Detection of Glutamic Acid in Oilseed Rape Leaves Using Near Infrared Spectroscopy and the Least Squares-Support Vector Machine
Abstract
:1. Introduction
2. Results and Discussion
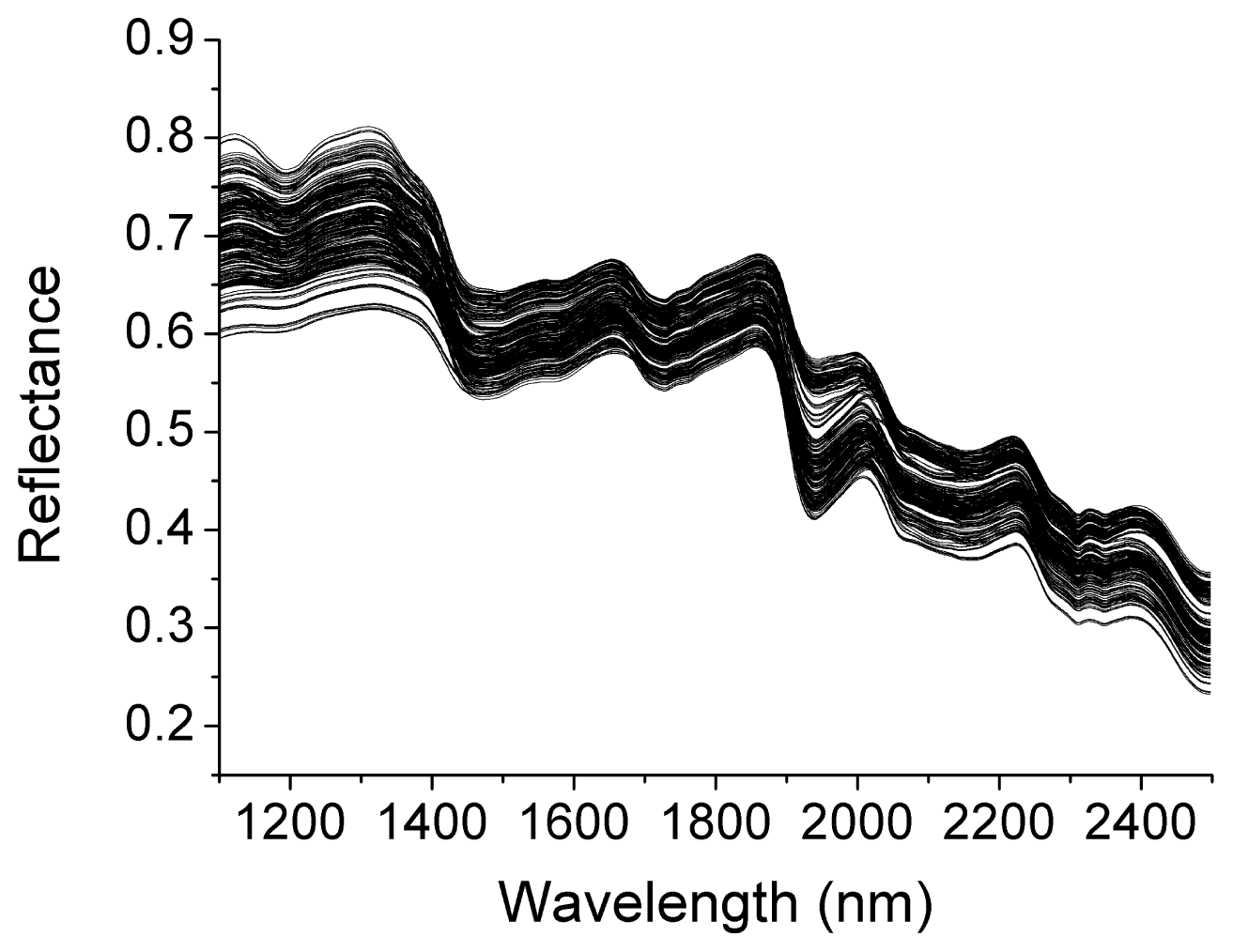
2.1. Spectral Features of Oilseed Rape
2.2. The Performance of PLS Models
2.3. EWs Selected by SPA
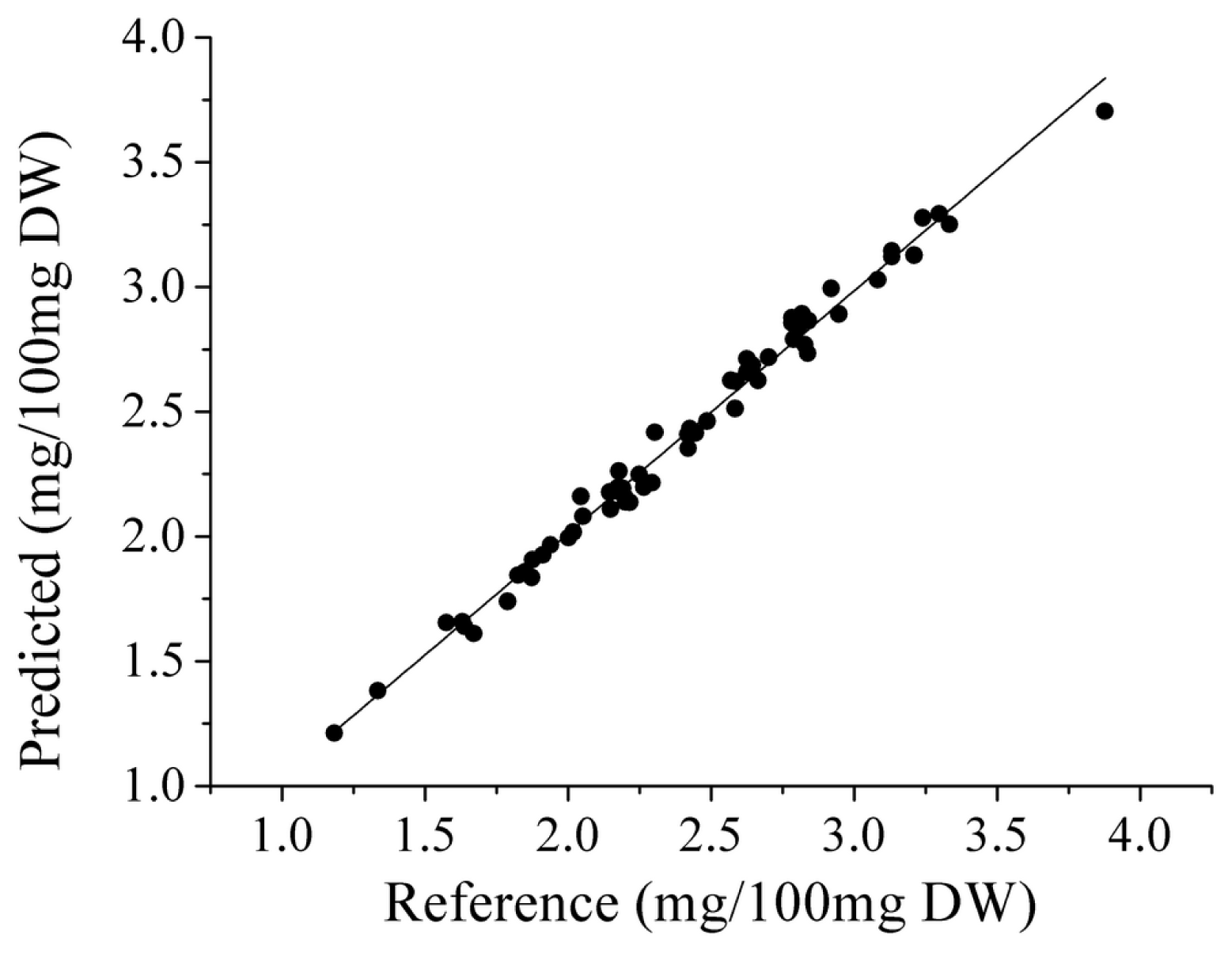
2.4 PLS and LS-SVM Models Based on SPA
3. Materials and Methods
3.1. Sample Preparation
3.2. Spectral Collection and Reference Method
3.3. Spectral Preprocessing and SPA
3.4. PLS and LS-SVM Methods
4. Conclusions
Acknowledgments
References
- Zhou, W.J. Oilseed Rape, Cultivation of Crops; Zhejiang University Press: Hangzhou, China, 2001. [Google Scholar]
- Yan, Y.L.; Zhao, L.L.; Han, D.H.; Yang, S.M. The Foundation and Application of Near Infrared Spectroscopy Analysis; China Light Industry Press: Beijing, China, 2005. [Google Scholar]
- Liu, F.; Nie, P.C.; Huang, M.; Kong, W.W.; He, Y. Nondestructive determination of nutritional information of oilseed rape leaves using visible/near infrared spectroscopy and multivariate calibrations. Sci. China Inf. Sci 2011, 54, 598–608. [Google Scholar]
- Liu, F.; Zhang, F.; Jin, Z.L.; He, Y.; Fang, H.; Ye, Q.F.; Zhou, W.J. Determination of acetolactate synthase activity and protein content of oilseed rape (Brassica napus L.) leaves using visible/near infrared spectroscopy. Anal. Chim. Acta 2008, 629, 56–65. [Google Scholar]
- Liu, F.; Fang, H.; Zhang, F.; Jin, Z.L.; Zhou, W.J.; He, Y. Nondestructive determination of acetolactate synthase in oilseed rape leaves using visible and near infrared spectroscopy. Chin. J. Anal. Chem 2009, 37, 67–71. [Google Scholar]
- Liu, F.; Jin, Z.L.; Naeem, M.S.; Tian, T.; Zhang, F.; He, Y.; Fang, H.; Ye, Q.F.; Zhou, W.J. Applying near-infrared spectroscopy and chemometrics to determine total amino acid in herbicide-stressed oilseed rape leaves. Food Bioprocess Technol 2011, 4, 1314–1321. [Google Scholar]
- Lu, J. Plant Nutrition, 2nd ed; China Agricultural University Press: Beijing, China, 2003. [Google Scholar]
- Cowe, I.A.; Koester, S.; Paul, C.; McNicol, J.W.; Cuthbertson, D.C. Principal component analysis of near-infrared spectra of whole and ground oilseed rape (Brassica napus L.) samples. Chemom. Intell. Lab. Syst 1998, 3, 233–242. [Google Scholar]
- Wang, J.; Sowa, M.G.; Ahmed, M.K.; Mantsch, H.H. Photoacoustic near-infrared investigation of homo-polypeptides. J. Phys. Chem 1994, 98, 4748–4755. [Google Scholar]
- Liu, F.; Jiang, Y.H.; He, Y. Variable selection in visible/near infrared spectra for linear and nonlinear calibrations: A case study to determine soluble solids content of beer. Anal. Chim. Acta 2009, 635, 45–52. [Google Scholar]
- Liu, F.; He, Y.; Wang, L. Comparison of calibrations for the determination of soluble solids content and pH of rice vinegars using visible and short-wave near infrared spectroscopy. Anal. Chim. Acta 2008, 610, 196–204. [Google Scholar]
- Williams, P.C. Near-Infrared Technology in the Agricultural and Food Industries; AACC International: St. Paul, MN, USA, 2001. [Google Scholar]
- Lisiewska, Z.; Kmiecik, W.; Korus, A. The amino acid composition of kale (Brassica oleracea L. var. acephala), fresh and after culinary and technological processing. Food Chem 2008, 108, 642–648. [Google Scholar]
- Chu, X.L.; Yuan, H.F.; Lu, W.Z. Progress and application of spectral data pretreatment and wavelength selection methods in NIR analytical technique. Prog. Chem 2004, 16, 528–542. [Google Scholar]
- Araújo, M.C.U.; Saldanha, T.C.B.; Galvão, R.K.H.; Yoneyama, T.; Chame, H.C.; Visani, V. The successive projections algorithm for variable selection in spectroscopic multicomponent analysis. Chemom. Intell. Lab. Syst 2001, 57, 65–73. [Google Scholar]
- Galvão, R.K.H.; Araújo, M.C.U.; Fragoso, W.D.; Silva, E.C.; José, G.E.; Soares, S.F.C.; Paiva, H.M. A variable elimination method to improve the parsimony of MLR models using the successive projections algorithm. Chemom. Intell. Lab. Syst 2008, 92, 83–91. [Google Scholar]
- Geladi, P.; Kowalski, B.R. Partial least-squares regression: A tutorial. Anal. Chim. Acta 1986, 185, 1–17. [Google Scholar]
- Suykens, J.A.K.; Vandewalle, J. Least squares support vector machine classifiers. Neural Process. Lett 1999, 9, 293–300. [Google Scholar]
| Set | No. | Range (mg/100 g DW) | Mean (mg/100 g DW) | S.D. (mg/100 g DW) |
|---|---|---|---|---|
| Calibration | 124 | 1.176–4.101 | 2.410 | 0.5252 |
| Validation | 62 | 1.189–3.654 | 2.411 | 0.5219 |
| Prediction | 62 | 1.183–3.877 | 2.411 | 0.5313 |
| Model | Treatment | LV/EW/(γ, σ2) | Calibration | Validation | Prediction | RPD | |||
|---|---|---|---|---|---|---|---|---|---|
| Rc | RMSEC | Rv | RMSEV | Rp | RMSEP | ||||
| PLS | Raw | 8/700/- | 0.9474 | 0.1674 | 0.9603 | 0.1462 | 0.9591 | 0.1530 | 3.6 |
| SG | 8/700/- | 0.9471 | 0.1678 | 0.9602 | 0.1463 | 0.9591 | 0.1528 | 3.6 | |
| SNV | 7/700/- | 0.9414 | 0.1765 | 0.9542 | 0.1552 | 0.9519 | 0.1628 | 3.4 | |
| MSC | 7/700/- | 0.9413 | 0.1765 | 0.9546 | 0.1546 | 0.9524 | 0.1621 | 3.4 | |
| 1-Der | 6/700/- | 0.9629 | 0.1412 | 0.9694 | 0.1282 | 0.9678 | 0.1335 | 4.1 | |
| 2-Der | 4/700/- | 0.9603 | 0.1459 | 0.9585 | 0.1483 | 0.9576 | 0.1527 | 3.5 | |
| Detrending | 7/700/- | 0.9507 | 0.1623 | 0.9598 | 0.1463 | 0.9550 | 0.1571 | 3.6 | |
| DOSC | 4/700/- | 0.9361 | 0.1840 | 0.9460 | 0.1692 | 0.9436 | 0.1752 | 3.1 | |
| SPA-PLS | Raw | 8/19/- | 0.9490 | 0.1649 | 0.9607 | 0.1458 | 0.9557 | 0.1591 | 3.6 |
| 1-Der | 3/10/- | 0.9487 | 0.1654 | 0.9575 | 0.1501 | 0.9528 | 0.1608 | 3.5 | |
| SPA-LS-SVM | Raw | -/19/ (1.5 × 104, 59.9) | 0.9911 | 0.0700 | 0.9966 | 0.0431 | 0.9943 | 0.0569 | 12.2 |
| 1-Der | -/10/ (38.6, 29.5) | 0.9869 | 0.0846 | 0.9952 | 0.0514 | 0.9787 | 0.1100 | 10.2 | |
| Preprocessing | No. | Selected EWs (nm) |
|---|---|---|
| Raw | 19 | 2252, 2228, 1404, 2268, 2178, 1434, 2426, 1844, 1692, 1190, 1344, 1636, 1730, 1892, 1234, 1546, 2409, 2046, 1776 |
| 1-Der | 10 | 1678, 2266, 2486, 2234, 2296, 1272, 1534, 2444, 2208, 1718 |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bao, Y.; Kong, W.; Liu, F.; Qiu, Z.; He, Y. Detection of Glutamic Acid in Oilseed Rape Leaves Using Near Infrared Spectroscopy and the Least Squares-Support Vector Machine. Int. J. Mol. Sci. 2012, 13, 14106-14114. https://doi.org/10.3390/ijms131114106
Bao Y, Kong W, Liu F, Qiu Z, He Y. Detection of Glutamic Acid in Oilseed Rape Leaves Using Near Infrared Spectroscopy and the Least Squares-Support Vector Machine. International Journal of Molecular Sciences. 2012; 13(11):14106-14114. https://doi.org/10.3390/ijms131114106
Chicago/Turabian StyleBao, Yidan, Wenwen Kong, Fei Liu, Zhengjun Qiu, and Yong He. 2012. "Detection of Glutamic Acid in Oilseed Rape Leaves Using Near Infrared Spectroscopy and the Least Squares-Support Vector Machine" International Journal of Molecular Sciences 13, no. 11: 14106-14114. https://doi.org/10.3390/ijms131114106








