, 543–548. [Google Scholar]
- Cho, S.; Won, C.H.; Lee, D.H.; Lee, M.J.; Lee, S.; So, S.H.; Lee, S.K.; Koo, B.S.; Kim, N.M.; Chung, J.H. Red ginseng root extract mixed with Torilus fructus and Corni fructus improves facial wrinkles and increases type I procollagen synthesis in human skin: a randomized, double-blind, placebo-controlled study. J. Med. Food 2009, 12, 1252–1259. [Google Scholar]
- Kim, J.H.; Kang, S.A.; Han, S.M.; Shim, I. Comparison of the antiobesity effects of the protopanaxadiol- and protopanaxatriol-type saponins of red ginseng. Phytother. Res 2009, 23, 78–85. [Google Scholar]
- Ortet, R.; Regalado, E.L.; Thomas, O.P.; Pino, J.A.; Fernandez, M.D. Chemical composition and antioxidant activities of the essential oil from Tornabenea bischoffii (Apiaceae). Nat. Prod. Commun 2011, 6, 1179–1182. [Google Scholar]
- Sharififar, F.; Derakhshanfar, A.; Dehghan-Nudeh, G.; Abbasi, N.; Abbasi, R.; Gharaei, R.R.; Koohpayeh, A.; Daneshpajouh, M. In vivo antioxidant activity of Zataria multiflora Boiss essential oil. Pak. J. Pharm. Sci 2011, 24, 221–225. [Google Scholar]
- Kim, S.S.; Kim, J.E.; Hyun, C.G.; Lee, N.H. Neolitsea aciculata essential oil inhibits drug-resistant skin pathogen growth and Propionibacterium acnes-induced inflammatory effects of human monocyte leukemia. Nat. Prod. Commun 2011, 6, 1193–1198. [Google Scholar]
- Campelo, L.M.; Goncalves, F.C.; Feitosa, C.M.; de Freitas, R.M. Antioxidant activity of Citrus limon essential oil in mouse hippocampus. Pharm. Biol 2011, 49, 709–715. [Google Scholar]
- Kim, H.J.; Kang, H.J.; Seo, J.Y.; Lee, C.H.; Kim, Y.S.; Kim, J.S. Antiobesity effect of oil extract of ginseng. J. Med. Food 2011, 14, 573–583. [Google Scholar]
- Lee, S.D.; Park, S.K.; Lee, E.S.; Kim, H.M.; Lee, C.W.; Lee, K.; Lee, K.H.; Kang, M.R.; Lee, K.S.; Lee, J.; Hwang, W.I.; Kim, D.C. A lipid-soluble red ginseng extract inhibits the growth of human lung tumor xenografts in nude mice. J. Med. Food 2010, 13, 1–5. [Google Scholar]
- Meerson, F.Z.; Kagan, V.E.; Kozlov Yu, P.; Belkina, L.M.; Arkhipenko Yu, V. The role of lipid peroxidation in pathogenesis of ischemic damage and the antioxidant protection of the heart. Basic Res. Cardiol 1982, 77, 465–485. [Google Scholar]
- Abe, J.; Berk, B.C. Reactive oxygen species as mediators of signal transduction in cardiovascular disease. Trends Cardiovasc. Med 1998, 8, 59–64. [Google Scholar]
- Rigoulet, M.; Yoboue, E.D.; Devin, A. Mitochondrial ROS generation and its regulation: mechanisms involved in H(2)O(2) signaling. Antioxid. Redox Signal 2011, 14, 459–468. [Google Scholar]
- Lee, M.; Sorn, S.; Baek, S.; Jang, S.; Kim, S. Antioxidant and apoptotic effects of korean white ginseng extracted with the same ratio of protopanaxadiol and protopanaxatriol saponins in human hepatoma HepG2 cells. Ann. NY Acad. Sci 2009, 1171, 217–227. [Google Scholar]
- Chen, X.; Zhong, Z.; Xu, Z.; Chen, L.; Wang, Y. No protective effect of curcumin on hydrogen peroxide-induced cytotoxicity in HepG2 cells. Pharmacol. Rep 2011, 63, 724–732. [Google Scholar]
- Ray, G.; Husain, S.A. Oxidants, antioxidants and carcinogenesis. Indian J. Exp. Biol 2002, 40, 1213–1232. [Google Scholar]
- Liu, D.H.; Chen, Y.M.; Liu, Y.; Hao, B.S.; Zhou, B.; Wu, L.; Wang, M.; Chen, L.; Wu, W.K.; Qian, X.X. Rb1 protects endothelial cells from hydrogen peroxide-induced cell senescence by modulating redox status. Biol. Pharm. Bull 2011, 34, 1072–1077. [Google Scholar]
- Naval, M.V.; Gomez-Serranillos, M.P.; Carretero, M.E.; Villar, A.M. Neuroprotective effect of a ginseng (Panax ginseng) root extract on astrocytes primary culture. J. Ethnopharmacol 2007, 112, 262–270. [Google Scholar]
- Li, Y.G.; Ji, D.F.; Zhong, S.; Shi, L.G.; Hu, G.Y.; Chen, S. Saponins from Panax japonicus protect against alcohol-induced hepatic injury in mice by up-regulating the expression of GPX3, SOD1 and SOD3. Alcohol Alcohol 2010, 45, 320–331. [Google Scholar]
- Neergheen, V.S.; Bahorun, T.; Taylor, E.W.; Jen, L.S.; Aruoma, O.I. Targeting specific cell signaling transduction pathways by dietary and medicinal phytochemicals in cancer chemoprevention. Toxicology 2011, 278, 229–241. [Google Scholar]
- Zhang, H.S.; Wang, S.Q. Notoginsenoside R1 inhibits TNF-alpha-induced fibronectin production in smooth muscle cells via the ROS/ERK pathway. Free Radic. Biol. Med 2006, 40, 1664–1674. [Google Scholar]
- Jang, Y.J.; Kim, M.E.; Ko, S.Y. n-Butanol extracts of Panax notoginseng suppress LPS-induced MMP-2 expression in periodontal ligament fibroblasts and inhibit osteoclastogenesis by suppressing MAPK in LPS-activated RAW264.7 cells. Arch. Oral Biol 2011, 56, 1319–1327. [Google Scholar]
- McGregor, D.; Lang, M. Carbon tetrachloride: genetic effects and other modes of action. Mutat. Res 1996, 366, 181–195. [Google Scholar]
- Shim, J.Y.; Kim, M.H.; Kim, H.D.; Ahn, J.Y.; Yun, Y.S.; Song, J.Y. Protective action of the immunomodulator ginsan against carbon tetrachloride-induced liver injury via control of oxidative stress and the inflammatory response. Toxicol. Appl. Pharmacol 2010, 242, 318–325. [Google Scholar]
- Gum, S.I.; Jo, S.J.; Ahn, S.H.; Kim, S.G.; Kim, J.T.; Shin, H.M.; Cho, M.K. The potent protective effect of wild ginseng (Panax ginseng C.A. Meyer) against benzo[alpha]pyrene-induced toxicity through metabolic regulation of CYP1A1 and GSTs. J. Ethnopharmacol 2007, 112, 568–576. [Google Scholar]
- Horie, Y.; Kikuchi, M.; Yamagishi, Y.; Umeda, R.; Ebinuma, H.; Saito, H.; Kato, S.; Ishii, H.; Hibi, T.; Han, J.Y. Effect of a herbal medicine on fatty liver in rats fed ethanol chronically. Nihon Arukoru Yakubutsu Igakkai Zasshi 2009, 44, 636–648. [Google Scholar]
- Lee, H.U.; Bae, E.A.; Han, M.J.; Kim, D.H. Hepatoprotective effect of 20(S)-ginsenosides Rg3 and its metabolite 20(S)-ginsenoside Rh2 on tert-butyl hydroperoxide-induced liver injury. Biol. Pharm. Bull 2005, 28, 1992–1994. [Google Scholar]
- Brattin, W.J.; Glende, E.A., Jr; Recknagel, R.O. Pathological mechanisms in carbon tetrachloride hepatotoxicity. J. Free Radic. Biol. Med. 1985, 1, 27–38. [Google Scholar]
- Thomas, C.E.; Aust, S.D. Free radicals and environmental toxins. Ann. Emerg. Med 1986, 15, 1075–1083. [Google Scholar]
- Zhang, Q.H.; Wu, C.F.; Duan, L.; Yang, J.Y. Protective effects of ginsenoside Rg(3) against cyclophosphamide-induced DNA damage and cell apoptosis in mice. Arch. Toxicol 2008, 82, 117–123. [Google Scholar]
- Iida, C.; Fujii, K.; Kishioka, T.; Nagae, R.; Onishi, Y.; Ichi, I.; Kojo, S. Activation of mitogen activated protein kinase (MAPK) during carbon tetrachloride intoxication in the rat liver. Arch. Toxicol 2007, 81, 489–493. [Google Scholar]
- Taniguchi, M.; Takeuchi, T.; Nakatsuka, R.; Watanabe, T.; Sato, K. Molecular process in acute liver injury and regeneration induced by carbon tetrachloride. Life Sci 2004, 75, 1539–1549. [Google Scholar]
- Jiao, H.L.; Zhao, B.L. Cytotoxic effect of peroxisome proliferator fenofibrate on human HepG2 hepatoma cell line and relevant mechanisms. Toxicol. Appl. Pharmacol 2002, 185, 172–179. [Google Scholar]
- Carrillo, M.C.; Kanai, S.; Nokubo, M.; Kitani, K. (−) deprenyl induces activities of both superoxide dismutase and catalase but not of glutathione peroxidase in the striatum of young male rats. Life Sci. 1991, 48, 517–521. [Google Scholar]
- Bogdanska, J.J.; Korneti, P.; Todorova, B. Erythrocyte superoxide dismutase, glutathione peroxidase and catalase activities in healthy male subjects in Republic of Macedonia. Bratisl. Lek. Listy 2003, 104, 108–114. [Google Scholar]
- Liu, F.; Ooi, V.E.; Chang, S.T. Free radical scavenging activities of mushroom polysaccharide extracts. Life Sci 1997, 60, 763–771. [Google Scholar]
- Ohkawa, H.; Ohishi, N.; Yagi, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem 1979, 95, 351–358. [Google Scholar]


© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bak, M.-J.; Jun, M.; Jeong, W.-S. Antioxidant and Hepatoprotective Effects of the Red Ginseng Essential Oil in H2O2-Treated HepG2 Cells and CCl4-Treated Mice. Int. J. Mol. Sci. 2012, 13, 2314-2330. https://doi.org/10.3390/ijms13022314
Bak M-J, Jun M, Jeong W-S. Antioxidant and Hepatoprotective Effects of the Red Ginseng Essential Oil in H2O2-Treated HepG2 Cells and CCl4-Treated Mice. International Journal of Molecular Sciences. 2012; 13(2):2314-2330. https://doi.org/10.3390/ijms13022314
Chicago/Turabian StyleBak, Min-Ji, Mira Jun, and Woo-Sik Jeong. 2012. "Antioxidant and Hepatoprotective Effects of the Red Ginseng Essential Oil in H2O2-Treated HepG2 Cells and CCl4-Treated Mice" International Journal of Molecular Sciences 13, no. 2: 2314-2330. https://doi.org/10.3390/ijms13022314




