Preparation and Physicochemical Properties of Vinblastine Microparticles by Supercritical Antisolvent Process
Abstract
:1. Introduction
2. Results and Discussion
2.1. Morphology
2.2. Optimum Parameters of Supercritical Antisolvent Process by L16 (45) Orthogonal Design
2.3. Physicochemical Properties of Vinblastine Microparticles
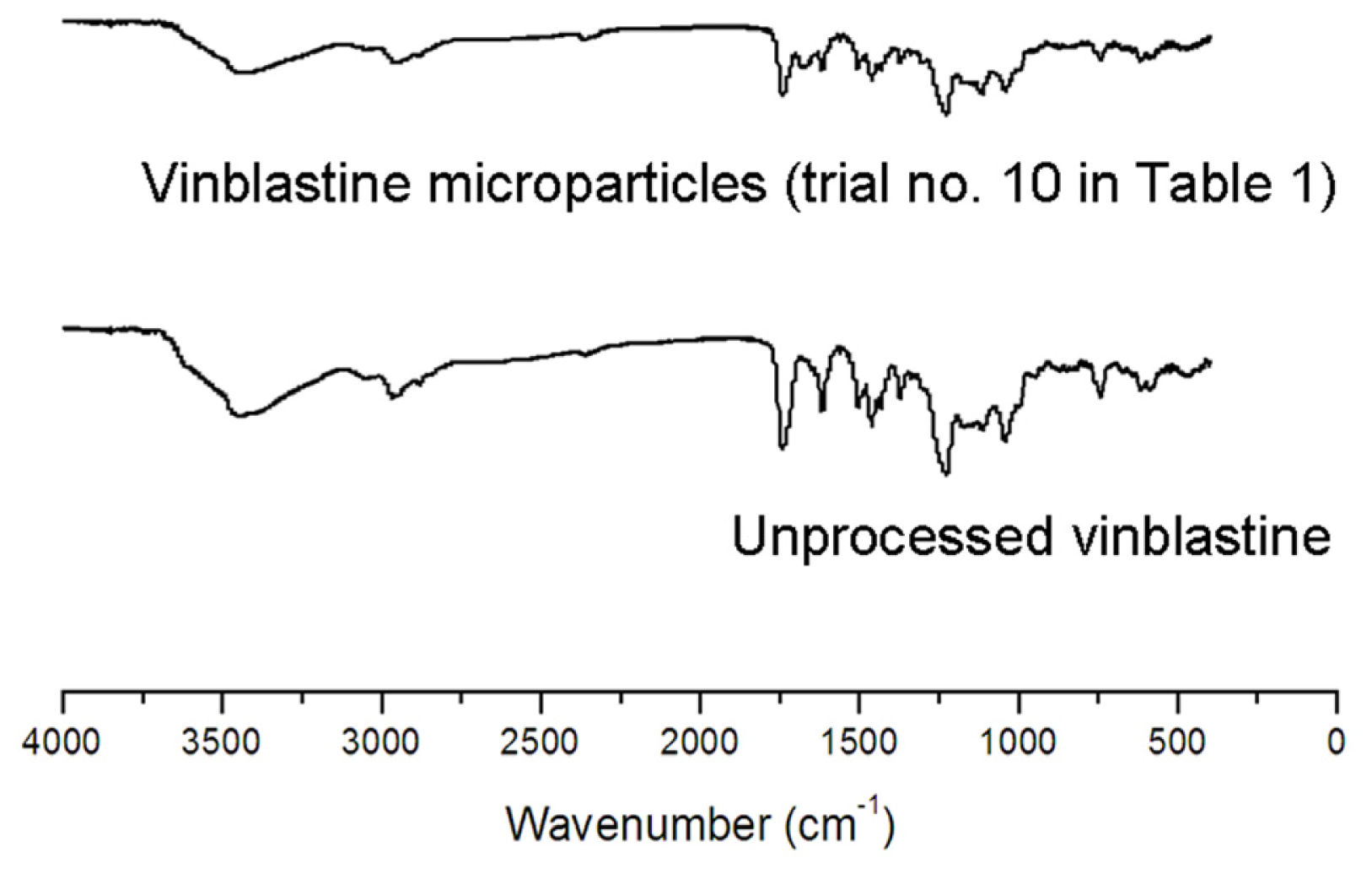
2.3.1. Fourier-Transform Infrared (FTIR) Spectroscopy Analysis
2.3.2. Mass Spectra Analysis
2.3.3. X-ray Powder Diffraction (XRD) Analysis
2.3.4. Dissolution Studies in Vitro
3. Experimental Section
3.1. Materials
3.2. Apparatus
3.3. Supercritical Antisolvent Micronization
3.4. Optimization of Supercritical Antisolvent Process
3.5. Powder Characterization
4. Conclusions
Acknowledgment
References
- Ganapathi, G.; Kargi, F. Recent advances in indole alkaloid production by Catharanthus roseus (Periwinkle). J. Exp. Bot 1990, 41, 259–267. [Google Scholar]
- Yang, L.; Wang, H.; Zu, Y.; Zhao, C.; Zhang, L.; Chen, X.; Zhang, Z. Ultrasound-assisted extraction of the three terpenoid indole alkaloids vindoline, catharanthine and vinblastine from Catharanthus roseus using ionic liquid aqueous solutions. Chem. Eng. J 2011, 172, 705–712. [Google Scholar]
- Noble, R.L. The discovery of the vinca alkaloids—Chemotherapeutic agents against cancer. Biochem. Cell Biol 1990, 68, 1344–1351. [Google Scholar]
- Miura, Y.; Hirata, K.; Kurano, N. Isolation of vinblastine in callus culture with differentiated roots of Catharanthus roseus. Agric. Biol. Chem 1987, 51, 611–614. [Google Scholar]
- Cheng, Y.Y.; Li, M.H.; Xu, T.W. Potential of poly (amidoamine) dendrimers as drug carriers of camptothecin based on encapsulation studies. Eur. J. Med. Chem 2008, 43, 1791–1795. [Google Scholar]
- Shavi, G.V.; Kumar, A.R.; Usha, Y.N.; Armugam, K.; Ranjan, O.; Ginjupalli, K.; Pandey, S.; Udupa, N. Enhanced dissolution and bioavailability of gliclazide using solid dispersion techniques. Int. J. Drug Delivery 2010, 2, 49–57. [Google Scholar]
- Gwak, H.; Choi, J.; Choi, H. Enhanced bioavailability of piroxicam via salt formation with ethanolamines. Int. J. Pharm 2005, 297, 156–161. [Google Scholar]
- Noble, C.O.; Guo, Z.; Hayes, M.E.; Marks, J.D.; Park, J.W.; Benz, C.C.; Kirpotin, D.B.; Drummond, D.C. Characterization of highly stable liposomal and immunoliposomal formulations of vincristine and vinblastine. Cancer Chemother. Pharmacol. 2009, 64, 741–751. [Google Scholar]
- Prabagar, B.; Yoo, B.K.; Woo, J.S.; Kim, J.A.; Rhee, J.D.; Piao, M.G.; Choi, H.G.; Yong, C.S. Enhanced bioavailability of poorly water-soluble clotrimazole by inclusion with β-cyclodextrin. Arch. Pharm. Res 2007, 30, 249–254. [Google Scholar]
- Yin, Y.; Cui, F.; Mu, C.; Choi, M.; Kim, J.S.; Chung, S.; Shim, C.; Kim, D. Docetaxel microemulsion for enhanced oral bioavailability: Preparation and in vitro and in vivo evaluation. J. Controlled Release 2009, 140, 86–94. [Google Scholar]
- Brigger, I.; Dubernet, C.; Couvreur, P. Nanoparticles in cancer therapy and diagnosis. Adv. Drug Delivery Rev 2002, 54, 631–651. [Google Scholar]
- Yang, L.; Huang, J.; Zu, Y.; Ma, C.; Wang, H.; Sun, X.; Sun, Z. Preparation and radical scavenging activities of polymeric procyanidins nanoparticles by a supercritical antisolvent (SAS) process. Food Chem 2011, 128, 1152–1159. [Google Scholar]
- Yang, L.; Sun, Z.; Zu, Y.; Zhao, C.; Sun, X.; Zhang, Z.; Zhang, L. Physicochemical properties and oral bioavailability of ursolic acid nanoparticles using supercritical anti-solvent (SAS) process. Food Chem 2012, 132, 319–325. [Google Scholar]
- Zhao, X.; Zu, Y.; Li, Q.; Wang, M.; Zu, B.; Zhang, X.; Jiang, R.; Zu, C. Preparation and characterization of camptothecin powder micronized by a supercritical antisolvent (SAS) process. J. Supercrit. Fluid 2010, 51, 412–419. [Google Scholar]
- Sui, X.; Wei, W.; Yang, L.; Zu, Y.; Zhao, C.; Zhang, L.; Yang, F.; Zhang, Z. Preparation, characterization and in vivo assessment of the bioavailability of glycyrrhizic acid microparticles by supercritical anti-solvent process. Int. J. Pharm 2012, 423, 471–479. [Google Scholar]
- Marco, I.D.; Reverchon, E. Influence of pressure, temperature and concentration on the mechanisms of particle precipitation in supercritical antisolvent micronization. J. Supercrit. Fluid 2011, 58, 295–302. [Google Scholar]
- Nelson, K.G. The Kelvin equation and solubility of small particles. J. Pharm. Sci 1972, 61, 479–480. [Google Scholar]
- Chattopadhyay, P.; Gupta, R.B. Production of griseofulvin nanoparticles using supercritical CO2 antisolvent with enhanced mass transfer. Int. J. Pharm 2001, 228, 19–31. [Google Scholar]

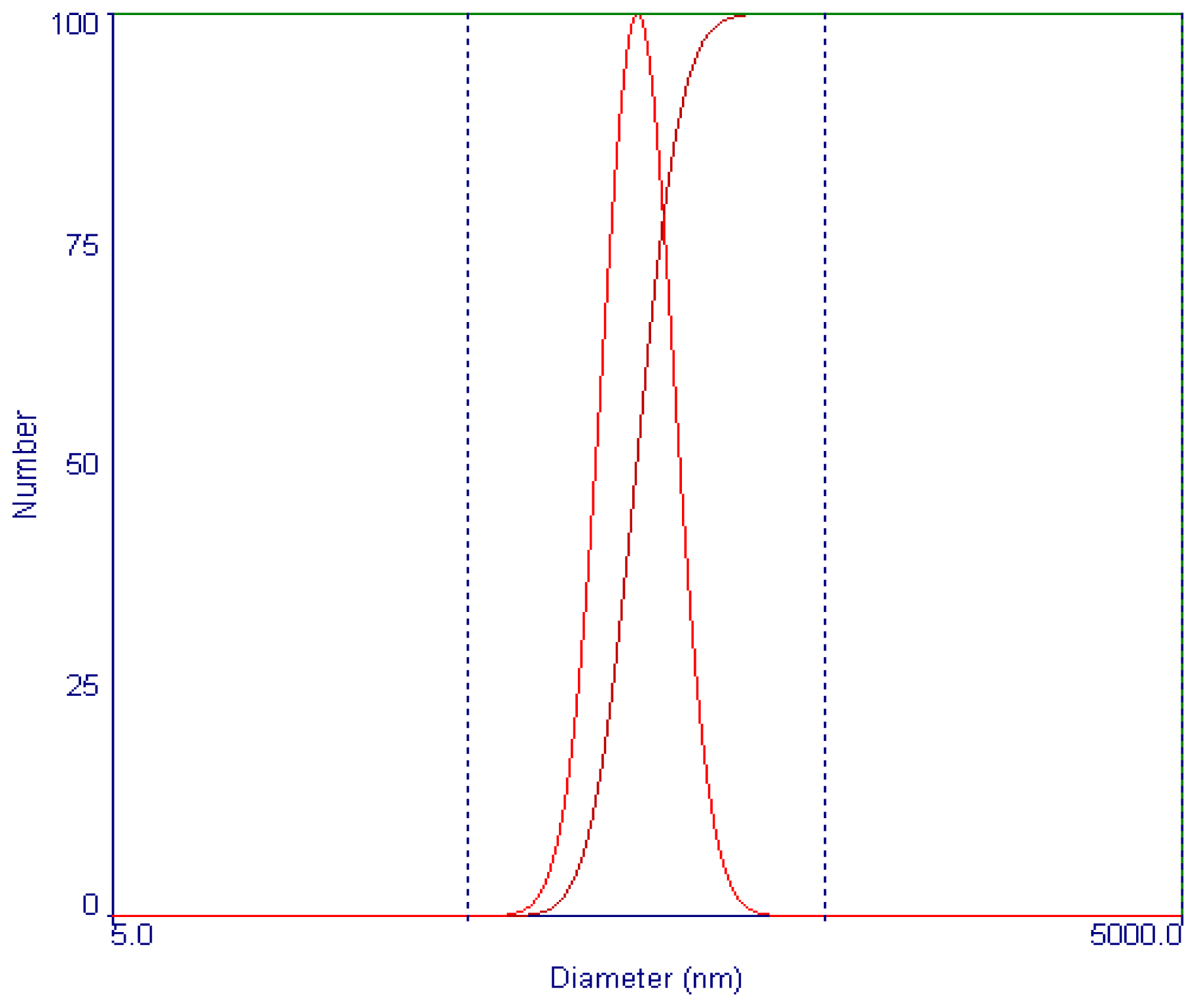



| Trial No. | A: Vinblastine concentration (mg/mL) | B: Drug solution flow rate (mL/min) | C: Precipitation pressure (MPa) | D: Precipitation temperature (ºC) | MPS (nm) (n = 3) |
|---|---|---|---|---|---|
| 1 | 1.25 | 3.3 | 10 | 40 | 480 |
| 2 | 1.25 | 6.7 | 15 | 50 | 372 |
| 3 | 1.25 | 10.0 | 20 | 60 | 240 |
| 4 | 1.25 | 13.3 | 25 | 70 | 159 |
| 5 | 2.50 | 3.3 | 15 | 60 | 256 |
| 6 | 2.50 | 6.7 | 10 | 70 | 254 |
| 7 | 2.50 | 10.0 | 25 | 40 | 258 |
| 8 | 2.50 | 13.3 | 20 | 50 | 301 |
| 9 | 3.75 | 3.3 | 20 | 70 | 355 |
| 10 | 3.75 | 6.7 | 25 | 60 | 121 |
| 11 | 3.75 | 10.0 | 10 | 50 | 347 |
| 12 | 3.75 | 13.3 | 15 | 40 | 287 |
| 13 | 5.00 | 3.3 | 25 | 50 | 284 |
| 14 | 5.00 | 6.7 | 20 | 40 | 213 |
| 15 | 5.00 | 10.0 | 15 | 70 | 366 |
| 16 | 5.00 | 13.3 | 10 | 60 | 438 |
| K1a | 312.75 | 343.75 | 379.75 | 309.50 | - |
| K2 | 267.25 | 240.00 | 320.25 | 326.00 | - |
| K3 | 277.50 | 302.75 | 277.25 | 263.75 | - |
| K4 | 325.25 | 296.25 | 205.50 | 283.50 | - |
| Rb | 58.00 | 103.75 | 174.25 | 62.25 | - |
| Optimal level | A2 | B2 | C4 | D3 | - |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhang, X.; Zhao, X.; Zu, Y.; Chen, X.; Lu, Q.; Ma, Y.; Yang, L. Preparation and Physicochemical Properties of Vinblastine Microparticles by Supercritical Antisolvent Process. Int. J. Mol. Sci. 2012, 13, 12598-12607. https://doi.org/10.3390/ijms131012598
Zhang X, Zhao X, Zu Y, Chen X, Lu Q, Ma Y, Yang L. Preparation and Physicochemical Properties of Vinblastine Microparticles by Supercritical Antisolvent Process. International Journal of Molecular Sciences. 2012; 13(10):12598-12607. https://doi.org/10.3390/ijms131012598
Chicago/Turabian StyleZhang, Xiaonan, Xiuhua Zhao, Yuangang Zu, Xiaoqiang Chen, Qi Lu, Yuliang Ma, and Lei Yang. 2012. "Preparation and Physicochemical Properties of Vinblastine Microparticles by Supercritical Antisolvent Process" International Journal of Molecular Sciences 13, no. 10: 12598-12607. https://doi.org/10.3390/ijms131012598





