A Systems Biology Approach to Understanding the Mechanisms of Action of Chinese Herbs for Treatment of Cardiovascular Disease
Abstract
:1. Introduction
2. Results and Discussion
2.1. OB Prediction and Analysis of Pharmacological Mechanisms
2.1.1. Ligusticum chuanxiong Hort
2.1.2. Dalbergia odorifera T. Chen
2.1.3. Corydalis yanhusuo WT Wang
2.2. Target Identification and Validation
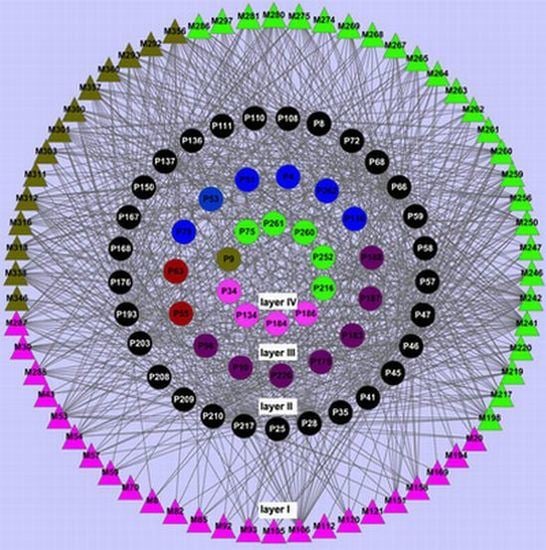
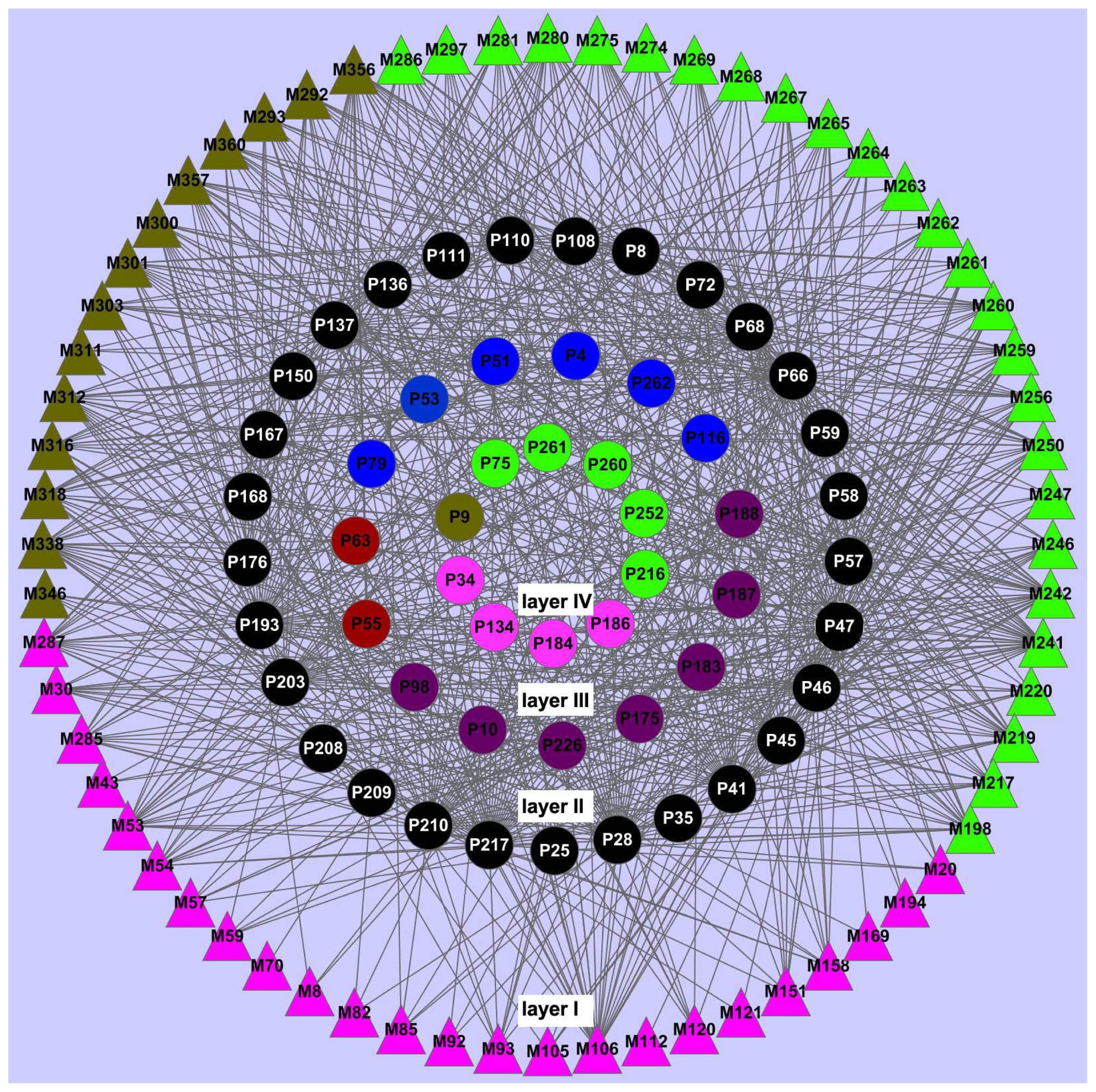
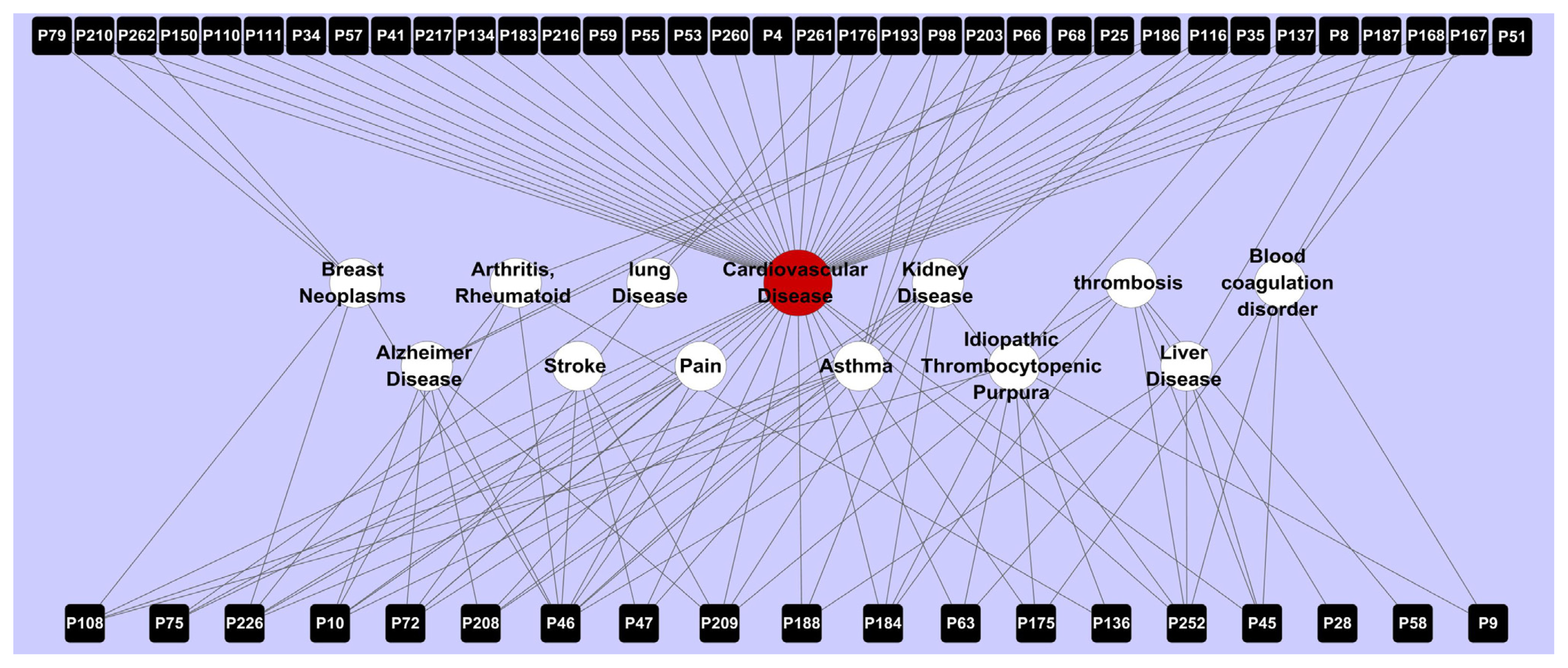
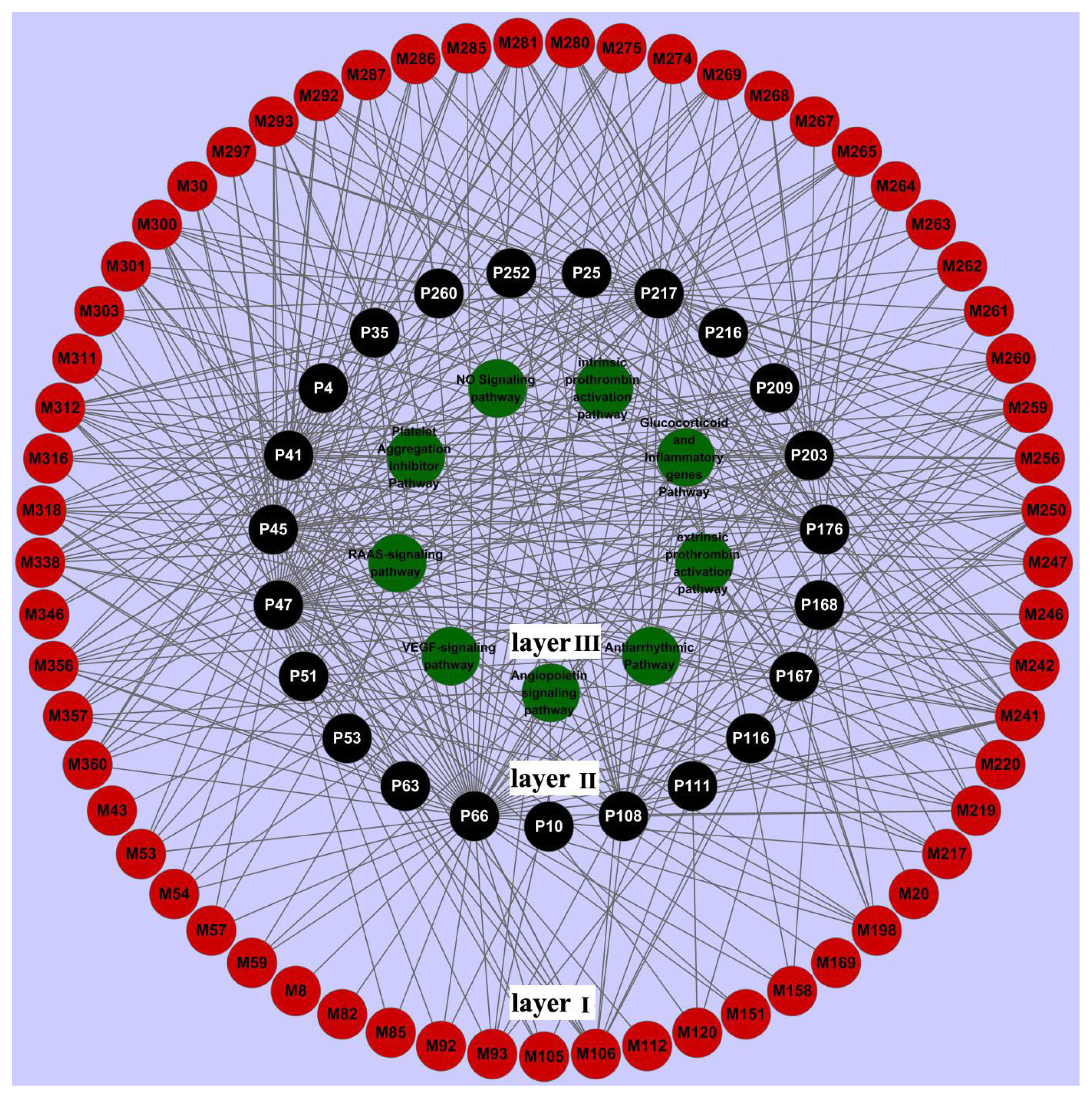
2.3. Network Construction and Analysis
2.3.1. Ligand-Candidate Target and Ligand-Potential Target Networks
2.3.2. Target-Disease Network
2.3.3. Target-Pathway Network
3 Materials and Method
3.1. Dataset Construction
3.2. Oral Bioavailability Prediction
3.3. Target Prediction
3.4. Target Validation
3.5. Network Construction
4. Conclusions
Supplementary Materials
ijms-13-13501-s001.pdfAcknowledgments
References
- Knutsson, A.; Boggild, H. Shiftwork and cardiovascular disease: Review of disease mechanisms. Rev. Environ. Health 2000, 15, 359–372. [Google Scholar]
- Xu, J.; Wu, H.Y. Chinese herbal medicine and acupuncture for the treatment of cardiovascular disease. J. Geriatr. Cardiol 2009, 6, 56–61. [Google Scholar]
- Cheung, F. TCM: Made in China. Nature 2011, 480, S82–S83. [Google Scholar]
- Tachjian, A.; Maria, V.; Jahangir, A. Use of herbal products and potential interactions in patients with cardiovascular diseases. J. Am. Coll. Cardiol 2010, 55, 515–525. [Google Scholar]
- Ran, X.; Ma, L.; Peng, C.; Zhang, H.; Qin, L.-P. Ligusticum chuanxiong Hort: A review of chemistry and pharmacology. Pharm. Biol 2011, 49, 1180–1189. [Google Scholar]
- Liang, M.-J.; He, L.-C.; Yang, G.-D. Screening, analysis and in vitro vasodilatation of effective components from Ligusticum chuanxiong. Life Sci 2005, 78, 128–133. [Google Scholar]
- Tao, Y.; Wang, Y. Bioactive sesquiterpenes isolated from the essential oil of Dalbergia odorifera T. Chen. Fitoterapia 2010, 81, 393–396. [Google Scholar]
- Steimle, A.E.; Lange, R.A.; Hillis, L.D. Antiplatelet therapy for ischemic heart disease. N. Engl. J. Med 2004, 350, 2101–2102. [Google Scholar]
- Liu, J.L.; Liu, H. The protective effects of dl-tetrahydropalmatine on isolated rat heart against ischemic/reperfusion damage exacerbated by extrinsic radical generating system. Zhongguo Yaoxue Zazhi 1994, 29, 462–464. [Google Scholar]
- Ling, H.; Wu, L.; Li, L. Corydalis yanhusuo rhizoma extract reduces infarct size and improves heart function during myocardial ischemia/reperfusion by inhibiting apoptosis in rats. Phytother. Res 2006, 20, 448–453. [Google Scholar]
- Yuan, C.-S.; Mehendale, S.R.; Wang, C.-Z.; Aung, H.H.; Jiang, T.; Guan, X.; Shoyama, Y. Effects of Corydalis yanhusuo and Angelicae dahuricae on cold pressor-induced pain in humans: A controlled trial. J. Clin. Pharmacol 2004, 44, 1323–1327. [Google Scholar]
- Li, X.X.; Wang, J.N.; Yu, H.; Yang, H.J.; Xu, H.Y.; Tang, S.H.; Li, Y.; Wang, Y.H.; Yang, L.; Huang, L.Q.; et al. Investigation into the mechanisms of action of traditional Chinese medicine from chemical, genomic and pharmacological data in an integrated framework. PLoS One 2012, in press. [Google Scholar]
- Yu, H.; Chen, J.X.; Xu, X.; Li, Y.; Zhao, H.H.; Fang, Y.P.; Li, X.X.; Zhou, W.; Wang, W.; Wang, Y.H. A Systematic prediction of multiple drug-target interactions from chemical, genomic and pharmacological data. PLoS One 2012, in press. [Google Scholar]
- Chan, L.M.S.; Lowes, S.; Hirst, B.H. The ABCs of drug transport in intestine and liver: Efflux proteins limiting drug absorption and bioavailability. Eur. J. Pharm. Sci 2004, 21, 25–51. [Google Scholar]
- Yan, R.; Ko, N.L.; Li, S.L.; Tam, Y.K.; Lin, G. Pharmacokinetics and metabolism of ligustilide, a major bioactive component in Rhizoma chuanxiong, in the rat. Drug Metab. Dispos 2008, 36, 400–408. [Google Scholar]
- Ko, W.C. A newly isolated antispasmodic butylidenephthahde. Jpn. J. Pharmacol 1980, 30, 85–91. [Google Scholar]
- Wei, Y.; Hu, J.; Li, H.; Liu, J.G. Preparative isolation and purification of senkyunolide-I, senkyunolide-H and ferulic acid from Rhizoma chuanxiong using counter-current chromatography. J. Sep. Sci 2011, 34, 3426–3432. [Google Scholar]
- Che, M.T.; Wen, Y.C.; Wun, C.K.; Ouyang, C.H. Antiplatelet effect of butylidenephthalide. Biochim. Biophys. Acta Gen. Subj 1987, 924, 375–382. [Google Scholar]
- Ozaki, Y.; Sekita, S.; Harada, M. Centrally acting muscle relaxant effect of phthalides (ligustilide, cnidilide and senkyunolide) obtained from Cnidium officinale Makino. Yakugaku Zasshi 1989, 109, 402–406. [Google Scholar]
- Jasicka-Misiak, I.; Lipok, J.; Nowakowska, E.M.; Wieczorek, P.P.; Mlynarz, P.; Kafarski, P. Antifungal activity of the carrot seed oil and its major sesquiterpene compounds. Z. Naturforsch. C J. Biosci 2004, 59, 791–796. [Google Scholar]
- Graf, E. Antioxidant potential of ferulic acid. Free Radic. Biol. Med 1992, 13, 435–448. [Google Scholar]
- Hou, Y.Z.; Yang, J.; Zhao, G.R.; Yuan, Y.J. Ferulic acid inhibits vascular smooth muscle cell proliferation induced by angiotensin II. Eur. J. Pharmacol 2004, 499, 85–90. [Google Scholar]
- Zhao, Z.H.; Moghadasian, M.H. Chemistry, natural sources, dietary intake and pharmacokinetic properties of ferulic acid: A review. Food Chem 2008, 109, 691–702. [Google Scholar]
- Jackson, S.P. The growing complexity of platelet aggregation. Blood 2007, 109, 5087–5095. [Google Scholar]
- Yadav, V.R.; Prasad, S.; Sung, B.; Aggarwal, B.B. The role of chalcones in suppression of NF-κB-mediated inflammation and cancer. Int. Immunopharmacol 2011, 11, 295–309. [Google Scholar]
- Kim, Y.W.; Zhao, R.J.; Park, S.J.; Lee, J.R.; Cho, I.J.; Yang, C.H.; Kim, S.G.; Kim, S.C. Anti-inflammatory effects of liquiritigenin as a consequence of the inhibition of NF-κB-dependent iNOS and proinflammatory cytokines production. Br. J. Pharmacol 2008, 154, 165–173. [Google Scholar]
- Yu, X.L.; Wang, W.; Yang, M. Antioxidant activities of compounds isolated from Dalbergia odorifera T. Chen and their inhibition effects on the decrease of glutathione level of rat lens induced by UV irradiation. Food Chem 2007, 104, 715–720. [Google Scholar]
- Lin, W.W.; Karin, M. A cytokine-mediated link between innate immunity, inflammation, and cancer. J. Clin. Invest 2007, 117, 1175–1183. [Google Scholar]
- Aggarwal, B.B.; Shishodia, S.; Sandur, S.K.; Pandey, M.K.; Sethi, G. Inflammation and cancer: How hot is the link? Biochem. Pharmacol 2006, 72, 1605–1621. [Google Scholar]
- Ding, B.; Zhou, T.; Fan, G.; Hong, Z.; Wu, Y. Qualitative and quantitative determination of ten alkaloids in traditional Chinese medicine Corydalis yanhusuo W.T. Wang by LC-MS/MS and LC-DAD. J. Pharm. Biomed. Anal 2007, 45, 219–226. [Google Scholar]
- Khan, I.A.; Alam, S.S.; Haq, A.; Jabbar, A. Biochemistry of resistance in chickpea against wilt disease caused by Fusarium osxysporum F.SP. ciceris. Pak. J. Bot 2005, 37, 97–104. [Google Scholar]
- Maurya, S.; Srivastava, J.S.; Jha, R.N.; Panday, V.B.; Singh, U.P. Effect of tetrahydropalmatine, an alkaloid on spore germination of some fungi. Mycobiology 2001, 29, 142–144. [Google Scholar]
- Kurahashi, K.; Fujiwara, M. Adrenergic neuron blocking action of dehydrocorydaline isolated from Corydalis bulbosa. Can. J. Physiol. Pharmacol 1976, 54, 287–293. [Google Scholar]
- Li, H.L.; Han, T.; Liu, R.H.; Zhang, C.; Chen, H.S.; Zhang, W.D. Alkaloids from Corydalis saxicola and their anti-hepatitis B virus activity. Chem. Biodivers 2008, 5, 777–783. [Google Scholar]
- Gray, G.A.; Webb, D.J. The endothelin system and its potential as a therapeutic target in cardiovascular disease. Pharmacol. Ther 1996, 72, 109–148. [Google Scholar]
- Douglas, S.A.; Ohlstein, E.H. Human urotensin-II, the most potent mammalian vasoconstrictor identified to date, as a therapeutic target for the management of cardiovascular disease. Trends Cardiovasc. Med 2000, 10, 229–237. [Google Scholar]
- Linsel-Nitschke, P.; Tall, A.R. HDL as a target in the treatment of atherosclerotic cardiovascular disease. Nat. Rev. Drug Discov 2005, 4, 193–205. [Google Scholar]
- Moens, A.L.; Kass, D.A. Tetrahydrobiopterin and cardiovascular disease. Arterioscler. Thromb. Vasc. Biol 2006, 26, 2439–2444. [Google Scholar]
- Liu, P.Y.; Death, A.K.; Handelsman, D.J. Androgens and cardiovascular disease. Endocr. Rev 2003, 24, 313–340. [Google Scholar]
- Yusuf, S.; Sleight, P.; Pogue, J.; Bosch, J.; Davies, R.; Dagenais, G. Effects of an angiotensin-converting-enzyme inhibitor, ramipril, on cardiovascular events in high-risk patients. N. Engl. J. Med 2000, 342, 145–153. [Google Scholar]
- Sun, D.Z.; Xu, L.; Wei, P.K.; Liu, L.; He, J. Syndrome differentiation in traditional Chinese medicine and E-cadherin/ICAM-1 gene protein expression in gastric carcinoma. World J. Gastroenterol 2007, 13, 4321–4327. [Google Scholar]
- Hishikawa, K.; Nakaki, T.; Fujii, T. Connective tissue growth factor induces apoptosis via caspase 3 in cultured human aortic smooth muscle cells. Eur. J. Pharmacol 2000, 392, 19–22. [Google Scholar]
- Lee, K.H.; Itokawa, H.; Kozuka, M. Oriental Herbal Products: The Basis for Development of Dietary Supplements and New Medicines in the 21st Century. In Oriental Foods and Herbs; Ho, C.T., Lin, J.K., Zheng, Q.Y., Eds.; Oxford University Press: Washington, WA, USA, 2003; pp. 2–31. [Google Scholar]
- Small, K.M.; Wagoner, L.E.; Levin, A.M.; Kardia, S.L.R.; Liggett, S.B. Synergistic polymorphisms of b1-and a 2C-adrenergic receptors and the risk of congestive heart failure. N. Engl. J. Med 2002, 347, 1135–1142. [Google Scholar]
- Kelly, H.B. Coronary artery disease in aviation. Routine examination of aircrew. J. R. Soc. Med 1979, 72, 374–377. [Google Scholar]
- Miles, E.A.; Zoubouli, P.; Calder, P.C. Differential anti-inflammatory effects of phenolic compounds from extra virgin olive oil identified in human whole blood cultures. Nutrition 2005, 21, 389–394. [Google Scholar]
- Cheng, W.H.; Lu, P.J.; Ho, W.Y.; Tung, C.S.; Cheng, P.W.; Hsiao, M.; Tseng, C.J. Angiotensin II inhibits neuronal nitric oxide synthase activation through the ERK1/2-RSK signaling pathway to modulate central control of blood pressure. Circ. Res 2010, 106, 788–795. [Google Scholar]
- Grosser, T.; Fries, S.; FitzGerald, G.A. Biological basis for the cardiovascular consequences of COX-2 inhibition: therapeutic challenges and opportunities. J. Clin. Invest 2006, 116, 4–15. [Google Scholar]
- Houslay, M.D.; Baillie, G.S.; Maurice, D.H. cAMP-Specific phosphodiesterase-4 enzymes in the cardiovascular system: A molecular toolbox for generating compartmentalized cAMP signaling. Circ. Res 2007, 100, 950–966. [Google Scholar]
- Xu, Q. Role of heat shock proteins in atherosclerosis. Arterioscler. Thromb. Vasc. Biol 2002, 22, 1547–1559. [Google Scholar]
- Atlas, S.A. The renin-angiotensin aldosterone system: Pathophysiological role and pharmacologic inhibition. J. Manag. Care Pharm 2007, 13, 9–20. [Google Scholar]
- Chou, M.T.; Wang, J.; Fujita, D.J., Sr. c kinase becomes preferentially associated with the VEGFR, KDR/Flk-1, following VEGF stimulation of vascular endothelial cells. BMC Biochem 2002, 3. [Google Scholar] [CrossRef]
- Bouloumie, A.; Schini-Kerth, V.B.; Busse, R. Vascular endothelial growth factor up-regulates nitric oxide synthase expression in endothelial cells. Cardiovasc. Res 1999, 41, 773–780. [Google Scholar]
- Fontana, J.; Fulton, D.; Chen, Y.; Fairchild, T.A.; McCabe, T.J.; Fujita, N.; Tsuruo, T.; Sessa, W.C. Domain mapping studies reveal that the M domain of hsp90 serves as a molecular scaffold to regulate Akt-dependent phosphorylation of endothelial nitric oxide synthase and NO release. Circ. Res 2002, 90, 866–873. [Google Scholar]
- Traditional Chinese Medicine Systems Pharmacology Database and Analysis Platform. Available online: http://tcmspnw.com/login_clearSession accessed on 1 November 2011.
- Xu, X.; Zhang, W.X.; Li, Y.; Huang, C.; Yu, H.; Wang, Y.H.; Duan, J.Y. In silico prediction of human oral bioavailability based on molecular properties integrated with metabolism. Int. J. Mol. Sci 2012, 13, 6964–6982. [Google Scholar]
- Cosconati, S.; Forli, S.; Perryman, A.L.; Harris, R.; Goodsell, D.S.; Olson, A.J. Virtual screening with autodock: Theory and practice. Expert Opin. Drug Discov 2010, 5, 597–607. [Google Scholar]
- Smoot, M.E.; Ono, K.; Ruscheinski, J.; Wang, P.L.; Ideker, T. Cytoscape 2.8: New features for data integration and network visualization. Bioinformatics 2011, 27, 431–432. [Google Scholar]

| No. | Molecule | OB | Herbs * |
|---|---|---|---|
| M8 | Carotol | 149.03 | Lc |
| M20 | 1-Acetyl-carboline | 67.12 | Lc |
| M30 | 2,3,5-Trihydroxymethyl-6-methylpyrazine | 73.44 | Lc |
| M43 | 3,4-Dihydroxybenzoic acid | 40.10 | Lc |
| M53 | 4,7-Dihydroxy-3-butylphthalide | 105.83 | Lc |
| M54 | 4-hydroxy-3-butylphthalide | 63.44 | Lc |
| M57 | 5-hydroxymethyl-3,6-dimethylpyrazine-2-methanioc-acid | 59.09 | Lc |
| M59 | 7-oxabicyclo-2/1,4-Cineole | 60.87 | Lc |
| M70 | Aromadendrene-oxide-2 | 64.71 | Lc |
| M82 | Bornyl-acetate | 65.53 | Lc |
| M85 | 3-n-Butylphthalide | 71.28 | Lc |
| M92 | Cis-sabinenehydrate | 94.32 | Lc |
| M93 | Cnidilide | 77.55 | Lc |
| M105 | Ferulic acid | 86.58 | Lc |
| M106 | Folic-acid | 70.51 | Lc |
| M112 | Globulol-(−)- | 81.85 | Lc |
| M120 | Ligustilide | 50.10 | Lc |
| M121 | Ligustrazine | 29.64 | Lc |
| M151 | Senkyunolide-D | 83.37 | Lc |
| M158 | Senkyunolide-K | 61.86 | Lc |
| M169 | Spathulenol | 82.37 | Lc |
| M194 | Vanillic-acid | 64.27 | Lc |
| M285 | Vestitone | 78.21 | Lc |
| M287 | Xenognosin-B | 73.99 | Lc |
| M198 | (3R)-vestitol | 71.33 | Do |
| M217 | 2′,6-dihydroxy-4′-methoxy-2-arylbenzofuran | 67.09 | Do |
| M219 | 2′-O-methylisoliquiritigenin | 79.15 | Do |
| M220 | 3′,4′,7-Trihydroxyflavone | 67.84 | Do |
| M241 | Butein | 78.38 | Do |
| M242 | Butin | 52.91 | Do |
| M246 | Clausseguinone | 60.89 | Do |
| M247 | Dalbergin | 78.57 | Do |
| M250 | Duartin | 61.17 | Do |
| M256 | Hydroxyobtustyrene | 78.44 | Do |
| M259 | Isoliquiritigenin | 61.38 | Do |
| M260 | Isomucronustyrene | 78.92 | Do |
| M261 | Koparin | 68.87 | Do |
| M262 | Liquiritigenin | 67.19 | Do |
| M264 | Melilotocarpan-A | 87.37 | Do |
| M265 | Melilotocarpan-C | 78.88 | Do |
| M267 | Methyl-2-hydroxy-3,4-dimethoxybenzoate | 89.23 | Do |
| M268 | Methylnissolin | 66.53 | Do |
| M269 | Mucronulatol | 64.93 | Do |
| M274 | Odoricarpin | 60.17 | Do |
| M275 | Odoriflavene | 84.49 | Do |
| M280 | Prunetin | 43.44 | Do |
| M281 | Sativanone | 73.01 | Do |
| M286 | Violanone | 71.83 | Do |
| M297 | Bicuculline | 76.96 | Do |
| M263 | Medicarpin | 76.02 | Cy |
| M292 | 13-methyldehydrocorydalmine | 72.22 | Cy |
| M293 | 13-methylpalmatrubine | 70.97 | Cy |
| M300 | Capaurine | 71.12 | Cy |
| M301 | Caseanidine | 93.87 | Cy |
| M303 | Clarkeanidine | 92.55 | Cy |
| M311 | Corynoloxine | 68.61 | Cy |
| M312 | Coryphenanthrine | 61.15 | Cy |
| M316 | Dehydrocavidine | 47.59 | Cy |
| M318 | Dehydrocorydaline | 60.36 | Cy |
| M338 | Norglaucine | 61.25 | Cy |
| M346 | Pseudoprotopine | 63.63 | Cy |
| M356 | Tetrahydropalmatine | 75.78 | Cy |
| M357 | Tetrahydroprotopapaverine | 64.29 | Cy |
| M360 | Yuanhunine | 84.94 | Cy |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Li, B.; Xu, X.; Wang, X.; Yu, H.; Li, X.; Tao, W.; Wang, Y.; Yang, L. A Systems Biology Approach to Understanding the Mechanisms of Action of Chinese Herbs for Treatment of Cardiovascular Disease. Int. J. Mol. Sci. 2012, 13, 13501-13520. https://doi.org/10.3390/ijms131013501
Li B, Xu X, Wang X, Yu H, Li X, Tao W, Wang Y, Yang L. A Systems Biology Approach to Understanding the Mechanisms of Action of Chinese Herbs for Treatment of Cardiovascular Disease. International Journal of Molecular Sciences. 2012; 13(10):13501-13520. https://doi.org/10.3390/ijms131013501
Chicago/Turabian StyleLi, Bohui, Xue Xu, Xia Wang, Hua Yu, Xiuxiu Li, Weiyang Tao, Yonghua Wang, and Ling Yang. 2012. "A Systems Biology Approach to Understanding the Mechanisms of Action of Chinese Herbs for Treatment of Cardiovascular Disease" International Journal of Molecular Sciences 13, no. 10: 13501-13520. https://doi.org/10.3390/ijms131013501
APA StyleLi, B., Xu, X., Wang, X., Yu, H., Li, X., Tao, W., Wang, Y., & Yang, L. (2012). A Systems Biology Approach to Understanding the Mechanisms of Action of Chinese Herbs for Treatment of Cardiovascular Disease. International Journal of Molecular Sciences, 13(10), 13501-13520. https://doi.org/10.3390/ijms131013501