Photocatalytic Degradation of p-Cresol by Zinc Oxide under UV Irradiation
Abstract
:1. Introduction
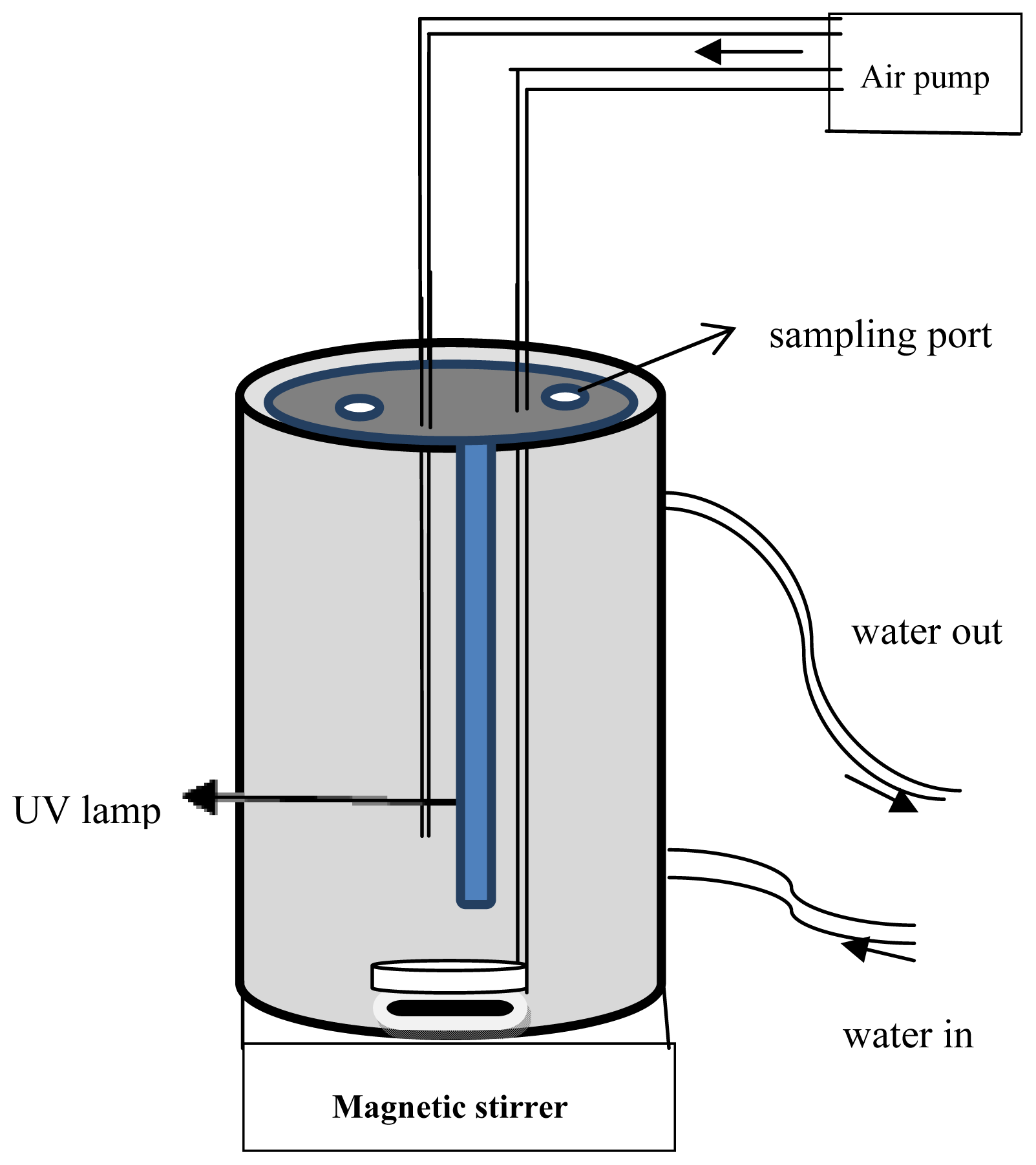
2. Materials and Methods
3. Results and Discussion
3.1. Effect of Photocatalyst Loading
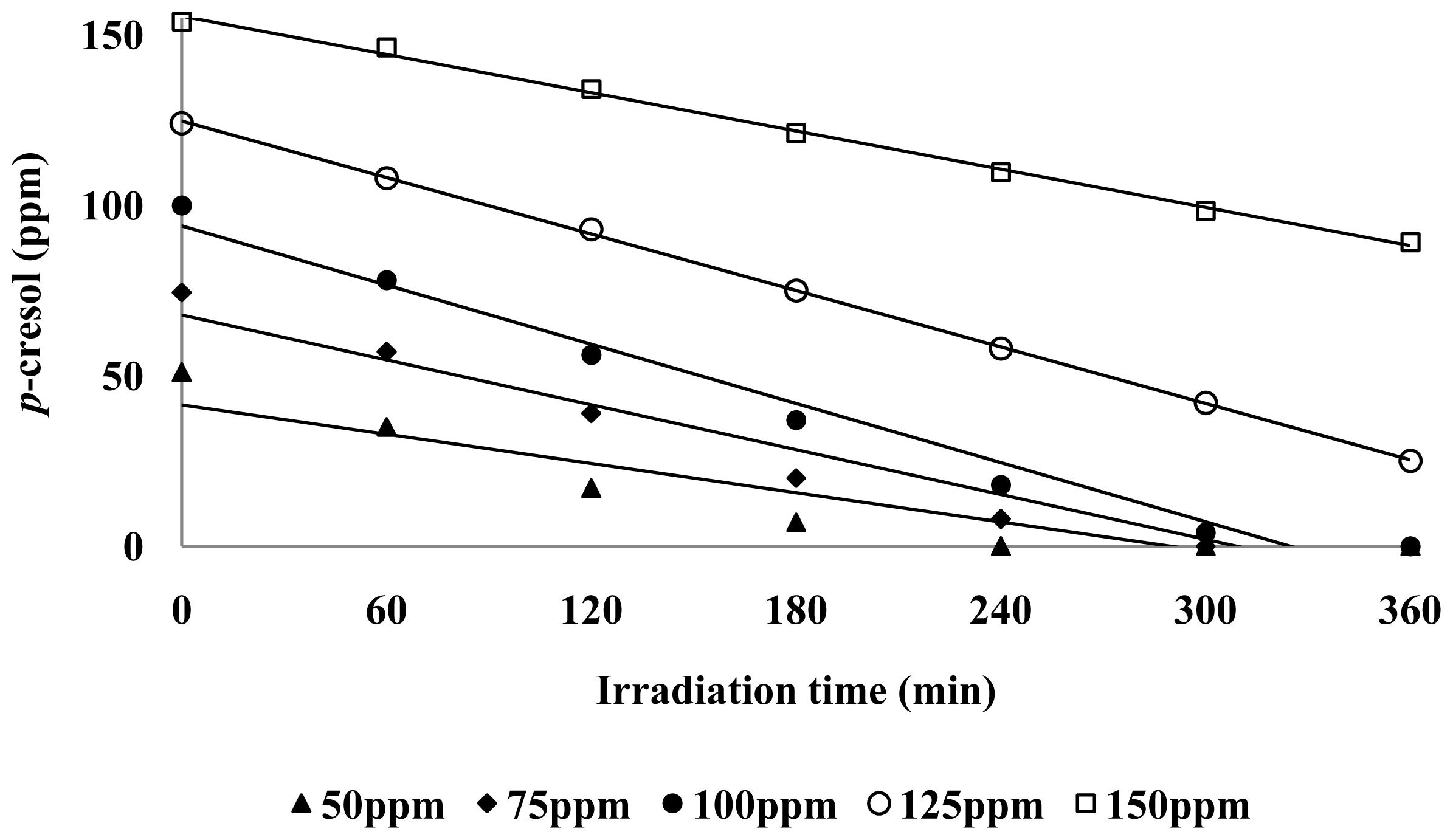
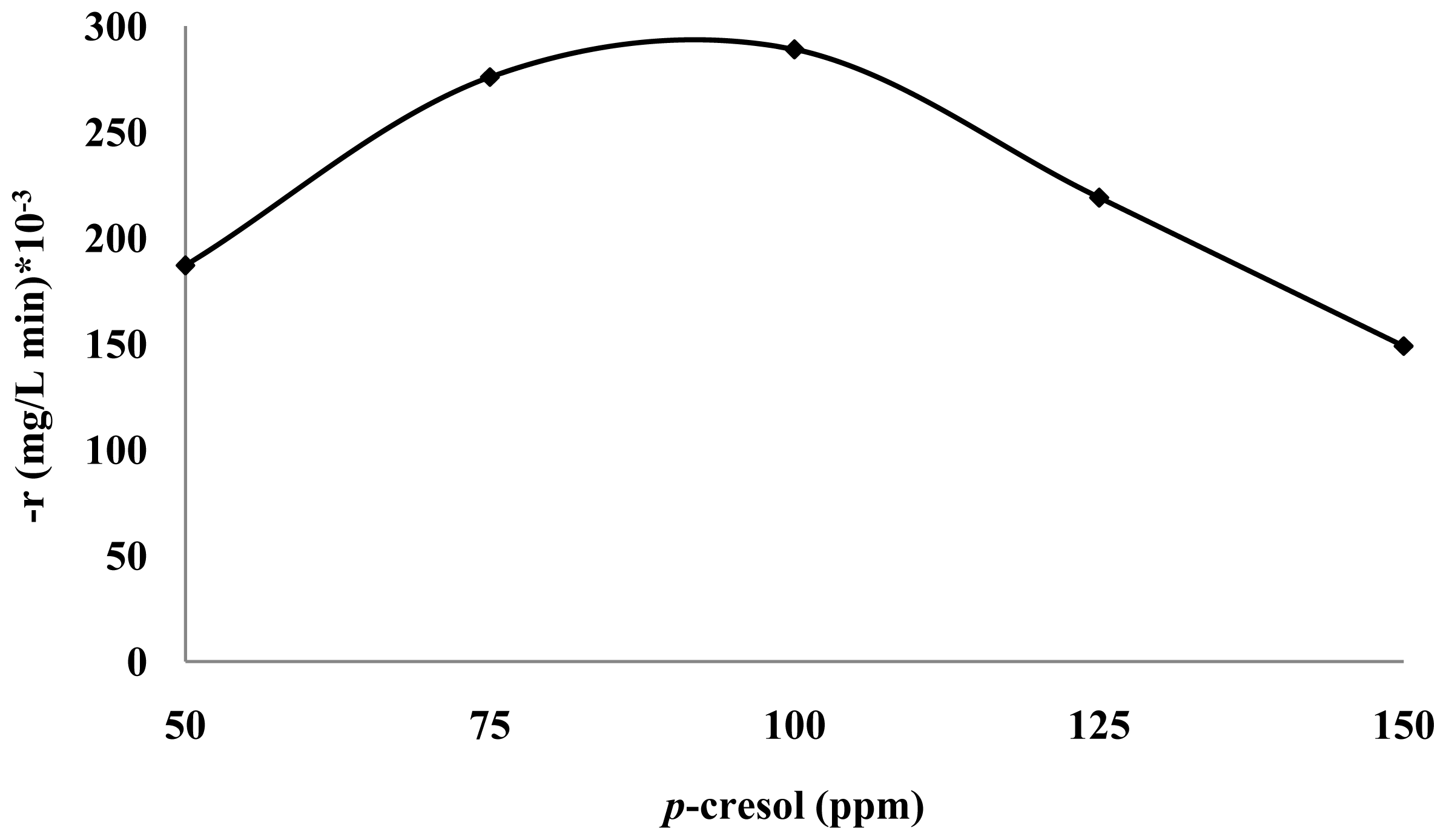
3.2. Effect of p-Cresol Concentration
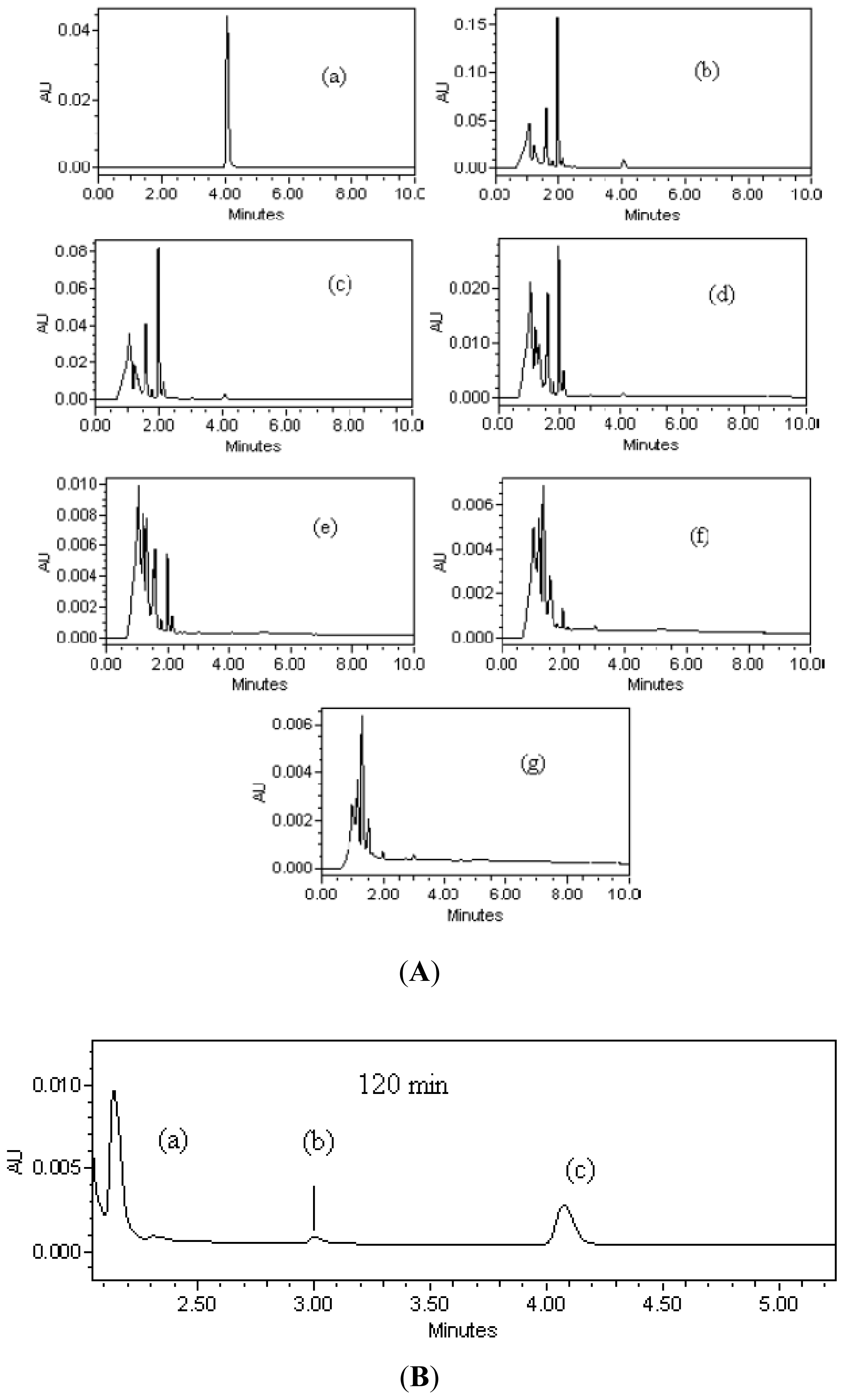
3.3. Photodecomposition Kinetics
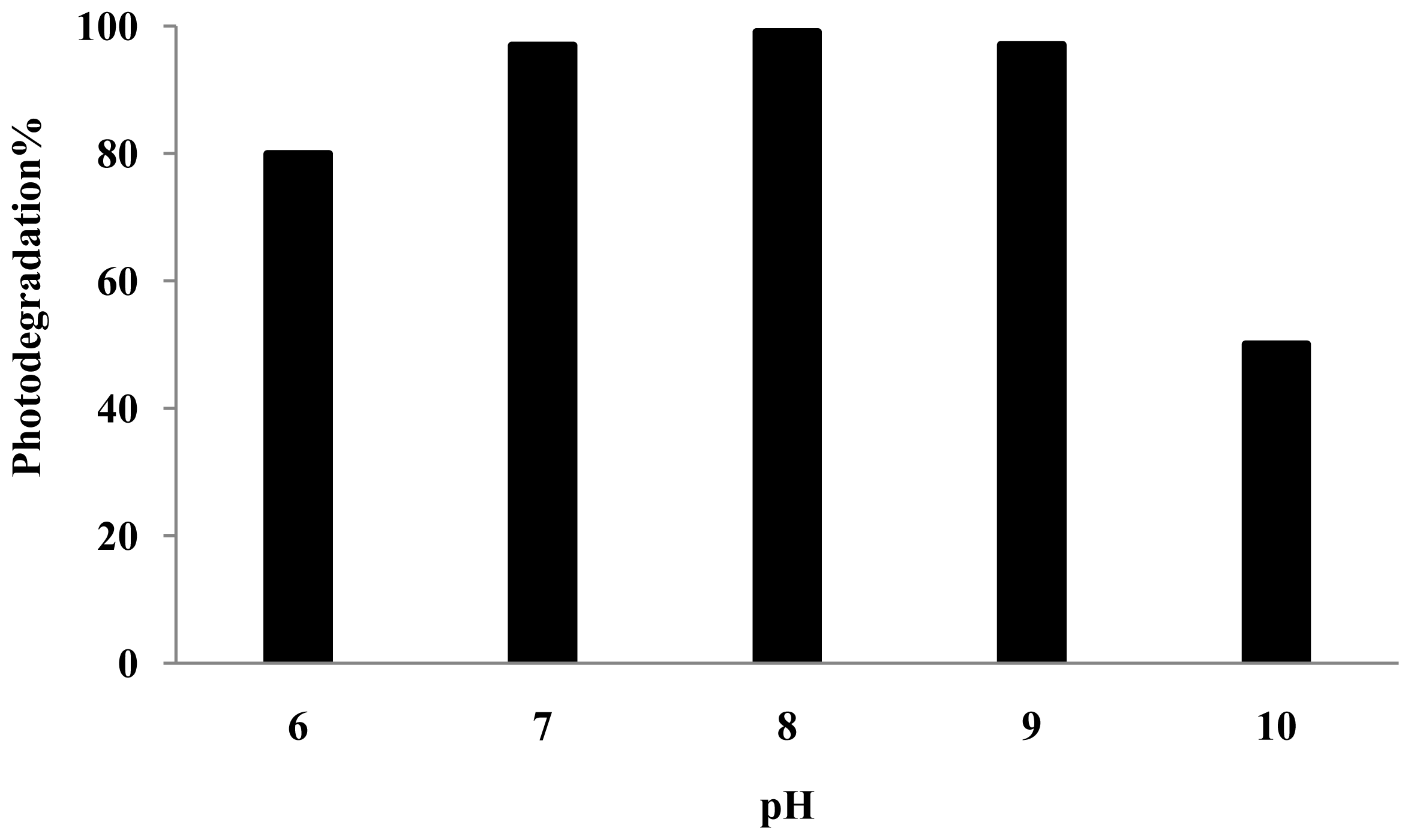
3.4. Effect of pH
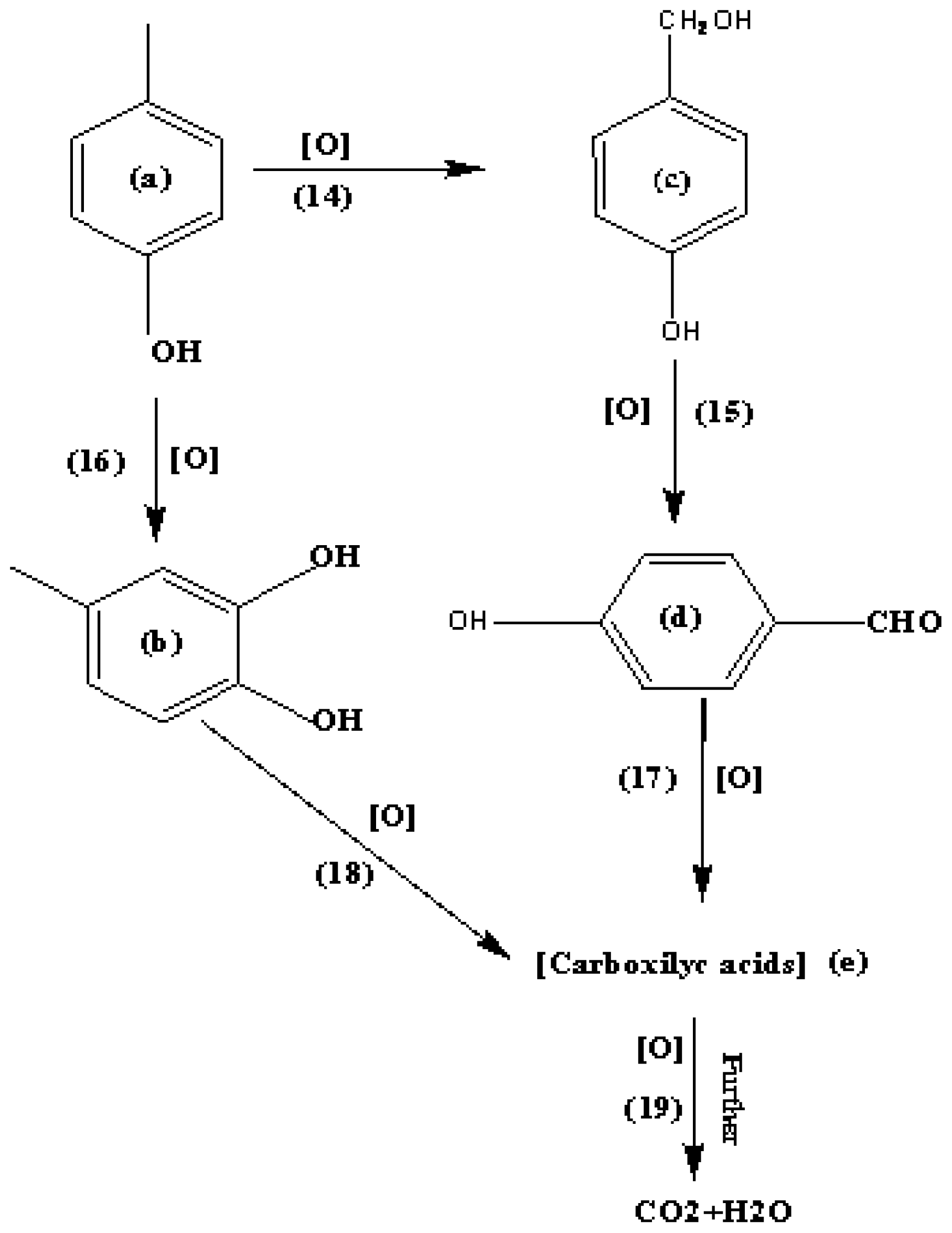
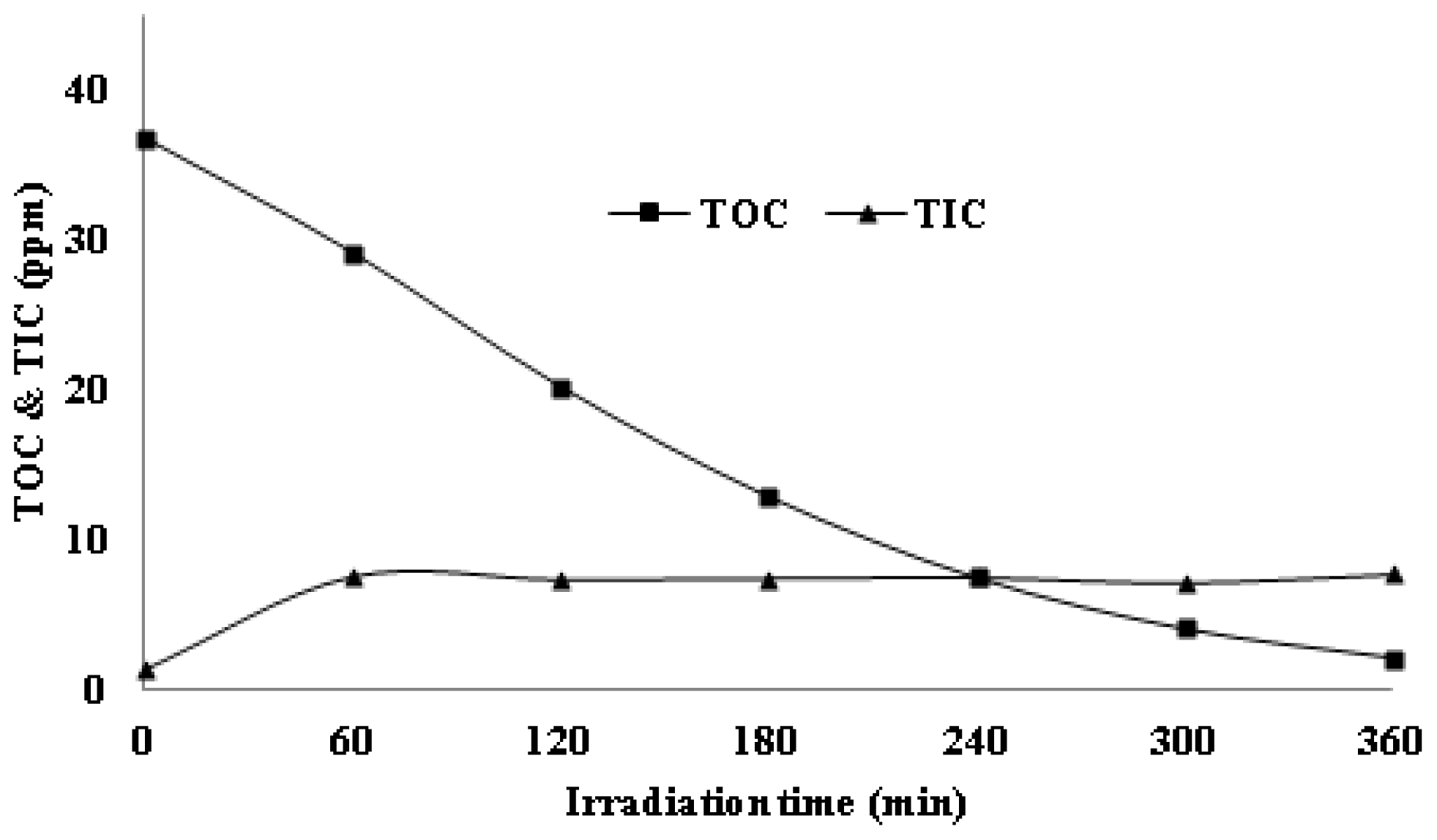
3.5. Mineralization
3.6. Reusability
4. Conclusion
References
- Peiró, A.M.; Ayllón, J.A.; Peral, J.; Doménech, X. TiO2-photocatalyzed degradation of phenol and ortho-substituted phenolic compounds. Appl. Catal. B: Environ 2001, 30, 359–373. [Google Scholar]
- Rathi, A.; Rajor, H.K.; Sharma, R.K. Photodegradation of direct yellow-12 using UV/H2O2/Fe2+. J. Hazard. Mater 2003, 102, 231–241. [Google Scholar]
- Schmelling, D.C.; Gray, K.A. Photocatalytic transformation and mineralization of 2, 4, 6-trinitrotoluene (TNT) in TiO2 slurries. Water Res 1995, 29, 2651–2662. [Google Scholar]
- Glaze, W. Drinking-water treatment with ozone. Environ. Sci. Technol 1987, 21, 224–230. [Google Scholar]
- Litter, M.I. Heterogeneous photocatalysis: Transition metal ions in photocatalytic systems. Appl. Catal. B: Environ 1999, 23, 89–114. [Google Scholar]
- Brezova, V.; Stasko, A. Spin trap study of hydroxyl radicals formed in the photocatalytic system TiO2-water-p-cresol-oxygen. J. Catal 1994, 147, 156–162. [Google Scholar]
- Pardeshi, S.K.; Patil, A.B. A simple route for photocatalytic degradation of phenol in aqueous zinc oxide suspension using solar energy. Solar Energ, 2008; 82, 700–705. [Google Scholar]
- Chen, C.C.; Lu, C.S.; Chung, Y.C. Photocatalytic degradation of ethyl violet in aqueous solution mediated by TiO2 suspensions. J. Photochem. Photobiol. A: Chem 2006, 181, 120–125. [Google Scholar]
- Kandavelu, V.; Kastien, H.; Thampi, K.R. Photocatalytic degradation of isothiazolin-3-ones in water and emulsion paints containing nanocrystalline TiO2 and ZnO catalysts. Appl. Catal. B: Environ 2004, 48, 101–111. [Google Scholar]
- Lachheb, H.; Puzenat, E.; Houas, A.; Ksibi, M.; Elaloui, E.; Guillard, C.; Herrmann, J.M. Photocatalytic degradation of various types of dyes (Alizarin S, Crocein Orange G, Methyl Red, Congo Red, Methylene Blue) in water by UV-irradiated titania. Appl. Catal. B: Environ 2002, 39, 75–90. [Google Scholar]
- Mai, F.; Chen, C.; Chen, J.; Liu, S. Photodegradation of methyl green using visible irradiation in ZnO suspensions: Determination of the reaction pathway and identification of intermediates by a high-performance liquid chromatography-photodiode array-electrospray ionization-mass spectrometry method. J. Chromatogr. A 2008, 1189, 355–365. [Google Scholar]
- Özgür, Ü.; Alivov, Y.; Liu, C.; Teke, A.; Reshchikov, M.; Do an, S.; Avrutin, V.; Cho, S.; Morkoc, H. A comprehensive review of ZnO materials and devices. J. Appl.Phys. 2005, 98, 041301. [Google Scholar]
- Akyol, A.; Bayramoglu, M. Photocatalytic degradation of Remazol Red F3B using ZnO catalyst. J. Hazard. Mater 2005, 124, 241–246. [Google Scholar]
- Akyol, A.; Yatmaz, H.C.; Bayramoglu, M. Photocatalytic decolorization of Remazol Red RR in aqueous ZnO suspensions. Appl. Catal. B: Environ 2004, 54, 19–24. [Google Scholar]
- da Silva, C.G.; Faria, J.L. Photochemical and photocatalytic degradation of an azo dye in aqueous solution by UV irradiation. J. Photochem. Photobiol. A: Chem 2003, 155, 133–143. [Google Scholar]
- Guillard, C.; Disdier, J.; Herrmann, J.M.; Lehaut, C.; Chopin, T.; Malato, S.; Blanco, J. Comparison of various titania samples of industrial origin in the solar photocatalytic detoxification of water containing 4-chlorophenol. Catal. Today 1999, 54, 217–228. [Google Scholar]
- Height, M.; Pratsinis, S.; Mekasuwandumrong, O.; Praserthdam, P. Ag-ZnO catalysts for UV-photodegradation of methylene blue. Appl. Catal. B: Environ 2006, 63, 305–312. [Google Scholar]
- Konstantinou, I.K.; Albanis, T.A. TiO2-assisted photocatalytic degradation of azo dyes in aqueous solution: kinetic and mechanistic investigations: A review. Appl. Catal. B: Environ 2004, 49, 1–14. [Google Scholar]
- Mehrotra, K.; Yablonsky, G.S.; Ajay, K. Kinetic studies of photocatalytic degradation in a TiO2 slurry system: Distinguishing working regimes and determining rate dependences. Ind. Eng. Chem. Res 2003, 42, 2273–2281. [Google Scholar]
- Callahan, M.; Slimak, M.; Gabel, N.; May, I.; Fowler, C.; Freed, J.R.; Jennings, P.; Dupree, R. Water-Related Environmental Fate of 129 Priority Pollutants. Volume 1: Introduction and technical background, metals and inorganics, pesticides and PCBs; Final Report 069443; U.S. Environmental Protection Agency: Washington, DC, USA, 1979. [Google Scholar]
- Cooper, E. On the Relations of Phenol and Meta-Cresol to Proteins; A Contribution to our knowledge of the Mechanism of Disinfection. Biochem. J 1912, 6, 362–387. [Google Scholar]
- Kavitha, V.; Palanivelu, K. Destruction of cresols by Fenton oxidation process. Water Res 2005, 39, 3062–3072. [Google Scholar]
- Flox, C.; Cabot, P.-L.; Centellas, F.; Garrido, J.A.; Rodríguez, R.M.; Arias, C.; Brillas, E. Solar photoelectro-Fenton degradation of cresols using a flow reactor with a boron-doped diamond anode. Appl. Catal. B: Environ 2007, 75, 17–28. [Google Scholar]
- Guyer, H. Industrial Processes and Waste Stream Management; John Wiley & Sons Inc: New York, NY, USA, 1998. [Google Scholar]
- Abdollahi, Y.; Abdullah, A.H.; Zainal, Z.; Yusof, N.A. Photodegradation of m-cresol by Zinc Oxide under Visible-light Irradiation. Int. J. Chem 2011, 3, 31–43. [Google Scholar]
- Abdollahi, Y.; Abdullah, A.H.; Zainal, Z.; Yusof, N.A. Photodegradation of o-cresol by ZnO under UV irradiation. J. Am. Sci 2011, 7, 165–170. [Google Scholar]
- Fox, M.; Dulay, M. Heterogeneous photocatalysis. Chem. Rev 1993, 93, 341–357. [Google Scholar]
- Konstantinou, I.K.; Albanis, T.A. TiO2-assisted photocatalytic degradation of azo dyes in aqueous solution: kinetic and mechanistic investigations: A review. Appl. Catal. B: Environ 2004, 49, 1–14. [Google Scholar]
- Lathasree, S.; Rao, A.; SivaSankar, B.; Sadasivam, V.; Rengaraj, K. Heterogeneous photocatalytic mineralisation of phenols in aqueous solutions. J. Mol. Catal. A: Chem 2004, 223, 101–105. [Google Scholar]
- Al-Ekabi, H.; De Mayo, P. Surface photochemistry: on the mechanism of the semiconductor photoinduced valence isomerization of hexamethyl Dewar benzene to hexamethylbenzene. J. Phys. Chem 1986, 90, 4075–4080. [Google Scholar]
- Hatipoglu, A.; San, N.; ÇInar, Z. An experimental and theoretical investigation of the photocatalytic degradation of meta-cresol in TiO2 suspensions: a model for the product distribution. J. Photochem. Photobiol. A: Chem 2004, 165, 119–129. [Google Scholar]
- Melián, E.P.; Díaz, O.G.; Araña, J.; Rodríguez, J.M.D.; Rendón, E.T.; Melián, J.A.H. Kinetics and adsorption comparative study on the photocatalytic degradation of o-, m- and p-cresol. Catal. Today 2007, 129, 256–262. [Google Scholar]
- Bangun, J.; Adesina, A.A. The photodegradation kinetics of aqueous sodium oxalate solution using TiO2 catalyst. Appl. Catal. A: Gen 1998, 175, 221–235. [Google Scholar]
- Lea, J.; Adesina, A.A. The photo-oxidative degradation of sodium dodecyl sulphate in aerated aqueous TiO2 suspension. J. Photochem. Photobiol. A: Chem 1998, 118, 111–122. [Google Scholar]
- Lea, J.; Adesina, A.A. Oxidative degradation of 4 nitrophenol in UV illuminated titania suspension. J. Chem. Technol. Biotechnol 2001, 76, 803–810. [Google Scholar]
- Hudaya, T.; Qazaq, A.S.; Lucien, F.P.; Adesina, A.A. Design of CeyCoxTi (1x) O3+ Perovskite for Photocatalysis: A Statistical Study. J. Adv. Oxid. Technol 2009, 12, 16–28. [Google Scholar]
- Sakthivel, S.; Neppolian, B.; Shankar, M.V.; Arabindoo, B.; Palanichamy, M.; Murugesan, V. Solar photocatalytic degradation of azo dye: comparison of photocatalytic efficiency of ZnO and TiO2. Solar Energ. Mater. Solar Cells 2003, 77, 65–82. [Google Scholar]
- Degen, A.; Kosec, M. Effect of pH and impurities on the surface charge of zinc oxide in aqueous solution. J. Eur. Ceram. Soc 2000, 20, 667–673. [Google Scholar]
- Reed, J.S. Introduction to the Principles of Ceramic Processing; John Wiley & Sons: New York, NY, USA, 1986. [Google Scholar]
- Zhao, J.; Wu, T.; Wu, K.; Oikawa, K.; Hidaka, H.; Serpone, N. Photoassisted degradation of dye pollutants. 3. Degradation of the cationic dye rhodamine B in aqueous anionic surfactant/TiO2 dispersions under visible light irradiation: Evidence for the need of substrate adsorption on TiO2 particles. Environ. Sci. Technol 1998, 32, 2394–2400. [Google Scholar]
- Nouri, S.; Haghseresht, F.; Lu, G.Q.M. Comparison of adsorption capacity of p-Cresol & p-Nitrophenol by activated carbon in single and double solute. Adsorption 2002, 8, 215–223. [Google Scholar]
- Shukla, G. Magnetic and optical properties of epitaxial n-type Cu-doped ZnO thin films deposited on sapphire substrates. Appl. Phys. A: Mater. Sci. Process 2009, 97, 115–118. [Google Scholar]
- Ullah, R.; Dutta, J. Photocatalytic degradation of organic dyes with manganese-doped ZnO nanoparticles. J. Hazard. Mater 2008, 156, 194–200. [Google Scholar]
- Davis, A.; Huang, C. Removal of phenols from water by a photocatalytic oxidation process. Water Sci. Technol 1989, 21, 455–464. [Google Scholar]
- Wang, K.H.; Hsieh, Y.H.; Chen, L.J. The heterogeneous photocatalytic degradation, intermediates and mineralization for the aqueous solution of cresols and nitrophenols. J. Hazard. Mater 1998, 59, 251–260. [Google Scholar]
- Gaya, U.I.; Abdullah, A.H.; Zainal, Z.; Hussein, M.Z. Photocatalytic Degradation of 2, 4-dichlorophenol in Irradiated Aqueous ZnO Suspension. Int. J. Chem 2010, 2, 180–189. [Google Scholar]
- Minero, C.; Mariella, G.; Maurino, V.; Vione, D.; Pelizzetti, E. Photocatalytic transformation of organic compounds in the presence of inorganic ions. 2. Competitive reactions of phenol and alcohols on a titanium dioxide-fluoride system. Langmuir 2000, 16, 8964–8672. [Google Scholar]
- Heiland, G.; Mollwo, E.; Stöckmann, F. Electronic processes in zinc oxide. In Solid State Physics; Frederick, S., David, T., Eds.; Academic Press: Darmstadt, Germany, 1959; pp. 191–323. [Google Scholar]



© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Abdollahi, Y.; Abdullah, A.H.; Zainal, Z.; Yusof, N.A. Photocatalytic Degradation of p-Cresol by Zinc Oxide under UV Irradiation. Int. J. Mol. Sci. 2012, 13, 302-315. https://doi.org/10.3390/ijms13010302
Abdollahi Y, Abdullah AH, Zainal Z, Yusof NA. Photocatalytic Degradation of p-Cresol by Zinc Oxide under UV Irradiation. International Journal of Molecular Sciences. 2012; 13(1):302-315. https://doi.org/10.3390/ijms13010302
Chicago/Turabian StyleAbdollahi, Yadollah, Abdul Halim Abdullah, Zulkarnain Zainal, and Nor Azah Yusof. 2012. "Photocatalytic Degradation of p-Cresol by Zinc Oxide under UV Irradiation" International Journal of Molecular Sciences 13, no. 1: 302-315. https://doi.org/10.3390/ijms13010302



